Abstract
Subcutaneous osmotic pump implantation connected to a venous catheter is a well-established method for delivering compounds intravenously for an intermediate duration (approximately two weeks). When prolonged release is desired (approximately four weeks) reduced flow rate is needed with a similar pump volume. With a fixed intra-pump compound concentration, reduced flow rate results in unwanted reduced bioavailability of the compound. Prolonged intravenous delivery would therefore need a pump replacement, resulting in increased discomfort and confounding effects on experimental outcome. To overcome this, we describe a method to double the compound infusion rate for four weeks by implanting two low-flow rate osmotic pumps (2.5 µL/h for 28 days) connected to a jugular vein catheter in a single rat. Rats implanted with a single high-flow rate pump (5 µL/h for 14 days) served as controls. Double pump-implanted rats displayed similar post-operative weight gain and physical activity indicating similar levels of discomfort when compared with single pump-implanted rats. Double pump-implanted rats had an increased risk of pump-related complications (four delivery failures [double pump] versus one delivery failure [single pump]). Our data show that double pump implantation is a feasible alternative to changing pumps or the use of extracorporeal pump systems connected via a long wire to partly restrained animals.
In experimental animal research intravenous administration of therapeutic agents for prolonged durations, especially fast-degradable compounds with low bioavailability and with unknown pharmacokinetics or pharmacodynamics, is often desired. To this end, intravenous catheter systems connected to an external pump have been developed. This method requires restraint of the animal. Tight restraint is however unwanted as it causes stress and harms the animal, making it unethical. 1 The use of long tubes partly overcomes these disadvantages and allows relative freedom of movement for the rats.2,3 Common complications are tubes which are bitten through or twisted, causing blockade of continuous infusion or fatal bleeding.
Another technique is the use of a subcutaneously implanted osmotic minipump connected to a venous catheter. 4 Osmotic minipumps with a fixed reservoir volume can have different duration rates of infusion, implying that increasing delivery duration can only be established by lowering the flow rate. In principle this could be overcome by increasing the concentration of the compound, however some compounds do not dissolve at higher concentrations. Prolonged infusion can be achieved by replacement of fast-flow rate pumps during the experiment. Although implantation of osmotic minipumps is generally considered to be a mild surgical procedure, it definitely impacts on animal welfare, especially when combined with another major surgical intervention. Therefore, pump exchange results in additional discomfort and certainly compromises proper comparison with rats that do not undergo replacement. To circumvent this, we developed a method of simultaneously implanting two long-lasting, low-flow rate pumps combined with another major surgical intervention (i.e. aortic transplantation). This method allows comparative kinetic studies (two versus four weeks infusion) by introducing only one variable with fixed pump reservoir compound concentration and without the need for pump exchange.
Animals
Male Brown Norway rats (BN, n = 32) weighing 230–270 g were obtained from Janvier (St Berthevin Cedex, France), and female Wistar rats (n = 2 used for pilot implantations) weighing 280–310 g were obtained from our own breeding colony. The rats were kept under clean conventional conditions and fed standard rat chow and water ad libitum. All experimental procedures were performed according to European Commission guidelines and Dutch laws and were approved by the animal ethics committee of the University of Groningen (DEC 6624B).
Materials and methods
Catheter and osmotic minipumps
To achieve a pump delivery rate of 5 µL/h for four weeks, two 2ML4 pumps (2.5 µL/h for 28 days) from Alzet (DURECT Corporation, Cupertino, CA, USA) were used. One day prior to pump implantation, the pumps were filled with 5% Tween 80 in saline with or without additional N-octanoyl-dopamine (NOD, 11.16 mg/mL), attached to a saline-filled jugular vein catheter (DURECT Corporation), cut after 1.5 cm and connected to an in-house made Y-tube (Figure 1a). Filled pumps were incubated in saline overnight at 37℃ according to the supplier’s instruction. NOD was our compound of interest, but this will not be further discussed as it is beyond the scope of this article. For comparison, conventional single pump implantation was performed using an Alzet 2ML2 pump (5 µL/h for 14 days) without using the Y-tube.
