Abstract
Implantable radiotelemetry methodology has been used to continuously monitor pulmonary hemodynamics including right ventricular pressure (RVP) or pulmonary arterial pressure (PAP) in conscious, untethered and freely moving animals such as mice and rats. The use of implantable radiotelemetry to monitor RVP or PAP has never previously been described in rabbits. The aim of the present study was to use implantable radiotelemetry to continuously monitor RVP in conscious adult rabbits. Telemetry transmitters were implanted in 44 adult male New Zealand rabbits using a trans-diaphragm approach for the catheter placement. RVP, heart rate (HR) and activity were monitored every 15 min for 20 s. Body mass was recorded once a week. A total of 39 (88%) rabbits were successfully implanted. Thirty rabbits survived the surgical procedure resulting in an overall survival rate of 73%. RVP, HR and activity were long-term monitored in 17 rabbits for an average period of 103 ± 15 days. Weekly body mass follow-up showed that implantable radiotelemetry did not impair the normal development of the animal. Twenty-four-hour period monitoring of RVP, HR and activity showed concomitant changes in RVP, HR and activity according to the dark/light cycle applied to the rabbits. To conclude, implantable radiotelemetry methodology can be safely used to continuously monitor RVP in conscious rabbits.
Pulmonary hypertension is a medical condition mainly affecting the pulmonary circulation. Various rodent animal models have been developed to better understand the physiopathology of pulmonary hypertension and to test potential new drugs. 1 Hemodynamic data from these animal models are usually restricted to a single measurement of pulmonary arterial pressure (PAP) and/or right ventricular pressure (RVP), in anesthetized, mechanically ventilated and often open-chest animals. 2,3 More recently, high-resolution echocardiography and magnetic resonance imaging (MRI) techniques have been developed to accurately estimate hemodynamics in small animals including rats, mice and rabbits, and to allow for serial monitoring of experimental pulmonary hypertension. 4,5 Although these techniques are non-invasive, they are performed on anesthetized animals, which implies respiratory and cardiovascular depression resulting in various artifacts. Continuous monitoring of RVP and PAP in conscious, untethered and freely moving animals was introduced by Hess et al. in the 1990s and has consequently and considerably improved hemodynamic pulmonary measurement in rodent models of pulmonary hypertension. 6 Since then, telemetry has been widely described as a method for measuring RVP and PAP in rodent models of pulmonary hypertension. 7 –10 However, the original procedure of implantable radiotelemetry methodology included extremely invasive thoracic surgery resulting in significant stress, potentially disturbing the cardiovascular system, and in delayed recovery. More recently, a trans-diaphragm surgical technique has been described in rats, avoiding any surgical invasion of the thorax, and resulting in significantly faster recovery of the animals after the surgery. 11
Although left ventricular pressure has been measured by telemetry in rabbits, 12 the use of telemetry to measure RVP in rabbits has thus far not been described. In the present study, we describe the use of telemetry to measure RVP in conscious and freely moving rabbits using the trans-diaphragm surgical approach to implant the telemetry device.
Animals
Adult male New Zealand rabbits were obtained from the animal facility (KU Leuven, Belgium). All the rabbits were housed one per cage (surface: 0.36 m2) under controlled conditions: room temperature of 18–21℃, 12 h/12 h light–dark cycle, with free access to water, standard rabbit chow once a day and dry grass once a week. The study protocol was approved by the Institutional Ethics Committee of KU Leuven.
Materials and methods
Telemetry system
The implantable radiotelemetry equipment for conscious, freely moving laboratory animals was purchased from Data Science International (St Paul, MN, USA) and included an implantable transmitter (model TA11PA-D70), a receiver (model RMC-1), a data processing device (Data Exchange Matrix) and an ambient pressure reference monitor (APR-1).The pressure catheter was a 35 cm fluid filled catheter with a terminal sensing region containing a non-compressible fluid and a plug of biocompatible gel. The sensing region relayed pressure waves to the transmitter. Calibration of the transmitter was accomplished by placing it into a sealed pressure chamber with measurements taken at 750, 850, and 950 mmHg. The output was read as a raw frequency output and was used as the calibration values for the transmitter. All data were computed using an analysis program (Dataquest 4.1). The data acquisition system, which has a sampling frequency of 500 Hz, was programmed to record the following parameters: heart rate (HR), activity, systolic and diastolic pressures. The transmitters were turned on before surgery and kept in sterile 0.9% NaCl solution (B Braun, Melsungen, Germany) until implantation. Data were continuously monitored during the transmitter implantation procedure to ensure proper placement of the pressure catheter into the right ventricle. Once the surgery was completed the transmitter was switched off for about 2 weeks to let the animals recover from the surgery. Thereafter, the transmitter was switched on and data were monitored and recorded every 15 min for 20 s.
