Abstract
Drug administration to experimental rodents is often invasive and stressful, thus reducing animal welfare and potentially confounding experimental results. Methods of oral drug delivery in which rodents cooperate voluntarily minimize stress, pain and morbidity. We herein describe a method for oral administration through voluntary intake of strawberry jam, developed for C57BL/6J mice. During a 3-day habituation period, animals were placed in individual cages once daily and presented with a drop of jam. Five days later, the jam was again offered with admixed drug. Mice ingested it in less than 5 min, with latency times below 1 min, confirming the suitability of the administration method.
Multiple routes are available for drug administration in mice. Many of them can be painful, or at least stressful, and may even require sedation. Injections, for example, induce stress responses and transient pain, and can result in tissue damage or inflammation.1,2 Physical restraint is itself a stressful procedure for mice 1 and affects numerous behavioural and physiological processes, introducing intra- and inter-individual variability.
The oral route is one of the most relevant and desirable to explore, due to translatability issues. In animal research this administration is typically achieved by orogastric gavage, which allows the delivery of precise doses. However, in addition to being technically challenging (particularly in small animals such as mice), gavage triggers a significant stress reaction in rodents3–5 and can occasionally cause oesophageal or gastric injuries, reflux, aspiration pneumonia and death.6,7Gavage-associated effects should be taken into consideration in experimental design, especially in extended and repeated dosing, as they may influence the phenotype of disease models 8 or cause immunologic sequelae 9 that may confound experimental outcomes.
Several studies demonstrate that oral administration by voluntary ingestion is associated with lower stress responses in mice than subcutaneous or intraperitoneal treatments, or gavage.4,5,10 Nonetheless, some stress-free options, like dissolving the drug in drinking water or mixing it with laboratory chow, also present disadvantages, such as lack of precise control over the amount and timing of drug ingestion. Therefore, the development of alternative non-invasive oral methods of drug delivery should be a priority, both for animal welfare reasons and for accomplishing more robust experiments.
Methods successfully applied in mice have been increasing. These include: chocolate and hazelnut spread,11–13 peanut butter, 5 flavoured gelatin,14,15 oat flakes 10 or bacon-flavoured dough. 4 A commercially available aqueous gel has also been investigated, 16 but the willingness of mice to ingest it was low.
We herein present the protocol for an alternative method of drug administration by voluntary oral intake in mice, using strawberry jam. This protocol was developed in the context of a project on the role of the specific pro-resolving mediator Maresin-1 (MaR1) as a therapeutic target in neuropathic pain (NP), approved by the local Animal Welfare Committee (ORBEA-FMUP_57_2017/1812). Twenty-two male and 22 female C57BL/6J mice (11-week-old specific pathogen-free, purchased from Charles River, France) were housed in groups of four, in a normal light:dark cycle. Food and water were provided ad libitum. After being randomly allocated within sex to two groups – MaR1 (n = 24) and Vehicle (n = 20) – all animals were subjected to NP-inducing surgery. Sample size calculation was based on the primary experimental outcome assessed on the project (changes in von Frey thresholds) and will be presented elsewhere. The experimental unit is the individual mouse.
MaR1 (Cayman Chemical, USA, https://www.caymanchem.com/pdfs/10878.pdf) is an ω-3 fatty acid derivative, with low aqueous solubility (ca. 0.05 mg/ml in PBS, pH 7.2), supplied as an ethanol solution. The jam (Doce extra Morango Froiz, Dulces y Conservas Helios, Spain) is composed of strawberries, sugar, glucose-fructose syrup, pectin and citric acid. Consideration of eventual palatability issues, as well as individual preferences for flavours, is necessary in any protocol for drug delivery via food. 17 In a small pilot study (eight animals), we first trialled whether mice would voluntarily eat the selected jam and whether the presence of MaR1 and its vehicle would affect that ingestion. The volume of jam was also selected to be completely ingested by each mouse in a short period of time.