Procedure of double pump implantation. (a) Osmotic minipumps, catheters and Y-tube ex vivo. Two osmotic minipumps are connected to a Y-tube that is attached to a jugular vein catheter. For implantation, the jugular vein catheter is replaced, separately implanted and connected to the Y-tube in situ again. (b) Submandibular skin incision for jugular vein catheterization. (c) Jugular vein is exposed by the ligation of the cranial end of the dissected vein. (d) Vein is punctured by a bent G20 needle and jugular catheter is introduced into the vein. (e) Catheter is fixed by the caudal suture (*). Cranial suture is knotted around the catheter (#) and loose ends of the cranial and caudal sutures are fixed together crosswise. (f) The jugular catheter is tunneled to the neck incision and subcutaneous pockets for pump implantation are made. (g) Scheme of the implanted double pump: dotted line: neck incision, red: jugular vein, blue: catheters and osmotic minipumps, black: Y-tube, dashed line: subcutaneous pockets. (h) Weight curves of animals. Note that pictures were taken without sterile drapes for pedagogic purposes only; the use of sterile drapes is highly recommended for this kind of surgery. Colour references for online edition only.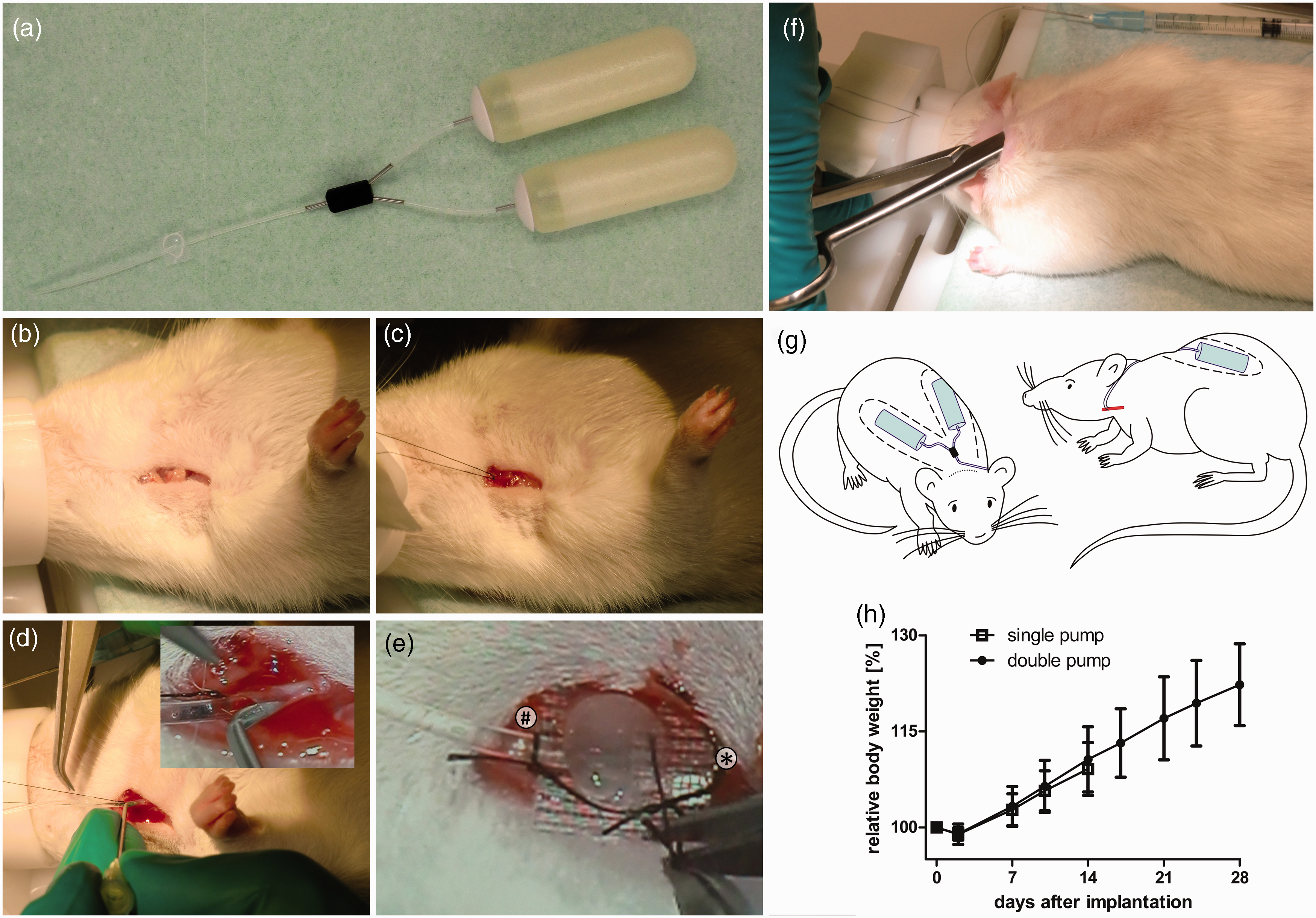
Implantation procedure
Rats were anaesthetized with isoflurane and 0.01 mg/kg body weight buprenorphine and placed in a supine position. On post-operative day 1, the same dosage of buprenorphine was used as analgesia. Aortic transplantation (using Dark Agouti donors) was performed as described previously. 5 For jugular vein cannulation, a 1 cm long incision at the right shoulder close to the base of the neck was made (Figure 1b). The jugular vein was bluntly dissected and the cranial end was tightly ligated using a 5–0 non-absorbable suture, loose ends were fixed at the table to expose the vein (Figure 1c). A second suture was loosely placed at the caudal end. The