Abstract
The use of appropriate analgesia in laboratory mice may be suboptimal because of concerns about adverse events (AE). Target Animal Safety trials were conducted to determine the safety of an extended-release suspension of buprenorphine. Drug or control suspensions were injected subcutaneously in surgically-treated BALB/c mice anesthetized with ketamine–xylazine to mimic post-operative conditions in which the compound might commonly be administered. Single and repeat five-fold (5×) excesses of the 3.25 mg/kg intended dose were used to provoke potential AE. Trials included prospective measurements of weight changes, blood chemistry, hematology, and histopathology. Clinical and histopathology findings were similar in drug-treated and control mice in a four-day trial using a single 16.25 mg/kg, 5× overdose of the drug. In a 12-day trial, which used a total buprenorphine dose of 48.75 mg/kg, clinical and histopathology values were also similar in control and drug-treated female mice. In the male arm of the repeat-overdose trial, two of eight mice died on the morning of day 12, three days following the third 16.25 mg/kg overdose administration. Histopathology did not reveal a cause of death. In a 14-month trial using a single 3.25 mg/kg dose of the drug, no significant findings identified potential AE. These findings indicate a high tolerance to an extended-release buprenorphine suspension administered post-operatively in mice with appropriate husbandry.
Analgesia for the management of any pain in laboratory animals is a scientific standard. Yet questions remain about the use of post-surgical opiate therapy in rodents. 1 Management challenges associated with providing repeated subcutaneous (SC) injections to surgically-treated small animals create one set of issues. The effective duration of SC morphine in mice and rats is limited to 2–3 h; buprenorphine is effective for only 3–5 h in mice and 6–8 h in rats. 2 Repeated injections stress small animals and consume significant personnel time. A second controversy is based on potential side-effects and whether adverse events (AE) could confound research outcomes. Concerns about AE generally focus on two side-effects common with opiate therapy: respiratory depression and nausea. Specific questions have persisted about the effect of opiates on weight gain and gastric distress in laboratory rodents. 3 Rates of post-surgical weight gain frequently serve as markers for recovery. 4 While appetite suppression is common in the surgical recovery process, there are reports of morbid gastric stress in opiate-treated rodents secondary to pica. 5
Growing evidence indicates that management issues can be addressed by the use of long-acting analgesic implants,6,7 and, perhaps, by using food-based pharmaceuticals.8–10 Clark and coworkers have demonstrated that the morbid effects of pica in buprenorphine-treated rats are exacerbated by wood chip bedding and could be mitigated by appropriate husbandry. 11 We hypothesized that an extended-release suspension of buprenorphine could be used safely in mice housed on soft contact bedding. Buprenorphine has a ceiling effect on respiratory depression and there is considerable evidence that its use in veterinary medicine is safe.12–14
To examine this hypothesis, surgically-treated male and female mice were challenged with significant overdoses of a proprietary buprenorphine suspension and monitored for AE according to US Food and Drug Administration (FDA) guidelines for assessing the safety of veterinary pharmaceutical products. 15 Similar guidelines are used by European regulatory authorities to determine the effects of drugs on major organ systems in target animal species. 16 Hematology and serum chemistry values were measured in mice treated with a surgical procedure using ketamine–xylazine (KX) anesthesia and challenged by single and repeat trials using one to five-fold doses (3.25–48.75 mg/kg) of an injectable buprenorphine suspension. Body weights, organ weights, and histopathology were performed on all mice. All mice were subjected to a surgical procedure (see Materials and methods). KX anesthesia was used based on 20 years of experience of the Johns Hopkins Hunterian Laboratories in implanting long-acting drug delivery systems in mice and rats.
The results of these trials demonstrate that an extended-release lipid–buprenorphine suspension can be used safely in male and female (20–22 g) adult BALB/c mice for analgesia following a surgical procedure. We believe this is the first study of a drug specifically designed for the management of pain in mice.
Materials and methods
Animals
Studies were approved by the Johns Hopkins University (JHU) Institutional Animal Care and Use Committee. The study was carried out in the JHU Department of Molecular and Comparative Pathobiology (Baltimore, MD, USA). Male and female BALB/c (6–8 weeks old and weighing 20–22 g) mice were obtained from Charles River Laboratories (Wilmington, MA, USA). The mice were inspected for general health conditions before being housed at a density of 4–5 mice per cage in Smart Bio-Pak cages (Allentown, NJ, USA) with Tek-Fresh bedding (Harlan, Madison, WI, USA), and were allowed free access to Teklad Global Rodent Diet chow (Harlan) and Baltimore City water. Mice were held for 14 months to enable complete studies using older mice. A total of 36 female and 36 male mice were used for the four-day, 12-day, and 14-month studies described below.
Study design
The study design was based on Target Animal Safety (TAS) protocol guidance to determine the safety of a generic drug. 17 The bioequivalent target range was selected from published reports demonstrating that buprenorphine blood levels greater than 0.5 ng/mL produce positive tail-flick responses in mice, 18 thermal latency in dogs, 19 and responses in human volunteers. 20 In a series of dose finding studies, male and female mice were injected with increasing doses of a lipid–drug suspension containing up to 25 mg/kg buprenorphine. A dose of 3.25 mg/kg, which afforded blood level concentrations of more than 1 ng buprenorphine/mL for at least two days, was selected for the TAS study.
In TAS studies, the mice were housed one per cage. For statistical purposes the experimental unit was the cage. No significant differences in weight, hematology, clinical chemistry, and clinical observations, were observed in the male and female mice dosed at 0 (control), 3.25, and 9.75 mg/kg in pilot studies. Thus, safety studies were conducted with five-fold excesses of the intended dose. The study period was four days, being one day more than the three-day elimination period. Eight male and eight female mice per group were used in the first TAS study comparing a 0× (control) and a 5× dose (16.25 mg/kg) challenge. Eight male and female mice per group were used in the second TAS study comparing 0× and 5× doses repeated at three four-day intervals. In a 14-month study, four male and four female mice were injected with a 1×, 3.25 mg/kg dose of the drug. No vehicle controls were used in this trial. Parameters evaluated in the four- and 12-day trials included body weight, hematology, clinical chemistry, clinical observations, and gross and histopathology. Daily clinical observations were not performed in the 14-month trial. The hypothesis tested was that the data for these parameters would be different in mice with 0× and 5× doses of such drugs.
