Abstract
Individual and permanent identification of experimental animals is a common and often essential research practice. There is little information available on the short-term effects of these procedures on the animals. In this study, seven rats were implanted with telemetric devices. The effects of three different identification methods (ear tattoo, ear notching and microtattoo) were compared. Cardiovascular data were collected for 24 h after the procedures. Time periods of 0–1, 1–4, 4–16 h (dark) and 16–24 h after the procedure were analysed separately. The most pronounced differences in measured parameters were observed during the first hour after the procedures were performed. Mean arterial pressure (MAP) was significantly higher (P < 0.012) following the ear tattoo than the microtattoo procedure by a difference of approximately 5 mmHg. Heart rate (HR) was significantly elevated (P < 0.001) after ear tattoo compared with both ear notching (Δ = 31 beats per minute [bpm]) and microtattoo (Δ = 44 bpm). During the 1–4 h period and the following dark period, the MAP was highest in the ear notching group, but no differences were observed in the HRs. During the following dark period (4–16 h) and the next day (16–24 h) differences in MAP and HR were minor. In conclusion, microtattoo appears to cause the mildest changes in HR and blood pressure. Based on these results, ear tattoo and ear notching should be replaced by microtattoo whenever possible.
Individual identification (ID) of experimental animals is an essential part of most animal experiments. Permanent ID methods include tattooing of ears, 1 foot pads, 2–4 tail 5,6 and body, 7 removal of parts of the ears, 8 toes 8,9 and tail 10 and adding identification devices to the body, such as microchips 11–13 or ear tags. Freeze marking has also been used, but it is only applicable to coloured animals. 14 According to the FELASA working group on identification methods, the most commonly used ID methods in rodents in both Europe and USA are ear notching and ear tagging. 15
Identification is one of the most common procedures to which experimental animals are subjected. Even small differences in pain and disturbance and possible long-term effects of ID methods should be considered as important, since such a large number of animals are subjected to ID procedures. An optimal identification method ‘should be simple, easy to apply, easy to read, harmless to the animal and offer no potential interference with experimentation or interpretation of the data’. 16 All of the ID methods described have their shortcomings, but efforts should be made to use only the most reliable and least harmful of the existing methods.
The reported long-term adverse effects of ID fall mainly into three categories: inflammation, neoplastic changes and chronic pain. These problems are mainly associated with ID methods involving the implantation of foreign objects. For example, ear tags have been associated with chronic inflammation 17,18 and neoplastic lesions. 19,20 Neoplastic lesions have also been reported following the placement of microchips. 21–23 However, foreign material is also introduced into the body by tattooing. The inflammatory reaction and tissue trauma resulting from tattooing have been reported to last for two weeks in mice. 24 Nevertheless, tail tattooing has been demonstrated to have no affect on the behaviour or growth of mice. 6,25 The methods based on removing small parts of the body have been claimed not to cause long-term adverse effects on the animals. Apparently, there are no changes in the growth and development of rats and mouse pups subjected to toe clipping, although these animals might perform more poorly in behavioural tests requiring the use of the removed toes, e.g. grip strength test. 26–28
The long-term effects of tail amputation depend on the size of the piece removed. Removal of a 2.5 mm piece, e.g. in genotyping, does not evoke changes in the growth or behaviour of mice, 25 whereas the amputation of 25 mm has been shown to result in long-lasting hyperalgesia. 29 However, tail amputation is not commonly used for ID. The most commonly used ID method involving removal of tissue is ear notching. We could not locate any publications indicating long-term adverse effects in animals subjected to ear notching. Ear notching is generally considered a less severe procedure than toe clipping or tail amputation.
The short-term impacts of the ID methods consist of the combination of pain, fear and disturbance caused by the procedure. The relative aversiveness of the different ID methods has not yet been established. There are some studies characterizing the immediate effects of tissue biopsy techniques used in genotyping. Some biopsy techniques used in genotyping are similar to the commonly used ID methods. It has been shown that the response to these procedures (as measured by heart rate [HR], body temperature and locomotor activity) lasts about one hour. However, the effects of the procedure cannot be distinguished from the effects of the handling and immobilization associated with the procedure. The stress reaction following simple handling and restraint without the actual biopsy procedure appears to be comparable. 30–32
The aim of this study was to characterize and compare the short-term effects of three different ID methods: ear notching, ear tattoo and microtattoo on the foot pads. Ear notching and ear tattooing were chosen for the study because they are common methods of rodent identification; microtattooing was chosen for its potential as a less stressful and more humane method of identification. The response of the rats to these techniques has been assessed by telemetric monitoring of HR and blood pressure with the goal being to determine which method is least disturbing to the animals and also to establish the duration of the reactions caused by these ID methods.