Preoperative care and anesthesia
Preoperative analgesia was induced by an intramuscular injection of 25 mg/kg ketamine (Anesketin, Eurovet, Heusden-Zolder, Belgium) and 0.5 mg/kg medetomidine (Domitor, Janssen Pharmaceutica, Beerse, Belgium). The trachea was blindly intubated using a 2.5 mm diameter tracheal tube without balloon (Mallinckrodt Medical, Athlone, Ireland).Ventilation was performed in the presence of oxygen (100% medical oxygen, Stroombeek, Meise, Belgium) using a rodent ventilator (Model 683, Harvard Apparatus, Holliston, MA, USA) at a rate of 55–65 breaths/min and with a tidal volume of 20 mL. General anesthesia was first induced by an intravenous bolus of 5 mg/kg propofol (Diprivan 2%, AstraZeneca, Brussels, Belgium) through a 22 G butterfly catheter within the left ear marginal vein and further maintained by a continuous intravenous infusion of 13 mg/kg/h propofol using an infusion pump (Perfusor® Secura, B Braun, Melsungen, Germany). During the surgical procedure, the rabbit’s body temperature was maintained using a heating pad. To prevent the rabbit’s eyes from drying, sterile 0.9 % NaCl solution was applied once at the beginning of the surgical procedure. HR and arterial blood oxygen saturation of the animal were continuously monitored using a pulse oxymeter (GE CarescapeV100, Uno, Zavenaar, The Netherlands) through a clip placed on the right ear central artery.
Surgical procedure
A sterile field was maintained around the animal using sterile foil (Steri-Drape, 3M, Diegem, Belgium). The operator wore sterile gloves and a mouth surgical mask. All surgical instruments were sterile. Telemetry catheter implantation into the right ventricle was performed using a trans-diaphragm approach. The abdominal cavity was opened by making a 12 cm middle line incision. Abdominal muscles were lifted 2 cm from the xiphoid process using suspension wires (Silk 4–0, Ethicon, Livingston, UK) bound to a metal bar placed over the animal to provide optimal exposure of the diaphragm and right ventricle during the whole procedure. During the surgical procedure, warm sterile 0.9% NaCl solution was administered within the abdominal cavity. A 2 cm incision was made within the diaphragmatic membrane, as described elsewhere.
11
The pericardium around the right ventricular wall was opened and a string purse suture was made using string suture (Silk 4–0, Ethicon) within the right ventricular wall, close to the apex (Figure 1a), as previously described.
12
A puncture towards the pulmonary artery was further performed within the string purse suture using a 14 G needle (Figure 1b). Any bleeding from the puncture side was stopped with cotton stick until the catheter tip of the telemetry device was properly inserted. The purse string suture was closed with a knot to tightly fix the tip of the catheter (Figure 1c). The purse string suture was bound carefully with string suture (Silk 3–0, Ethicon) placed beforehand at 1.5 cm from the tip of the catheter, to tightly maintain the catheter in place. The diaphragm incision was further closed using running suture (Vicryl 6–0, Ethicon). To obtain full expansion of the lungs, 10–20 mL air was removed from the thoracic cavity using a 24 G winged catheter bound to a 10 mL syringe inserted through the closed incision of the diaphragm into the thoracic cavity. Suspension wires were removed. The body of the transmitter was placed under the right abdominal muscles and sutured at two different points to the muscles using 3–0 silk suture. Abdominal muscles were sutured with running suture (Vicryl 5–0, Ethicon). Before suturing the skin using running suture (Vicryl 4–0, Ethicon), continuous anesthesia using propofol was stopped.