Trial structure
Single 0 and 5× doses, four-day trial: In the four-day trial, eight mice of each gender were anesthetized, subjected to a sham surgical procedure, and dosed on day 0 with single 0 and 5× doses (16.25 mg/kg) of the drug, or the same volume (0.250 mL) of the control preparation. Repeat, three 0 and 5× doses, 12-day trial: In the 12-day dose-repeat trial, eight mice of each gender were anesthetized, subjected to a sham surgical procedure, and dosed with 0 and 5× doses (16.25 mg/kg) of the drug, or the control suspension, on days 0, 4, and 8. Single 1× dose, 14-month trial: In the 14-month trial, four mice of each gender were anesthetized, subjected to a sham surgical procedure, and dosed on day 0 with a single 1× dose (3.25 mg/kg) of the drug. No vehicle control was used in this trial.
The mice in the single-dose and repeat-dose trials were evaluated by daily clinical observations for signs of distress by an observer female veterinarian who was blinded to the treatment groups. At the midpoint of the first two trials, day 2 or day 6, half of the mice were weighed, euthanized, and then exsanguinated to collect blood for hematology and clinical chemistry testing. At the endpoint of each trial, day 4, day 12, and month 14, the remaining mice were euthanized to measure body weight, hematology, clinical chemistry, and anatomical pathology.
Coagulation studies: A 12-day dose-repeat trial with eight control and eight female mice treated with a 48.75 mg/kg dose, the highest dose to be tested, was conducted to furnish sufficient blood for coagulation studies. Mice in this study were not examined by histopathology or daily clinical observations.
Drug and control preparations
The cholesterol–buprenorphine drug powder was supplied by Animalgesic Laboratories Inc (Millersville, MD, USA). The drug powder contained United States Pharmacopeia (USP) grade buprenorphine HCl (Noramco, Wilmington, DE, USA), cholesterol, and glycerol tristearate, (Sigma, St Louis, MO, USA). Drug preparations were verified for purity and content by AAI Pharma (Wilmington, NC, USA). Negative control, drug-free, powder was prepared by dry, tumble blending a mixture of cholesterol and glycerol tristearate (96/4, w/w) for 48 h at 5℃.
Drug delivery
Injectable suspensions of the drug powder and the control were prepared by suspending 80 mg of powder per milliliter of medium chain triglyceride (MCT) oil (Miglyol 812; Sasol, Hamburg, Germany) followed by a brief shaking to make a homogeneous suspension containing approximately 4 mg of powder per 0.05 mL of MCT oil. Suspensions were generally prepared within 1–2 days of use and stored at 2–8℃. A single (1×) dose consisted of a 0.05 mL drug suspension. In the study comparing 0 and 5× doses, the control groups were given 0.25 mL of the control suspension. One-milliliter syringes with 1 inch 20 gauge needles were used to inject suspensions (described below) of the cholesterol–triglyceride–buprenorphine powder and the cholesterol–triglyceride control powder.
Buprenorphine blood concentration measurements
Blood concentrations of buprenorphine were measured by an LC–MS/MS method (described below). Eight-week-old male and female mice (24 per sex) and housed three per cage were anesthetized and injected with a 3.25 mg/kg dose of the drug. Cages were changed daily to prevent re-dosing by coprophagy. Because the LC–MS/MS assay required 0.25 mL of plasma, mice were euthanized at the time points reported in ‘Results’. Blood samples were collected at 6 h and subsequent 23–25 h intervals after the time of the SC analgesic injection. Day 0 values were obtained from untreated male and female BALB/c mice weighing 20–22 g.
Surgical procedure
The surgical procedure used was based on the procedure used to implant Alzet mini-osmotic pumps in mice and rats. A video of the procedure, which is briefly described below, is available at the Alzet website. 21
Mice were given intraperitoneal (IP) anesthesia with a solution containing 25 mg/mL ketamine plus 2.5 mg/mL xylazine and 14.25% ethanol in saline. The dose of anesthesia was 0.15 mL/20 g per mouse.
When anesthesia was established, approximately 1 cm2 of mid dorsal skin, away from the interscapular area, was shaved, washed with ethanol, and then coated with Betadine. Mice were transferred to a procedural table that was cleaned with 70% ethanol solution and covered with a clean disposable towel. A sterile disposable no. 10 blade was used to make a 4–5 mm incision through the skin only. Bleeding, if any, was controlled with sterile gauze and light pressure. Sterile forceps were used to separate the skin and to create an approximately 2 × 4 cm subcutaneous pocket. The skin was then apposed and stapled with 9 mm Autoclips (Kent Scientific, Torrington, CT, USA). After the skin was stapled, mice were injected with a drug or control suspension into the interscapular subcutis. All mice in the study were treated to this ‘sham surgical procedure’.
After the procedure, the mice were moved to a holding cage. This cage contained a 37℃ heating pad covered with a clean disposable towel. Each mouse was placed in a clean cage after it regained consciousness, as demonstrated by movement and the absence of signs of distress, which included, but were not limited to, sluggish movement, abnormal paw movements, efforts to scratch the incision site, and cowering in a corner of the holding cage.
Euthanasia
The mice were asphyxiated with carbon dioxide. After a one-minute absence of respiration they were weighed. The heart was exposed. The mice were exsanguinated via cardiac puncture to obtain approximately 0.8 mL of blood for hematology and clinical chemistry testing. They were then perfused through the heart with a syringe-driven 10% neutral buffered formalin.