vein was punctured with a bent G20 needle. Through the curved part of the needle the saline-filled catheter was introduced 3.8 cm into the vein (Figure 1d), such that the catheter tip ended in the cranial cava vein just before the right atrium. The catheter was fixed by tightening the caudal suture. The cranial suture was unfixed and knotted around the catheter (Figure 1e). Intravascular access was checked by withdrawing blood and flushing with saline. The jugular vein catheter was tunneled subcutaneously to the dorsal side.
For implantation of the double pump, the rat was placed in a prone position. A 2 cm long skin incision was made across the neck (Figure 1f). Subsequently, a subcutaneous pocket was made at each flank and pumps were implanted, connected to the Y-tube. Finally the Y-tube was connected to the jugular vein catheter and skin incisions were closed. Figure 1g shows a schematic representation of the anatomical location of the implanted pumps connected to the jugular vein catheter.
Results
List of complications.
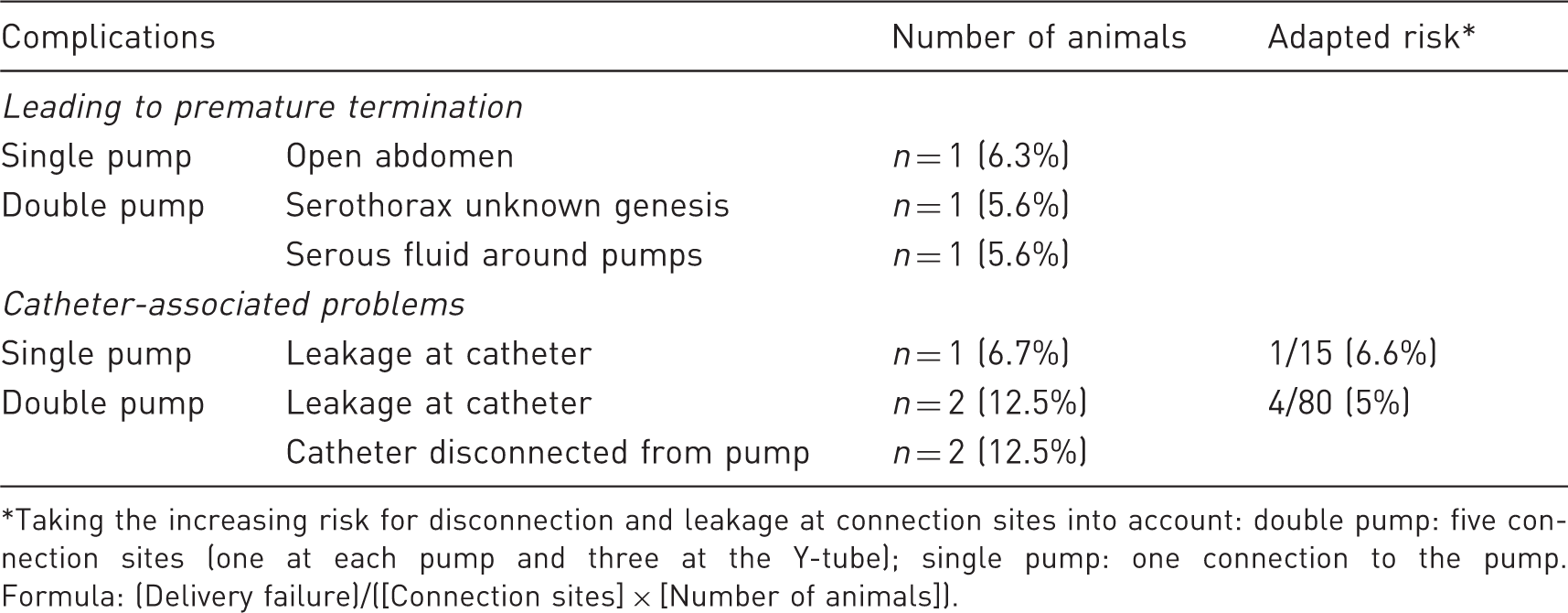
Taking the increasing risk for disconnection and leakage at connection sites into account: double pump: five connection sites (one at each pump and three at the Y-tube); single pump: one connection to the pump. Formula: (Delivery failure)/([Connection sites] × [Number of animals]).
In the remaining cases, the pumps were removed at 14 days (15 rats; 15 catheter connections [single pump]) or 28 days (16 rats, 80 catheter connections [double pump]). Inspection at sacrifice revealed that in the single pump group, 6.7% (1 out of 15) of the connections were leaking, whereas in the double pump group, 2.5% (2 out of 80) were leaking and another 2.5% (2 out of 80) were disconnected from the pump (total failure rate: 5% [4 out of 80]). No significant differences in weight gain (Figure 1h) or physical activity were detected between both groups within the first two weeks.
Discussion
