Abstract
The aim of this study was to evaluate a newly developed infrared side-stream capnograph with minimal sample volume for the continuous measurement of end-tidal carbon dioxide (CO2) concentrations in small rodents. Thirty-four male Wistar rats (weight 345 ± 70 g) were treated in accordance with the National Institutes of Health (NIH) guidelines for animal care. All experiments were performed with approval of the local animal care and use committee. Sepsis was induced by implanting an 18 gauge stent into the colon 24 h prior to the experiments, allowing a constant fecal leakage into the peritoneal cavity (25 septic and nine control animals). Hemodynamic variables and end-tidal CO2 were recorded continuously and arterial blood (5 × 120 µL) was sampled periodically for arterial blood gas analysis. After baseline controlled mechanical ventilation was randomized and titrated to either normocapnia (35–45 mmHg) or hypercapnia (65–75 mmHg) with exogenous application of CO2. A total of 155 paired CO2 measurements comparing end-tidal and arterial partial pressure were conducted. Side-stream capnography underestimated the CO2 partial pressure with a bias of −6.1 mmHg and a 95% limit of agreement from 6.7 to –19.1 mmHg. Our results suggest that side-stream end-tidal CO2 monitoring with a low dead space could be utilized in rats as a surrogate for the arterial CO2 measurement over a wide range of partial pressures in normal and septic animals.
Controlled mechanical ventilation is an essential component of clinical practice. The injured lung is especially vulnerable to barotrauma, stretch injury and alveolar hypo- or hyperinflation. Therefore, controlled mechanical ventilation with a tight surveillance of ventilator settings, peak pressures and ventilation efficacy is essential not only in clinical practice but also in experimental studies.
As animal researchers progress to develop suitable animal models for investigation of critical care treatment options, new limitations arise due to technical challenges resulting from the animals’ sizes. Rats have long been accepted in experimental studies due to their availability and low cost making them ideal models for invasive interventions. However, the low tidal volumes of small animals exceed just a few milliliters and, therefore, monitoring devices that add additional dead space, like in-line capnography, jeopardize effective and efficient alveolar ventilation. In particular, mechanically controlled alveolar ventilation 1 over several hours needs to be monitored carefully. 2 While measurement of subcutaneous hemoglobin oxygenation provides easily accessible information about the adequacy of oxygenation, monitoring the efficacy of alveolar ventilation and measurement of end-tidal carbon dioxide (CO2) proved to be much more difficult. The gold standard for the assessment of ventilation has been the analysis of arterial blood gas oxygen (O2) and CO2 partial pressures. However, arterial blood gas analysis (BGA) requires heparinized arterial blood, and the sampling volume greatly depends on the analyzer used. Average samples are required to contain about 120 µL blood, and the blood volume of an adult rat with a body weight of about 200–400 g averages 6–7 mL/100 g. 3 Thus, the necessity of obtaining serial blood samples in chronic experimental models implies the potential to induce significant hemorrhagic or, if replaced with crystalloid fluids, hemodilution effects.
For continuous monitoring of controlled ventilation in small animals, an ideal monitoring setting would consist of a pressure-controlled ventilation, transcutaneous measurement of hemoglobin oxygenation and assessment of end-expiratory carbon dioxide concentration (etCO2). Side-stream capnography analyzes a fixed sample volume of expiratory air and measures CO2 content via infrared light absorption. The sample volumes required are fairly high compared with the low minute volume of rodent ventilation (we used a tidal volume of about 2 mL and a frequency of 70/min) and this effect is exacerbated with an even smaller animal size. 4 The discrepancy of sample and tidal volumes and the limited blood volume for intermittent BGA greatly restrict the controlled mechanical ventilation of small rodents over a longer period of time.
In order to develop animal models for deepening our understanding of the underlying pathophysiology and developing adequate treatment options our models need to mimic the clinical setting as closely as possible. The aim of this study was to evaluate a newly developed infrared side-stream capnograph with minimal sample volume for the continuous measurement of end-tidal CO2 concentrations in small rodents. In order to qualify for experimental use in critical care the etCO2 measurement needs to be accurate in healthy animals as well as in septic conditions with possible end-organ impairment and to cover a wide range from normocapnic to hypercapnic ventilation.
