Abstract
Preliminary studies have suggested a correlation between postsurgical pain and nest building behaviour in laboratory mice. However, there is no standardized measure for estimating pain by means of nest building performance. Here, we investigated nest building under various conditions, and scored nest complexity to assess postsurgical pain. Mice of both sexes, different strains [C57BL/6J, DBA/2J, and B6D2-Tg(Pr-mSMalphaActin)V5rCLR-25], and kept under different housing conditions, showed no differences in their latency to use the offered nest material. Healthy female C57BL/6J mice were engaged 4.3% of the day with nest building and showed three peaks of this behaviour: in the beginning and middle of the light phase, and in the second half of the dark phase. For assessment of postsurgical pain, female C57BL/6J mice underwent a sham embryo transfer +/− different doses of the analgesic carprofen or control treatment. Nest complexity scoring at 9 h after the experimental treatments (i.e. at the end of the light phase) resulted in less than 10% of animals with noticeably manipulated nest material (nestlet) after surgery and more than 75% of healthy mice having built identifiable-to-complex nests or had noticeably manipulated nestlets, while animals after anaesthesia-only showed intermediate nest complexity. Carprofen analgesia resulted in no (5 mg/kg) or only slight (50 mg/kg) improvement of nest complexity after surgery. Thus, nest complexity scoring can be incorporated into daily laboratory routine and can be used in mice as a sensitive tool for detecting reduced wellbeing and general condition, but probably not for determining the efficacy of pain treatment.
The construction of nests is common in rodent species. Wild house mice build nests to provide heat conservation; shelter from elements, predators, and competitors; and to allow successful reproduction. 1 –3
The motivation and ability to perform this complex behavioural sequence culminating in a finished nest persist also in domesticated mice and those in laboratory animal facilities. Aside from ‘brood’ or maternal nests, built specifically for reproduction, if provided with suitable nest building materials, laboratory mice of both sexes build ‘sleeping’ or non-maternal nests. 4,5 In the laboratory setting, nests might allow the mouse to shield itself from conspecifics, as well as humans and external stimuli, e.g. direct light. 6 Also, as most animal facilities have ambient temperatures below their thermoneutral temperature, mice might build nests for thermoregulatory reasons. 7,8 The motivation for nest building is high, and nest building material is highly valued by laboratory mice. 9,10
Spontaneous, species-specific behaviours, such as nest building, that are performed in the animals' home cage have been proposed in the last decade as useful indicators for welfare assessment in small laboratory animal species, and might be used as simple, non-invasive and reproducible indicators for estimating, e.g. neurological dysfunction or pain. A reduction in these behaviours may signal a change in the motivational state of the animal and good performance seems to be indicative of good wellbeing in animals. 11 –15
Nest building performance has been shown to be sensitive to several diseases and neurological impairments. A decrease in this spontaneous behaviour correlates with brain lesions, 16,17 and genetic mutations, 18 as well as the progression of scrapie and Alzheimer’s disease. 19,20 Nest building is compromised by the systemic injection of MPTP, a Parkinson model, 14 and in a model of Rett syndrome. 21 Also, in LPS-treated mice, maternal nest building is decreased significantly. 22
A previous study from our laboratory revealed a correlation between postsurgical pain and nest building performance in laboratory mice. 12 However, a standardized protocol for the assessment of pain by means of nest building performance has not been developed so far.
In the present study we used ‘latency to nest building’ and a nest complexity scoring scale similar to already published nest building assessment methods. 1,2,14,23,24
We analysed the general nest building performance of healthy mice of different strains, sexes and housing conditions to evaluate genetic and environmental influences on nest building.
We assume that successful assessment of nest building performance depends on the right testing time points, as mice tend to destroy and rebuild their nests in a circadian rhythm. Therefore, to determine a suitable testing time we analysed the normal nest building rhythm in healthy C57BL/6J mice.
Using this determined time point, we aimed to prove the feasibility and reliability of nest complexity scoring as a method for detecting mild to moderate postsurgical pain in laboratory mice, and to standardize this method for routine laboratory use. For this purpose, individually-housed female C57BL/6J mice underwent a sham embryo transfer with or without different doses of the analgesic carprofen or underwent control treatment only.
We hypothesized that nest complexity is a sensitive and reliable tool that can be used to assess and grade postsurgical pain in mice.
