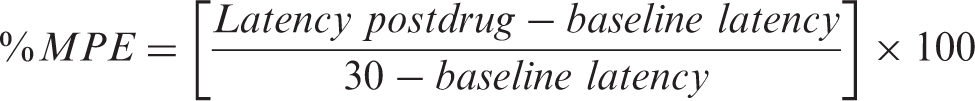Abstract
Postsurgical oral self-administration of analgesics in rodents is an interesting technique of providing analgesia, avoiding the negative effects of manipulation. Several strategies, using gelatin or nutella, have already been described. However, rodents require some habituation period to reach a good intake because of their neophobic behavior. The current study aimed to explore whether buprenorphine when mixed with an extruded diet offers a potential treatment option in the pain management of mice using a triple approach: by measuring the spontaneous intake in healthy animals; by using the hot-plate test; and finally by assessing the drug’s ability to provide postoperative analgesia in a surgical intervention of moderate severity (intra-utero electroporation). Mice consumed during 20 hours, similar amounts of extruded diet alone, mixed with glucosaline, and mixed with buprenorphine (0.03 mg per pellet) or meloxicam (0.25 mg per pellet) both of which were diluted in glucosaline, showing that no neophobia was associated with these administrations. Relative increase from baseline latency (% maximal possible effect) in the hot-plate test at 20 h of administration was significantly higher for oral buprenorphine in diet 0.03 mg/pellet, and diet 0.15 mg/pellet, compared with placebo and no differences were found between those oral administrations and subcutaneous buprenorphine 0.1 mg/kg measured 3 h later. The treatment was also effective in attenuating the reductions in food consumption and body weight that occur after surgery. These data suggest that providing buprenorphine with the diet is a feasible and effective way of self-administration of analgesia in mice and does not cause neophobia and may easily contribute to the refinement of surgical procedures.
The prevention and effective treatment of pain in laboratory animals must be warranted due to ethical, legal and scientific reasons. 1 Buprenorphine is a widely used analgesic in rodents.2–4 It is a partial agonist at the μ-opioid receptor, which has been shown to be effective in a variety of pain models and has a wide safety margin compared with full μ-opioid receptor agonists such as fentanyl. However, it must be administered subcutaneously at least 2–3 times a day to maintain effective therapeutic levels. 1 This requires human interaction and therefore added stress on animals undergoing different surgical procedures that might imply moderate to high levels of stress. 5 Surgical stress triggers the release of corticosteroids from the adrenal cortex. Persistent elevated corticosteroid levels are associated with impairment of the immune system, impaired reproduction and altered physiological and endocrinological functions. 6 Adequate analgesia reduces postoperative stress in rats. 7 Therefore, efforts to reduce postsurgical stress are desirable in improving animal welfare and easing recovery. In this sense, postsurgical per oral self-administration of buprenorphine seems to be a very attractive option because the animal can receive analgesia without the negative effects of handling during subcutaneous injections.
Several techniques have been described for a non-stressing oral administration of buprenorphine, for example mixing it with gelatin, 8 nutella®,9–11 or water. 12 Food neophobia is a difficulty in the use of oral administration of analgesics because rodents need to be habituated to new food items for a period of between one to five days in order to reach the necessary intake for therapeutic levels to be effective. 13 However, Jacobsen et al. reported that although buprenorphine mixed with nutella was eaten significantly slower the first time when mice found it, it did not affect the total serum concentration 17 h after the exposure. 14 This food neophobia has also been described in rats with water supplemented with cherry-flavored children's acetaminophen solution15 and with buprenorphine. 12
The current study aimed to explore whether buprenorphine when mixed with an extruded diet offers a potential treatment option in the pain management of mice using a triple approach: (1) by measuring the spontaneous intake in healthy animals; (2) by using a standard pain assessment tool (thermal analgesiometry); and (3) by assessing the drug’s ability to provide postoperative analgesia in mice that have undergone a surgical intervention of moderate severity.
We hypothesized that mice would spontaneously eat enough analgesic-mixed food to provide clinically relevant analgesia and thus improve welfare.
Animals
Three different experiments were conducted in mice: a food intake study with 32 females (Experiment 1), a thermal nociception response study with 30 females (Experiment 2), and a study of the efficacy of analgesia in a surgery performed routinely in our institution with 20 pregnant females (Experiment 3).
The operated mice used in Experiment 3 were included in two different studies with the same procedure of intra-utero electroporation. This study was approved by the ethics committee of our center, in accordance with current Spanish legislation.
