Abstract
To master the skills needed for microsurgery techniques, residents must enrol in a long and complex training program that includes manipulations on simulators, on ex vivo tissues and finally in vivo training. This final step consists of performing vascular anastomoses on murine models. We propose here a simulation program designed to decrease the number of rats used during the final in vivo training. Our study presents the materials used, the various exercises proposed and their evaluations. Two identical student groups were compared in the framework of the University Diploma of Microsurgery. Group A (seven students) followed a classic training program, all of whom achieved permeable vascular anastomoses. A total of 149 rats were needed for this group. Group B (seven students) first validated their manipulations on the task-trainer simulation program. A mean of 6 h was necessary to obtain this validation. All these students achieved the required permeable vascular anastomoses but only 77 rats were used for this group. This simulation program spared 72 rats, abiding by the Russell and Burch concept of a humane experimental technique, namely the 3R principles. This home-made, cost-efficient and easy-to-use task trainer included various exercises with increasing difficulty levels and a progressive scoring system. We believe that microsurgery training needs to include both simple and sophisticated tools in order to reduce the number of animals used to master these surgical skills.
Microsurgery is a surgical technique used by many specialists. Since Buncke and O’Brien, it has now spread worldwide and covers several surgical specialties. 1 Mastering these skills has become a prerequisite for many surgical residents. Usually, part of the training program is based on animal models. 2 Most training programs include different learning stages, i.e. manipulation on simulators such as the PVC-Rat® task trainer (Braintree Scientific, Inc, Braintree, MA, USA), training with tissue ex vivo, and then the final in vivo training phase which consists of performing vascular anastomoses on animal models. 3 Several animal models are used (pig, 4 rabbit, 5 rat 6 ) in order for students to master the surgical skills to perform vascular anastomoses under a microscope.
In the light of reducing the number of animals used for this training and of abiding by the Russell and Burch 3R concept proposed in 1959: ‘replace, reduce, refine’, 7 we propose here a simple approach with a cost-efficient and easy to build task-trainer that can be used in microsurgery labs. This device consists of sewing needles stuck into a polystyrene support. 8 Students must perform a predetermined number of exercises to master the skills they would have acquired by training on an animal model.
Material and methods
Animals
We used Sprague-Dawley male rats aged 8–12 weeks, with conventional sanitary status and housed in conventional animal facilities. During training the animals were under anesthesia and analgesia with ketamine (75 mg/kg) + xylazine (10 mg/kg) via intraperitoneal administration. During the postoperative period nalbuphine (1 m/kg) was administered subcutaneously when the surgery did not exceed 2 h. If the procedure exceeded 2 h or if the animal had already undergone two procedures it was euthanized at the end of the procedure with sodium pentobarbital 100 (via intraperitoneal injection). 9 The entire protocol was validated by the local ethics committee under the authority of the French Ministry of Agriculture in accordance with animal welfare guidelines.10,11
Participants
Details of the participants in both groups.