Materials and methods
Ethics statement
The animal housing and experimental protocols were approved by the Cantonal Veterinary Department, Zurich, Switzerland, under licence no. ZH 120/2008, and were in accordance with Swiss Animal Protection Law. Housing and experimental procedures also conform to the European Convention for the Protection of Vertebrate Animals used for Experimental and other Scientific Purposes (Council of Europe No. 123 Strasbourg 1985) and to the Guide for the Care and Use of Laboratory Animals (Institute of Laboratory Animal Resources, National Research Council, National Academy of Sciences, 1996).
Animals
The animals were 48 female and eight male C57BL/6J, eight female DBA/2J, and eight female B6D2-Tg(Pr-mSMalphaActin)V5rCLR-25 mice, obtained from our in-house breeding facility at the age of 6–8 weeks.
The animals’ health status was monitored throughout the experiments by a health surveillance programme according to Federation of European Laboratory Animal Science Associations (FELASA) guidelines. The mice were free of all viral, bacterial, and parasitic pathogens listed in the FELASA recommendations, except for Helicobacter species. 25
All the animals were housed in groups of three to six animals for at least three weeks prior to testing in our animal room. The animals were kept in Eurotype III clear-transparent plastic cages (425 mm × 266 mm × 155 mm) with autoclaved dust-free sawdust bedding and one nestlet™ (5 cm × 5 cm), consisting of cotton fibres (Indulab AG, Gams, Switzerland) as nest building material. They were fed a pelleted and extruded mouse diet (Kliba No. 3436, Provimi Kliba, Kaiseraugst, Switzerland) ad libitum and had unrestricted access to sterilized drinking water. The light/dark cycle in the room consisted of 12/12 h with artificial light (approximately 40 Lux in the cage). The temperature was 21 ± 1℃, with a relative humidity of 45 ± 10%, and with 15 complete changes of filtered air per hour HEPA H13 filter, Camfil AG, Unterägeri, Switzerland. The animal room was insulated to prevent electronic and other noise. Disturbances, e.g. visitors or unrelated experimental procedures in the animal room, were not allowed.
Experiments
Latency to nest building: effects of strain, sex and housing conditions
Latency to first nest building activity was determined in mice of the three different strains, both sexes and under different housing conditions to analyse effects of these factors on nest building performance.
Eight female and eight male C57BL/6J, eight female DBA/2J, and eight female B6D2-Tg(Pr-mSMalphaActin)V5rCLR-25 mice, housed individually, and eight pairs of female C57BL/6J mice were tested in their familiar home cages. Additionally, eight female C57BL/6J mice, housed individually, were transferred to a new and clean cage directly before testing.
The mice were housed individually or in pairs three days prior to and during the observations. At the start of the 3-day adaptation phase, one nestlet was placed in the cage. Prior to observation at the beginning of the light phase, nestlet material was removed and a new nestlet was placed in the cage at the beginning of the light phase. Animals were video recorded for 24 h with an infrared-sensitive camera fixed above the cage.
All video recordings were analysed with ObserverXT™ 9 software (Noldus, Wageningen, The Netherlands). Nest building activity was defined as manipulating or carrying the nestlet or nestlet material for more than 3 s, and the latency to nest building was recorded in seconds.
Assessment of circadian nest building rhythm
The 24 h observations of eight individually-housed female C57BL/6J mice in their familiar home cage (the same mice as used in the assessment of latency to nest building) were analysed to assess normal circadian nest building rhythms in order to determine the optimal time point for nest complexity scoring.
All video recordings were analysed with ObserverXT™ 9 software, and nest building duration in seconds was recorded continuously. The remaining behaviours were separated into resting (inactive) and other activities and measured in seconds. Additionally nest complexity was scored by carefully approaching the cage without disturbing the animal at eleven time points using the scale described in Figure 1.
Nest complexity scoring: Score 0 = nestlet not manipulated, possibly dragged around the cage; Score 1 = nestlet slightly manipulated, more than 80% of nestlet intact, possibly a few shreds picked out; Score 2 = nestlet noticeably manipulated, less than 80% of nestlet intact, shreds spread around or in one area; Score 3 = noticeable nest site; less than 80% of nestlet intact, shreds are placed mostly in the nest site, hollow in bedding, mice start building walls; Score 4 = flat nest, hollow in bedding, walls mainly higher than mice and encasing the nest less than 50%; Score 5 = complex nest, more than 50% shreds picked out, bowl-shaped nest, walls higher than mice and encasing the nest by more than 50%.
