Abstract
Haemarthrosis is the most common clinical manifestation of haemophilia and is responsible for significant morbidity in haemophilic patients. The murine experimentally-induced knee bleeding model is an important model in haemophilia research but it is currently unknown if the use of analgesia in this model might impact on the inflammatory response. The aim was to investigate the inflammatory response after a needle induced knee bleed in haemophilia A mice treated with buprenorphine or saline. One hundred and sixty mice were randomized into two groups to blindly receive buprenorphine or saline. All the mice were anaesthetized and knee injury was induced by inserting a 30 G needle into the right knee joint. At t = 6, 24, 48 and 72 h, 20 mice from each group were terminated and the following parameters were assessed: change in body weight and joint diameter, visual bleeding score (VBS), white blood counts, haematocrit, platelet concentrations, haemoglobin, plasma haptoglobin and plasma and synovial fluid levels of 23 cytokines. Twenty mice were terminated at t = 0 receiving no injury or treatment to provide baseline measures. Twenty-one cytokines in plasma and 22 cytokines in synovial fluid, joint diameter change, VBS and blood parameters were not significantly altered by the administration of buprenorphine. Slight alterations of plasma haptoglobin at t = 48 h, body weight, plasma and synovial eotaxin and plasma G-CSF were found in buprenorphine-treated mice. We demonstrated that buprenorphine does not overall impact on the inflammatory response, and the use of buprenorphine in the knee bleeding model in haemophilic mice should be continued.
Haemarthrosis is the most common clinical manifestation of haemophilia and is responsible for significant morbidity in haemophilic patients.1,2 After an intra-articular bleeding episode blood is removed by absorption to the synovial tissue 3 and with recurrent bleeding episodes degradation products of red blood cells accumulate in the tissue, triggering an inflammatory response.3,4 This inflammatory response is an important part of the immune response, and secreted cytokines provide signals between immune cells to coordinate the inflammatory response. 5
Cytokines have been shown to be involved in a number of arthritic disorders such as haemarthrosis, 6 osteoarthritis (OA),7,8 rheumatoid arthritis (RA),9–13 inflammatory arthritis 14 and acute joint trauma.15–21
The pro-inflammatory cytokines, IL-1β, IL-6 and TNF-α, are considered major players in both RA and OA.7,9,10,12,22 In addition, other cytokines such as IL-8, IL-15, IL-17, IL-18 and IL-21 have been described as being involved in both OA and RA,7,10,11,22–26 albeit at different levels; synovial fluid cytokine levels are in general significantly increased in patients with RA compared to patients with OA.10,27–29
Limited research has been published describing cytokine involvement in haemophilic arthritis but we have previously shown that haemophilic mice suffering from induced haemarthrosis demonstrate increased levels of IL-1β, IL-6, KC and MCP-1 in synovial fluid. 6 Haemarthrosis is also present in acute traumatic joint diseases such as anterior cruciate ligament (ACL) rupture and this has been more widely studied: TNF-α, IL-1β, IL-6, IL-8, IL-10, MCP-1, IFN-γ, MIP-1β have all been found to be elevated in the haemorrhagic synovial fluid of ACL.17,18,20
Animal models of haemophilia are invaluable tools in studies concerning therapeutic intervention, as well as mode of action studies. In the work with animal models evaluating inflammatory response as a scientific goal, the impact of analgesia is always a challenge and must be considered and evaluated to ensure there is little or no impact.
The murine experimentally-induced knee bleeding model30,31 is a very important model in haemophilia research. In this model a joint bleed is induced by inserting a needle into the knee joint cavity of F8 knock-out (F8-KO) mice, resulting in a consistent and severe joint bleed. In human patients intra-articular bleedings are known to cause acute pain which is usually treated with non-steroidal anti-inflammatory drugs (NSAIDs) or opioids. 32 Mice suffering from haemarthrosis are expected to experience the same kind of pain and should be treated accordingly. Buprenorphine is the most commonly used analgesic in laboratory rodents 33 and the drug of choice for analgesia in this model but the impact on inflammatory response is unknown. Buprenorphine is a semi-synthetic opioid and has been found to function as a full µ-agonist for analgesia 34 and its analgesic potency has been found to be 25–40 times that of morphine in mice when administered parenterally. 35 Most research on opioids and their effect on the inflammatory response concern morphine and heroin and describe increased production of pro-inflammatory cytokines.36–39 Buprenorphine however, has been found not to modify cytokine release in several models40,41 but results are inconsistent. 42 As the inflammatory response is an important part of the pathogenesis of haemophilic arthropathy any administration of analgesics should ideally not alter this response. For animal welfare and ethical reasons however, the full analgesic protocol in this model should be continued and the response of buprenorphine on the inflammatory response should be evaluated. In addition to cytokines we also evaluated a number of chemokines as well as haptoglobin. Chemokine MCP-1 has previously been found to be elevated in haemophilic mice suffering from induced haemarthrosis, 6 and haptoglobin has been found to be elevated in rats with arthritis. 43
The objective of this study was to investigate the inflammatory response after a needle induced knee bleed in haemophilia A mice treated with either buprenorphine or saline. Thus, we hypothesized that treating the haemophilic mice with buprenorphine in conjunction with a needle-induced joint bleed should not influence the inflammatory response.
Materials and methods
Animals
One hundred and eighty F8-KO mice, female:male 50:50 (B6;129S4-F8tm1Kaz/J) from Taconic, Denmark were enrolled in the study. The mice were between 12 and 16 weeks of age when included in the study and were acclimatized for at least seven days at Novo Nordisk A/S, Måløv under standardized conditions including 12/12 h light/dark cycle, 21℃ and 60% relative humidity. The mice were fed (Altromin 1320 diet; Brogaarden, Gentofte, Denmark) and watered ad libitum and twice a week they were given corn and wheat mixed with their bedding material at the bottom of the cage. The mice were housed in cages (Tecniplast Makrolon type IV cages, Scanbur; Karlslunde, Denmark) with raised lids providing the animals with a cage bottom area of 1800 cm2 and a cage height of 25 cm. For environmental enrichment the animals were provided with bedding (Tapvei aspen bedding, Tapvei, Kortteinen, Finland) and nesting (Enviro-Dri® paper-based nesting material; Lillico, Surrey, UK) materials, hides, a wood biting stick (Tapvei, Kortteinen, Finland) and a 30 cm hemp rope (Fyns Kranudstyr, Odense, Denmark) with a 6 mm diameter.