Housing conditions
The animals were maintained at the barrier facility of Ratones Modificados Genéticamente–Servicio de Experimentación Animal (RMG–SEA) in the Instituto de Neurociencias de Alicante, Universidad Miguel Hernández–Consejo Superior de Investigaciones Científicas (IN, UMH–CSIC). The facility was free of the following agents: mouse rotavirus (EDIM), mouse hepatitis virus (MHV), mouse parvovirus (MPV), minute virus of mice (MVM), pneumonia virus of mice (PVM), Sendai virus, Theiler’s murine encephalomyelitis virus (TMEV), ectromelia virus, lymphocytic choriomeningitis virus (LCMV), mouse adenovirus 1 and 2, mouse cytomegalovirus (MCMV), Hantaan virus, Citrobacter rodentium, Corynebacterium kutscheri, Clostridium piliforme, Mycoplasma pulmonis/arthritidis, Salmonella spp., Pasteurella spp., Streptobacillus moniliformis, Streptococcus beta haemolyticus, Streptococcus pneumoniae, Helicobacter spp. and endo- and ectoparasites. Three out of 15 rooms were positive for murine norovirus (MNV).
Hsd:ICR (CD-1®) female mice from Harlan Laboratories (Horst, Netherlands) were used in this study. They were housed in II-L Makrolon cages (Tecniplast, Buguggiate, Italy). Poplar woodchips (Lignocel Select Fine®; Rettenmaier & Söhne GmbH, Rosenberg, Germany) were used as bedding and autoclaved paper was provided as environmental enrichment. Room temperature was maintained at 22 ± 2 ℃, relative humidity was 30–60%, and the light regimen was a 12:12 h dark:light cycle (lights on at 08:00 h). Autoclaved water was provided ad libitum as well as irradiated 2914 Teklad Global 14% Protein Rodent Maintenance Diet (Harlan Laboratories), except after surgery when medicated pellets were administered. Medicated pellets were made from 2919 Teklad Global 19% Protein Extruded Rodent Diet (Harlan Laboratories). 2914 Teklad Global is the standard maintenance diet in our institution. It is presented in a pellet form, which does not allow for easy mixing with liquids. However, 2919 Teklad Global is extruded, which has quite a porous texture and may be easily mixed with liquids for medicated pellets to be prepared.
Materials and methods
Medicated pellets
Buprenorphine (Buprex®; Schering Plough, Kenilworth, New Jersey, USA) was dissolved in glucosaline (BBraun®, Melsungen, Germany) at 0.03 mg/mL to prepare the buprenorphine pellets, and meloxicam (Loxicom®; Norbrook Laboratories, Newry, Northern Ireland) was dissolved at 0.25 mg/mL to prepare the meloxicam pellets. One milliliter of the solution was mixed with each pellet in a 50 mL tube, so each pellet contained 0.03 mg of buprenorphine or 0.25 mg of meloxicam. Pellets for the control group were prepared in the same way with just one milliliter of glucosaline. In the hot-plate experiment (Experiment 2) we also tested a higher dose of buprenorphine by mixing one milliliter of a 0.15 mg/mL buprenorphine solution resulting in a pellet with 0.15 mg of buprenorphine. No previous contact with medicated pellets or any drug had been produced by any of the animals in any of the experiments in this article. In all the experiments, only the medicated pellets were presented in the food tray.
Experiment 1: Food intake
In order to measure the acceptance of the diet by the animals, 32 healthy non-pregnant Hsd:ICR (CD-1®) female mice were individually housed and the food intake was measured 20 h later. They were divided into four groups of eight animals each: group 1 received the diet without any additions, group 2 received the diet with glucosaline, group 3 received the buprenorphine diluted in glucosaline and group 4 received the diet with meloxicam diluted in glucosaline. The daily food consumption was calculated by subtracting the measured weight of food at the end of the 20 h period from the initial weight.
Experiment 2: Hot-plate test
The hot-plate test was used to assess the antinociceptive effects of oral buprenorphine, as described previously, 16 with minor modifications. In brief, Hsd:ICR (CD-1®) female mice were individually placed on the surface of a hot-plate (IITC Inc Life Science, Woodland Hills, CA, USA) maintained at 55 ℃ and the latency was recorded from starting to the endpoint of jumping, licking or shaking hind paws. A cut-off time of 30 s was imposed to prevent the possibility of tissue damage.
For subsequent analysis, latencies in the hot-plate test were converted to percentages of maximal possible effect (% MPE), a way of expressing the absolute latencies relative to baseline (0% MPE) and to cut-off time (100% MPE), according to the formula, as previously described:17,18
Mice were divided into five experimental groups each containing six mice. The experimental groups were: group 1: diet with glucosaline; group 2: subcutaneous buprenorphine 0.1 mg/kg; group 3: oral buprenorphine in diet 0.03 mg/pellet; group 4: oral buprenorphine in diet 0.15 mg/pellet; and group 5: subcutaneous buprenorphine 0.1 mg/kg plus oral buprenorphine in diet 0.03 mg/pellet. Baseline thermal latency data and body weights were measured on day 1, before different treatments were provided. The treatment thermal values were evaluated 20 h later in all groups, except in group 2, which was evaluated 3 h later. Group 5 received buprenorphine 0.1 mg/kg subcutaneously after the baseline measure and was also provided with the diet containing buprenorphine 0.03 mg/pellet, which was the treatment received by the mice in the surgery study.