Materials and methods
A total of 155 successful paired CO2 measurements comparing end-tidal and arterial partial pressures were conducted in 25 septic and nine control male Wistar rats (weight 345 ± 70 g) treated in accordance with the National Institutes of Health (NIH) guidelines for animal care. Out of the 170 expected paired measurements, 15 could not be used for evaluation due to the death of the animals or technical problems (12 in septic animals, three in sham animals). All the experiments were performed with the approval of the local animal care and use committee.
Polymicrobial sepsis was induced by colon ascendens stent peritonitis surgery, as previously reported, 5 allowing a constant fecal leakage into the peritoneal cavity. Briefly, an approximately 2 cm long median laparotomy was performed under general anaesthesia (3.0 vol% sevoflurane, FiO2 0.5 and buprenorphine 0.05 mg/kg subcutaneously). The cecum was located and an 18 gauge stent of about 8 mm length was sutured into the colonic wall 1.5 cm distal from the ileocecal valve, ensuring a constant leakage of feces into the peritoneal cavity. The control group (sham) received the same treatment as stated above, but instead of the stent projecting into the intestinal lumen it was just sutured on the outside wall of the tunica serosa, and thus not inducing fecal leakage. After replacing the intestine in its former position, 1.5 mL sterile saline solution was applied into the abdomen, the abdominal wall was closed and the animals placed in their cages to recover from anaesthesia. In order to avoid unnecessary suffering all animals were carefully examined every 4 to 6 h after surgery, applying a scoring system approved by the local animal care and use committee. However, no animal had to be excluded from the experiments.
After 24 h the animals were anaesthetised with pentobarbital (60 mg/kg intraperitoneally), tracheotomized and placed on a controlled warming pad, ensuring a constant body temperature of 37–38℃. The volume-controlled and pressure limited mechanical ventilation (pressure limit 35 cmH2O) was set at a constant frequency of 70/min and the volume was individually adapted (approximately 2 mL) to maintain a minimum pO2 of 90 mmHg and pCO2 of 35–45 mmHg. No positive end-expiratory pressure (PEEP) was used in this experimental setting. The right external jugular vein was cannulated for a continuous infusion of 10 mg/kg/h pentobarbital, 4 mL/h Ringer’s solution and 3 mg/kg pancuronium every 2 h. The left external carotid artery was carefully isolated from its surrounding vagal nerve and cannulated for continuous recording of arterial blood pressure. One milliliter of arterial blood was extracted singularly for further analysis and 120 µL of blood for intermittent BGA throughout the experiment. After each BGA, 4 mL Ringer’s solution was administered to compensate for the volume depletion and to flush the catheters. The laparotomy was reopened and the macroscopic manifestation of the progressive peritonitis recorded. The removed blood was analyzed for white cell count and interleukin levels to assess the severity level of sepsis.
After instrumentation and 30 min of baseline ventilation with a normocapnic arterial pCO2 target of 35–45 mmHg, exogenous CO2 was added to the inspiratory air (FiO2 0.3) in the hypercapnic group to establish a target arterial pCO2 of 65–75 mmHg and minimum pO2 of 90 mmHg. Ventilator settings remained unchanged henceforth and ventilation was continued for 120 min.1
Experimental protocol
Systemic hemodynamic and oxygenation variables
Mean arterial pressure (MAP) and heart rate (HR) were measured via a Gould–Statham pressure transducer (P23ID, Elk Grove, IL USA). Arterial blood was sampled every 30 min for measurement of O2 and CO2 partial pressures, hematocrit and electrolyte balance (ABL 715, Radiometer, Copenhagen, Denmark).
Respiration
Pressure, frequency and volume provided by Inspira Advanced Pressure Controlled Safety Ventilator were recorded continuously (MA1 55-7059; Hugo Sachs Elektronik–Harvard Apparatus GmbH, March-Hugstetten, Germany).