Schema of the implantation of the catheter in the right ventricle. (a) String purse suture. (b) Insertion of the catheter in the apex of the right ventricle. (c) Correct placement of the catheter into the right ventricle.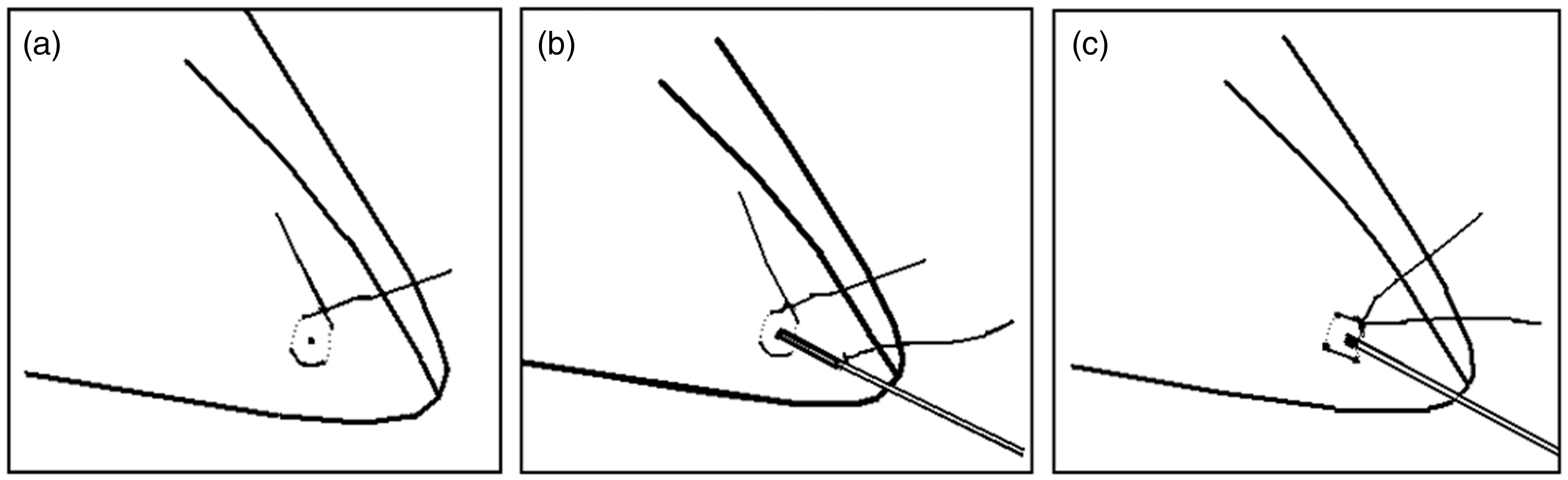
Postoperative care
After skin closure, the animals received a subcutaneous injection of 0.03 mg/kg buprenorphine (Temgesic, Schering-Plough, Brussels, Belgium) and of 10 mg/kg enrofloxacin (Baytril 5%, Bayer, Brussels, Belgium). Mechanical ventilation was stopped as soon as the animals could breathe spontaneously. Until fully awakened, the animals were supplied with oxygen through a mask, maintained on a heating pad, and HR and blood oxygen saturation were continuously monitored. When eye and pedal reflexes were observed, the animals were placed into an individual cage. The first day following the surgery, the animals were further checked several times a day and any problems were reported. The animals received a second subcutaneous injection of 0.03 mg/kg buprenorphine 6 h after the end of the surgical procedure, twice 0.02 mg/kg buprenorphine and once 10 mg/kg enrofloxacin on day 1 after the surgery and twice 0.01 mg/kg buprenorphine and once 10 mg/kg enrofloxacin on day 2 after the surgery. As a general practice, the animals were checked twice a day. Body mass was monitored every week following the surgery to investigate any side-effects of telemetry device implantation on animal development.
Autopsy
Any adverse effects of telemetry transmitter chronic implantation were investigated at the end of the observation period, after having euthanized the animals by intravenous injection of 100 mg/kg pentobarbital (Nembutal, Ceva, Brussels, Belgium), or after spontaneous early death. Macroscopic examination of the right ventricle, lungs and diaphragm were performed. The right ventricle was opened along the ventricle septum and correct implantation of the transmitter device catheter was checked.
Statistics
Statistical analyses were performed using GraphPad Prism 4.01 (GraphPad Software Inc, La Jolla, CA, USA). Means were compared between groups using analysis of variance (ANOVA) test followed by post hoc test (Tukey) and proportions using chi-square test. Results were expressed as mean ± SD. Survival Kaplan–Meier curves were established, censoring the animals at the end of the observation period. A value of P < 0.05 was considered statistically significant. All P values were for 2-sided tests.