Food was weighed to calculate the intake and the dose of buprenorphine provided.
Experiment 3: Surgical procedure
Pregnant female mice, 11–12 days after the observation of their vaginal plug, (E11–E12) underwent intra-utero electroporation as previously described 19 under aseptic conditions. Mice were anesthetized with isoflurane (5% in induction and 1–2% for maintenance). They all received ritodrine (0.1 mL of a 14 mg/mL solution) and buprenorphine (Buprex®, Schering Plough) 0.1 mg/kg subcutaneously before surgery. After shaving and disinfection of the surgical site, a 1.5–2 cm medium laparotomy was performed. The uterine horns were exposed and continuously irrigated with sterile tempered saline. The DNA solution (1.5 µg/µL test DNA + 0.9 µg/µL GFP or mCherry expressing vector) was injected into the brain of each embryo using a graduated pulled-glass micropipette (World Precision Instruments, Sarasota, Florida, USA). The head of each embryo was placed between tweezer-type electrodes (CUY650-P5; Nepa Gene, Chiba, Japan) and five square electric pulses (38 V, 50 ms) were passed at 950 ms intervals using an electroporator (CUY21SC, Nepa Gene). The wall and skin of the abdominal cavity were sutured and closed with silk.
After recovery from surgery, the mice were individually housed and they received pellets with buprenorphine (treatment group) or glucosaline (control group).
Data from the animals were obtained the following day after surgery between 10:00 and 12:00 h by an investigator who was blinded to the treatment. The daily food consumption was calculated by subtracting the measured weight of food from the amount measured the previous day. Mice were weighed just after the surgery and the following day to calculate their weight loss. After the data recording, rescue subcutaneous buprenorphine 0.1 mg/kg was provided if pain signs were observed.
Twenty pregnant mice were included in this study, ten of them belonged to one of the research groups participating in the study and the other ten belonged to the other one. Twelve of them were included in the treatment group and eight in the control group.
Statistical analysis
Absolute and relative frequencies were used to describe qualitative variables. We determined if quantitative variables followed a normal distribution by means of the Kolmogorov-Smirnov test. The mean ± standard deviation (SD) or median (percentile 25-percentile 75) were used for the description of quantitative variables. Chi-square test was used to compare qualitative variables. Student’s t-test or analysis of variance (ANOVA) was used to compare qualitative with quantitative variables (two categories or more than two respectively).
Results
Experiment 1: Evaluation of the acceptance of the diet in healthy animals
Twenty hours after the administration of medicated pellets, healthy, non-operated mice had eaten the following amounts of food (mean ± SD): 4.37 ± 0.97 g in the diet without additions, 4.24 ± 0.57 g in the diet with glucosaline, 4.40 ± 1.28 g in the diet with buprenorphine diluted in glucosaline (0.03 mg/pellet), and 3.11 ± 1.71 g in the diet with meloxicam diluted in glucosaline. Thus, no neophobia was observed in any of the groups.
Experiment 2: Evaluation of the antinociceptive test
Food intake and dose of oral buprenorphine received according to the analgesic group in the hot-plate test (Experiment 2).