The experiments were performed according to guidelines from the Danish Animal Experiments Council, the Ministry of Food, Agriculture and Fisheries of Denmark.
Procedures
The mice were treated according to procedures previously described. 44 In brief, joint bleeding was induced by the insertion of a 30 G needle into the right knee joint of fully anaesthetized mice at an anterior approach. The left knee joint served as a control. Immediately after the knee injury the mice received a subcutaneous injection of buprenorphine (Temgesic® Vet, 0.3 mg/mL; Schering-Plough, Ballerup Denmark) at a dose of 0.1 mg/kg or an equivalent dose of 0.9% saline. The analgesic dosing regimen was decided as recommended by Hau et al. 45 At termination the knee joints were opened, inspected macroscopically, and bleeding was scored according to the visual bleeding score (VBS). 46 Body weight and joint diameter were measured before the induction of the joint bleed and again at termination. Blood was sampled from the retro-orbital plexus, and synovial fluid was sampled from both knee joints. At the end of the study the mice were euthanized under full anaesthesia.
In order to evaluate the effect of buprenorphine on the inflammatory response a number of clinical and biochemical parameters were assessed: change in body weight, change in joint diameter, VBS, haematological profile including white blood counts, haematocrit, platelets and haemoglobin, as well as plasma and synovial fluid levels of 23 different cytokines and, finally, plasma haptoglobin.
Study design
One hundred and sixty mice were randomized into two groups to blindly receive buprenorphine or vehicle. Males and females were equally distributed in each group. Buprenorphine (0.1 mg/kg corresponding to a volume of 0.007 mL/g) or an equal volume of 0.9% saline was administered subcutaneously in connection with the induction of the knee bleed. During the remainder of the study period buprenorphine was administered in the drinking water to all the mice in the buprenorphine group. Buprenorphine was provided 0.6 mg per 100 mL water, corresponding to an oral dose of 0.9 mg/kg per 24 h. The dosing regimen of buprenorphine was decided as previously described.
6
The study was blinded so the operator was unaware which mice were receiving buprenorphine and which were receiving saline by injection as well as water bottles. At day 0, 20 mice receiving no treatment or knee injury were sacrificed to obtain baseline measurements of white blood counts, haematocrit, platelet concentration and haemoglobin as well as haptoglobin and cytokine levels. In the remaining 160 mice, a knee bleed was induced as previously described. At 6, 24, 48 and 72 h after knee injury, 20 mice from each group were euthanized and processed as described in Figure 1. The group size was estimated based on results obtained in a previous study measuring cytokines in synovial fluid from haemophilic mice.
6
Based on the standard deviation (SD) and the ability to detect a 10-fold difference JMP® (Release 8; SAS Institute Inc, Cary, NC, USA) was used to carry out group size estimation.
Study time-line. One hundred and eighty mice were enrolled in the study. Twenty mice were sacrificed at t = 0 receiving no knee injury providing baseline measurements. The remaining 160 mice were injured in the right knee at t = 0 and were randomly and blindly assigned to receive buprenorphine or saline 0.9%. At the following time points after induction of the knee bleed t = 6 h, 24 h, 48 h and 72 h, 20 mice from each group were sacrificed.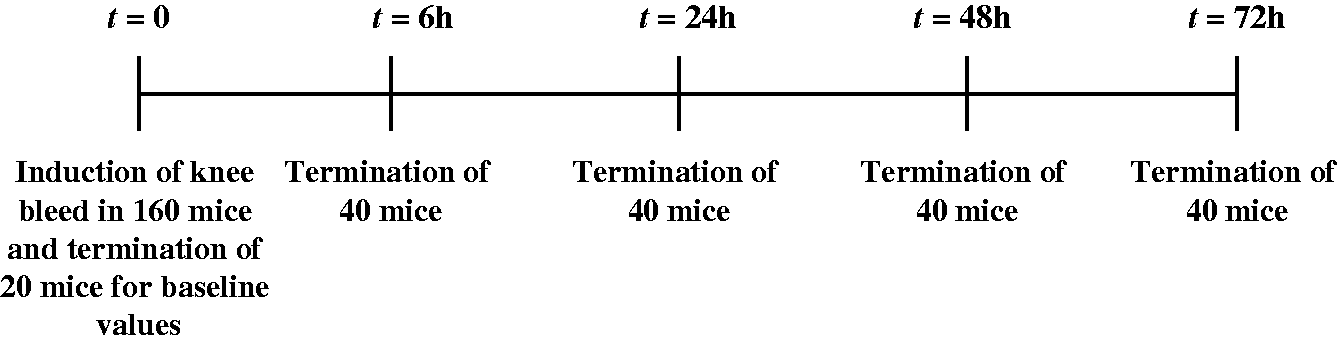
Measurement of joint diameter
The diameters of both right and left knee joints were measured before injury and again at termination with a digital caliper (Mitutoyo, Tokyo, Japan). The diameter was measured medially to laterally across the knee joint five times and the mean of the five measurements was recorded as the joint diameter.
Measurements of white blood counts, haematocrit, platelet concentration and haemoglobin
Blood was collected from the retro-orbital plexus using a 20 µL EDTA coated capillary tube for determination of white blood counts, haematocrit, platelet concentration and haemoglobin by the Medonic CA530 (Boule, Stockholm, Sweden).
Plasma samples for cytokine and haptoglobin determination
Blood was sampled from the retro-orbital plexus using a 25 µL capillary tube in an Eppendorf tube containing sodium citrate (0.13 M) (1:10). The blood and sodium citrate were mixed thoroughly but gently and then centrifuged at 4000 g for 5 min. Plasma was frozen immediately after centrifugation and stored at –80℃ until analysis.
Euthanasia
All mice were euthanized by cervical dislocation under full isoflurane/N2O/O2 (0.3 L/min O2 and 0.7 L/min N2O) anaesthesia.
Visual bleeding score
After inspection of the knee joint, the joint was dissected under a microscope. The skin was resected and the patellar ligament transected. Using microsurgery scissors an incision was placed along the knee joint on each side thereby exposing the joint cavity. The joint cavity was assessed and the presence of blood was noted. The joint was scored according to the VBS 46 based on the presence of blood and the degree of distension of the joint cavity.