Results
Animals
A total of 44 adult male New Zealand rabbits were implanted with a radiotelemetry transmitter to further measure continuous RVP.
Placement of the telemetry catheter tip within the right ventricle
Proper placement of the telemetry device catheter tip within the right ventricle was checked by continuously recording RVP by telemetry, by observing a typical wave shape as shown in Figure 2. Recording of RVP was monitored in 39 animals from the moment the telemetry device catheter was introduced in the right ventricle until closure of the diaphragm, restoration of thoracic negative pressure and fixation of the transmitter body to the abdominal muscles. Five animals died during the surgical procedure before introducing the telemetry catheter within the right ventricle. The average anesthesia duration was 73 ± 14 min (range 54 to 120 min).The average right ventricle systolic pressure (RVSP), right ventricle diastolic pressure (RVDP) and HR monitored by telemetry during 20 min following closure of the diaphragm, restoration of thoracic negative pressure, and fixation of the transmitter body to the abdominal muscles were 16.5 ± 3.3 mmHg (range: 8.4 to 24.8 mmHg), 4.2 ± 3.1 mmHg (range: –6.6 to 9.9 mmHg) and 165 ± 29 beats per minute (bpm) (range: 116 to 246 bpm), respectively. HR and arterial blood oxygen saturation measured in 36 of the animals using the oxymeter were 152 ± 28 bpm (range: 96 to 225 bpm) and 98.8 ± 2.3% (range: 89 to 100 %).