Several methods have been described for continuous intravenous administration of compounds in unrestrained rats. To increase infusion rates over a longer period of time, we showed that the simultaneous implantation of two slow-flow rate osmotic pumps connected to a jugular vein catheter is feasible and is not linked to additional signs of discomfort compared with single pump-implanted rats.
Both lethal complications in the double pump group were manifested at day 13 post implantation. As all single pump-implanted animals were terminated at day 14 (except for one case, not related to the pump), this indicated that premature loss was indeed associated with double versus single pump implantation. Double pump implantation appeared to be associated with a higher risk of pump-related complications leading to delivery failure or earlier termination of the animal. One should take into account that these rats had two catheters with five connection sites (one at each pump and three at the Y-tube) instead of one catheter with one connection to the pump, therefore with increased risk that one of these connections could fail. Adapted relative risk, as calculated for the individual rat, was comparable between both groups (6.6% [single pump] versus 5% [double pump] [Table 1]). We believe that delivery failure is related to the five-fold higher number of connections in double pump-implanted rats, although possible consequences of the longer follow-up time (28 versus 14 days) cannot be excluded.
In conclusion, simultaneous double pump implantation is a feasible method for achieving prolonged intravenous administration in unrestrained rats.
Footnotes
Acknowledgments
We thank Hans Thole, Research Appliance Manufacturer, University Medical Center Groningen, for technical support with development and production of the Y-tubes. This research received no specific grants from any funding agency in the public, commercial, or not-for-profit sectors.
