Abstract
Juvenile pigs are prone to hypoglycemia, nevertheless they are commonly used in preclinical studies. The optimal perioperative fluid management with crystalloid solutions in such settings has not yet been studied. The aim of the current study was to correlate the perioperative fluid administration of Ringer’s solution, with and without the addition of a minimal amount of glucose (0.4% glucose) to the blood glucose, and the incidence of perioperative hypoglycemia in 20 juvenile pigs (31.7 ± 5.2 kg) undergoing surgery for approximately one hour. Infusion of Ringer’s solution without glucose (group Ri, n = 10) led to a significant decrease of the serum blood glucose level from 105.7 ± 27.1 mg/dL at baseline compared with 87.8 ± 31.7 mg/dL post surgery. The pigs (50%) in group Ri were observed to be hypoglycemic (<72 mg/dL). The second group received Ringer’s solution containing a final concentration of 0.4% glucose (group Ri-Glu, n = 10) and showed a significant increase in blood glucose level from 104.4 ± 15.8 mg/dL at baseline compared with 122.3 ± 14.3 mg/dL post surgery. No animal in group Ri-Glu was observed to be hypoglycemic. In conclusion, we showed that even minor changes in perioperative crystalloid fluid management significantly influence blood glucose levels. In order to avoid hypoglycemia in juvenile pigs we recommend adding a minimal amount of glucose (0.4% final concentration) to the Ringer’s solution given perioperatively during surgery of approximately one hour.
Fluid management during surgery in humans, as well as in veterinary medicine, is essential to maintain intravascular fluid volume and organ perfusion. It is also used in order to balance electrolytes and to provide nutrients. Fluid imbalance can lead to critical neurological and cardiovascular incidents due to changes in pH, potassium, sodium or decreased calcium and glucose. 1 The best fluid regimen in human medicine is also still being discussed. 2 However, rational fluid and volume therapy should be goal-directed to ensure hemodynamic optimization and to meet the physical needs of patients in specific situations. 3 In pediatric surgery and anesthesia the prevention of hypoglycemia is of particular interest besides optimal perioperative volume therapy. 4
In experimental surgery, pigs are frequently used for preclinical studies as anatomical and physiological characteristics are comparable with humans for specific research questions. 5 Such porcine models are often used for preclinical studies in cardiovascular science, bone regeneration, trauma, shock, hernia repair, skin grafting and burns.6–11
For various mainly structural reasons, the usage of juvenile pigs in medical research is very common, however the optimal perioperative fluid management with crystalloid solutions in such settings has not yet been studied. Many different crystalloid solutions are currently available and used in clinical and preclinical settings. In our research laboratory plain Ringer’s solution is frequently used for perioperative fluid replacement. Ringer’s solution contains sodium (147.2 mmol/L) and chloride (155.7 mmol/L) but also calcium (2.25 mmol/L) and potassium (4.0 mmol/L). Furthermore, based on frequently observed cases of perioperative hypoglycemia, a small amount of glucose is added to this carrier solution to maintain blood glucose at a normal level in juvenile pigs. The use of pure glucose solutions (5%) is not recommended in the perioperative setting, because once glucose is metabolized, only free water remains, which distributes across all (extra- and intracellular) compartments, with negligible intravascular volume effects and a further risk of cellular oedema. In the case of accidental hyperinfusion of the mentioned concentrated glucose infusion several case reports have shown that this may lead to potentially fatal brain damage in humans.12–14 Larsen et al. have also shown in their study that the plasma glucose level of pigs is significantly affected by age and that young minipigs tend to develop hypoglycemia more frequently. 15
The aim of the current study was to compare the perioperative fluid administration of Ringer’s solution with or without the addition of a minimal amount of glucose (final concentration of 0.4% glucose) to juvenile pigs. The chosen glucose concentration was based on our daily clinical practice. We had frequently observed episodes of hypoglycemia which we had started to treat with different concentrations of glucose. We expected large perioperative variations of blood glucose levels, as may occur during surgery, but the main aim was to prevent hypoglycemic episodes with a low dose of glucose. Therefore the main study parameters of interest were the blood glucose level and the incidence of hypoglycemia. Further study parameters were changes in electrolytes and the acid–base balance.