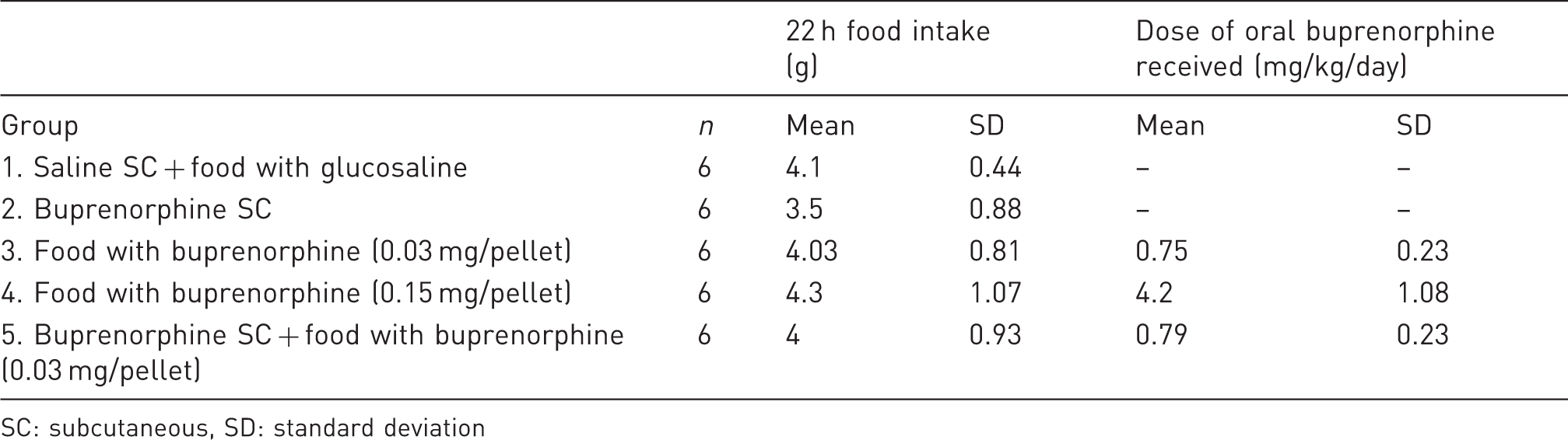
SC: subcutaneous, SD: standard deviation
Paw withdrawal latency times were measured before (baseline, time 0) and after the different treatments and percentages of MPE were calculated. The results are represented in Figure 1. The difference in relative paw withdrawal response between the placebo group and every of the other treatments was statistically significant (Mann-Whitney’s U-test, P < 0.05 for each one). No significant differences were found between the group of buprenorphine at 4 h and the rest of the groups where oral buprenorphine was provided (Mann-Whitney’s U-test, P = 0.8, P = 0.3 and P = 0.9 for group 2 compared with groups 3, 4 and 5, respectively).
Antinociceptive effect of buprenorphine following the administration with food using the hot-plate test (Experiment 2). Percentage of maximal possible effect (% MPE) in the hot-plate test for each treatment group. The overall difference in relative paw withdrawal response between the placebo group and every of the other treatments was statistically significant (Mann-Whitney’s U-test, P < 0.05 for each one). No significantly differences (Mann-Whitney’s U-test, P = 0.8, P = 0.3 and P = 0.9 for group 2 compared with groups 3, 4 and 5 respectively) were found between the group of buprenorphine at 4 h and the rest of the groups where oral buprenorphine was provided. Bup: buprenorphine, SC: subcutaneous, O: Open circle represents an extreme value.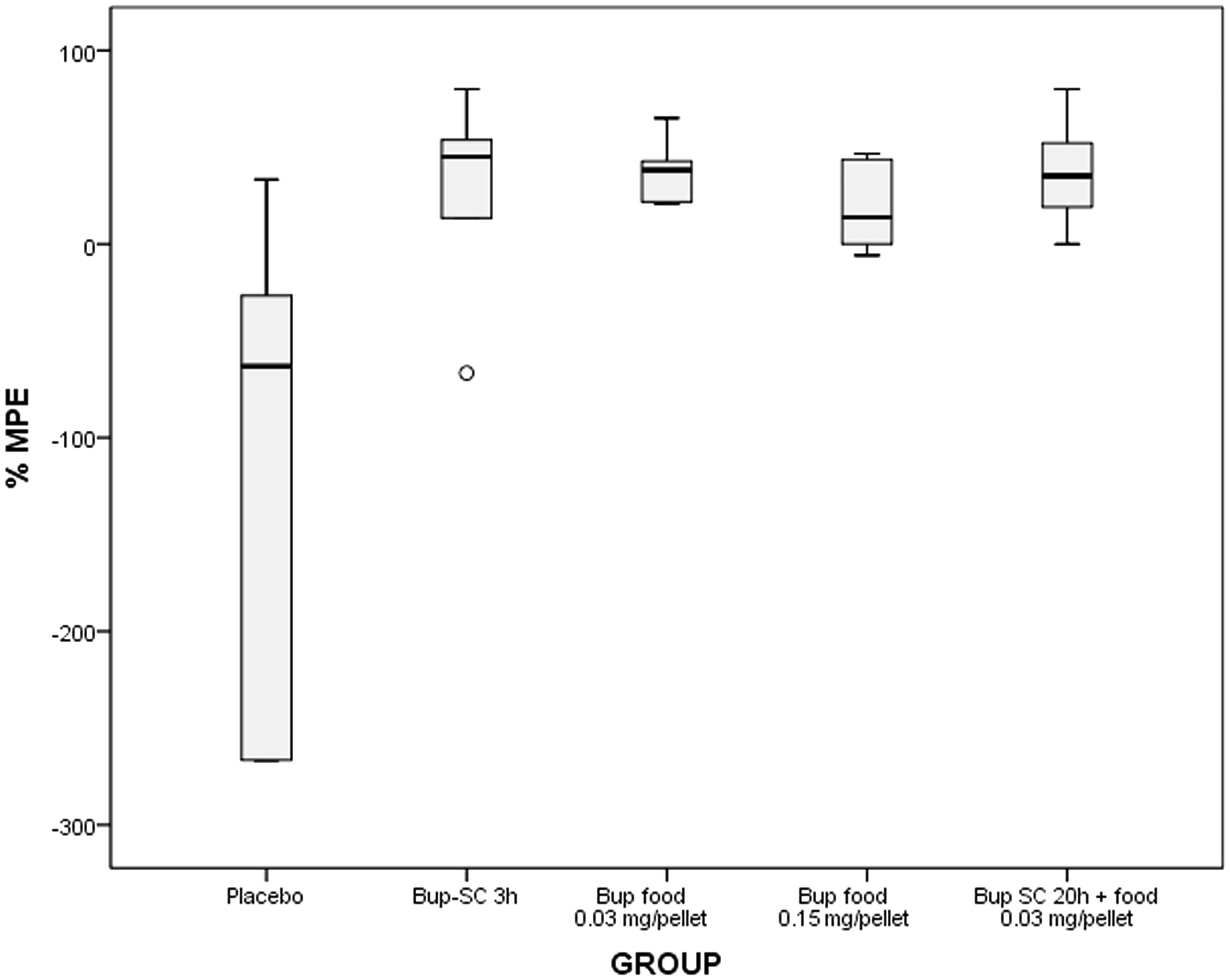
Experiment 3: Evaluation of the efficacy of the treatment in operated animals
All the mice were pregnant at E11–E12 and mean weight was 45.1 ± 6.25 g.