Collection of synovial fluid
After inspection and scoring of the knee joints samples of the synovial fluid were obtained from both right and left knee joints as previously described. 6 In brief, synovial fluid was obtained by saturation of a filter paper (diameter 2 mm) placed in the knee joint. To obtain comparable amounts of synovial fluid a standardized piece of filter paper was used and it was allowed to saturate. After collection the samples were individually placed in Eppendorf tubes and frozen immediately. The samples were stored at –80℃ until analysis.
Extraction of synovial fluid from filter paper
As previously described 6 the extraction of synovial fluid from the filter paper was accomplished by elution in a 90 µL extraction buffer (assay buffer containing ‘complete mini protease inhibitor cocktail with EDTA’ [Roche, Basel, Switzerland]; one tablet dissolved per 7 mL of assay buffer [PBS containing 5 mL/L Tween 20 and 10 g/L BSA, pH 7.4]). The samples were extracted for 60 min at room temperature in a microplate shaker (600 rpm).
Detection of cytokines/chemokines
Levels of 23 cytokines/chemokines: IL-1α, IL-1β, IL-2, IL-3, IL-4, IL-5, IL-6, IL-9, IL-10, IL-12(p40), IL-12(p70), IL-13, IL-17, eotaxin, G-CSF, GM-CSF, IFN-γ, KC, MCP-1, MIP-1α, MIP-1β, RANTES and TNF-α were determined in both synovial fluid and plasma at each time point by means of a Bio-plex® mouse cytokine kit (Bio-Rad Laboratories Ltd, Herts, UK).
Both anti- and pro-inflammatory cytokines were included in the study. IL-1α, IL-1β, IL-2, IL-6, IL-12(p40), IL-12(p70), IL-17, IFN-γ, KC and TNF-α represent the pro-inflammatory cytokines7,47 and IL-4, IL-5, IL-10 and IL-13 represent the anti-inflammatory cytokines. 5 A number of chemokines were also included in the study: MCP-1, MIP-1α, MIP-1β, RANTES and eotaxin.
Synovial fluid eluates and plasma samples were processed according to the protocol provided by the manufacturers and previously described. 48 In brief, samples were incubated with antibody-coupled beads, and after a washing step removing unbound protein, a biotinylated detection antibody was added for the creation of a sandwich complex. After another washing step streptavidin–phycoerythrin was added to bind to the biotinylated detection antibodies on the bead surface. The captured bead-complexes were subsequently read by the Bio-Plex® array reader (Bio-Rad Laboratories Ltd). Cytokine concentrations were calculated by reference to the standard curves generated on the supplied standards using instructions from the manufacturer. Synovial fluid cytokine levels were determined in 50 µL of the filter paper eluates. Plasma samples were diluted 1:4 with sample diluent provided by the manufacturer.
Detection of plasma haptoglobin
The haptoglobin concentrations in plasma were determined using a mouse haptoglobin ELISA test kit (Life Diagnostics Inc, West Chester, PA, USA). The plasma samples were diluted 1:50000 and processed according to the instruction manual as previously described. 49 The SpectraMax 340 (MDS Analytical Technologies, Sunnyvale, CA, USA) was used for plate reading, and haptoglobin concentrations were calculated by reference to the standard curves generated on the supplied standards using instructions from the manufacturer.
Statistical analysis
All results are expressed as mean ± SD. Statistical analyses were carried out using JMP®. For statistical analysis of the cytokine levels, out of range low values were set as 0.1 pg/mL to allow for log transformation of data. A two-way analysis of variance (ANOVA) and Tukey's honestly significant difference test or a Wilcoxon rank sum test were used to compare the levels of the cytokines in plasma and synovial fluid at different time points as well as comparison with the baseline level.
In order to compare differences and for comparison with baseline levels a two-way ANOVA and Tukey's honestly significant difference test were applied to the following parameters: joint diameter, body weight, white blood counts, haematocrit, platelets and haemoglobin. For plasma haptoglobin levels a Wilcoxon rank sum test was used for comparison between groups at different time points and for comparison with the baseline level. An ordinal logistic regression was used to compare VBS between groups at different time points. A 0.05 significance level was used. For graphic illustrations of the results GraphPad Prism 5® (Version 5, GraphPad Software, Inc, San Diego, CA, USA) was used. Applicable test assumptions were met for all statistically tests.
Results
Change in body weight
There was a minor but significant difference in body weight change between buprenorphine and saline-treated mice (P < 0.01). Mice receiving buprenorphine on average lost 1.32% of their body weight (mean –0.3 g, SD 0.8) during the study period whereas mice receiving saline on average gained 0.4% of their body weight (mean + 0.09 g, SD 0.68). When comparing body weight change between groups at the different time points, it was found only to be significantly different between groups at 24 h post injury.
Change in joint diameter
There was no significant difference in joint diameter change for mice receiving buprenorphine compared with mice receiving saline at any time point.
White blood counts, haematocrit, platelet concentration and haemoglobin
There was no significant difference in white blood counts, haematocrit, platelet concentration and haemoglobin for mice receiving buprenorphine compared with mice receiving saline at any time point. Furthermore, there was no significant difference for the buprenorphine or saline group compared with baseline at any of the time points included.
Visual bleeding score
In the buprenorphine and the saline-treated groups severe joint bleeds with a mean VBS of 2.1 (SD = 0.8) and 2.2 (SD = 0.8) were observed, respectively. Identical procedures done in normal non-haemophilic mice result in a mean VBS of 0–0.5 (SD = 0.5).31,44 At no time point was there any significant difference in VBS for mice receiving buprenorphine compared with mice receiving saline (Figure 2).