Materials and methods
The study protocol was reviewed and approved by the Animal Care and Use Committee of the University of Kuopio. This study was conducted in the Laboratory Animal Centre (LAC), University of Oulu and in the National Laboratory Animal Center (NLAC), University of Kuopio.
Animals and housing
A total of seven male barrier-bred rats were used in this study. The study population consisted of four Hsd:Sprague Dawley rats from LAC, Oulu, Finland and three were HsdBrlHan:WIST from NLAC, Kuopio, Finland. They were bred in barriers free of the pathogens listed in the FELASA recommendations for health monitoring. 33 The rats were housed in solid bottom Makrolon® polycarbonate cages (55 × 35 × 20 cm) with a wire-grid lid (Bayer AG, Leverkusen, Germany). Ambient temperature was kept at 20 ± 2°C and relative air humidity at 50 ± 10%. Room illumination followed a 12/12 h cycle, with lights on at 07:00 h. Food (R36, Lantmännen Ab, Södertälje, Sweden) and tap water in polycarbonate bottles were available ad libitum. Aspen chips (3 L) were used as bedding (Tapvei Oy, Kaavi, Finland) and rectangular aspen tubes (20 × 10.5 × 10.5 cm, wall thickness 1.5 cm) as enrichment (Tapvei Oy). Cages, bedding, aspen tubes and water bottles were renewed once a week. The animals were situated geographically in two locations: the four Hsd:Sprague Dawley rats in LAC, Oulu, Finland and the three HsdBrlHan:WIST rats in NLAC, Kuopio, Finland.
Study design
The rats were housed in groups of three. One, randomly chosen rat from each cage (7 rats altogether) was implanted with a telemetry transmitter (TA11PA-C40, Data Sciences International, St Paul, MN, USA) at the age of 11–12 weeks. Each study rat with the telemetric device was housed with two cage mates which did not participate in the study. The details of anaesthesia, analgesia, surgery and collection of the telemetric data have been described in a previous publication. 32 Prior to this study, these same animals were used for a non-invasive handling study described by Baturaite et al. 32 The rats were 25 weeks old when this study began. The animals were kept in the same groups of three throughout this experiment and the prior handling experiments. They were used to each other and also used to being handled. Care was taken to handle all rats in both locations in a similar way and frequency. All procedures were discussed thoroughly and practised among the technicians performing the procedures beforehand to ascertain that the procedures would be done in an identical manner in both locations and by all technicians.
The experiment used a crossover design. Each of the implanted animals was subjected to the three different identification methods. The order of the treatments was randomized. Between each treatment, the rats were allowed a washout period of one week.
Identification methods
Ear tattoo
The rat was removed from the cage and held firmly by encircling the body with one hand and supporting the body with the other hand. An assistant tattooed the actual number 15 on the rat's left ear using tattooing pliers (Ebeco AB, Göteborg, Sweden) and black tattooing paste (H Hauptner & Richard Herberholz GmbH & Co, Solingen, Germany) (Figure 1).
Ear tattooing procedure
Ear notching
The rat was removed from the cage and held firmly by encircling the body with one hand and supporting the body with the other hand. An assistant marked the number 7 on the rat's right ear (two holes on the periphery of the ear) using Kopferdam pliers (Dentaldepot Ltd, North Andover, MA, USA) (Figure 2).

Ear notching procedure
Microtattoo
The rat was removed from the cage and held firmly by encircling the body with one hand and supporting the body with the other hand. An assistant exposed the rear feet by holding them between the thumb and the index finger and tattooed the number 33 by piercing the distal toe pad of both third phalanges on the rear feet. The apparatus used was Aramis and it used green tattooing ink (Ketchum Manufacturing Inc, Ottawa, Canada) (Figure 3).
Microtattooing procedure
The identification procedures were done at 14:00 h on each occasion (all identifications were conducted between 14:00 and 14:07 h). Each identification procedure lasted less than one minute and was performed standing next to the animals' home cages. After the procedure, the rats were immediately placed back into their home cage. The cage mates of each experimental animal were not subjected to any procedures, but remained in the home cage when the implanted animals were removed from the cage.