Capnography
300 µL of end-expiratory air were sampled per breath, using the same probe of a newly developed capnograph for the continuous monitoring of the etCO2 in each rat, adding an additional dead space of just 58 µL. The capnograph was equipped with a highly sensitive infrared spectroscopy CO2 sensor and compensated for pressure and temperature changes. The sampling volume was 21 mL/min with smooth averaging over four breath cycles (Type 340, Hugo Sachs Elektronik–Harvard Apparatus GmbH).
Statistical methods
All hemodynamic and respiratory variables were continuously recorded on a personal computer after analog to digital conversion (Powerlab, ADInstruments, Castle Hill, Australia) and analyzed via Chart v 5 for Windows (AD Instruments GmbH, PowerLab®, Spechbach Germany).
Data are presented as means ± SD, Bland–Altman plot, analysis with GraphPad Prism (v 5.01, GraphPad Software, Inc, San Diego, CA, USA).
Results
The technique of exogenous application of CO2 with otherwise unchanged ventilator settings enabled the tight control of pCO2 and reliably induced hypercapnia and respiratory acidosis in all the investigated animals. The mean pCO2 was 44 ± 6 mmHg in the normocapnia group and 72 ± 7 mmHg in the hypercapnia group. All animals remained hemodynamically stable during the experiments and neither the septic nor control animals required any pharmacological support. Results on interleukin levels, white cell counts and markers of inflammation are to be reported elsewhere.
Bland–Altman analysis of the end-tidal CO2 measurements in comparison with the arterial blood gas values showed that the side-stream capnography underestimated the CO2 partial pressure with a bias of −6.1 mmHg and a 95% limit of agreement from 6.7 to −19.1 mmHg (Figure 2).
Experimental protocol. Bland–Altman analysis of end-tidal CO2 measurements and arterial blood gas analysis in all animals (n = 34).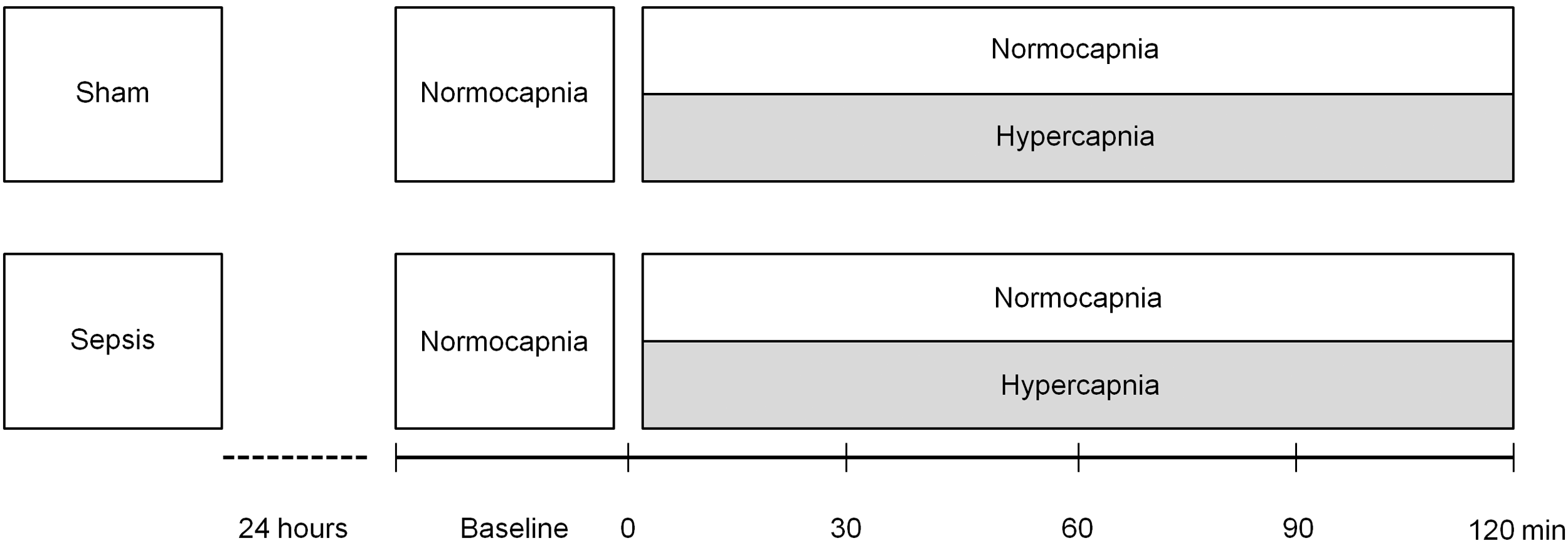
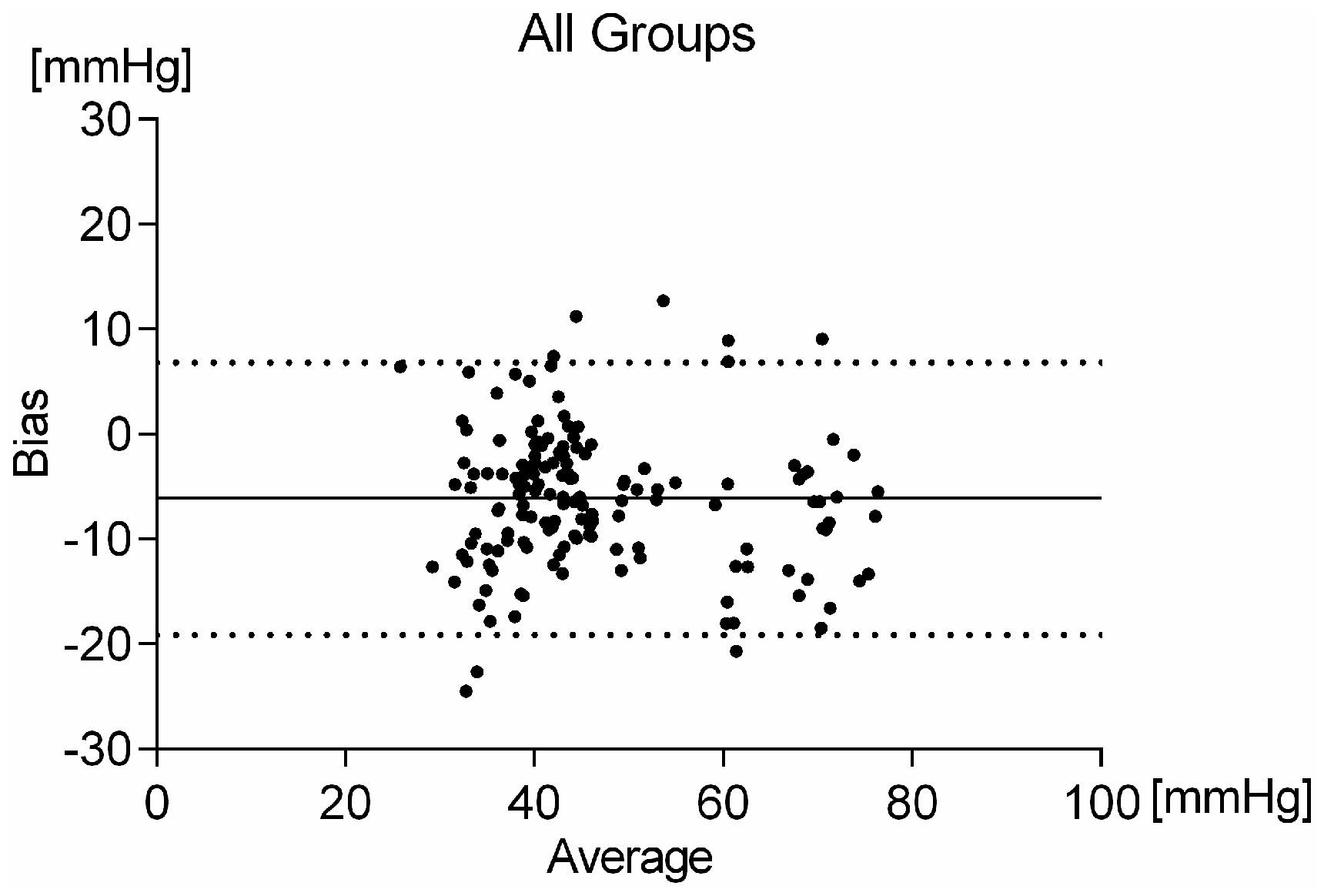
Bland–Altman analysis of sham animals revealed a bias of −4.5 mmHg, a 95% limit of agreement from 4.2 to −13.1 mmHg (Figure 3).