Clinical observations
A female veterinarian blinded to the test groups observed the mice between 07:00 and 08:00 h and between 17:00 and 18:00 h daily. Observation forms were designed for the entry of ‘Yes/No’ scores, and numerical grading of signs and symptoms including respiration, tremors, motor activity, movement, nasal and ocular findings, bleeding, and surgical site erythema, edema and infection. Space was available for comments. The same forms were used for both TAS studies: the single-dose four-day observation period, and the three repeated-dose 12-day observation period. Cage conditions, motor activity, ocular findings, and the appearance of fur were observed twice daily.
Clinical laboratory tests
Blood chemistry, hematology, and histopathology were performed at the Johns Hopkins Phenotyping Core. 22 Hematology tests were performed on a Hemavet 950 Hematology System (Drew Scientific, Waterbury, CT, USA). Values obtained included white blood cell, neutrophil, lymphocyte, monocyte, eosinophil, basophil, red blood cell and platelet counts; hemoglobin concentration, hematocrit, mean corpuscular volume, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration, red cell distribution width, and mean platelet volume. A VetACE Clinical Chemistry system (Alfa Wassermann, West Caldwell, NJ, USA) was used to measure blood chemistry profiles: cholesterol (Chol), triglycerides (Tri), uric acid (UA), total bilirubin (TBili), glucose (Glu), total protein (Tpr), calcium (Ca), urea nitrogen (BUN), creatinine (Creat), albumin (Alb), high density lipoproteins (HDL), direct bilirubin (DBili) and the enzymes creatine kinase (CK), lactic dehydrogenase (LDH), alkaline phosphatase (ALP), amylase (Amy), gamma glutamyl transferase (GGT), alanine aminotransferase (ALT), and aspartate aminotransferase (AST).
Coagulation studies were performed at the JHU Department of Laboratory Medicine within 1–2 h of sample acquisition. Groups of eight female mice were injected with three doses of 5× drug suspension at days 0, 4, and 8, or control suspension as described in the repeat, three doses, 12-day trial format. Four mice from each group were euthanized at the midpoint, day 6, and at the endpoint, day 12. Exsanguination provided approximately 0.8 mL of blood that was gently mixed with 0.08 mL of citrate anticoagulant solution.
Body and organ weights
The mice were weighed in procedure rooms with a calibrated Taconic mouse scale (Taconic, Hudson, NY, USA) before they were assigned to a treatment group and no more than 24 h before they were injected with one or more doses of the drug or control suspensions on day 0. All subsequent weights were measured blind to the dose group. Mice scheduled for euthanasia were sedated with carbon dioxide and weighed on an electronic Ohaus micro balance (Ohaus, Parsippany, NJ, USA). Tissues were weighed on an Ohaus micro balance.
Histopathology
Summary of experimental design: histopathology phase.

: male;
: female.
Tissues evaluated by light microscopy.

Lymph nodes included submandibular superficial cervical collected with salivary glands from the neck; mesenteric and pancreaticoduodenal collected with mesentery and pancreas.
Parathyroid glands were evaluated when present in the plane of section of the thyroid gland.
Two male mice in the 12-day repeat 48.75 mg/kg dose trial were found dead on day 12 and were not perfused. Their tissues were collected and examined as for the other animals. After trimming, the tissues were processed, embedded in paraffin, sectioned, mounted on glass slides and stained with hematoxylin and eosin (H&E). Tissues listed in Table 2 were evaluated by light microscopy.
Statistics
Analyses of treatment group blood concentrations were made using GraphPad Prism Software version 5.04 (La Jolla, CA, USA). Microsoft Excel version 2007 was used to generate average and standard deviation (SD) data of hematology and clinical chemistry values.
Graham StarTox version 3.1.0 software was generously made available (Druquest International, Leeds, AL, USA) to manage histology data.
Results
Blood concentrations of drug
Buprenorphine blood concentration in mice treated with a single dose (3.25 mg/kg).
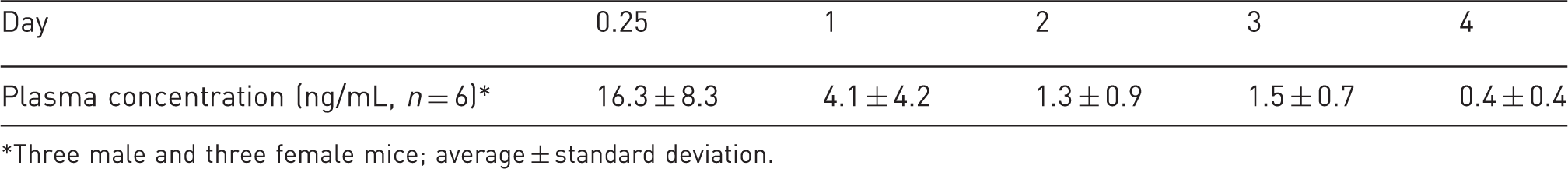
Three male and three female mice; average ± standard deviation.
Clinical observations
Approximately 2640 clinical observation entries were recorded for the mice in the four-day trial, and 7920 entries were recorded for the mice in the 12-day trial. There were no significant differences in signs of pain or distress between male and female mice in the drug treatment and control groups in either trial.
Body weight
Mean body weights in the 12-day dose-repeat Target Animal Safety trial, 48.75 mg/kg total dose.