Because of their neophobic nature, mice must be habituated to ingest the novel food before beginning the treatment. Therefore, mice were trained to voluntarily ingest the jam for three consecutive days. In each training session, each mouse was placed for 10 min into an empty cage lined with a surgical underpad and presented with a 60 µl drop of jam in a small Petri dish. This period could be extended up to 20 min, if the jam had not been ingested in the initial 10-min period. Latency time to ingestion significantly decreased between the first and the second training sessions and remained constant thereafter (Figure 1). By the end of the habituation period, all mice completely ingested the jam within less than 5 min, with latency times below 1 min in 95% of cases.
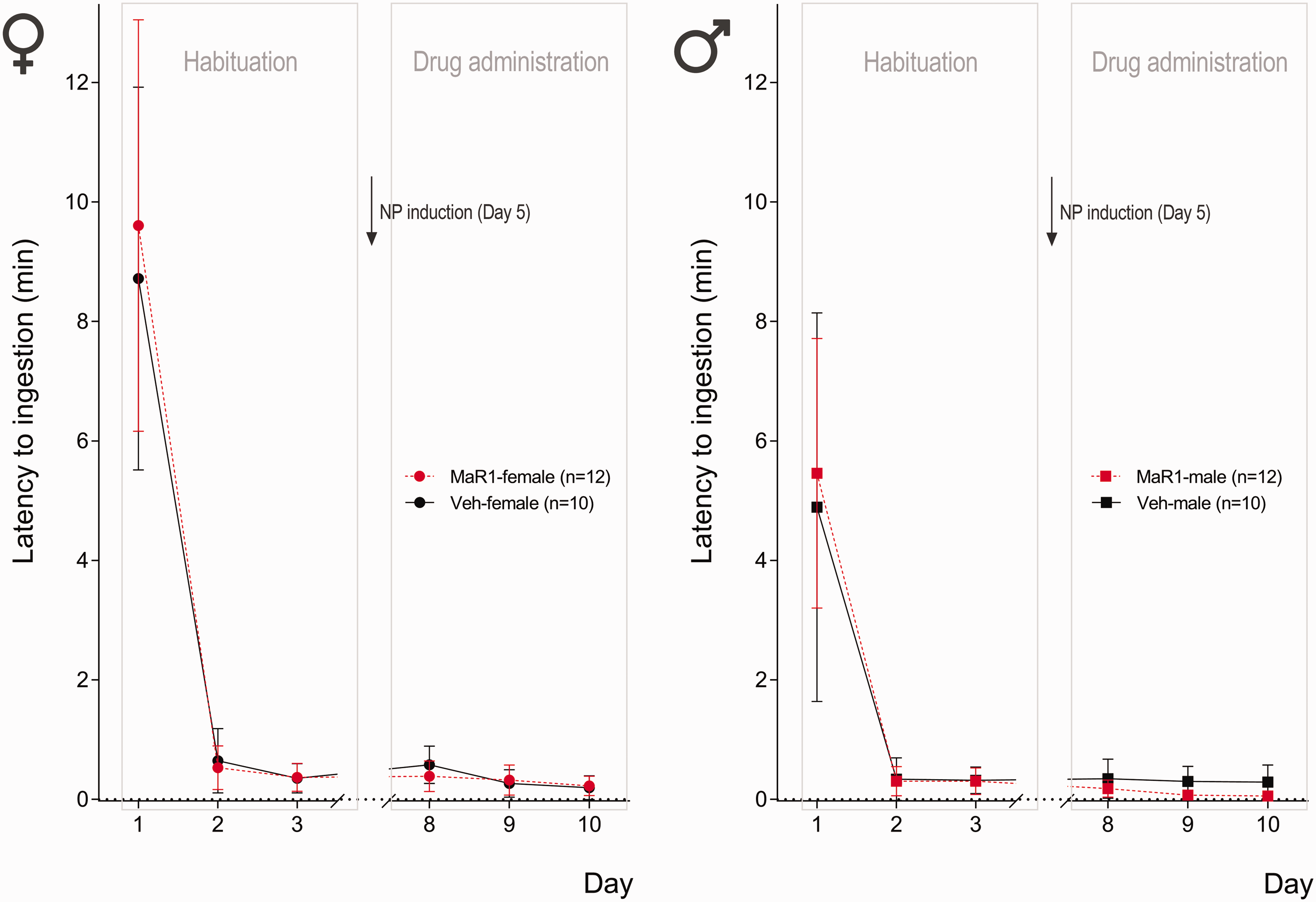
Latency to ingest the jam and the jam plus admixed drug (MaR1, n = 24) or vehicle (Veh, n = 20), during habituation and drug administration periods, respectively. Results are presented as 95% confidence intervals centred on the means.