Bland–Altman analysis of the end-tidal CO2 measurements and arterial blood gas analysis in sham animals (n = 9).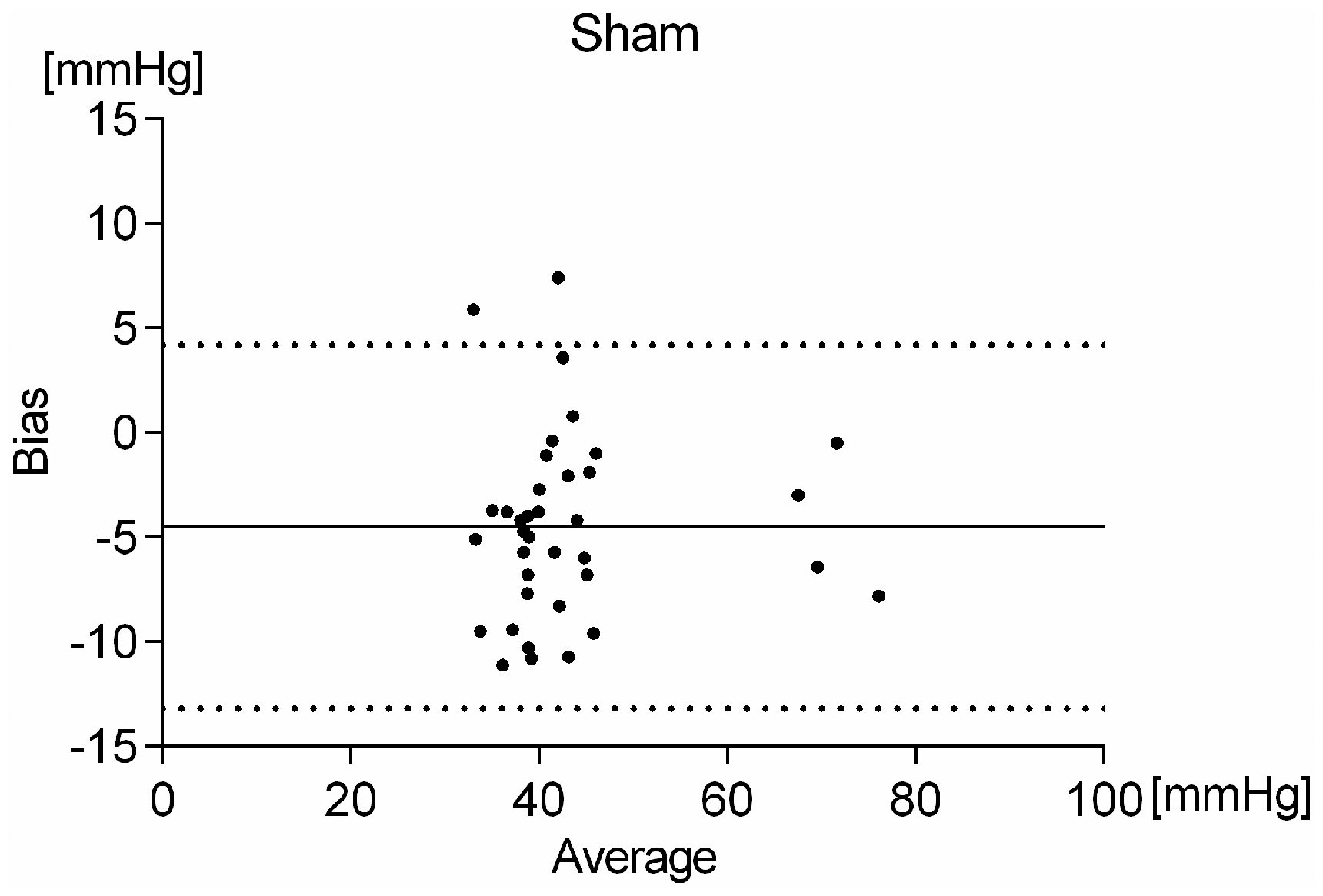
Bland–Altman analysis of septic animals showed a bias of −6.7 mmHg, a 95% limit of agreement from 7.2 to −20.7 mmHg (Figure 4).