The telemetric receivers were under the rats' home cages. The procedures were clocked accurately for each animal in order to analyse the right time windows from the telemetric raw data. All recordings were obtained from the animals' home cages. All animals from NLAC, University of Kuopio were housed in one animal room and all animals from LAC, University of Oulu were housed together in another animal room. None of the animals were removed from the rooms at any point during this study. The rooms were dedicated to this experiment and all other confounding disturbances to the animals were minimized.
Data processing and statistical analyses
The telemetric data were recorded for 10 s every 5 min. The mean arterial pressure (MAP) and the HR recordings were collected. The mean MAP and HR values of the time periods analysed were calculated for each individual rat using 5 min means extracted from the raw data. These individual means were then used in the statistical analysis.
The different identification methods were compared with each other using linear mixed models. The individual was included as a random effect in the model to account for possible dependencies within the data. The results are presented as model-based estimates of the means and their standard errors (SEM).
The equality of variances between the different identification methods was investigated by comparing the coefficients of variation (CV) of each variable from each time period using linear mixed models. Each individual rat had its own CV value calculated from the 5 min means from the raw data.
SPSS 14.0 for Windows (SPSS Inc, Chicago, IL, USA) was the statistical software package used to process and analyse the data. The graphs were created with SigmaPlot 11.0 software (Systat Software Inc, San Jose, CA, USA).
Results
During the first 60 min after the identification procedure, the model-based estimate of the mean MAP was 112.1 (1.3) mmHg after the ear tattoo, 109.4 (1.6) mmHg after the ear notching and 107.1 (1.5) mmHg after the microtattoo. The MAP was significantly higher after the ear tattoo than after the microtattoo (P = 0.012). During the 1–4 h period, the mean MAP was 102.2 (0.7) mmHg after the ear tattoo, 107.1 (0.8) mmHg after the ear notching and 104.1 (0.8) mmHg after the microtattoo. All of the differences between the different identification methods were statistically significant (P < 0.05). During the dark period (4–16 h), the mean MAP