Materials and methods
Animals
This study was carried out during surgery for the preparation of a hemorrhagic shock model in pigs, which was approved by the local Animal Investigation Committee of Vienna, Austria. Twenty juvenile male pigs (Landrace, Münichsthal, Austria), ranging from 12 to 16 weeks of age and weighing around 31.7 ± 5.2 kg, were investigated in this study. They were housed in groups of two pigs on straw. Providing optimal living conditions and animal welfare should ensure happy animals and good science 16 and is therefore considered important in our research laboratory during the whole course of any research project. The animal husbandry was controlled once a day by the veterinarian on duty (logbook recording: ear tag, gender, arrival time, driver, farmer, project, miscellaneous). All pigs were fed ad libitum with farmer’s domestic produced shredded diet (Münichsthal, Austria): 40% maize, 19% barley, 17% soya, 5% peas, 10% wheat bran, 4% Porkovit-F and 5% triticale and tap water was also available ad libitum. In addition to the three water dispensers in the stable the pigs had a plastic box (30 × 40 cm), filled with fresh water several times a day, to provide better hydration but also for secondary activity and play. The housing was enriched with rubber balls, straw and wooden sticks (size L; Abedd, Vienna, Austria). The average stable temperature was maintained between 19 and 23℃, relative humidity at approximately 55 ± 10% (daily logbook record). The stable was kept on a 12 to 12 h light to dark cycle (light was dimmable). The pigs had two days to acclimatize for surgery and before that they were kept at a farmhouse stable in groups of 10 to 15 with indoor runs. All animals were fasted overnight in order to avoid vomiting during premedication time, but had free access to water before anesthesia and surgery, which was carried out in the morning of the next day.
Anesthesia
All pigs were premedicated by injecting a mixture of butorphanol (0.1 mg/kg; Alvetra and Werfft AG, Vienna, Austria), medetomidine (0.03 mg/kg; Eurovet Animal Health, Bladel, The Netherlands) and midazolam (0.5 mg/kg; Nycomed, Linz, Austria) intramuscularly. The stable light was dimmed to the lowest mode during this procedure. In order to put the pigs into a deeper sedation, each animal additionally received intravenous ketamine (0.07 mg/kg; Pfizer, Vienna, Austria) once the auricular vein access was established. After tracheal intubation (6.5 tracheal tube) anesthesia was initially maintained with 1% isoflurane (Abbott GmbH, Vienna, Austria) inhalation. Starting after the baseline the animals were continuously infused with rocuronium (5 mg/kg/h; Organon, Oss, The Netherlands) and sufentanil (0.008 mg/kg/h; Janssen, Vienna, Austria) intravenously via the auricular vein. Until final preparation of the vena jugularis externa all animals inhaled 1% isoflurane. After the placement of the jugularis catheter (see below for procedure), anesthesia was maintained totally thereafter with midazolam (0.8 mg/kg/h), norepinephrine (5 µg/kg/h; Sanofi-Aventis, Frankfurt, Germany), rocuronium and sufentanil (doses as above) intravenously. The animals were kept in a supine position on a vacuum mattress. Using a volume-controlled ventilator (Draeger Primus, Lübeck, Germany), ventilation was carried out to obtain normocapnia with FiO2 of 30%, tidal volume of 10 mL/kg/body weight, 20 breaths/min, positive end-expiratory pressure of 3 mmHg and an end-tidal CO2 of 4.5–5.5%. Monitoring was carried out by means of a pulse oximeter (SpO2, heart rate), electrocardiogram (ECG), and temperature sensor which was placed orally. Depth of anesthesia was checked by monitoring blood pressure and heart rate throughout the experimental procedure.
Study protocol
After premedication all pigs were randomly assigned into two groups with 10 animals each: Group 1 (Ri) was given Ringer’s solution (Fresenius Kabi, Graz, Austria). Group 2 (Ri-Glu) was given Ringer’s solution enriched with 0.4% glucose (Glucosteril® 40%; Fresenius Kabi, Bad Homburg, Germany).