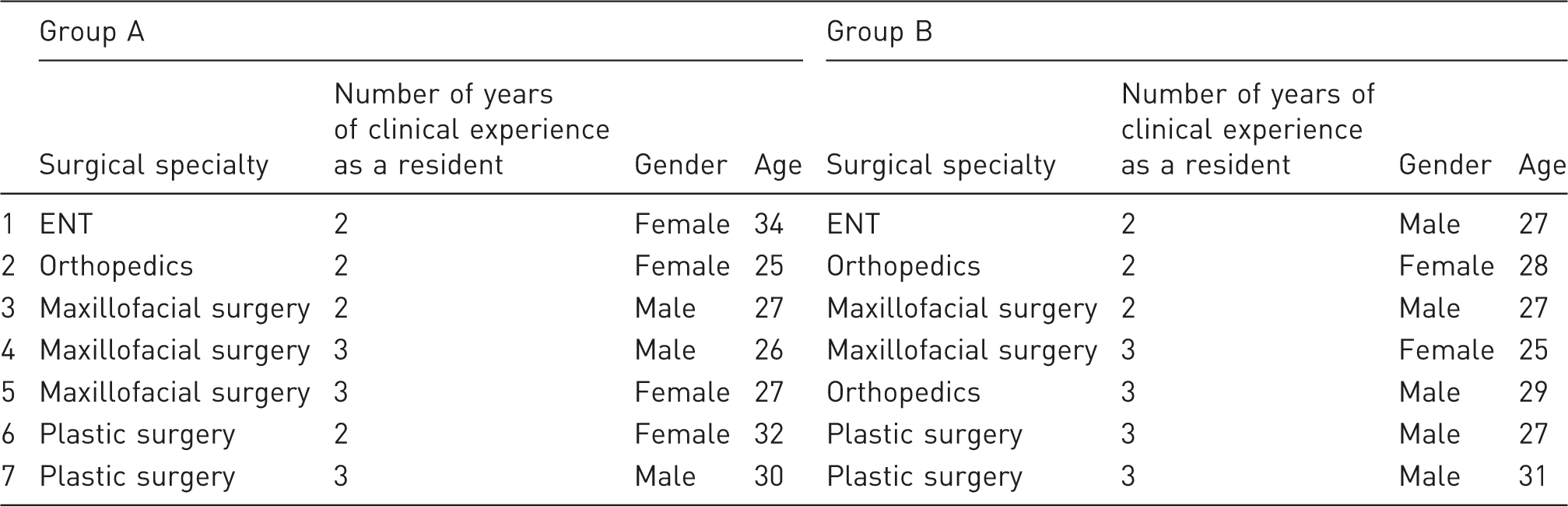
Simulator device
The task trainer was made up of sewing needles placed in a circle or double circle as reported by Dumont and Chan.8,12 This simulator enabled residents to perform different types of exercises. They had to maneuver the thread through all the eyes of the needle in a regular pattern. The needles were placed either on the outside or inside of a 3 cm circle; the various exercises are depicted in Figure 1.
Devices consisted of sewing needles placed either on the perimeter or the diameter of a 3 cm circle. Different exercises were proposed. The resident was timed when maneuvering the thread through the eyes of the needles according to a predetermined course.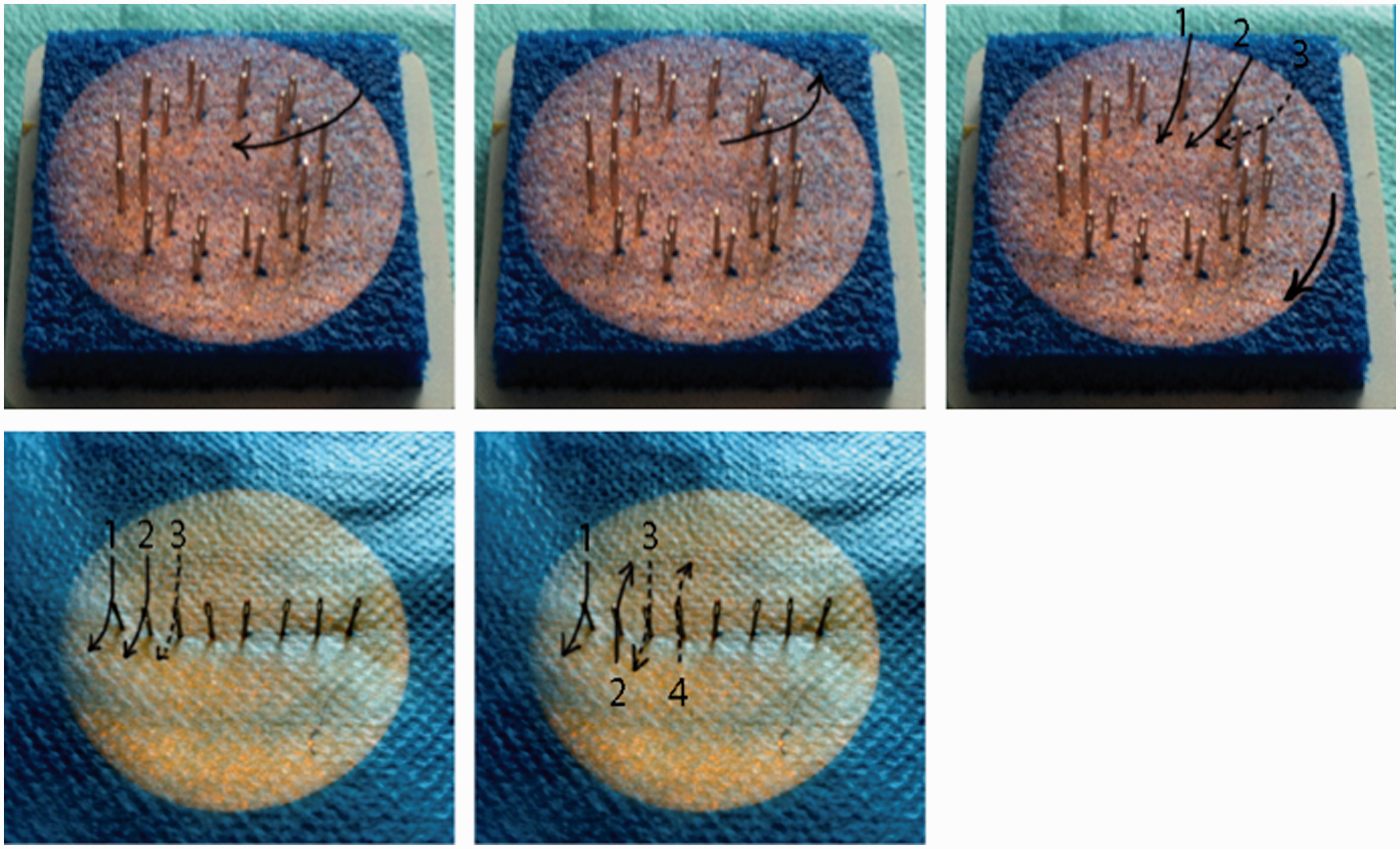
Simulation program contents
Exercises 1 and 2 consisted in maneuvering forehanded or backhanded a needle fitted with Ethilon 10/0 thread through the eyes of two needles set at 1 mm apart from one another. The time required to complete the knot and its solidity were evaluated with this scoring grid, which was voluntarily very simple in order to allow for a simple self-assessment while giving a ‘playful’ approach to the exercise.

Finally, for Exercise 5, 24 sewing needles were placed in a double circle identical to the design reported by Dumont et al. 8 The goal for the residents was to maintain a regular pattern while maneuvering the surgical needle through the 24 eyes of the needles stuck in the task trainer from the outer circle to the inner circle going clockwise (for right-handed students) or counter clockwise (for left-handed students), some parts of the exercises were performed forehanded and others backhanded. The following criteria were used to evaluate the exercise: time to complete the task, movement precision and manipulation of the surgical needle reflected by the aspect of the needle at the end of the course, i.e. the curve of the needle had to be intact.
Program validation method
Two homogeneous groups of surgical residents, enrolled in the University Diploma of Microsurgery were compared. These surgical residents came from different specialties: plastic surgery, maxillofacial surgery, ENT surgery, and neurosurgery. The groups were determined according to the residents’ level of medical training and their surgical specialty (Table 1). Group A (seven students) followed the in vivo training program consisting of performing vascular anastomoses on rats. Thirty manipulations were required to achieve seven different permeable anastomoses: carotid artery, jugular vein, aorta, vena cava, carotid and internal jugular shunts, and carotid bypass. Since this type of in vivo training program is standard in many centers, it represented the control group for our study.
A second group, group B (seven students) benefited from the task trainer simulation program before moving on to performing their seven different permeable anastomoses on rats. The main criteria for evaluating the benefits gained from the task were: the success rate in mastering the skills for these seven required manipulations on live animals as well as the quality and patency of anastomoses (tested by the examiner both in anterograde and retrograde directions and permeability was validated 30 min after unclamping the artery or vein).
Other criteria used to assess the potential advantages of this technique were: the number of rats used for each student to graduate as well as time required to complete an end-to-end anastomosis of the carotid artery.