In order to measure the ingestion of buprenorphine during the first 24 h, we measured the weight of the pellets consumed during that period. Mice from the treatment group consumed significantly more food than mice from the control group (mean ± SD): 1.75 ± 1.55 versus 0.45 ± 0.33 g respectively (Student’s t-test, P < 0.05, Figure 2). The mean buprenorphine dose during the first 24 h after surgery in the treatment group was 0.3 ± 0.25 mg/kg.
Food intake in the control and treatment groups in operated animals (Experiment 3). Mice from the treatment group, which received one pellet with 0.03 mg of buprenorphine dissolved in glucosaline, consumed significantly more food than mice from the control group, which received one pellet with glucosaline (mean ± SD): 1.75 ± 1.55 versus 0.45 ± 0.33 g respectively (Student’s t-test, P < 0.05). Both groups received 0.1 mg/kg of buprenorphine subcutaneously post surgery. Rescue subcutaneous buprenorphine was provided 24 h after the surgery if needed. *represents extreme values in mice number 16 and 15.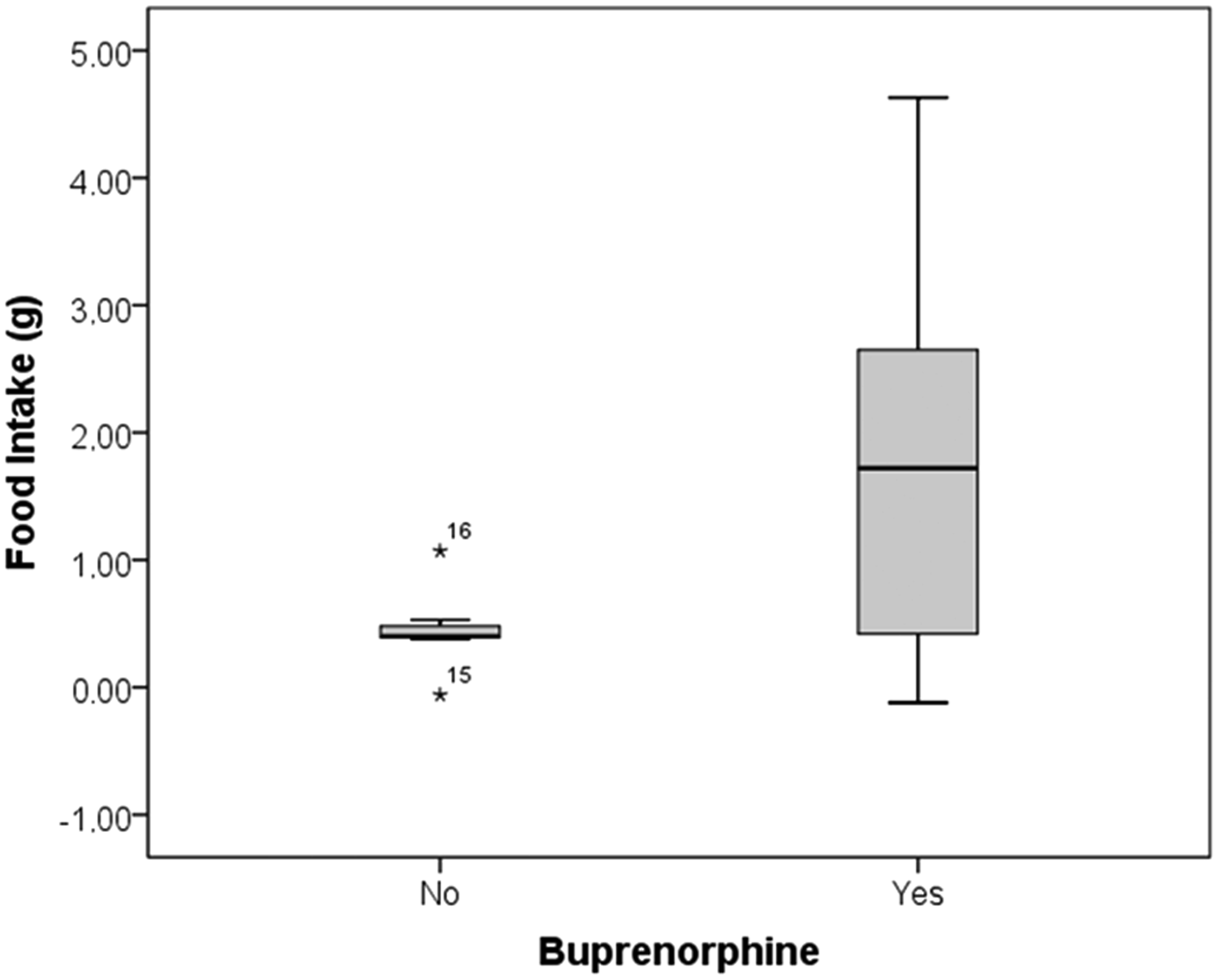
It has been described as consistent weight loss in all operated animals after surgery.
20
To determine the effect of buprenorphine treatment on this weight loss, we measured their values at surgery time and 24 h later. Weight loss in the animals of the treatment group was lower than that of the mice from the control group (mean ± SD): 6.19 ± 5.8% versus 12 ± 6.29% respectively, Student’s t-test, P < 0.05 (Figure 3).
Weight loss after surgery in the control and treatment groups (Experiment 3). Weight loss in the mice of the treatment group, which received one pellet with 0.03 mg of buprenorphine dissolved in glucosaline, was lower than that of the mice from the control group, which received one pellet with glucosaline (mean ± SD): 6.19 ± 5.8% versus 12 ± 6.29% respectively (Student’s t-test, P < 0.05). O: Open circle represents an extreme value in mouse number 14.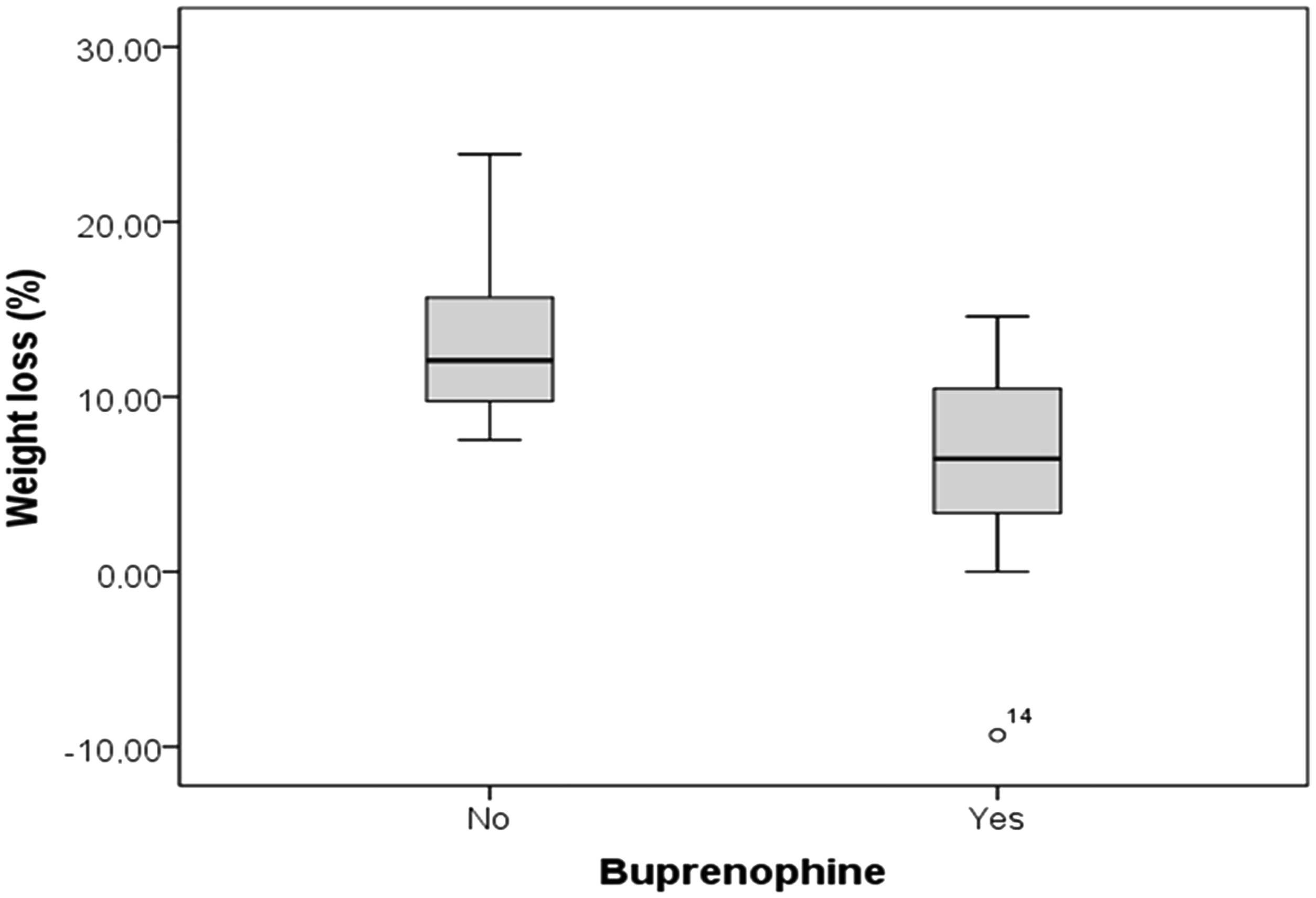