Pain assessment with nest complexity scoring
Experimental design: Forty-eight individually-housed female C57BL/6J mice were tested (partly the same mice as used in the previous observations). Animals were tested before (baseline) and after an experimental procedure (experimental). Eight mice were allocated randomly to one of six experimental groups: (1) surgery + anaesthesia (mice underwent anaesthesia and surgery without analgesic treatment), (2) surgery + anaesthesia + low dose analgesia (mice underwent anaesthesia and surgery with 5 mg/kg carprofen), (3) surgery + anaesthesia + high dose analgesia (mice underwent anaesthesia and surgery with 50 mg/kg carprofen), (4) anaesthesia only, (5) anaesthesia + low dose analgesia, (6) anaesthesia + high dose analgesia.
Baseline data acquisition: Mice were housed individually three days prior to and during testing. At the start of the 3-day adaptation phase, one nestlet was placed in the cage. Prior to testing, the nestlet material was removed and a new nestlet was placed in the home cage at the beginning of the light phase.
Nest scoring (Figure 1) was carried out in the animal room by blinded observers 9 h after providing the nestlet, as this was found to be the optimal time point for nest complexity scoring in the analysis of circadian nest building rhythm (see also Results).
Experiments and experimental data acquisition: Experimental scoring of nest complexity was performed 2 days after baseline measurements. The experiment began at 1.5 h before the start of the light phase with a subcutaneous injection of 2 µL/g body weight of phosphate buffered saline (PBS) for the surgery + anaesthesia and anaesthesia-only groups. In the surgery + anaesthesia + analgesia and anaesthesia + analgesia groups, 5 or 50 mg/kg body weight of the analgesic carprofen (Rimadyl™, Pfizer Inc, New York, NY, USA) was diluted in PBS and injected subcutaneously as 2 µL/g body weight. Forty-five minutes later, the animals were transferred in individual transport cages to the operating theatre, which was located nearby. Mice were anaesthetized with sevoflurane (Sevorane™, Abbott, Baar, Switzerland) as a mono-anaesthesia. The anaesthetic gas was provided with a rodent inhalation anaesthesia apparatus (Provet, Lyssach, Switzerland); oxygen was used as a carrier gas. After induction of anaesthesia in a Perspex induction chamber (8% sevoflurane, 600 mL/min gas flow) animals were transferred to a warming mat (Gaymar, TP500, Orchard Park, NY, USA) set at 39 ± 1℃ to ensure constant body temperature, and anaesthesia was maintained via a nose mask (6–7% sevoflurane, 600 mL/min gas flow). The fur was clipped and the operating field disinfected with ethanol in all animals. Mice of surgery groups underwent a one-side sham embryo transfer. The incision in the abdominal muscle wall was closed with absorbable sutures (Vicryl™, 6/0 polyglactin 910, Ethicon Ltd, Norderstedt, Germany) and the skin was closed using skin staples (Precise™, 3 M Health Care, St Paul, MN, USA). Surgery was completed within 6–8 min in the surgery groups. Anaesthesia lasted 14–16 min in all groups. Animals were allowed to recover for 15–20 min on the warming mat before being transferred back to the animal room for subsequent behavioural testing.
The testing began at the beginning of the light phase after removing the used nestlet and adding a new nestlet by returning each mouse from its transport cage to its home cage. At 9 h after providing the new nestlet, nest scoring was carried out by carefully approaching the cage without disturbing the animal.
Statistical data analysis
Statistical analyses were performed using SPSS 20.0 software (IBM, Armonk, NY, USA). All data were tested for normal distribution and homogeneity of variance and met the necessary assumptions for parametric analyses. Mean and standard error of the mean (SEM) of latency to nest building, duration of nest building, and nest complexity scores for baseline and experimental measurements were calculated. Latencies to nest building were compared between different strains or housing conditions with a one-way analysis of variance (ANOVA). To compare the effect of sex on latency to nest building, an independent two-sample t-test was used. To test for significant differences between nest scores general linear model for repeated measures with time as within- and treatment as between-subject factor was used; post hoc testing was conducted with the Bonferroni test. Significance for all statistical tests was established at P < 0.05.
Results
Latency to nest building: effects of strain, sex and housing conditions
Latency to nest building: mean latency (SEM) of all strains, sexes and housing conditions.

Circadian nest building rhythm
Mice were occupied with nest building for, on average, 62.3 (SEM 22.6) min during 24 h of observation, which is equivalent to 4.3% of the day.