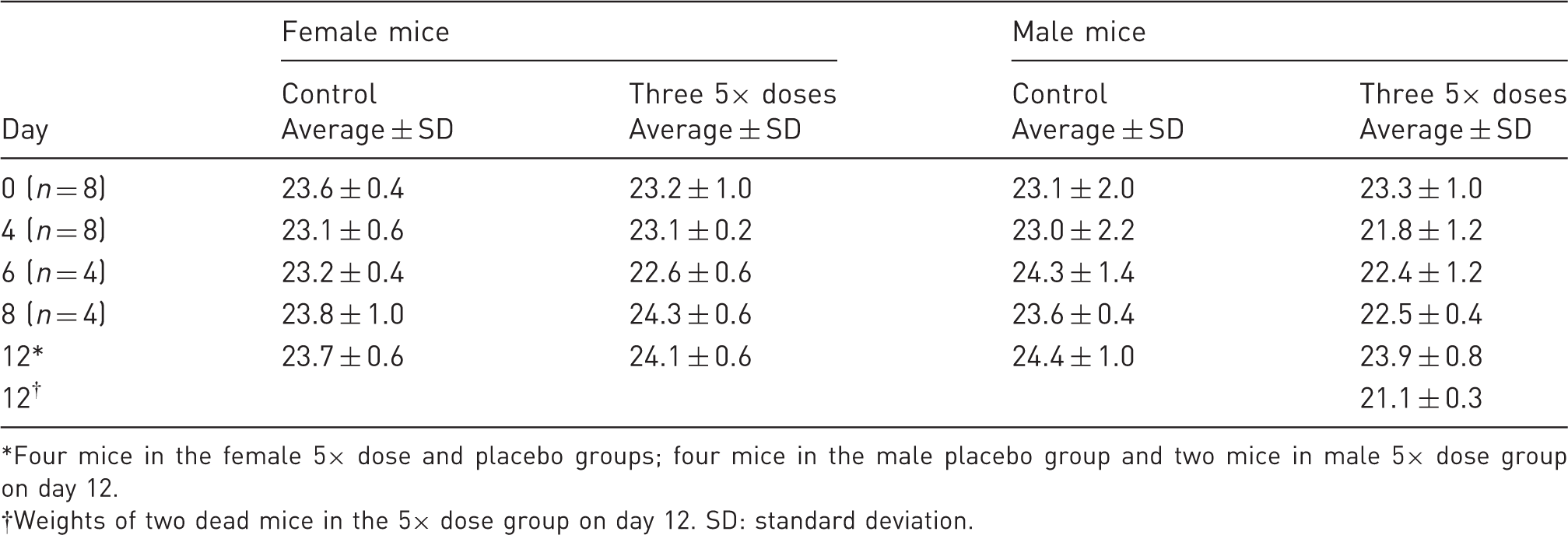
Four mice in the female 5× dose and placebo groups; four mice in the male placebo group and two mice in male 5× dose group on day 12.
Weights of two dead mice in the 5× dose group on day 12. SD: standard deviation.
Organ weight
Heart, kidney, and spleen weights were similar in the drug and control groups of both sexes in the single-dose four-day trial. On average, livers weighed approximately 0.1 g less in the 5× dose groups in males and females. The differences, however, did not reach statistical significance using t-tests. The P value for a treatment difference in the male group was 0.129; the value for a difference in the female group was 0.130.
Mean organ weights in the 12-day dose-repeat Target Animal Safety trial.
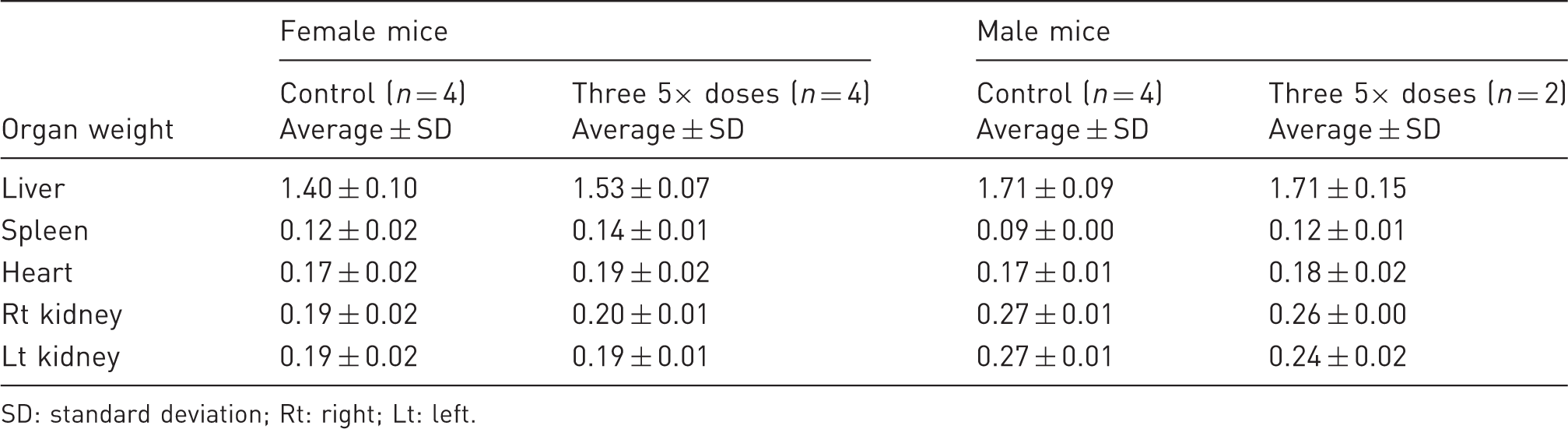
SD: standard deviation; Rt: right; Lt: left.
Clinical pathology
Summary, 12-day 3-dose (48.75 mg/kg) and 14-month 3.25 mg/kg dose challenge: endpoint hematology.