Five days after the last habituation session (3 days after NP-induction), 50 µg/kg MaR1 or vehicle were administered once daily for 3 days. On each treatment day, a 12–15 µl ethanolic drug aliquot was thawed per animal and further prepared by evaporating the ethanol with a gentle stream of nitrogen down to a final volume of approximately 5 µl, to which 6 µl of 0.1 M PBS pH 7.4 was added. After placing 60 µl of jam in a Petri dish, the freshly prepared MaR1 solution was added and mixed, and each mouse was placed in the respective training cage and offered the mixture for immediate ingestion. The control mice were offered 60 µl jam mixed with 6 µl PBS plus 5 µl ethanol. Animals were removed from the cages shortly after they had ingested the mixture.
After three training days, mice maintained their interest for the jam when re-introduced later. The addition of drug/vehicle did not interfere with latency times (Figure 1). Mice behaviour was observed continuously to confirm the full ingestion of the mixture and Petri dishes were visually inspected at the end. In general, males immediately ingested the entire volume in a single attempt whereas females preferred a slower ingestion, although all animals completely ingested the jam mixture in a voluntary manner and within a 5-min interval.
Contrary to other methods, ours does not require food/water deprivation or single-housing during the whole protocol (either habituation or treatment period).10,12,14,15 Instead, each mouse was kept individually in a cage, without access to water and their usual food pellets, solely during the short periods (up to 20 min) required for daily jam/drug administration. Furthermore, this procedure is not very time-consuming, as a single experimenter can perform it with multiple mice simultaneously. Moreover, other options applied to mice describe longer familiarization periods with the novel food,5,14,15 longer preparation time (they require, for example, drying the formulation),4,10 need specialized equipment,4,5 or are not appropriate for some compounds (because of solubility and thermal stability).5,14,15
The caloric contribution of the 60 µl volume of jam amounts to 0.17 kcal, which corresponds to between 1 and 1.7% of the average daily caloric intake for a 25 g mouse. We therefore do not expect it to cause significant diet changes, but the suitability of this method to long-term dosing nonetheless will depend on the experimental model studied and scientific hypothesis under investigation. Ingestion of the full dose could be compromised in models that induce appetite suppression or clinically significant gastrointestinal complications. 17
Methods for drug delivery through voluntary intake may be affected by potential sex and strain differences in taste preferences. Indeed, the preference for sweet substances is known to differ among strains of laboratory mice,18,19 and genetic factors also seem to be significantly involved. 20 In studies that compare several mouse strains, the strain we used (C57BL/6) has consistently been one of the strains that display the strongest preference and acceptance for sweeteners and ingest the greatest amounts thereof.21,22 However, mice of different strains have been shown to significantly increase their avidity for sucrose when retested, whereby experience attenuates and/or even eliminates strain differences in sucrose preference.23,24 Differences between strains are also less evident at high sweetener concentrations.20,21 Therefore, considering the high sugar content of the jam and the habituation period that is included in our protocol, we expect our method to be effective for drug delivery in other mouse strains as well.
This method was developed for treating mice with MaR1, but it may be applied to other experimental compounds, provided that any potential aversive taste is effectively masked by the palatable flavour of the jam. Also, compounds must be free from immediate adverse effects or else animals will learn to refuse the presented food. 25
In conclusion, this non-invasive method of oral administration through voluntary intake of strawberry jam affords an effective, cheap and less-stressing alternative to injections or gavage, reducing potential injuries and allowing control of dose and ingestion timing. We expect it to be of interest especially for the controlled oral administration of compounds otherwise difficult to solubilize or disperse in biologically compatible media.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Universidade do Porto/FMUP and European Social Fund [NORTE-08-5369-FSE-000011-Doctoral Programmes] and by Fundação Grünenthal [Bolsa para Jovens Investigadores em Dor 2018]