Discussion
Pain is difficult to measure in laboratory rodents. They are prey species and they do not normally show it as a survival strategy in the wild. This greatly complicates the postsurgical veterinary management and probably contributes to the underuse of postoperative analgesics. 21 Many attempts to assess pain in rodents have been done. 4 Analgesiometric tests, such as tail flick and hot-plate tests, are used to determine analgesic potency by measuring antinociceptive effects, but their use alone to make dosage recommendations is controversial, 4 because the neurological mechanisms involved may not be equivalent to those responsible for clinical pain. Other authors, 21 however, have criticized the indirect measures of analgesia (e.g. feeding or locomotion) because they consider that these are not validated enough and can be confounded by independent effects of opioids. For this reason we have used two approaches to assess analgesia in this study. We have used the hot-plate test, which is a classic nociceptive test, 22 and we have also evaluated it in a routine surgery of moderate severity, measuring some parameters, such as food intake and weight, suggested in the bibliography as indicators of surgical pain. We have found evidence of analgesic efficacy with this method of oral analgesia in both, analgesiometric test and clinical indicators in routine surgery.
The efficacy of oral administration of buprenorphine has been discussed since buprenorphine underwent first-pass liver metabolism.4,23 In 1981, Brewster et al. 24 suggested that an oral dose of buprenorphine 10 times higher than the effective parentally administered dose would be necessary to be effective. They based their work in pharmacokinetic studies, but they did not address analgesic efficacy directly. Thompson et al. concluded that the recommended dose of 0.5 mg/kg dose 25 was not effective as an analgesic in oral administration, and the 5 mg/kg was