On average, mice started with nest building within the first 2 h after nest material was provided. This phase with high nest building activity (Figure 2a) was followed by resting phases with short disruptions for several behavioural activities and short nest building periods for rebuilding or maintaining the nest. In this late light phase nest scores reached high values. During the start of the dark phase, the mice normally had periods of locomotor activity with short breaks for occasional resting. In this active phase, the nest was usually destroyed or flattened out in the bedding by running over the nest site or digging in the bedding, which resulted in a decrease of nest complexity. From the middle to the end of the dark phase, mice started to rebuild their nests, interrupted by resting periods. On average, mice showed higher nest building activity towards the end of the dark phase with increasing nest scores.
(a) Normal nest building activity during 24 h in female individually-housed C57BL/6J mice. Mean duration of nest building in minutes per observed hour is shown on the vertical axis. (b) Determination of optimal scoring time: Mean nest scores ( + SEM) at 11 scoring time points. Scores increased during light phase, decreased at the onset of activity during the dark phase and increased towards a maximum at the end of the dark phase.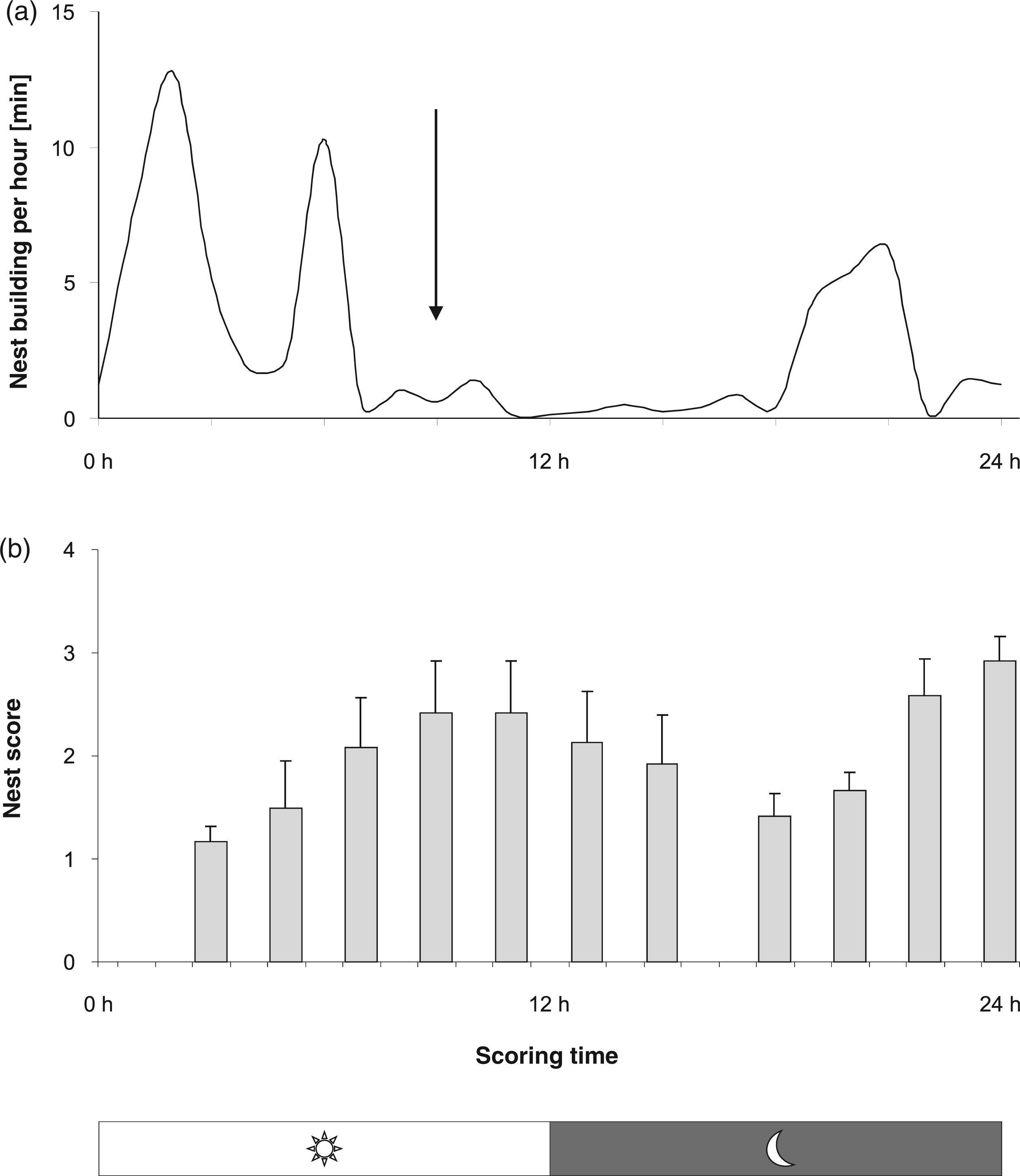
Regarding the analysis of the video sequences and the nest scoring, a single time point, 9 h after providing the new nestlet and the start of the light phase, was chosen for determination of the nest complexity scores for pain assessment in the following experiments (Figure 2a and b, arrow).