Bland–Altman analysis of the end-tidal CO2 measurements and arterial blood gas analysis in septic animals (n = 25).
Discussion
The aim of this study was to evaluate whether the measured etCO2 of a newly developed side-stream capnography is a useful surrogate of arterial CO2. To be suitable for experimental research in small rodents, capnography needs to be reliable both in healthy and septic animals and needs to cover a wide range from normocapnic to hypercapnic ventilation.
Controlled mechanical ventilation in small rodents has been established for many years now. Yet, the monitoring of respiratory settings is essential and requires frequent and invasive arterial BGA, inducing a significant blood loss if ventilation is to be continued for several hours. Non-invasive monitoring of hemoglobin oxygenation can be achieved reliably with transcutaneous pulse oximetry. The similar technique of transcutaneous CO2 has severe limitations, such as sensor-associated thermal burns, long calibration and stabilization intervals and an overall poor correlation with arterial CO2 partial pressure 6 and has therefore only been slowly introduced into clinical practice. 7 Capnography in patients requiring mechanical ventilation has become the gold standard and an essential part of good clinical practice, measuring end-tidal CO2 concentrations and allowing continuous and non-invasive surveillance of ventilation, even in neonates. 8
However, measurement of end-tidal CO2 in the experimental setting in small rodents is difficult. Because of their low weight and thus tidal volume, a low suction flow of the side-stream capnography is necessary to prevent dilution by the surrounding air which occurs when expiratory gas flow is lower than the suction rate. This technical limitation defines the longer delay time and a distortion of the CO2 signal as a consequence of gas mixing in the tube, which can lead to a clinically unacceptable under-recording of the alveolar CO2 by side-stream capnography. 9
The major difficulty of end-tidal CO2 monitoring arises due to the high impact of additional dead space in relation to the low tidal volumes of small rodents. With a tidal volume of only about 2 mL per breath the ventilator set-up requires the reduction of dead space to ensure effective alveolar ventilation. With the small sampling volume of just 300 µL per breath the side-stream capnography used in this study could reliably assess end-tidal CO2 concentration while adding only minimal dead space (58 µL) to the experimental setting. Thus, with a more precise measurement of expired CO2 it is now possible for capnography to closely reflect alveolar and arterial CO2. levels.
In addition to the technical hurdles which need to be overcome when measuring ventilation in the experimental setting, pathophysiological changes also need to be taken into account. The pathophysiology of sepsis is very complex, but in the lung tissue inflammation and subsequent tissue damage induce increased endothelial permeability and capillary leakage. Inflammatory exudates induce surfactant dysfunction and promote alveolar de-recruitment and ventilation heterogeneity. Furthermore, diffusion distances are increased and capillary perfusion is altered by endothelial dysfunction and leukocyte activation. This complex pattern of inflammatory changes results in ventilation and perfusion mismatches and thus increases the alveolar dead space, severely impairing pulmonary gas exchange. 10 Therefore, alterations in pulmonary gas exchange properties are expected to increase the bias of end-tidal CO2 measurement due to ventilation–perfusion mismatches. 11
Our results suggest that side-stream end-tidal CO2 monitoring with a low dead space could be utilized in rats as a surrogate for the arterial CO2 measurement over a wide range of partial pressures in normal and septic animals.
Footnotes
Acknowledgement
This study was supported by the research commission of the Medical Faculty of Heinrich-Heine University Düsseldorf, Germany.
Declaration of conflicting interest
Type 340 Capnograph was developed and provided Hugo Sachs Elektronik–Harvard Apparatus GmbH.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