Right ventricular pressure (RVP) measured at the time of implantation. RVP was measured for 4 s in four different animals: rabbit R34 (a), rabbit R36 (b), rabbit R48 (c), rabbit R54 (d) when the telemetry device catheter has been introduced in the right ventricle and after having closed the diaphragm, removed air from the thoracic cavity and fixed the transmitter body to the abdominal muscles. RVP was expressed in mmHg.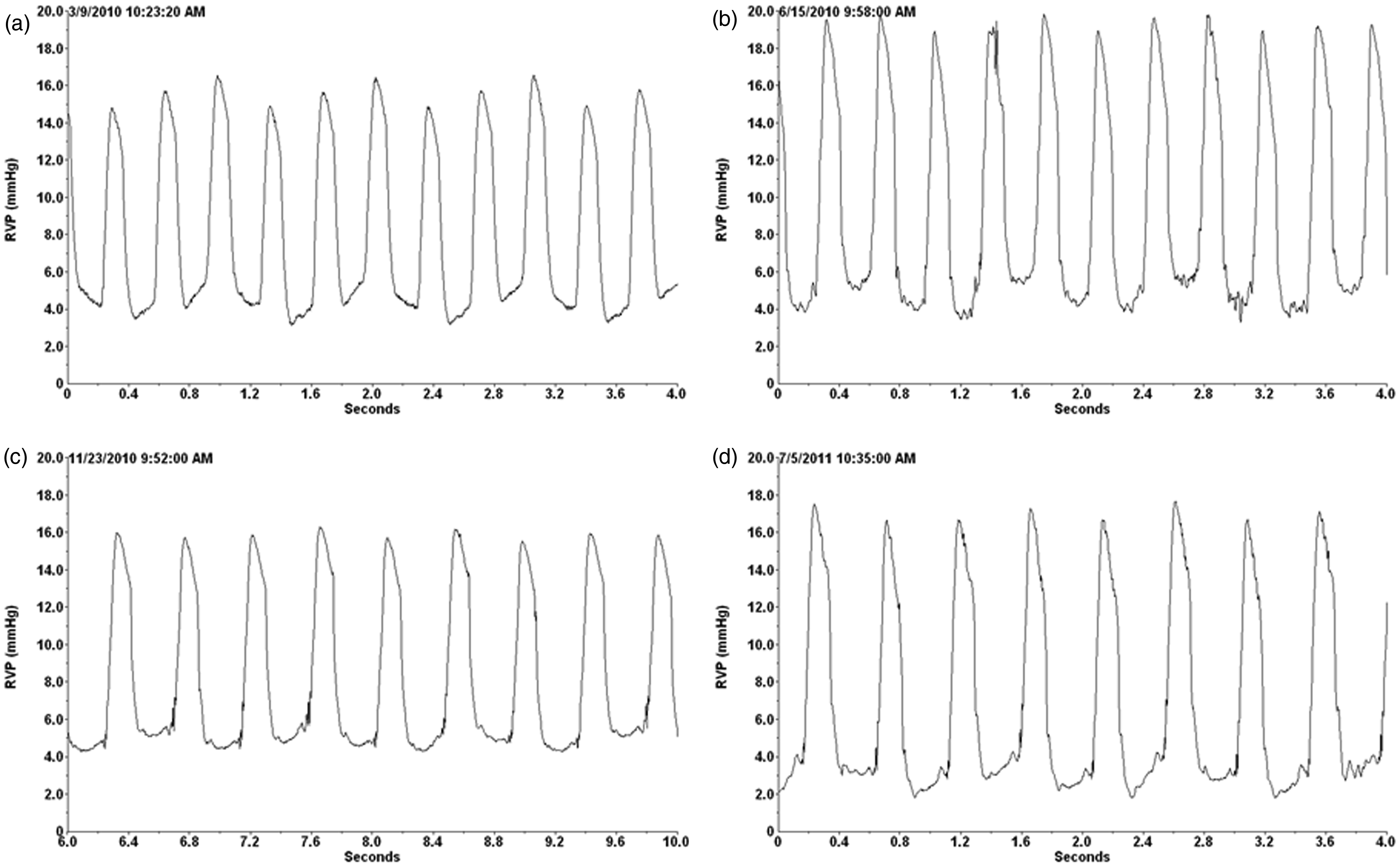
Surgery survival
Surgery survival.
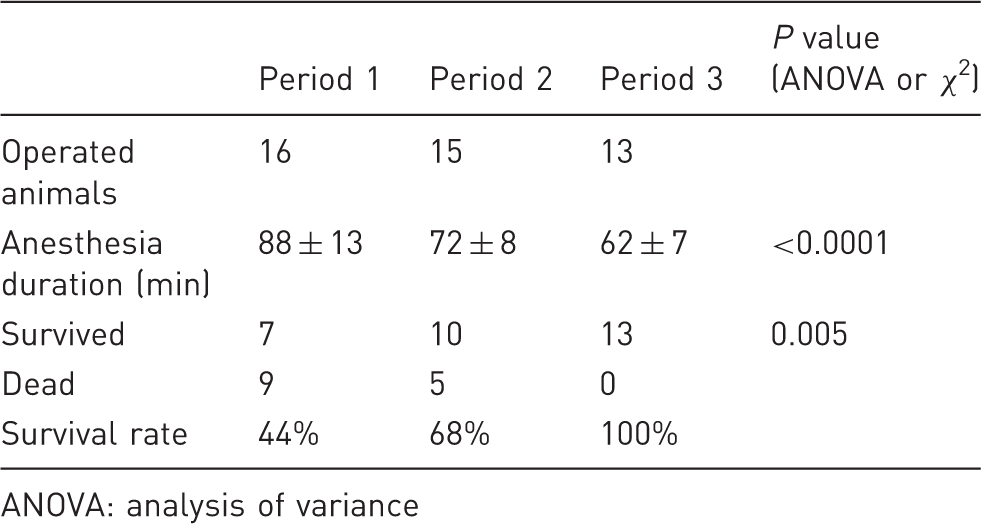
ANOVA: analysis of variance
Body mass follow-up
At the time of the surgery, body mass was 3.54 ± 0.32 kg (range: 3.00 to 4.25 kg). One week after the operation, body mass was measured in 23 rabbits: 4 (17%) did not display any change in their body mass, 2 (9%) gained 50 g and 17 (74%) lost an average of 208 ± 124 g (range: 50 to 550 g), representing a 5.7% loss of initial body mass. Three weeks after the operation 67% of the 30 surviving animals recovered their initial body mass or displayed an increase in their body mass, and this proportion increased to 73% of the animals after 4 weeks.
Figure 3a shows the body mass evolution of four different animals; rabbit R20 was not implanted with any telemetry device, whereas rabbits R18, R46 and R59 were implanted. This figure shows that telemetry device implantation did not impair the normal development of the animal.