Nest complexity scoring
Nest scores showed a gradation after experiments (Figure 3). While animals that underwent surgery without or with a low dose of analgesia did not construct noticeable nests, animals treated with higher doses of analgesia had nest scores comparable with anaesthesia-only animals. Animals that received anaesthesia and analgesic treatment only had the highest experimental nest scores.
Mean nest complexity scores (+SEM) of female individually-housed C57BL/6J mice at 9 h. Baseline and experimental measurements of all experimental groups are shown. Asterisks indicate within-subject effect of time (baseline versus experimental) at P < 0.001. A significant between-subject effect of treatment was found (P < 0.05). Different letters over the bars indicate the differences revealed by post hoc analysis between nest scores of experimental groups at P < 0.05 (a versus b).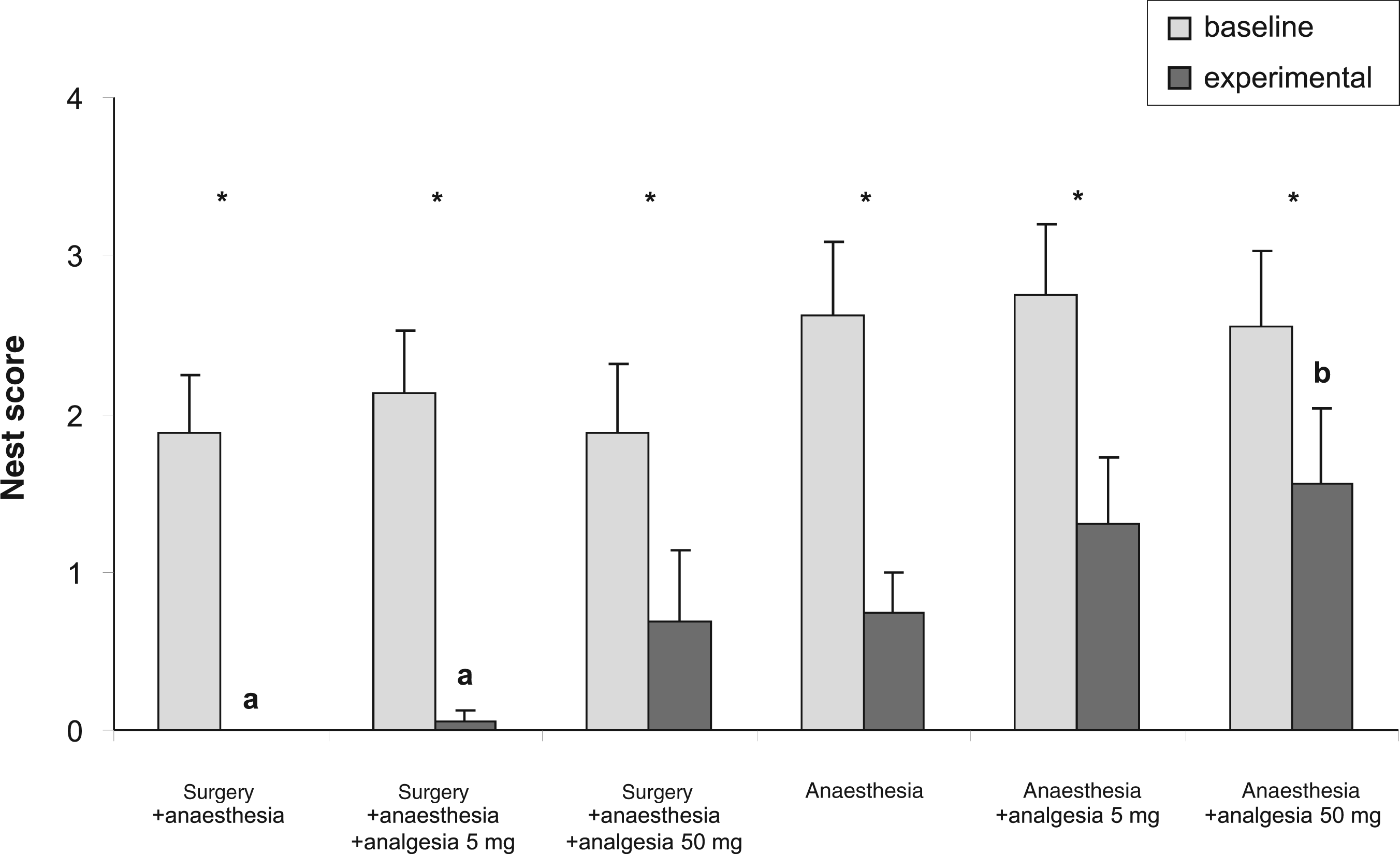
A significant within-subject effect of time (baseline versus experimental, P < 0.001) as well as a significant between-subjects effect of treatments (P = 0.024), but no interactions between time and treatment were found. Differences between nest scores of experimental groups were significant when comparing surgery + anaesthesia and surgery + anaesthesia + analgesia 5 mg with anaesthesia + analgesia 50 mg (P = 0.022; P = 0.031; Figure 3).
Overall, in baseline measurements 75–88% of all mice had noticeable-to-complex nests or at least their cages showed signs of noticeable nestlet manipulation, i.e. scores of two and higher, at the defined scoring time. After the experiments, the percentage of animals with noticeably manipulated nestlets and/or noticeable nests decreased to less than 63%, while animals after surgery without or with a low dose of analgesia never had noticeable nests and only less than 10% of these animals showed noticeable nestlet manipulation (Figure 4).