The visual bleeding score (VBS) for the buprenorphine and saline groups during the study period. There was no significant difference in VBS for mice receiving buprenorphine compared with mice receiving saline at any time points during the study period. Values are illustrated as means and SD.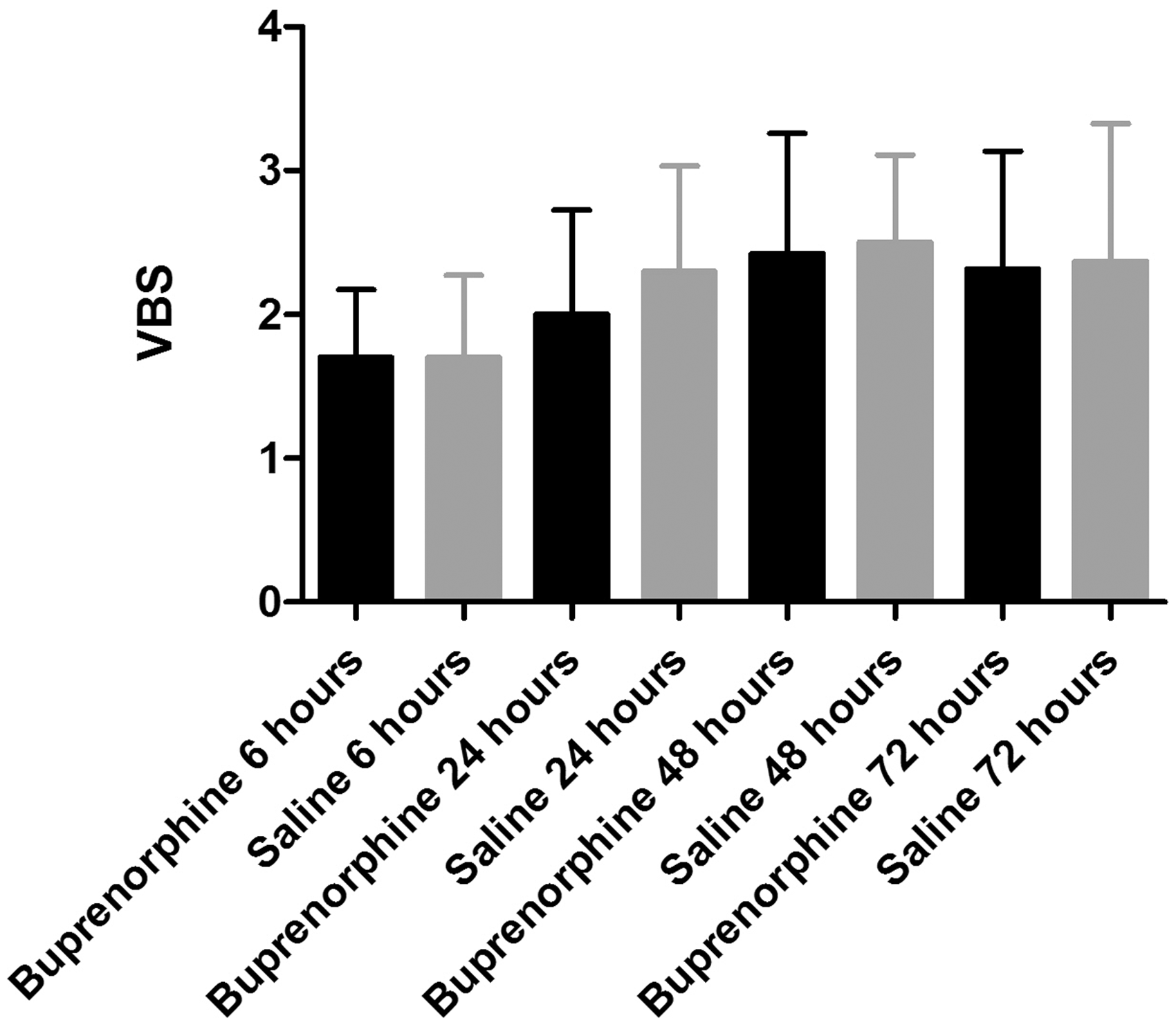
Cytokines
Overall, the cytokine responses were acute and generally highest at 6 h post injury. The difference in cytokine levels between groups was most pronounced at this time point. Because of this, only cytokine levels at 6 h post injury will be addressed in the following.
Cytokines in synovial fluid
For IL-1α, IL-2, IL-12(p40), IL-17, IFN-γ, TNF-α (pro-inflammatory cytokines), IL-5, IL-10, IL-13 (anti-inflammatory cytokines), MIP-1α, MIP-1β, RANTES (chemokines), GM-CSF and IL-9 no significant differences in synovial fluid levels between the buprenorphine and saline groups were seen. Further, none of these cytokines were significantly elevated in either the buprenorphine or the saline group compared with the baseline group.
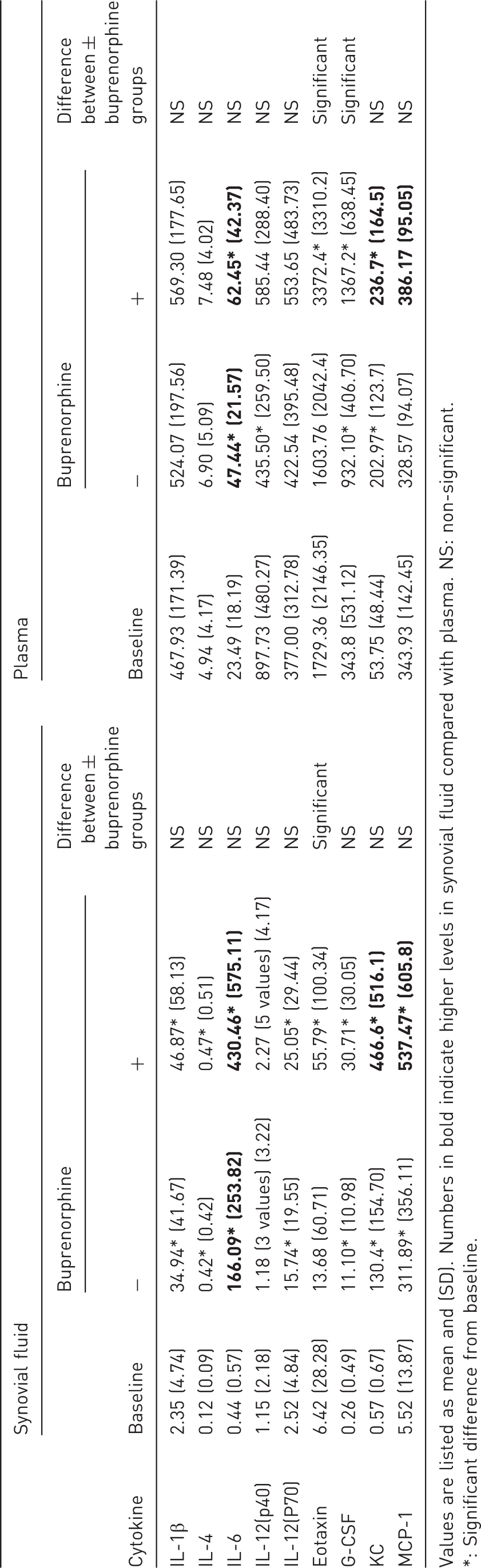
IL-1β (P < 0.05), IL-12(p70) (P < 0.05), KC (P < 0.0001) (pro-inflammatory cytokines), IL-4 (P < 0.01), IL-6 (P < 0.0001) (anti-inflammatory cytokines), MCP-1 (P < 0.0001) (chemokines) and G-CSF (P < 0.0001) were all significantly elevated in synovial fluid in both the buprenorphine and the saline groups compared with the baseline group. No significant differences were found between the buprenorphine and the saline groups for any of these cytokines (Figure 3 and Table 1).