Statistics
The results were analyzed with GraphPad Prism version 5.00 (GraphPad Software, San Diego, CA, USA). To compare qualitative data we used a Student’s t-test. The significance threshold was set at P < 0.05.
Results
From September 2011 to June 2012, 14 surgical residents were enrolled in the microsurgery training program in order to validate their diploma. Both groups were matched according to two criteria: number of years of surgical experience as a resident and surgical specialty.
In group A, all students mastered the skills needed to complete the seven required techniques. All vascular anastomoses were permeable 30 min after unclamping. Mean time to perform an end-to-end anastomosis of the carotid artery was 53 min. Overall, students needed 30 manipulations to master these skills. Mean number of rats used per student was 21.3, and mean number of manipulations per animal was 1.4.
Residents from group B benefited from the simulation training program before moving on to in vivo training on animals. To obtain the 15 stars required to complete the simulation program, students spent a mean of 6.3 h on the task-trainer device and a mean of 3.5 Ethilon nylon threads were used per student.
Adherence to the program was very strong; furthermore students gave very positive feedback and reported the exercises on task trainers as ‘fun’. The scoring grids and time needed to complete the exercises also reinforced this positive feedback. Residents deemed the exercises relevant to master the microsurgery skill needed to perform the vascular anastomoses.
These seven students all successfully performed their vascular anastomoses on rat models, and these anastomoses were permeable 30 min after unclamping. The time needed to complete the end-to-end anastomosis of the carotid artery with interrupted 10-0 Ethilon stitches was 50.5 min on average which was not significantly different from group A. Overall, 11 rats were needed per student to complete the representative anastomoses required for graduating from this microsurgery training, hence group B only sacrificed 77 rats, whereas group A used 149 (Figure 2). With this simulator program we observed a 48.3% decrease in the mean number of rats used per student. The time needed to complete the end-to-end anastomosis of the carotid artery was similar in both groups. The simulation program prepared residents in group B to perform their anastomoses in vivo while reducing the number of rats used.
In group A the mean number of rats needed per student was 21.3, whereas in group B only 11 rats were necessary for each student, i.e. a 48.3% reduction in the number of rats used.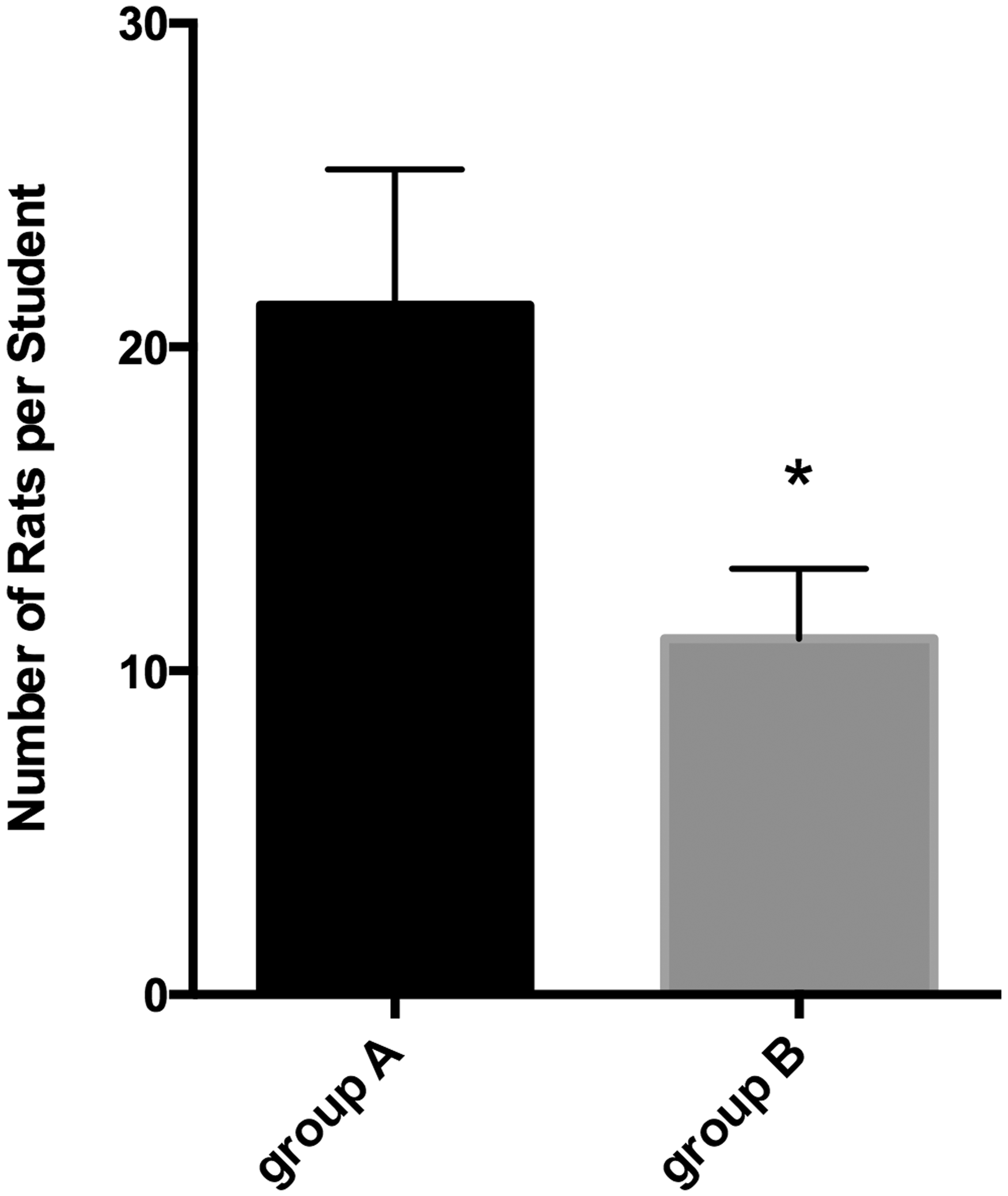