was 106.9 (0.4) mmHg after the ear tattoo, 107.6 (0.4) mmHg after the ear notching and 105.7 (0.4) mmHg after the microtattoo. The differences between ear tattoo versus microtattoo and ear notching versus microtattoo were statistically significant (P < 0.05). After the dark period (16–24 h), the mean MAP was 102.6 (0.5) mmHg after the ear tattoo, 102.9 (0.4) mmHg after the ear notching and 101.2 (0.5) mmHg after the microtattoo. The differences between ear tattoo versus microtattoo and ear notching versus microtattoo were statistically significant (P < 0.05) (Figure 4 and Table 1).
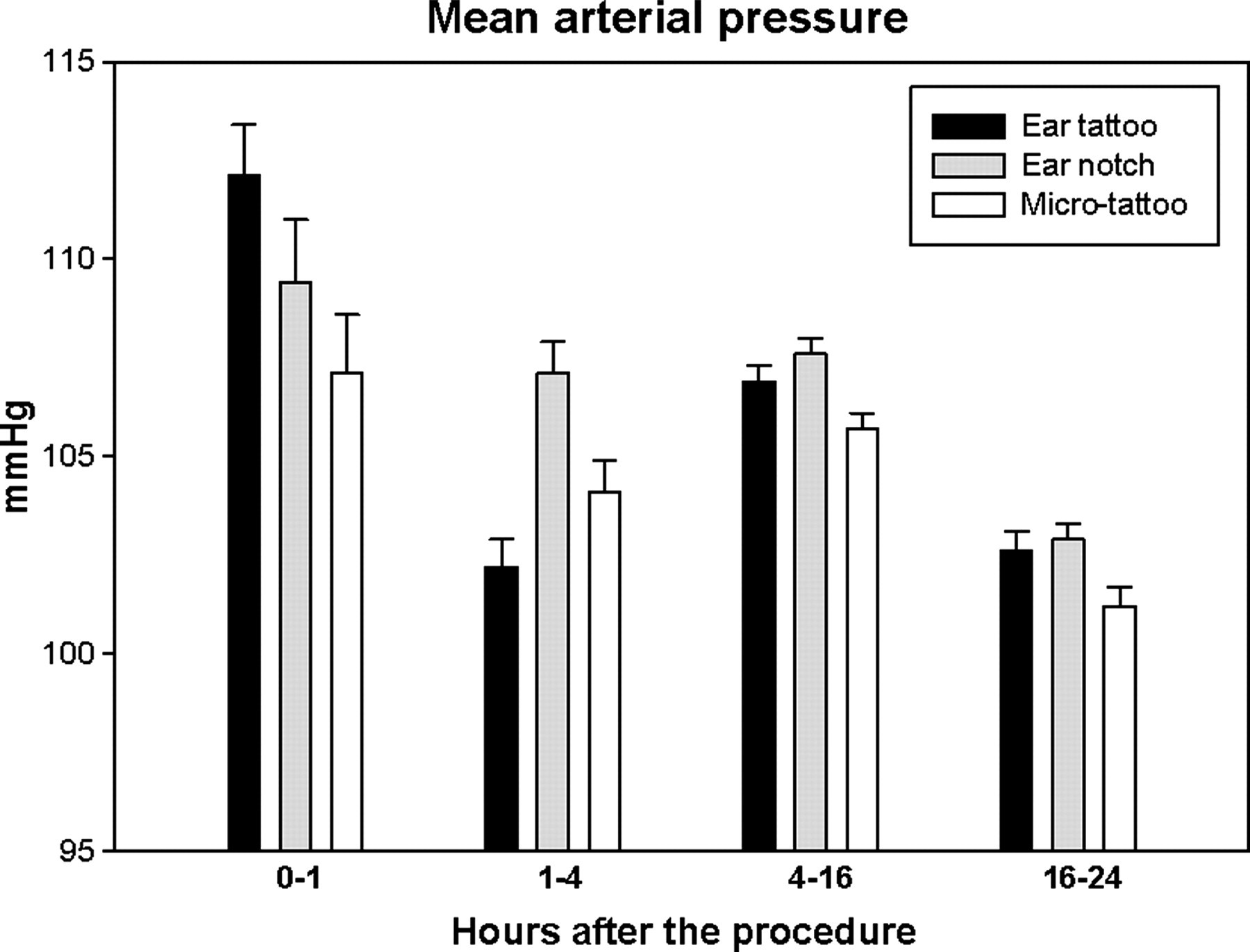
Mean arterial pressure during the first 24 h following ear tattoo, ear notching and microtattoo (n = 21). The dark period is situated in the time window 4–16 h. The error bars represent the standard errors of the means
Values of the heart rates (HR) and mean arterial pressures (MAP) at different times of the experiment (n = 21 in all of the measurements and statistical analysis)
The means are presented as model-based estimates of the means and their standard errors (SEM). The three treatments are compared with each other by presenting the P values. When the difference is statistically significant, the relative difference of the means (% diff., calculated by dividing the difference with the smaller value) and the biological significance (B.sig.) are also presented. The difference is interpreted as biologically significant (BS) if the difference is bigger than the overall mean difference of the same variables night and day values (4.5 mmHg in MAP and 50 bpm in HR). bpm: beats per minute; NS: not significant
During the first 60 min after the identification procedure, the model-based estimate of the mean HR was 378 beats per minute (bpm) (SEM 5.6) after the ear tattoo, 347 (5.8) bpm after the ear notching and 334 (4.7) bpm after the microtattoo. The differences between the ear tattoo and ear notching and between the ear tattoo and microtattoo were statistically significant (P < 0.001). No differences were found in the HR during the 1–4 h period and 4–16 h period after the identification procedures. In the 16–24 h period after the procedures, the model-based estimates for the HR were 322 (1.7) bpm for the ear tattoo, 309 (1.7) bpm for the ear notching and 315 (1.6) bpm for the microtattoo. The differences between all of the procedures were statistically significant (P < 0.05) (Figure 5 and Table 1).