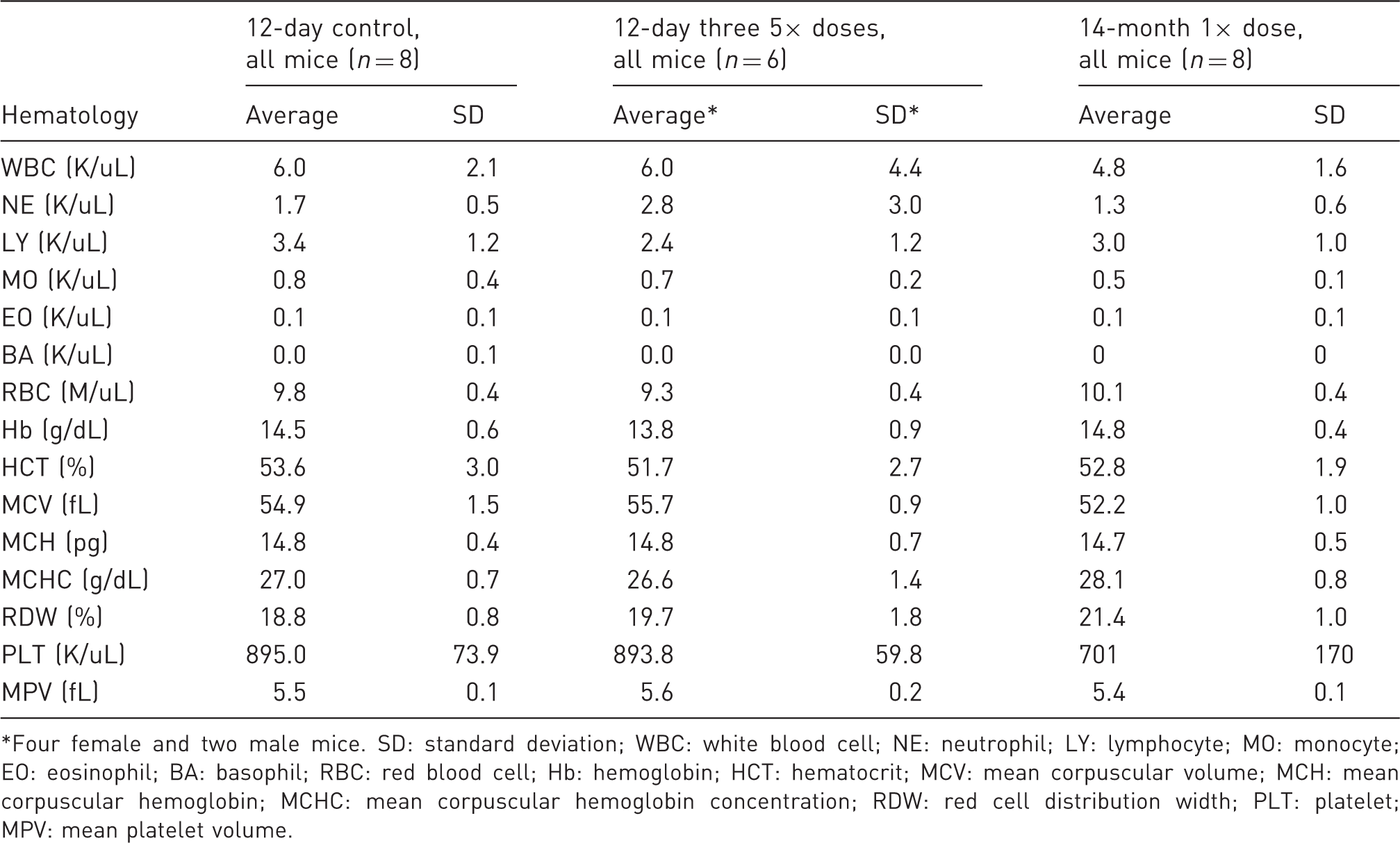
Four female and two male mice. SD: standard deviation; WBC: white blood cell; NE: neutrophil; LY: lymphocyte; MO: monocyte; EO: eosinophil; BA: basophil; RBC: red blood cell; Hb: hemoglobin; HCT: hematocrit; MCV: mean corpuscular volume; MCH: mean corpuscular hemoglobin; MCHC: mean corpuscular hemoglobin concentration; RDW: red cell distribution width; PLT: platelet; MPV: mean platelet volume.
Clotting factors and buprenorphine levels in female mice treated with three 5× doses of drug for mice.
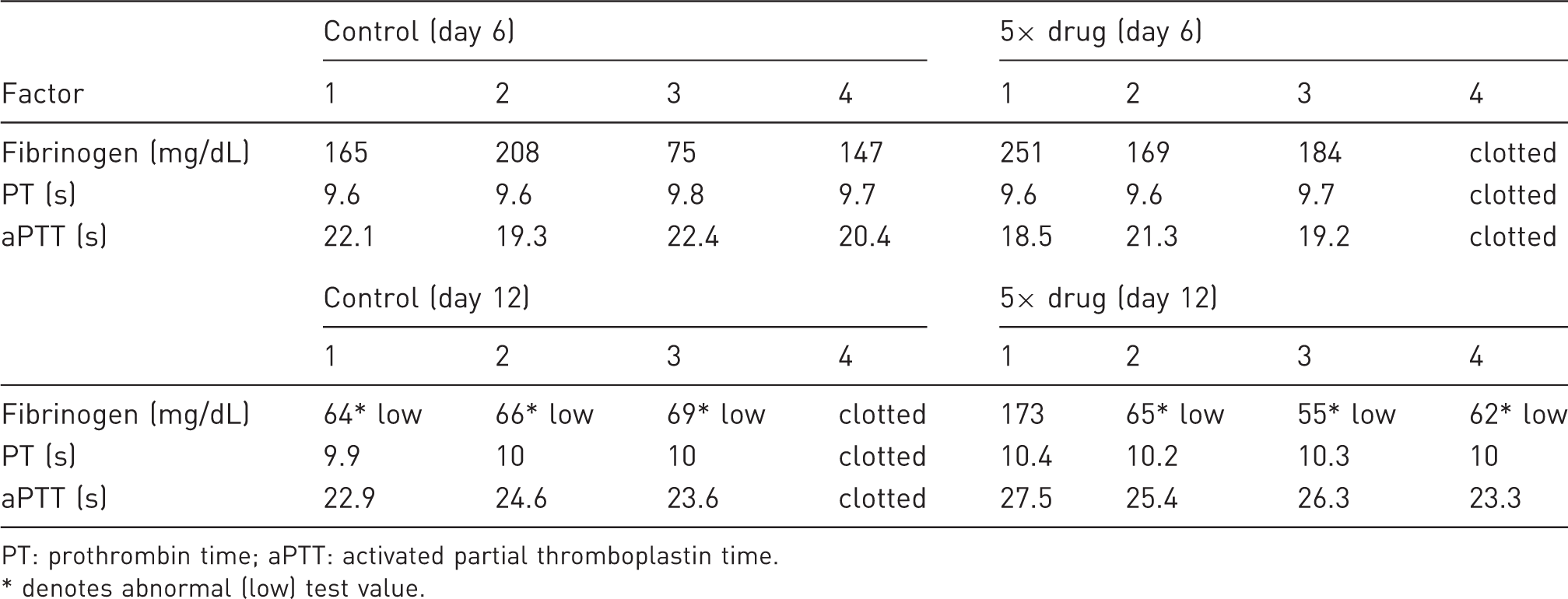
PT: prothrombin time; aPTT: activated partial thromboplastin time. * denotes abnormal (low) test value.