Synovial fluid level of IL-1β, IL-4, IL-6, IL-12(p70), G-CSF, KC, MCP-1 and eotaxin for the baseline group and for the buprenorphine and saline groups 6 h post injury. *P < 0.05, **P < 0.01, ***P < 0.0001. Values are illustrated as means and SD. There were significant elevated levels of IL-1β, IL-4, IL-6, IL-12(p70), G-CSF, KC and MCP-1 in synovial fluid in both the buprenorphine and the saline groups compared with baseline with no significant differences between the buprenorphine and saline groups. There was a significantly higher level of eotaxin in synovial fluid in the buprenorphine group compared with both the saline and the baseline groups and no significant differences in eotaxin synovial fluid levels were found between the saline and baseline groups. Synovial fluid and plasma levels of nine cytokines showing significant differences either between groups or compared with baseline value 6 h post injury. Values are listed as mean and (SD). Numbers in bold indicate higher levels in synovial fluid compared with plasma. NS: non-significant. *: Significant difference from baseline.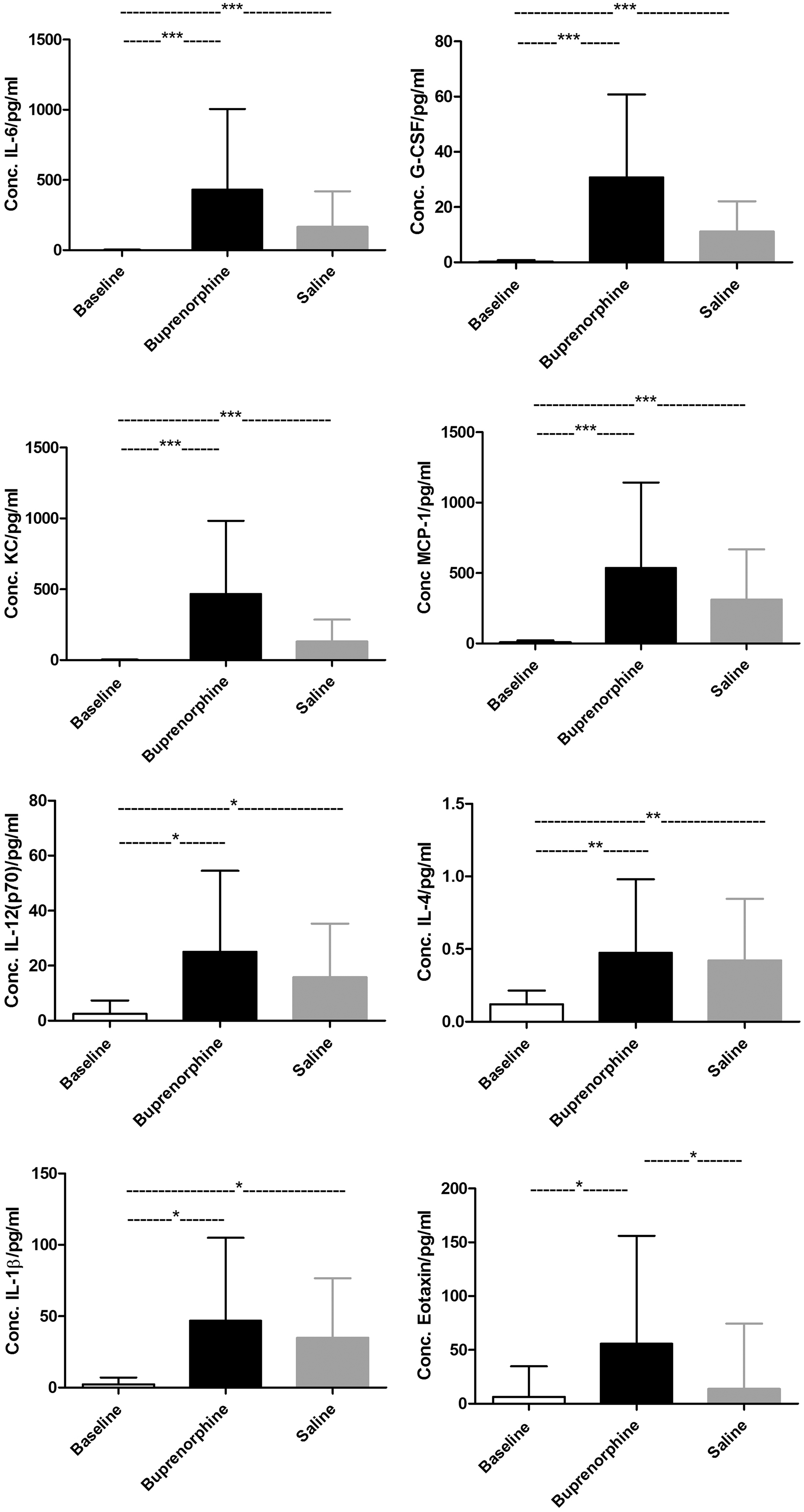
The chemokine, eotaxin, was found to be significantly elevated in synovial fluid in the buprenorphine group compared with both the saline (P < 0.05) and the baseline (P < 0.05) groups. No significant differences were found between the saline and baseline groups (Figure 3 and Table 1).