effective but rejected by the animals when administered in jello.21,23 They based their conclusions in the hot water tail-flick assay in rats. In our experiment, we found no differences in paw withdrawal latency times when comparing the hot-plate test at 3 h of subcutaneous injection, which according to the bibliography, it is supposed to have an analgesic effect, 26 with all the other regimens of oral administration 20 h later, including the 0.5 mg/kg questioned by Thompson et al. We have not found a decrease in the voluntary intake of medicated pellets compared with non-medicated, even with higher doses of buprenorphine (4 mg/kg), indicating that this method could be used to administer higher analgesic doses without rejection by the animals.
Jessen et al. studied the antinociceptive efficacy of buprenorphine administered through drinking water in rats compared with repeated subcutaneous injections at 8 h intervals. 12 Some degree of refusal to drink the medicated water was also found in this study. They found that buprenorphine in drinking water induced significant increases in paw withdrawal latency times in the hot-plate test during a three-day period of administration, with a maximal effect at 39 h after the start of buprenorphine administration, but the subcutaneous injection regimen did not increase the paw withdrawal latency times. One single injection followed by buprenorphine in the drinking water induced an earlier onset of antinociception and, thus, they recommended one single subcutaneous injection followed by buprenorphine in drinking water. Our study’s results also suggest that combining a postsurgical subcutaneous injection with oral buprenorphine with the diet would be a viable treatment option for the relief of pain in laboratory mice. In our surgery experiment, some of the animals did not eat any food. Rescue subcutaneous injection could be applied the following day if animals did not eat enough medicated food or if pain was observed.