Percentage of female individually housed C57BL/6J mice of different experimental groups with noticeably manipulated nestlet and/or noticeable nests (nest complexity score >1) before and after experimental treatment at 9 hours at the end of the light phase. After experiments (i.) < 10% animals with noticeably manipulated nestlet and/or noticeable nests were found in surgery groups, (ii.) 20 - 60% animals in anaesthesia groups, (iii.) and >75% animals in baseline groups (healthy mice).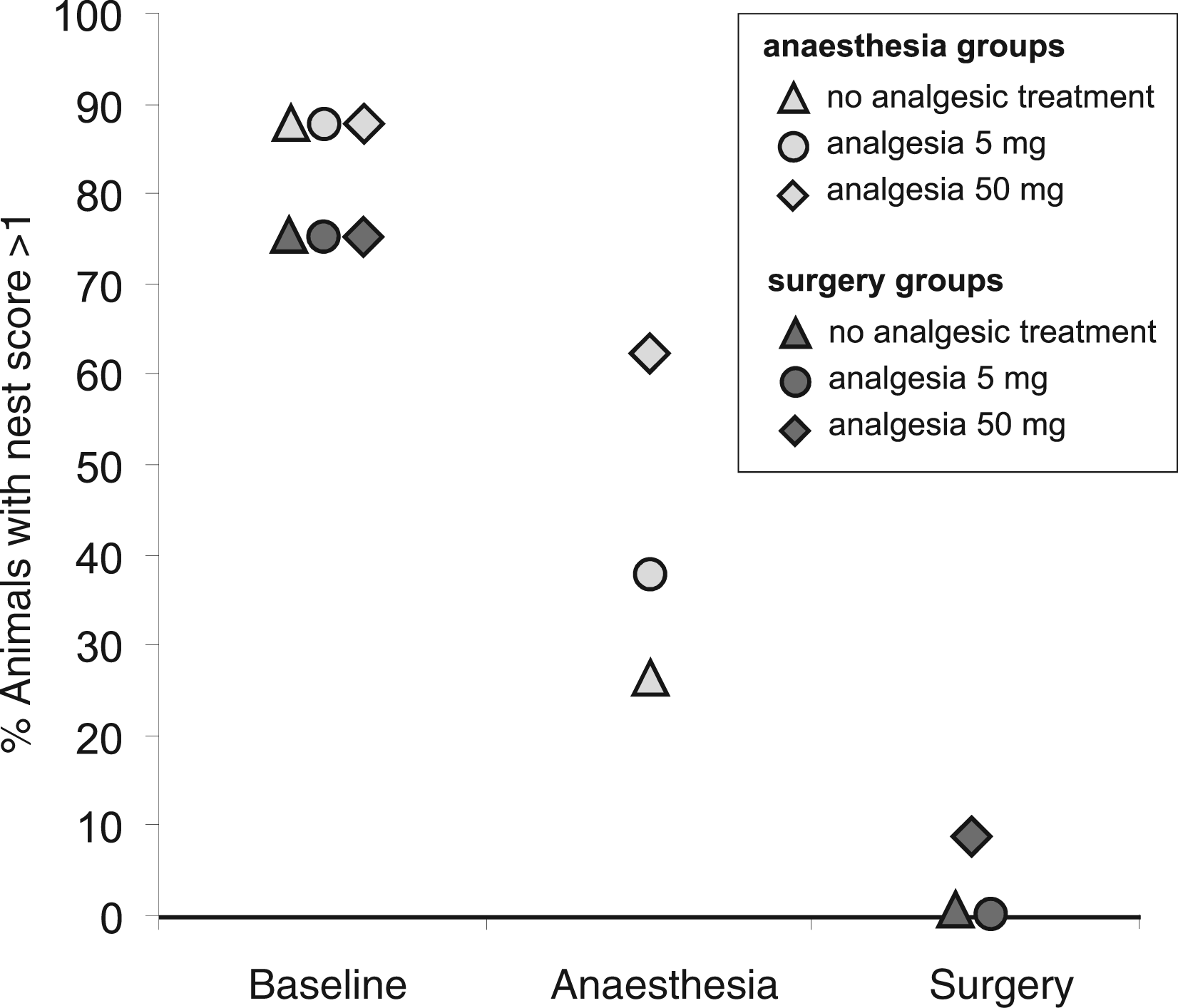
Discussion
All healthy mice investigated in this study exhibited complex nest building behaviour and constructed nests regardless of strain or sex and under all housing conditions tested. This situation changed distinctly when animals underwent a surgical or anaesthetic procedure: an incremental decrease of nest building performance was observed, correlating with the degree of invasiveness of the experiment.
We propose that changes in the highly motivated nest building behaviour can be used as a robust indicator of reduced animal wellbeing as also suggested previously by Arras et al. 12 and Deacon 2 . To establish a standardized protocol for the evaluation of postsurgical pain by nest building behaviour, we analysed common factors that could impact on the measurements and consequently influence the transferability of the monitoring protocol. Variability between sexes or genotypes regarding the amount of nest material used (e.g. weight of cotton fibres) or the shape and complexity of the nests have been reported. 4,26 However, in our study, the motivation to use the offered nest building material seemed to be high, and was comparable in different strains, sexes and under the different housing conditions used, as no difference in latency to nest building was observed in healthy mice. Most mice began to manipulate the nest material rapidly and spontaneously within minutes after the material was placed in the cage. Although small inter-individual differences in the shape and complexity of the nests appeared, individuals appeared to be consistent – an observation also described by others. 4