Cytokines in plasma
For IL-1α, IL-1β, IL-2, IL-12(p40), IL-12(p70), IL-17, IFN-γ, TNF-α (pro-inflammatory cytokines), IL-4, IL-5, IL-10, IL-13 (anti-inflammatory cytokines), MCP-1, MIP-1α, MIP-1β, RANTES (chemokines), IL-3, IL-9 and GM-CSF no significant differences in plasma levels between the buprenorphine and the saline groups were seen. Further, none of these cytokines were significantly elevated in either the buprenorphine or the saline group compared with the baseline group.
As in synovial fluid KC (pro-inflammatory cytokine), IL-6 (anti-inflammatory cytokine) and G-CSF were found at significantly higher levels in plasma for both the buprenorphine and saline groups compared with the baseline group (P < 0.0001). No significant differences were found between the buprenorphine and the saline groups for IL-6 and KC. For G-CSF however, the buprenorphine group had a significantly elevated plasma level compared with the saline group (P < 0.05) (Figure 4 and Table 1).
The plasma levels of IL-6, G-CSF, KC and eotaxin for the baseline group and the buprenorphine and saline groups at 6 h post injury. *P < 0.05, ***P < 0.0001.Values are illustrated as means and SD. There were significantly elevated plasma levels of IL-6, G-CSF and KC in both the buprenorphine and saline groups compared with baseline. Further, a significantly higher plasma G-CSF level in the buprenorphine group compared with the saline group was found. There was a significantly higher plasma level of eotaxin in the buprenorphine group compared with both the saline and the baseline groups. No significant differences in plasma eotaxin levels were found between the baseline and the saline groups.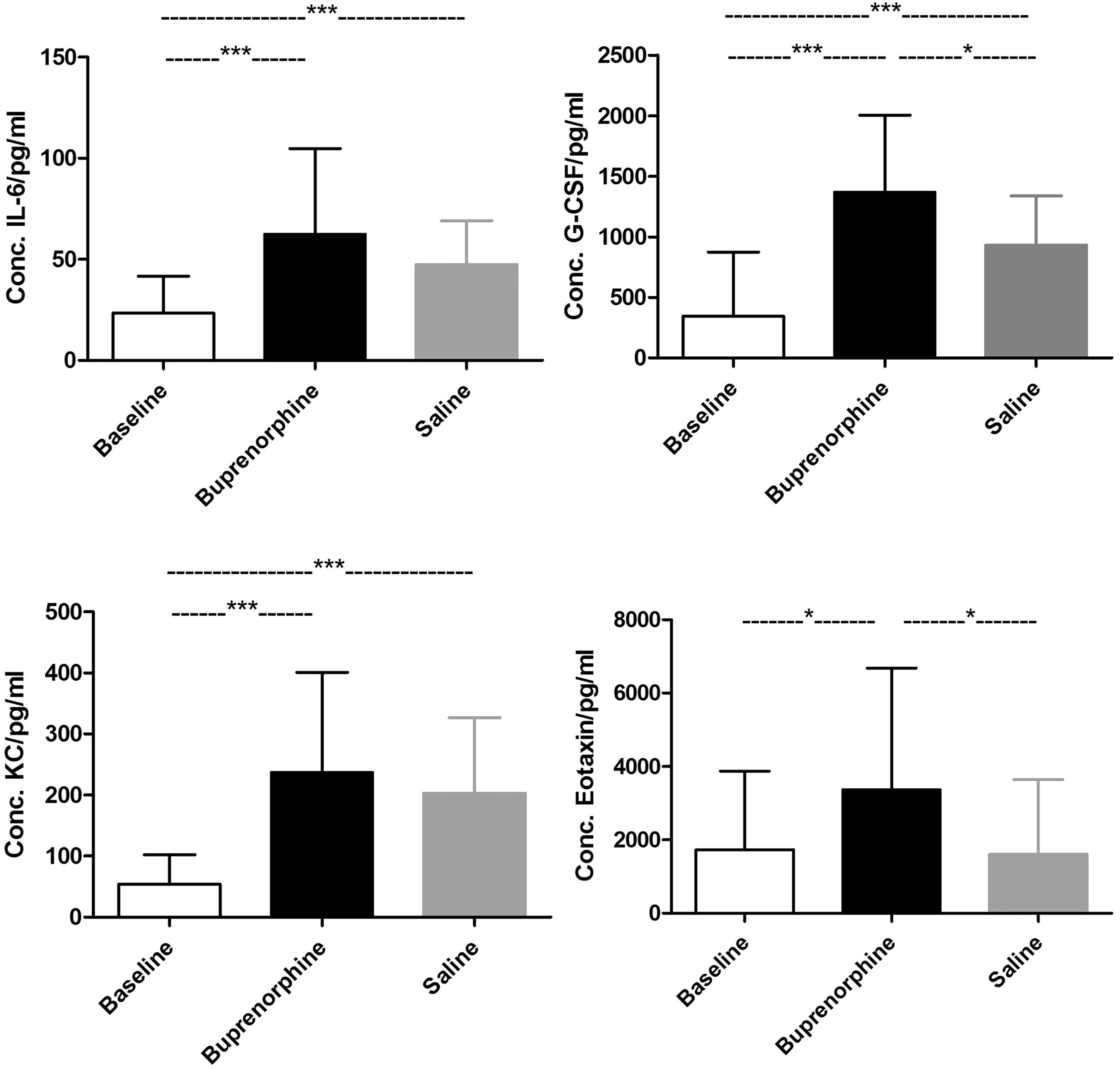
Likewise, the chemokine eotaxin was found to be significantly elevated in plasma in the buprenorphine group compared with both the baseline group and the saline group (P < 0.05). No significant differences were found between the baseline and the saline groups (Figure 4 and Table 1).
Plasma haptoglobin
For mice receiving buprenorphine significantly elevated plasma haptoglobin levels were found at 24, 48 and 72 h post injury compared with baseline. For mice receiving saline significantly elevated plasma haptoglobin levels were found 24 and 48 h post injury compared with baseline. Additionally, mice receiving buprenorphine had significantly higher plasma haptoglobin levels at 48 h post injury compared with mice receiving saline. At no other time point was any significant difference found between the buprenorphine and saline groups (Figure 5).