Twelve-day 3-dose and 14-month 1× challenge: endpoint serum chemistry.
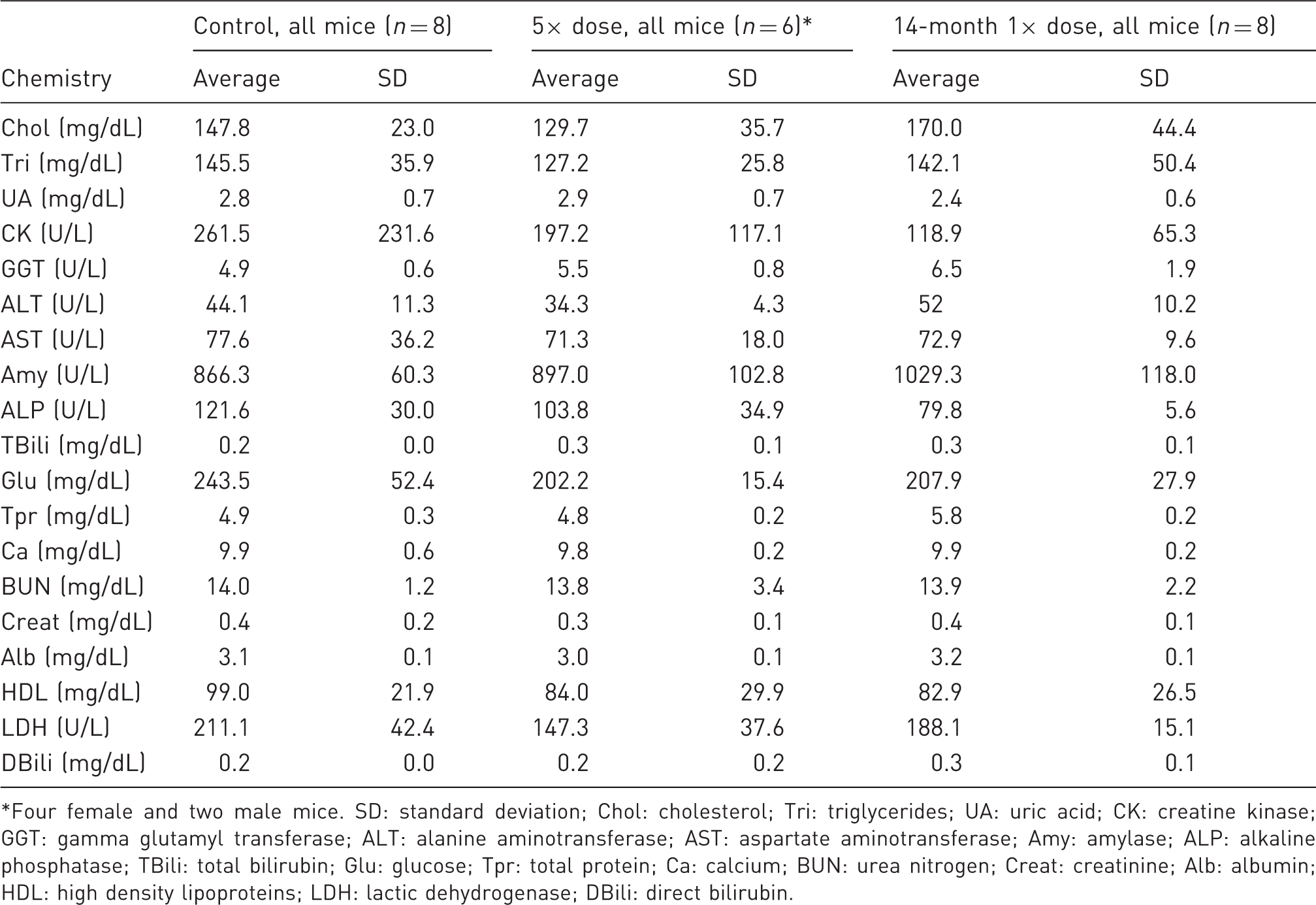
Four female and two male mice. SD: standard deviation; Chol: cholesterol; Tri: triglycerides; UA: uric acid; CK: creatine kinase; GGT: gamma glutamyl transferase; ALT: alanine aminotransferase; AST: aspartate aminotransferase; Amy: amylase; ALP: alkaline phosphatase; TBili: total bilirubin; Glu: glucose; Tpr: total protein; Ca: calcium; BUN: urea nitrogen; Creat: creatinine; Alb: albumin; HDL: high density lipoproteins; LDH: lactic dehydrogenase; DBili: direct bilirubin.
Histopathology
Day 4, single 5× dose
Minimal Kupffer cell hypertrophy was present in four of four males and in four of four females, but not in the control animals. This finding, which may represent a response to the physical nature of the drug as a suspension rather than as a solution, was considered to be non-adverse.
Macrophage infiltrates in lymph node sinuses (sinus histiocytosis) were more prominent in treated than in control animals and were also considered to be a non-adverse response to the physical nature (suspension) of the test article.
Mild lymphoid hyperplasia in mandibular lymph nodes was present in one of four males (mild) and none of the four females in the four-day single-dose control group, and was present (minimal to mild) in three of four males and in three of four females in the four-day single dose test article group. Because this occurred in the region of the incision where the drug was administered it was considered to be a non-adverse response to the sham surgical procedure.