We then tested the feasibility of nest complexity scoring in a set-up, with female C57BL/6 J mice housed individually in their familiar home cages. Since we observed in our daily work that mice in general destroyed their nests during locomotor activity periods, we considered the appropriate choice of testing time to be an important prerequisite for successful complexity scoring. Nest complexity scoring is often done in the morning after material having been provided the day before (see Ref 2 for example), but surgery or other experimental procedures are often conducted during the day, and pain or other impairment are likely to be most significant directly after such procedures. In experiments with minor invasiveness, it is possible that nest scoring 24 h post procedure might miss the most pronounced signs of reduced wellbeing, as nest scores seemed to increase in our study during the dark phase (i.e. at 22 and 24 h after the experiment) up to baseline values (data not shown). Therefore we aimed to identify the appropriate testing time, considering the time point of the experiment and adapting to laboratory routine schedules.
The results of our analyses of behavioural circadian rhythmicity were comparable with the findings of other studies, showing that mice normally build a new nest or repair an old nest at the end of the dark phase. 27,28 Nest building peaks of our mice were shifted towards the beginning of the light phase, which was perhaps associated with the fact that fresh nest building material was provided in this phase. After pronounced nest building activity, animals rested in their nests and nest complexity scores were high and remained relatively constant in this phase. Additionally, this phase with constant nest scores fell within the normal working hours of laboratory personnel, which is a relevant argument for a method that should be applicable under routine laboratory conditions.