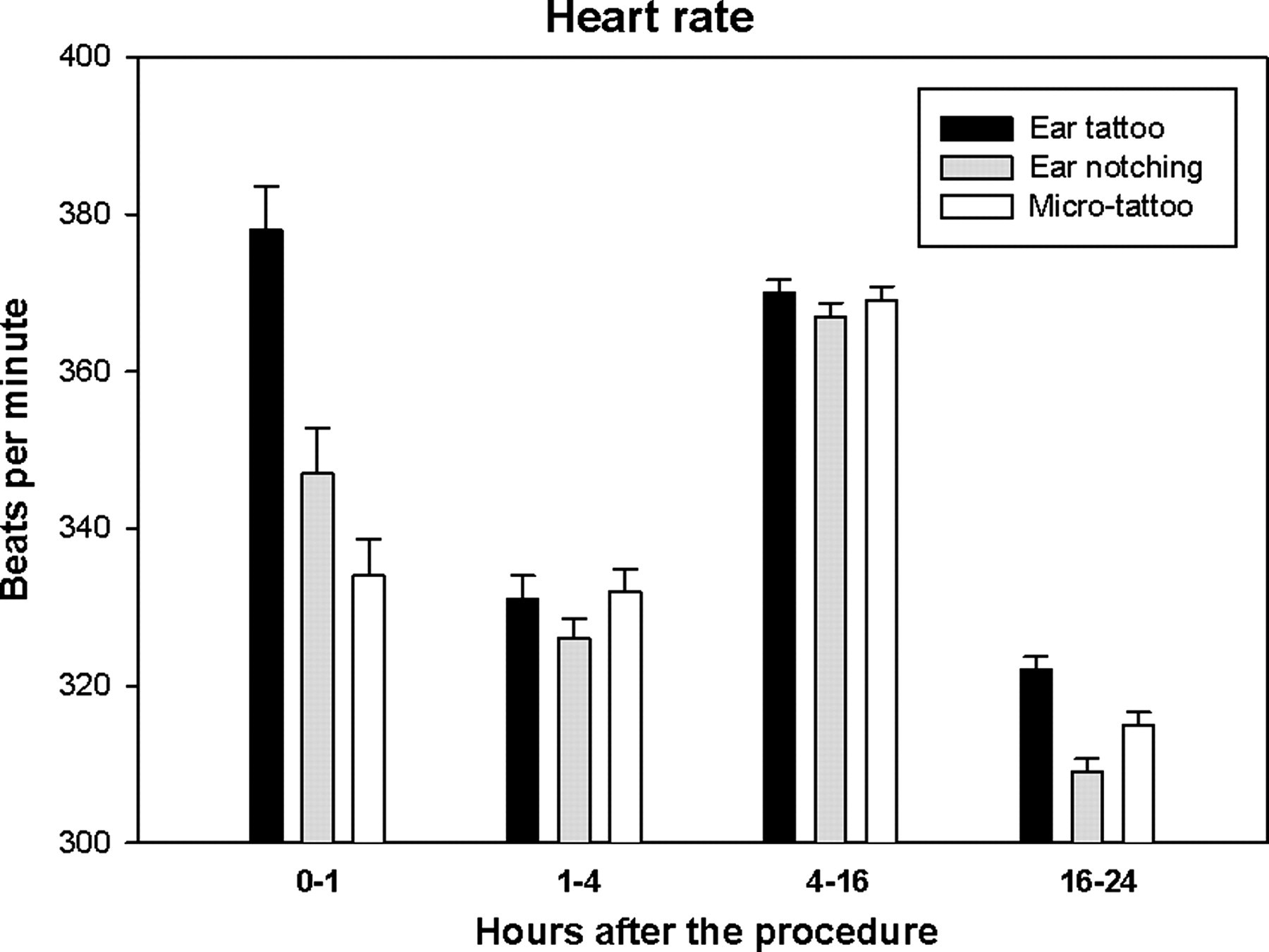
Heart rate during the first 24 h following ear tattoo, ear notching and microtattoo (n = 21). The dark period is situated in the time window 4–16 h. The error bars represent the standard errors of the means
Discussion
In this study the cardiovascular reactions to three different identification methods were compared. The experiment was done using two stocks and locations to increase the applicability of the results. The interaction between the stock and treatment was not statistically significant, nor did the results differ considerably or in any consistent fashion. Therefore the results are presented and discussed without separating them according to the stock.
The animals used in this study were considerably older than animals usually subjected to ID procedures. This was due to the fact that telemetric devices cannot be implanted into very young animals. The tissues of older animals could be thicker and might also heal slower. It could be hypothesized that younger animals could recover more quickly from ID procedures. On the other hand, these rats were used to handling, which might not be the case in young rats close to the age of weaning.
The HR and MAP were measured with telemetric monitoring for 24 h after each ID procedure. The values were compared between the different ID groups. The data were analysed in four different time windows: during the first hour after the procedure (immediate reaction), during the following 3 h until the dark period (recovery phase), during the 8 h of the dark period (dark period) and finally during the first 8 h of the next light period (next day), until 24 h after the procedure.