Haptoglobin levels in plasma for the baseline group and the buprenorphine and saline groups during the study period. *P < 0.05, **P < 0.01 and ***P < 0.0001 indicating significant differences compared with baseline level. __*__ = P < 0.5 indicating significant differences between groups. In mice receiving buprenorphine, plasma haptoglobin levels were significantly increased at 24, 48 and 72 h post injury compared with the baseline level. Further, in mice receiving saline, plasma haptoglobin levels were significantly increased at 24 and 48 h post injury compared with the baseline level. At 48 h post injury mice receiving buprenorphine had a significantly elevated plasma haptoglobin level compared with mice receiving saline.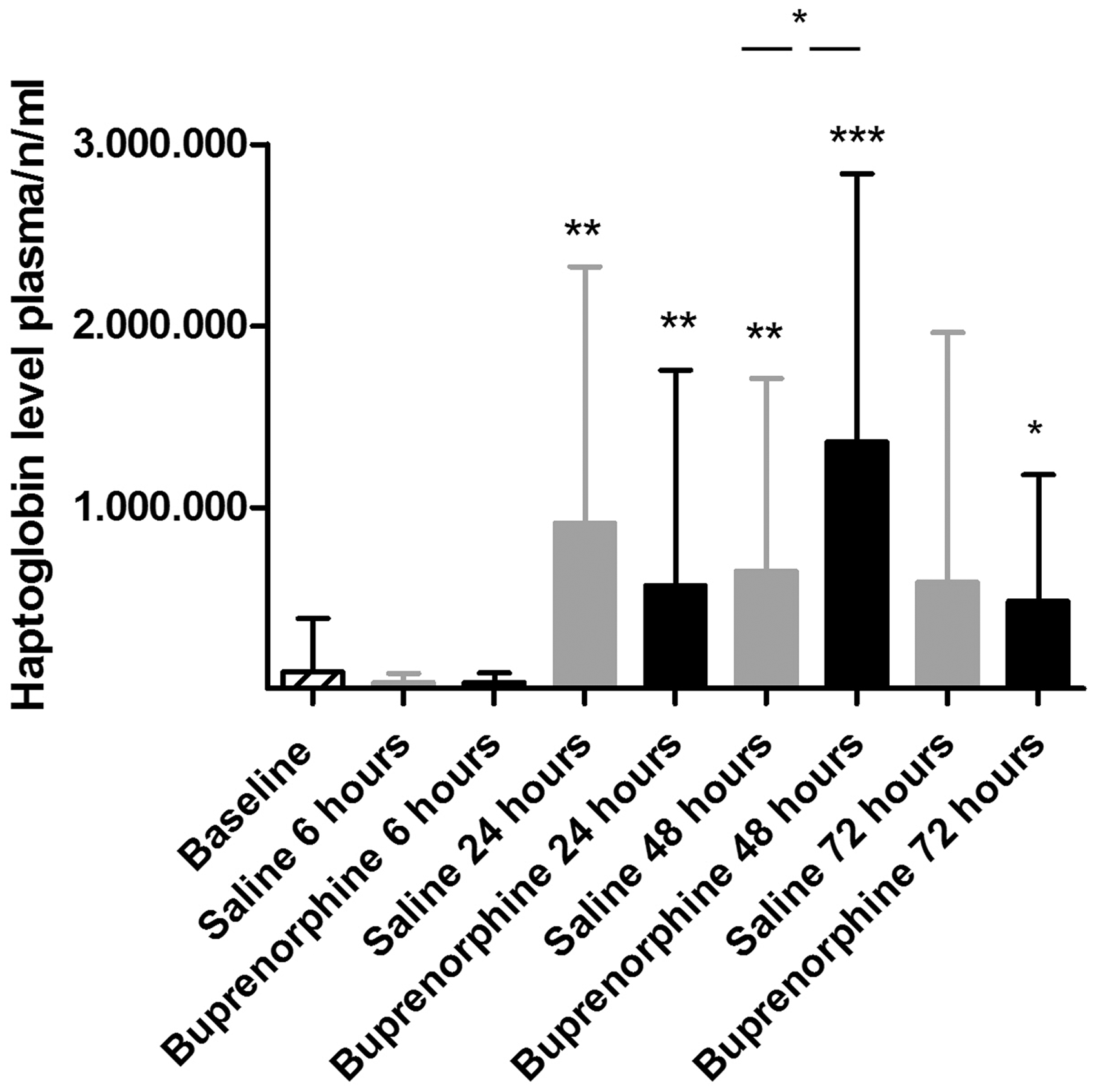
Discussion
In the current study we demonstrate that pain management using an analgesic protocol applying buprenorphine does not significantly alter the measured inflammatory responses in a needle-induced joint bleeding model in haemophilic mice, indicating that the buprenorphine analgesic protocol described may be used in future studies. A wide panel of inflammatory parameters was evaluated, and it was demonstrated that 21 of 23 cytokines in plasma and 22 of 23 cytokines in synovial fluid were not significantly altered by the administration of buprenorphine. Furthermore, no effects on joint diameter change, VBS, white blood counts, haematocrit, platelets or haemoglobin were determined by the administration of buprenorphine.
Plasma haptoglobin levels were found to be increased in both saline and buprenorphine-treated mice from 24 h after injury. At 48 h post injury the plasma haptoglobin level for the buprenorphine group was found to be significantly increased compared with the saline group. It may be speculated whether this difference represents an actual effect of buprenorphine or is caused by the large variation seen in the plasma haptoglobin levels. Haptoglobin is an acute phase protein produced primarily by hepatocytes in the liver but also in other tissues such as skin, lung and kidney. 50 Haptoglobin has also been found to be produced locally in inflamed joints in patients with juvenile idiopathic arthritis 51 and in affected joints in arthritic rats. 43 The systemic or local origin of haptoglobin is beyond the scope of this work, but should be further investigated.
Body weight was found to be only slightly, though significantly, affected by the administration of buprenorphine. Mice receiving buprenorphine lost on average 1.32% of their body weight during the study period whereas mice receiving saline gained on average 0.4% of their body weight. This difference in body weight change between groups was most pronounced at 24 h post injury. Buprenorphine has previously been described as having a negative effect on postoperative body weight in rats 52 and as decreasing food consumption in normal non-operated rats.53,54 Pica behaviour (illness response of rats similar to vomiting in other species) has been observed in normal non-operated rats and rats undergoing laparotomy.55,56 Thus, a possible cause of the slight weight loss observed in the buprenorphine-treated mice in our study could be decreased food consumption resulting from buprenorphine-induced nausea, but has to be placed in the perspective of the pain management achieved through the analgesic protocol.