During a standardized surgery of approximately one hour, one litre of the study fluid was infused intravenously. The surgery consisted of open procedure of the vena jugularis externa (for intraoperative drug administration) and inserting a saline-filled perfusor catheter (Braun Perfusor Line, Melsungen, Germany) intraluminally. It was fixed with two ligatures set around the vein. The same open procedure was carried out for the arteria carotis interna (for blood pressure control) on the same side of the neck. For the placement of the urinary catheter a laparotomy of about 5 cm for open procedure of the bladder was carried out. A small incision was made into the ventral apical bladder pole and a urinary catheter (Ureofix 500 classic, Braun, Melsungen, Germany; balloon catheter brilliant size 20, Rüsch, Austria) was inserted and fixed with a purse-string suture. Neck and laparotomy wounds were closed with sutures. The urinary output was monitored and sterile urine was collected once from each pig during the procedure time, for use in urine analysis (Combur 10 Test® M; Cobas, Roche, Germany). For this purpose glucose, ketone bodies and pH parameters were recorded.
Blood samples were taken twice: before surgery (baseline, BL) and after surgery (postSurg). Blood was taken from the auricular vein and collected in a one-milliliter heparinized syringe for baseline measurements, while the pigs were sedated. Arterial blood (arteria carotis interna) was collected in the same manner after surgery. The blood analysis was carried out with a blood gas analyser including additional measurements of electrolytes, lactate and glucose (ABL 870 Flex; Radiometer, Copenhagen, Denmark). Waldmann et al. described a reference range of 72–115 mg/dL for glucose in the plasma of pigs. They also reported that blood values in pigs vary due to genetic differences (breed or strain), gender, age, weight and feeding. 17
Besides glucose, as the main study parameter of interest, secondary parameters such as pH, base excess (BE), bicarbonate (