Overall 226 rats were used for this specific University Diploma meaning that 72 rats were spared that year. If this program were extended to all surgical residents in training, we could see a significant decreased in the number of animals used. We calculated that with this type of simulation program the number of rats needed for microsurgery training could decrease by 50%. This would amount to 150 rats being saved per year just for this specific Microsurgery University Diploma.
Discussion
Many surgical residents enrol in training programs to master the skills needed for microsurgery. The learning curve is slow and respecting the training steps is essential before performing any procedures on a patient. 14 Routine training procedures involve three stages: in vitro training, training with tissue ex vivo followed by training in vivo on animal models. 15 This last step remains essential yet we strongly believe that it is possible to reduce the number of animals used by designing simulation programs with task trainers.
We conducted this study during one full University Year in a microsurgery training lab using a simulation program. In fact, this very cost-efficient and easy-to-implement home-made task trainer showed a high reproducibility. Furthermore, the feedback we got from students was excellent. An average of 6 h of training was required to complete the simulation program which surprised us as being a relatively fast learning process. The fact that stars were awarded during each exercise made the use of the task trainer similar to a video game. In fact, just as with video games, residents wanted to move on to the next level. The different difficulty levels showed a soft progression curve so that residents did not stay on the same level for more than 90 min. The scoring grids were actually designed in a simple way with the student’s autonomy in mind. The use of validated scores such as the Structured Assessment of Microsurgery Skills questionnaire 16 seemed disproportionate for this type of simulation program. We deliberately chose this type of self-evaluation not related to a grade or score which appeared to us specifically adapted to task-trainer simulation programs used in microsurgery training.
This program is designed to train students to master the skills required for microsurgery while reducing the number of animals used and thus abiding by the 3 R concept of humane experimental technique presented by Burch and Russell. We estimate that this program could spare almost 50% of the rats used for this specific diploma in our Center. Students were completely satisfied with the program and its outcomes.
Simulation programs for microsurgery training are becoming readily available; 17 they could benefit from more sophisticated technologies such as numeric simulators which are already available for laparoscopic surgery or robot-aided surgery. 18 However, animal models remain essential for the ultimate training stage before performing the technical procedures in the operating room.
Footnotes
Acknowledgement
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