The plasma levels of IL-6, G-CSF and KC were significantly elevated in both the buprenorphine and saline-treated groups compared with baseline. These results demonstrate an acute systemic response to a local knee bleed in haemophilic mice.
IL-6, G-CSF and KC were also significantly elevated in the synovial fluid of both groups compared with baseline. Additionally, IL-1β, IL-4, IL-12(p70) and MCP-1 were significantly elevated in the synovial fluid but not in the plasma of both groups compared with baseline, suggesting an additional local response to the induced knee bleed.
This is in agreement with an earlier study where elevated levels of IL-1β, IL-6, KC and MCP-1 were found in the synovial fluid of haemophilic mice suffering from an experimentally-induced knee bleed. 6 In this study IL-12(p70) and G-CSF were not evaluated but IL-4 was found to be below the detection limit in all samples (24 h post injury). In the current study the inflammatory response was found to peak at 6 h post injury. Furthermore, we optimized procedures to inhibit degradation of cytokines by proteases during the elution procedure, which may explain the determination of increased IL-4 levels in the current study.
We found eotaxin to be significantly increased in both plasma and synovial fluid after buprenorphine treatment though no difference was seen between the saline and the baseline groups. Furthermore, a significant difference in plasma G-CSF levels between the buprenorphine and saline groups was observed. This may suggest an induction of eotaxin and G-CSF of buprenorphine in this inflammatory model, but the substantial variation in the groups has also to be considered. No such data have been published before and these findings have to be further explored.
We hypothesized that buprenorphine should not significantly alter the inflammatory response in the knee bleeding model in haemophilic mice. This was confirmed, and only minor differences were determined. A slight tendency for the buprenorphine group to have higher levels of measured cytokines compared with the saline group was determined; all plasma cytokines and 16 synovial fluid cytokines were higher in the buprenorphine group. However, these differences were not statistically significant for the majority of the cytokines and only for plasma levels of eotaxin and G-CSF and for synovial fluid level of eotaxin were these differences significant.
This is in agreement with a previous study in a rat burn model, where the impact of buprenorphine was investigated on six different cytokine plasma levels. Twenty-four hours after injury and treatment plasma IL-6 levels were found to be significantly elevated in the buprenorphine-treated group. Furthermore, a non-significant increase in IL-1β, CINC-2, TNF-α and MCP-1 plasma levels were determined in the buprenorphine-treated group. 42 By contrast Hugunin et al. found no effects of buprenorphine on plasma levels of IL-1β, IL-6, TNF-α and IL-10 in a mouse cecal ligation and puncture model 12 h after treatment 40 and Martucci et al. found no effects in normal mice (no inflammatory condition) on IL-2 and IFN-γ levels 60 min, 1, 3 or 7 days after buprenorphine treatment. 41
Thus it is possible that the effect of buprenorphine is dependent on the underlying inflammatory condition. This is supported by unpublished data from our laboratories measuring the inflammatory response in F8-KO mice subjected only to a subcutaneous injection of buprenorphine or saline. This study showed that all eight measured cytokines (IL-1β, IL-2, IL-4, IL-5, IL-10, GM-CSF, IFN-γ and TNF-α) in plasma were lower in the buprenorphine-treated mice compared with the saline-treated mice 6 h after injection. For IL-1β, IL-4, IL-5 and GM-CSF this difference was significant (P < 0.05).
Another explanation for the differences in the effects of buprenorphine on cytokine levels between studies could be caused by the difference in time from buprenorphine administration to sampling for cytokine measurement. Thus, opioids have previously been found to affect cytokine production in a highly time dependent manner in rats 57 as well as in mice. 58
In the current study the inflammatory response peaked at 6 h post injury, and a difference between the buprenorphine and saline groups was most pronounced at this time point and formed the basis for the evaluation.
Comparison between studies is always challenging, and should also take into account the impact of different analgesic protocols. This should be further evaluated in future studies.
The general finding of a tendency towards slight upregulation of the inflammatory response after administration of buprenorphine in the knee bleeding model in haemophilic mice may be explained by several factors. Mice not receiving analgesia are believed to experience pain, which is known to have a potential impact on inflammatory response. However, most literature has demonstrated that pain increases the inflammatory response.59,60 Hence, it seems unlikely that the inflammatory response should be upregulated in the buprenorphine group. Another explanation could be that mice not receiving analgesia exhibit decreased activity and less use of the affected leg due to the pain experienced. A study of immediate loading after ACL reconstruction in rats found an increased number of inflammatory cells at the healing surface in rats subjected to mechanical loading on the affected joint compared with rats subjected to immobilization of the affected knee joint. 61 Additionally, in dogs with acute joint injury, full weight-bearing resulted in significantly increased TNF-α in synovial fluid compared with minimal weight-bearing. 62 Hence, the pain relief in the current study may lead to increased activity and weight-bearing impact at the injured knee, which may then explain the slightly increased inflammatory response in these animals.
Finally, it could be speculated that buprenorphine acts on cells in the central nervous system to elicit cytokine release. This has been described for morphine where a significant release of pro-inflammatory cytokines was observed when the spinal cord section was incubated in vitro with morphine. 38 Similarly results were seen in vivo when rats were administered with an intrathecal injection of morphine or methadone once daily for seven days. 38 The significant increase in pro-inflammatory cytokines seen in several models after morphine administration36–39 is not observed after buprenorphine administration in this model, suggesting that buprenorphine does not possess the same pro-inflammatory properties as other opioids. However, the tendency for a slight upregulation of the inflammatory response in the buprenorphine group was diminished after 24 h, and a possible effect of buprenorphine on the inflammatory response is therefore only considered to be brief.
In conclusion we have demonstrated that buprenorphine does not significantly modulate the inflammatory response overall, as measured by changes in body weight and joint diameter, white blood counts, haematocrit, platelet concentration, haemoglobin, VBS, plasma and synovial fluid level of 23 different cytokines as well as plasma haptoglobin level in this haemophilic mouse model, and hence the use of buprenorphine for pain management in the knee bleeding model should be continued.
These data should prompt further investigation in other inflammatory in vivo models and influence ethical and 3R considerations when working with these models.
Footnotes
Acknowledgement
This work was carried out at the Translational Haemophilia Pharmacology Department at Novo Nordisk A/S, Novo Nordisk Park, 2760 Måløv, Denmark.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