After subjecting the pigs to an experimental protocol of hemorrhagic shock (not described) the animals were finally euthanized painlessly in deep anesthesia with an overdose of a barbiturate and T61 (Intervet GmbH, Vienna, Austria). The veterinarian on duty confirmed the death of the animals.
Statistical evaluation
Data were expressed as the mean and standard deviation (mean ± SD). Normal distribution of the data was confirmed using the Kolmogorov–Smirnov test. Based on this distribution the Student's t-test or the Mann–Whitney U-test was used to compare the group differences. A two-tailed P value of <0.05 was considered as significant. All statistical calculations were performed using the commercially available statistical software (GraphPad Prism 5; Graphpad Software Inc, San Diego, CA, USA).
Results
For all 20 pigs, surgery was carried out without any major problems and lasted 57 ± 5 min with no differences between the groups.
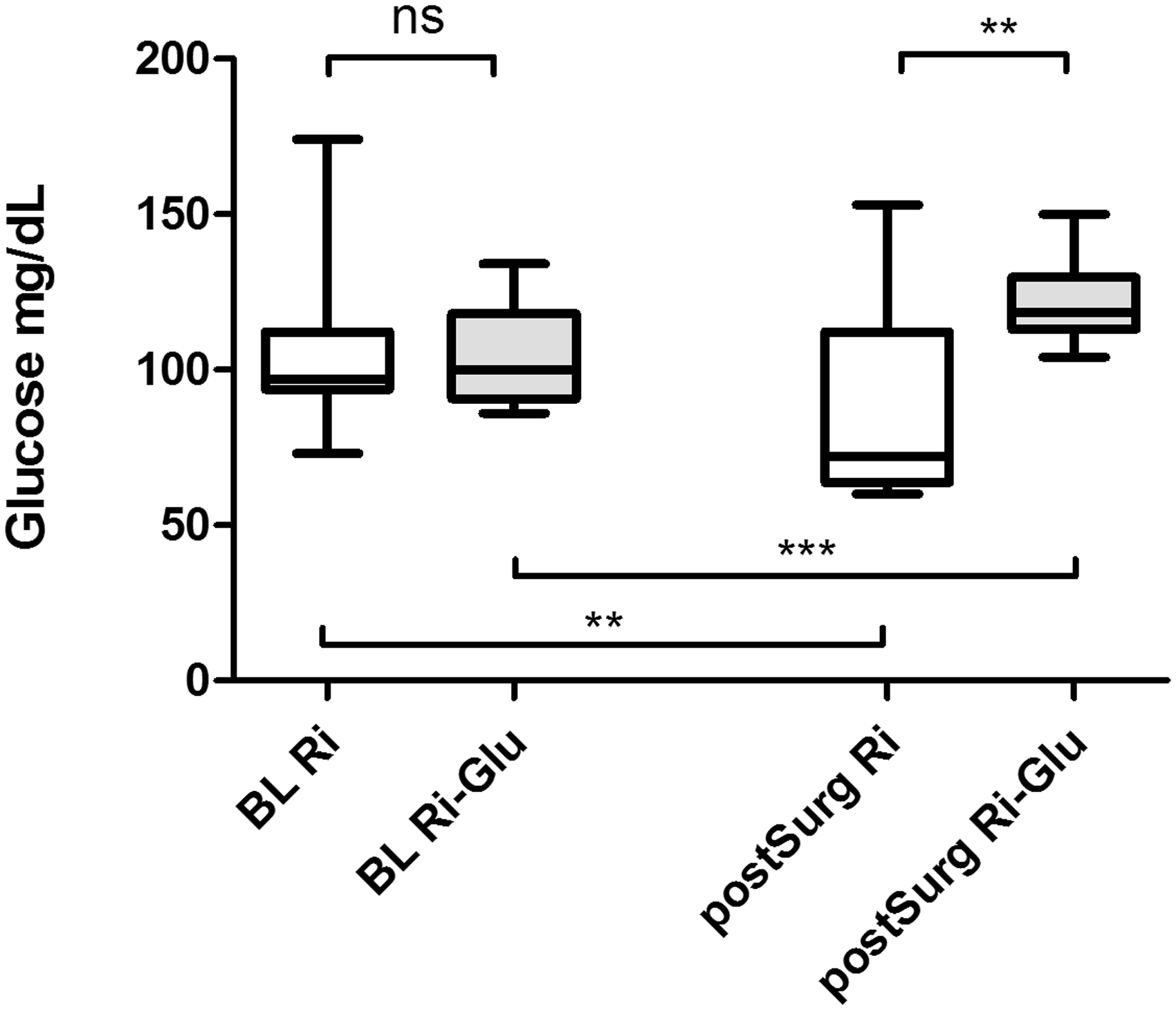
In group 1, the infusion of Ringer’s solution led to a decrease of baseline blood glucose levels of 105.7 ± 27.1 mg/dL to 87.8 ± 31.7 mg/dL (P < 0.01) after surgery. In group 2, the infusion of Ringer’s solution containing glucose, blood glucose levels rose from 104.4 ± 15.8 mg/dL to 122.3 ± 14.3 mg/dL (P < 0.001). Starting from equal baselines the two different infused fluids affected the blood glucose levels with a significant difference between group 1 (Ri) and group 2 (Ri-Glu) after surgery (P < 0.01) (Figure 1). In contrast to group 1 (Ri), where 50% of the pigs showed blood glucose levels below normal (threshold of 72 mg/dL) no pigs from group 2 (Ri-Glu) became hypoglycemic. The results from the other study parameters are shown in Tables 1 and 2. Sodium levels significantly decreased in both groups, but still within the normal range. In contrast to group 2, potassium levels significantly decreased in group 1. In both groups calcium levels increased significantly, but within the normal range.