The preference for oral administration of buprenorphine is also supported in the study by Kalliokoski et al. 27 They found that voluntary ingestion resulted in a higher serum concentration and a longer duration than via the widely recommended subcutaneous route. This is a clear advantage of the oral over the parenteral route. In Kalliokoski’s experiment, buprenorphine was administered with nutella, and animals were previously habituated for 18–24 h. The animals consumed the entire mixture within 15 min in each case. In our experiment, consumption was sustained during periods of up to 20 h. This presumably may contribute to reaching serum buprenorphine levels that are maintained over time. However, further studies are needed to assess this. As our study was made using female mice, further studies in rats and male mice would be needed to assess the palatability and efficiency of this analgesic presentation in these animals.
Sustained-release formulation of buprenorphine is another option of analgesia in rodents and was evaluated by Foley et al. in rats 1 and by Carbone et al. in mice. 18 They concluded that this formulation provides analgesia at least 72 h after the injection, which could be enough for most of the moderate surgical procedures. However this formulation was only available in Canada and the United States at the time of writing the manuscript. Both studies also describe some skin irritation including erythema and scabbing of the skin around the injection site developed 24 h after the injection in mice and also in rats. In our work, no side-effects were observed.
Flecknell and Liles have shown that postoperative reductions in food and water consumption, body weight and generalized locomotor activity may be used to assess pain in rats.4,20 Recent studies support these data and have shown that non-invasive measures such as food and water consumption and some behaviors are useful and reliable for measuring postsurgical pain in mice. 28 Our results clearly show an increase in food intake combined with a lower weight loss. Both indicators provide robust evidence that the oral administration of buprenorphine after a surgical procedure produces clear clinical improvement. The dose that was eventually consumed by the animals in our study and produced clinical improvement was 0.3 ± 0.25 mg/kg. This is consistent with the dose of 0.4 mg/kg that was found to be effective in other studies based on clinical evolution.9,25 Taken together the clinical and the analgesiometric tests, our results suggest that the dose of 0.5 mg/kg, questioned by Thompson et al. in rats, is useful to provide analgesia in mice by mixing the buprenorphine with their food.
One of the major complications of oral self-administration using jello and nutella is that mice tend to refuse them when they find them for the first time because of their neophobic behavior. Therefore, reaching the necessary intake requires habituation for some days. This is a major cause of failure with these methods. 28 Our experiments show that no neophobia is associated with this method of administration, probably because pellets are included in a normal diet. In our institution, this practice is commonly performed and very well accepted by researchers because it is easy to use. The animal house provides the dilutions of buprenorphine free of charge and the researchers mix them with the pellet. The day after the surgery, if the animal has not eaten any of the food or if signs of pain are observed, we provide a subcutaneous buprenorphine injection as an analgesic rescue therapy. This method contributes to the improvement of animal welfare in our institution.
Although the main objective of this study was to assess buprenorphine efficacy when administered with food, in Experiment 1 we also tested the acceptance of meloxicam. Although the intake was slightly lower than with buprenorphine, this difference was not significant, meaning that this method of administration could be applied to other analgesic drugs. However, further studies are needed to verify this.
Oral administration in pellet form also has other practical advantages. There is no problem introducing pellets in barrier facilities as they can be mixed with irradiated food. They can also be prepared considerably faster than gelatin. Martin et al. suggested that a change in pH following mixing buprenorphine with jello could have been associated with failures in the analgesic effect. 23 Moreover, buprenorphine should not be exposed to temperatures exceeding 40–60 ℃. 4 Pellet mixtures do not have any problems with pH or with temperature. Pellets also do not introduce differences in the diet and they are cheap.
Conclusion
Our data suggest that mixing buprenorphine with a standard pellet diet may be a useful method of non-stressful self-administration of analgesia in mice. Administration of buprenorphine with the pellet is easy to implement and does not require any habituation period to reach a good intake, making planning of experiments much easier. This approach has been demonstrated to be successful in increasing thermal latency in the hot-plate test, and in attenuating the reductions in food consumption and body weight that occur after surgery. The feasibility of this method may contribute to the improvement of animal welfare and thus the refinement of surgical procedures.
Footnotes
Acknowledgements
The authors would like to thank their colleagues in the Animalario de Ratones Modificados Genéticamente–Servicio de Experimentación Animal (UMH–CSIC) for their work and support; especially to Antonio Caler, Carmen Checa, Sandra González, Verónica Jiménez, Ana Lorena Marín, Gonzalo Moreno, Patricia Muñoz, Rebeca Ortiz, Raúl Pardo, Ángeles Soler, and Lucia Yuste. We would also like to thank Dr Juan Galcerán and Dr Jorge Brotons for their review of the manuscript, and to Clara Muñoz and Asunción Romero for their suggestions and advice.
Declaration of conflicting interest
Buprenorphine industrial diet production has been protected by a patent by the Consejo Superior de Investigaciones Científicas.
Funding
This research received no specific grant from any funding agency in the public, commercial, or non-profit sectors.