Based on these data, we chose a scoring time point of 9 h after experimental treatments at the end of the light phase. However, several hours before this time point also appear to be suitable for successful nest complexity scoring. By this means, nest scoring can act as a short-term retrospective indicator of impairment, which can be applied easily within the normal working day, particularly if procedures are conducted in the early morning. Thus, mice that have suffered, or are still suffering because of inefficient analgesic treatment can be identified easily and can be provided with rescue analgesic treatment.
A total of 75–88% of all healthy mice had identifiable nests or cages that showed at least noticeable nestlet manipulation leading to mean nest scores of 2–3. The maximum scores of 4–5 were difficult to reach within 9 h – in particular for a single-housed mouse – as the nestlet was a quadrate of tightly packed cotton fibres which thus needed intense work to reconstitute into a nest. Therefore we assume that nest scores of two and higher are normal nest scores for healthy individually housed mice after 9 hours.
In contrast to the substantial nest building performance of healthy mice, i.e. nest scores of 2 and above (see Figure 4), none of the animals undergoing surgery had noticeable nests 9 h after experiments and less than 10% of the animals manipulated the nestlet noticeably, suggesting that these groups may suffer from distress, impaired general condition or even pain.
As observed already in previous studies, 12,13,29 anaesthesia alone had a marked impact on behaviour. Here we observed a distinct effect of anaesthesia on nest building behaviour, which was significantly relative to the baseline, but clearly did not affect the animals as strongly as surgery, indicating only mild impairment. Animals that underwent anaesthesia and additional carprofen treatment of different doses had higher nest scores than anaesthesia-only groups. Whether carprofen can inhibit anaesthesia-induced behavioural aberrations, and thus have a nest building promoting effect, could not be clarified in this study. To our knowledge no behavioural effect of carprofen has been described that might explain these results.
Animals that received the low dose analgesic carprofen before surgery did not show a clear increase in nest complexity, which might be a sign that this dose was too low to relieve postsurgical pain, despite the fact that 5 mg/kg is a standard dose for mice 30 and has been shown to act as an effective analgesic protocol after surgery. 13 In the higher dose (50 mg/kg), we observed a slight tendency towards higher nest complexity comparable with the anaesthesia-only group, although not as high as in anaesthesia with analgesia groups.
As the clear and significant difference in nest complexity between healthy mice and mice that underwent surgery could not be alleviated by carprofen treatment in a significant manner, our study lacked a sound proof that postsurgical pain caused the massive decrease in nest complexity. Thus, other impacts of surgery might also affect nest complexity, e.g. physiological stress or motor impairment. However, it is very unlikely that carprofen in the used dose rates was not capable of pain relief as it has been proved to be effective after laparotomy in several studies (see Refs 13 and 31 for examples). Nest complexity scoring might therefore be a useful indicator of reduced wellbeing after surgery, but cannot be used to assess the efficacy of pain treatment. Nevertheless, the estimation, i.e. grading, of the impact of procedures on wellbeing and general condition is possible with nest complexity scoring.
Our results suggest that deficits of nest building are associated with reduced wellbeing and impaired general condition, 12 which can also include pain, and may trigger a competitive motivational system that makes the animal tend to be lethargic or be concerned with other behaviours like self grooming and with decreased motivation to engage in otherwise highly valued nest building behaviour. 22,32
Nest complexity scoring is based on the animal's normal behaviour performed in the animal's home cage, and does not require special apparatus or housing facilities. The test causes no additional stress to the animals, as nest building is a species-specific and complex form of active interaction with the environment. Providing nest material allows mice to structure their environment and gain more control over their living conditions, which is assumed to enhance their wellbeing. 10,20,33,34
In summary, nest complexity scoring can be implemented easily in any laboratory animal facility and can be applied in the daily routine for the detection and assessment of post-procedural impairment in laboratory mice. Even though the motivation to use the nest material was comparable under different conditions, it might be necessary to adapt the scoring system to other nest building material or housing conditions. As social housing is the preferred housing condition for mice and has been suggested to enhance postsurgical recovery in female mice, 15,29,35 further studies should focus on an adaption of the described assessment method to prevalent housing conditions like pair or group housing.
Footnotes
Acknowledgments
This work was sponsored by grants from the Federal Veterinary Office (Bern, Switzerland), and UBS Foundations. The authors would like to thank Robin Schneider and the staff of the Central Biological Laboratory for support in housing the mice. We thank Professor Kurt Bürki for generously providing research facilities and resources.