Blood glucose levels before and after perioperative administration of Ringer’s solution with or without the addition of 0.4% glucose. Ringer (Ri) versus Ringer with glucose (Ri-Glu) at baseline (BL) and post surgery (postSurg). There is a significant rise (***P < 0.001) in blood glucose levels from baseline to post surgery in group 2 (Ri-Glu) and a significant decrease (**P < 0.01) in group 1 (Ri) from baseline to post surgery. Furthermore there is a significant difference (**P < 0.01) in blood glucose levels between Ri and Ri-Glu post surgery. ns: not significant. Electrolytes, acid–base balance and glucose for Ri and Ri-Glu at baseline. Comparison of laboratory parameters at baseline (BL) expressed as means ± SD, Ringer’s solution (Ri, n = 10) and Ringer’s solution + 0.4% glucose (Ri-Glu, n = 10). BE: base excess,
Electrolytes, acid–base balance and glucose for Ri and Ri-Glu post surgery. Comparison of laboratory parameters post surgery (postSurg) expressed as means ± SD, Ringer’s solution (Ri, n = 10) and Ringer’s solution + 0.4% glucose (Ri-Glu, n = 10). BE: base excess,


Discussion
This study investigated the effects of two different crystalloid infusion regimens in juvenile pigs during surgery, showing that the administration of plain Ringer’s solution may lead to hypoglycemia, while the addition of as little as 0.4% glucose can prevent this adverse event. The limitation on our project was the fact that only male pigs were used because of the subsequent hemorrhagic shock model, as this trauma (in humans) predominantly affects males. Further studies should be carried out using a 50:50 ratio of female to male piglets.
In the perioperative setting isotonic crystalloids containing electrolytes are generally used, but without glucose. During neurosurgery in humans the use of glucose-free electrolyte solutions is strongly recommended. Huang et al. have also reported that intraoperative hypoglycemia occurs even in adult patients undergoing long neurological surgery with this fluid regimen. 18 An increased risk for perioperative hypoglycemia has also been reported in children. 19
Hypoglycemia is a serious adverse event and there are human studies which show that severe (<40 mg/dL) and moderate (<70 mg/dL) hypoglycemia in intensive care units (ICUs) are associated with poor outcomes – even if it lasts for just a short period of time. A retrospective case–control study by Krinsley et al. showed that there is a 2.3 times higher risk of death for patients who experience hypoglycemia during their stay in ICU than for those who do not. 20 The exact pathophysiology of the increased morbidity and poor outcome is unknown. There are suggestions that hypoglycemia can cause vascular dysfunction, oxidative stress, inflammation and hypercoagulability.21,22 Furthermore even moderate hypoglycemia for a short period of time acutely increases plasminogen activator inhibitor, vascular endothelial growth factor (VEGF) and vascular adhesion molecules in patients with diabetes and healthy individuals. 22 It is also known that low blood glucose level induces metabolic acidosis and lipid mobilization. 14
Our pigs were aged about three months and were frequently observed to become hypoglycemic during surgery when no glucose was added to the infusion. Strauß et al. have reported from the pediatric population that children have an increased risk of hypoglycemic episodes, because of their reduced capacity for gluconeogenesis. 4 Furthermore Lauritsen et al. have reported that hepatic gluconeogenesis has a greater influence on anesthesized pigs than on humans. Normoglycemia in pigs cannot be maintained by extrahepatic gluconeogenesis, in contrast to humans where this is possible. 23
The prevention of hypoglycemia is of particular interest in pediatric surgery and anesthesia. In order to avoid using 5% glucose (the standard glucose solution ready for infusion), Sümpelmann et al. have recommended the use of isotonic balanced electrolyte solutions with 1% glucose for intraoperative fluid therapy in children.24,25 In the current study we observed acceptable results with as little as 0.4% glucose being added to the Ringer’s solution during surgery of one hour in pigs.
With regard to changes in electrolytes we found a significant decrease of sodium levels after surgery in all animals regardless of infusion regimen, but still within the normal ranges. This could be explained by the fact that operative stress leads to an increased release of the antidiuretic hormone (ADH) from the pituitary gland. This causes retention of free water in the kidney and production of less but more concentrated urine with a higher sodium concentration. 4 In both groups of our research project blood calcium levels rose – probably due to the calcium concentration (2.25 mmol/L) in the Ringer’s solution.
Conclusion
Our study shows that minor changes in perioperative crystalloid fluid management significantly influence blood glucose levels in juvenile pigs. When using Ringer’s solution we recommend adding glucose (0.4% final concentration) to the perioperative infusion regimen during surgery of approximately one hour in juvenile pigs in order to avoid hypoglycemia. Further studies will be necessary with a main focus on surgery of a longer duration.
Footnotes
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