As assessed by both MAP and HR, ear tattoo evoked the greatest immediate reaction, while microtattoo resulted in the lowest MAP and HR. The reaction to ear notching fell between these two extremes. After the immediate reactions the data are more difficult to interpret. No differences were observed in HR during the recovery phase or the dark period. However, during the next day, the HR of the ear tattooed group was again significantly higher than that of the other groups. The MAP results revealed a different trend. After the immediate reaction, the MAP of the ear notching group remained highest throughout the remaining observation time (Table 1).
Telemetric monitoring can provide cardiovascular measures, such as HR and blood pressure, and also measures on body temperature and locomotor activity without the confounding effects of handling. 34–36 These parameters can be used to assess the stress responses of animals to different procedures and housing conditions. 37–39 It is recognized that the elevation of HR and blood pressure is a fundamental component of the stress reaction. 40–44 Pain has also been shown to cause a reaction in HR and blood pressure. 45 Both HR and blood pressure are often reported to rise in a similar manner after stressors. 39,46–49 However, there is evidence that HR is an even more acute measure of stress, declining more rapidly than blood pressure after the actual stressor has elapsed. 38,47 This could explain the discrepancy observed between HR and MAP in this study.
It should be noted that because telemetric monitoring yields an enormous amount of data, the statistical analysis often produces significant differences even when the absolute differences are very small. This problem of interpreting the true significance of results obtained with telemetry has been recognized by other authors. A 6% threshold is sometimes used as guideline for deciding whether an observed effect is truly significant. 38,50 Another parameter-specific threshold that the authors have used is the difference between the diurnal and nocturnal means. 51,52 If the treatment effect exceeds this value, it is said to have biological significance. If the 6% guideline is followed, only the differences during the immediate reaction in the HR between ear tattoo and the other methods are truly significant. Biological significance is observed only in MAP comparisons: during the immediate reaction, the difference between ear tattoo and microtattoo and also the difference between ear notching and ear tattoo during the recovery phase.
The effects of the ID methods cannot be completely distinguished from those of the handling involved in these techniques. In a previous study comparing handling techniques done on the same rats used in this study, mere handling provoked an elevation in HR and MAP which lasted about 30 min. The absolute values between these studies are not comparable due to differences in data collection techniques.
The effects of handling have generally waned within one hour, 30–32 but in this study there were still statistically significant differences occurring on the next day, with ear tattooed animals having the highest HR and ear notched animals the highest MAP. This could suggest that both ear notching and ear tattoo evoke pain which is still present after 16–24 h. It should be noted, however, that the differences observed during the next day did not exceed the 6% limit or the limit of biological significance. Even though the tissue reactions (wound healing and inflammation caused by the tattoo colour) last up to two weeks, 24 there is little evidence that tissue healing is associated with measurable pain or discomfort.
HR and MAP display prominent diurnal rhythms. Undisturbed rats have low MAP and HR during the light period, and higher values during their active phase, the dark period. 53,54 In this study, the MAP during the immediate reaction was higher than the mean dark period MAP in all ID groups. During the recovery phase, only the ear notching group had a MAP higher than the mean dark period MAP. HR was higher than the mean dark period HR only in the ear tattooed group during the immediate reaction phase. Throughout the next day, all groups had MAP and HR values substantially lower than the mean dark period values. These results indicate that the disturbance caused by the ID methods was mainly limited to the first hour after the procedures.
The results were also analysed from a reduction 55 point of view. The extent of interindividual variation is directly related to the number of animals needed in experiments. 56 In this study, no differences were observed in the variations of HR and MAP between any of the ID groups. Thus, none of the ID methods studied possesses a greater reduction potential than its counterparts.
In conclusion, microtattoo might be a preferable choice of ID over ear tattoo or ear notching. The animals subjected to microtattoo exhibited smaller reactions in HR and MAP, indicating less disturbance, stress or pain after the procedure. The benefits of microtattoo also include the reliability of the method; ear markings can be made unreadable, e.g. by fights with conspecifics. Moreover, if the markings fade or become unreadable, microtattoo can be renewed easily without the danger of obscuring the original numbering. Microtattoo also has its disadvantages; reading the tattoo usually requires handling of the animal. Tattooing does not offer the possibility of obtaining tissue biopsies as a side product of identification.
Footnotes
ACKNOWLEDGEMENTS
We thank research technicians Tuula Mäkinen (University of Oulu) and Heikki Pekonen (University of Eastern Finland) for carrying out the ID procedures.