Day 12, repeat 5× dose
No histological findings were attributed to the drug suspension article in the 12-day repeat-dose groups. By day 12, Kupffer cell hypertrophy in the liver, lymphoid hyperplasia in the mandibular and mesenteric lymph nodes and spleen, sinusoidal macrophage infiltrates in the mandibular and mesenteric lymph nodes, and hematopoietic hyperplasia in the bone marrow and spleen had generally similar incidences and severities in the control and test groups. These findings were considered to be due to the administration procedure (dorsal skin incision) of the control and the drug. Minimal or mild findings in other tissues were consistent with spontaneous findings in naïve mice, with no drug-related incidences or severities. These findings were considered to be incidental and unrelated to the drug.
Month 14, single 1× dose
When compared with the controls and the 5× dosed mice from both the four-day and 12-day trials, no histological findings attributed to the drug suspension article were identified in the 14-month single dose group. Mild histiocytic inflammation with cholesterol clefts was present at the surgical site of two single-dosed females and this was likely to have been associated with the surgical technique. Other incidental findings included a focal pulmonary adenoma (1 mm) in the lungs of an individual male and mild small intestinal amyloidosis in two females. These findings are relatively common in aged BALB/c mice and were considered to be spontaneous and unrelated to the drug.
Discussion
The present report describes the morbidity and mortality encountered in a TAS trial of an extended-release buprenorphine veterinary pharmaceutical product for moderate to severe pain therapy in surgically-treated mice. A sensitive and specific LC–MS/MS analysis demonstrated that a single, 3.25 mg/kg dose of the drug injected at the time of surgery afforded plasma concentrations of the drug of 1 ng/mL or more for 2–3 days (Table 3). These concentrations have been consistently associated with pain therapy in animal and human studies. 14 In separate studies, tests to measure efficacy using Hargreaves plantar analgesia methods showed that drug-treated female mice averaged about 15% more latency than did controls for four days. Significant latency responses were observed using tail-flick studies. Drug-treated male and female mice showed 2–3 fold slower tail-flick responses for two days. 25
Post-operative buprenorphine therapy has been associated with weight loss. 12 However, it appears possible that stress caused by repeated SC and IP injections could also play a role in suppressing appetite and weight gain. 3 Gastric distress independent of pica involving hardwood bedding may also be involved. 11 We saw no significant weight changes in control mice or mice dosed with 5× doses of the drug for at least eight days (Table 4). There were no significant weight changes in the heart, liver, spleen or kidney in male and female mice receiving 16.25 mg/kg of buprenorphine, and in female mice dosed with 48.75 mg/kg of the drug (Table 5). The necropsy studies showed no evidence of ingested bedding. The present results support the theory that stresses associated with repeated buprenorphine injections may be partially responsible for the reported weight losses. Nonetheless, the trend to weight loss in the four-day TAS trial indicates that buprenorphine itself can briefly suppress appetites in surgically-treated mice. A long-acting opiate preparation cannot eliminate concerns about weight gain. Further research may show that different surgical models and other strains of mice are more susceptible to appetite suppression. However, there is little evidence that opiate-associated weight loss in post-surgical therapy is clinically significant or challenging to research models with appropriate husbandry.
Prolonged administration of buprenorphine can decrease white blood cell counts in mice. Mice given daily IP injections of buprenorphine (300 µg/kg) for 60 consecutive days developed severe leukopenia. Leukocytes (WBC) returned to normal ranges within 45 days following withdrawal of the drug. 26 In the present study, no AE were observed in hematology studies. Blood counts, differentials, and hemoglobin measurements from the drug overdose and the carrier solution were similar in the two trials. Platelet counts and volumes were normal in mice treated three times with 5× doses of the drug (Table 6).
Additional information about the effects of the drug on hemostasis was obtained from female mice treated with repeat 5× doses of the drug in a 12-day trial. Fibrinogen levels, prothrombin time (PT), and activated partial thromboplastin time (aPTT) were analyzed at the midpoint of the trial and at day 12. Clotting factors were similar for the treated and control animals at both time points (Table 7). Although fibrinogen concentrations were abnormally low at day 12 in the control and 5× drug groups, repeated 5× challenge doses of the drug did not appear to affect coagulation parameters.
Because buprenorphine is metabolized in the liver, ALT, AST, and other liver enzyme concentrations can offer sensitive markers of buprenorphine hepatotoxicity. Average ALT and AST elevations were found in the 5× dose group of male and female mice at day 2 following the surgical procedure. Yet in the present trials the increases were within the normal range and were comparable with control data. At day 4 in the single 5× overdose trial, chemistry values were similar in the control and drug-treated groups. There was no evidence to support previous reports associating buprenorphine with adverse metabolic effects. 4 Liver chemistry values remained within normal limits in the second TAS trial (Table 8). Although two male mice in the dose-repeat group died at day 12, laboratory chemistries were normal at day 6, or two days after the mice received two 5× doses, or a total of 32.5 mg/kg of the drug in four days. Laboratory values for the two remaining male mice in the 12-day trial were within normal limits (Table 8).
Under the conditions of this study, there were no drug-related gross pathology findings. There was no evidence of skin lesions in any of the study groups. No histological findings in the tissues evaluated, including injection site skin, were attributed to the drug at 16.25 mg/kg. Average organ weight differences between the drug-treated and control mice in the four-day trial were not significant, and were not seen in the 12-day trial, which further indicated that differences observed in the four-day trial were as likely due to chance as a treatment effect. Histological findings attributed to the drug in the single-dose groups (day 4) included minimal hypertrophy of hepatic Kupffer cells, and minimal to mild inflammatory changes in all groups. Histopathology revealed that the combined volume MCT oil carrier solutions in the endpoint, day 12 mice, which totaled 0.75 mL, resulted in an MCT oil pool in dorsal SC spaces. The residual oil may account for the mild granulomatous inflammation noted in this site.
Generally mild granulomatous or pyogranulomatous inflammation, sometimes with acicular clefts and lipid-like materials attributed to injected material, was identified in all groups. Histology findings in the injection site at the skin, and increased hematopoiesis in bone marrow and spleen, had similar incidences in the control and drug groups and were considered to be responses to the dosing procedure and to be unrelated to the drug suspension. Findings in other tissues were consistent with spontaneous findings in naïve mice, and incidences or severities were not drug-related. These findings were considered to be incidental and unrelated to the drug suspension. Epicardial mineralization was present in one of four females exposed to the drug in both the four-day and 14-month single-dose trials, and in the repeated-dose groups. Based on this incidence and because it correlated with a common spontaneous finding in BALB/c mice, mineralization in the heart was considered to be incidental and unrelated to the drug suspension.
Histopathology of the two male mice that died on day 12 did not identify a cause of death. Autolysis compromised evaluation of several tissues. Histopathology findings were comparable with the non-drug treated control mice. Mortality in the 48.75 mg/kg dose group may have been related to hypotension caused by the combination of repeated KX anesthesia plus the high doses of buprenorphine. Roughan and colleagues have described a high mortality rate in rats that received weekly doses of ketamine/medetomidine anesthesia and 0.5 mg/kg SC of buprenorphine for six weeks. 27 KX combinations are widely used for anesthesia in mice and other species. The combination was selected for these trials to recapitulate surgical situations in which buprenorphine would be used for post-operative analgesia. KX combinations can induce bradycardia, cardiac depression, and hypotension. Buitrago and colleagues have tested four combinations of drugs – KX, ketamine–xylazine–acepromazine (KXA), ketamine–xylazine–buprenorphine, and ketamine–xylazine–carprofen for their ability to produce anesthesia in BALB/c mice. Induction time, anesthetic duration, blood pressure, pulse rate, and time to recovery were recorded. The KXA combination produced stable physiological parameters and was associated with the longest duration of anesthesia (40 ± 8 min); immobility was produced in all other groups (38 ± 5 min), but a surgical plane of anesthesia could not be confirmed. All KX anesthetic protocols produced significant hypotension. 28
Summary findings present with drug challenge versus control mice.
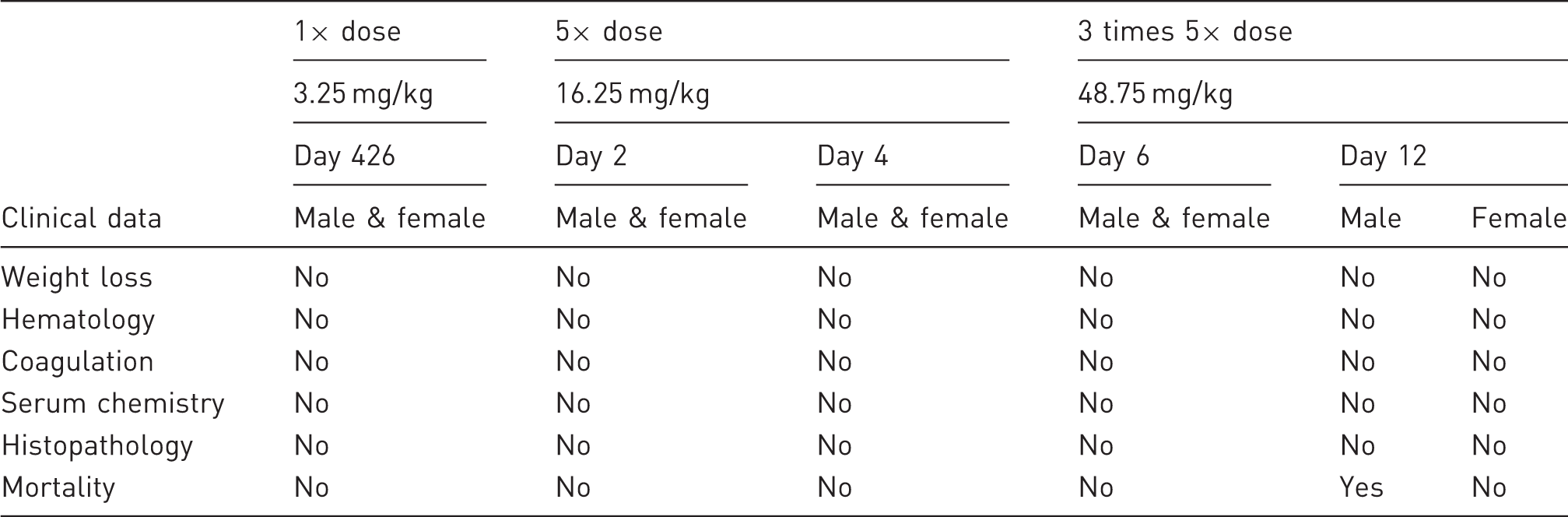
The safety profile of the extended-release buprenorphine suspension described in this report confirms previous studies of long-acting preparations of morphine in mice, 7 lipsome-encapsulated oxymorphone in mice, 6 and lipsome-encapsulated oxymorphone in rats.29,30 The data are also consistent with previous studies demonstrating that buprenorphine can be applied safely in commonly used research models.2,31–34
Footnotes
Funding and Conflicts of interest
J Romero is supported by a fellowship from Genentech organized by the American College of Veterinary Pathologists and Society of Toxicologic Pathology Coalition for Veterinary Pathology Fellows. Ward R Richter generously supplied advice and software for the evaluation of histopathology slides. C Chapleo of Reckitt Benckiser supplied buprenorphine for the early trials. Angela Wu and Gina Wilkerson helped prepare the manuscript. Funding for the present study was supplied by the Maryland Industrial Partnership, a State of Maryland fund to promote the development of products and processes through industry/university research partnerships. K Traul has served as a paid consultant to Animalgesic Labs Inc. M Guarnieri received additional funding from Animalgesic Labs Inc, and holds a significant financial interest in Animalgesic Labs.