Rabbit body mass follow-up and long-term survival curves. (a) Animal body mass was measured once a week starting one week after telemetry device implantation. Rabbits R18, R46 and R59 were implanted, whereas rabbit R20 was not. (b) Kaplan–Meier curves comparing survival rate during the three observation periods. Chi-square, P = 0.004.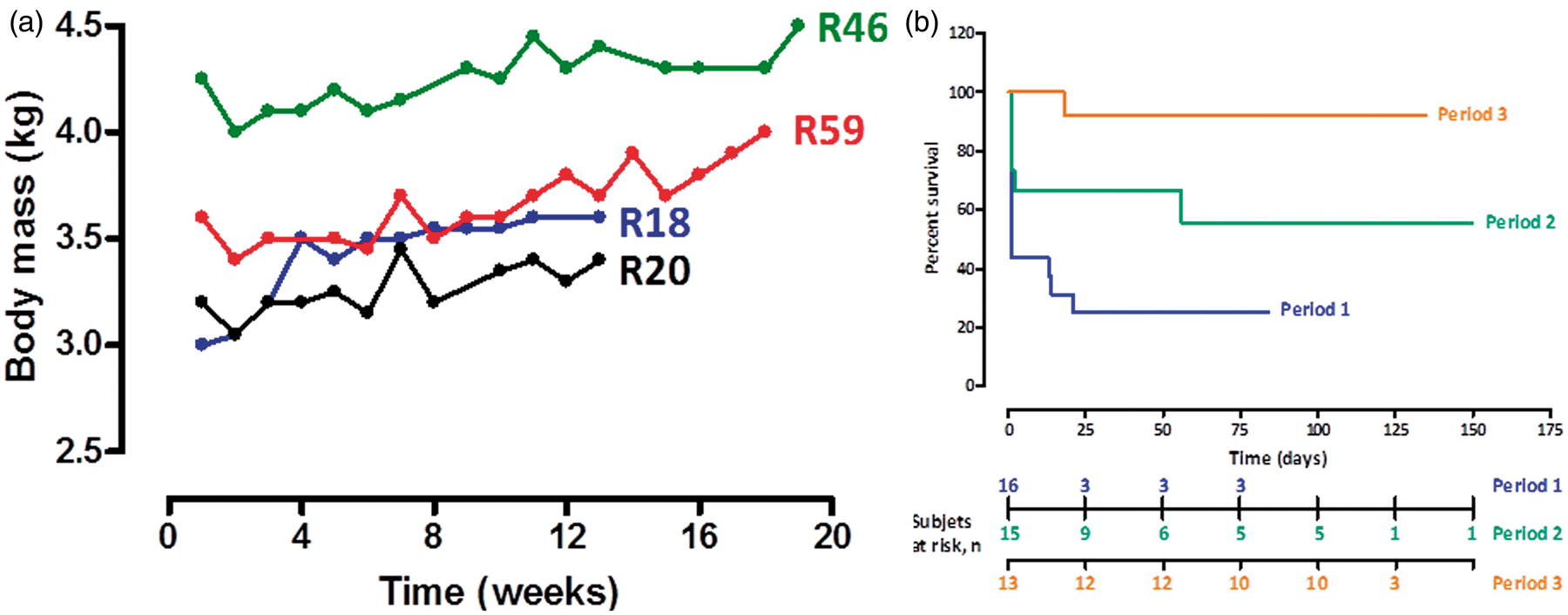
Telemetry data and long-term survival
The telemetry transmitter was switched on two weeks after being implanted into the animal. RVP, HR and activity were continuously monitored using a sample method consisting of recording periods of 20 s every 15 min. Of the 30 rabbits who survived the implantation surgery, three were euthanized because of unusual RVP waveforms, due to incorrect placement of the catheter within the right ventricle, which was confirmed at autopsy. One rabbit died of infection and one died of unknown causes. Long-term telemetry monitoring was performed in 17 animals, with an observation period of 63 to 121 days. Figure 3b shows the survival curve of rabbits implanted during periods 1, 2 and 3. The survival rate significantly improved within the successive periods (P = 0.004). The overall survival rates at 50 days were 25%, 67% and 92% for periods 1, 2 and 3, respectively; and at 100 days they were 25%, 56% and 92%, for periods 1, 2 and 3, respectively.
Figure 4 shows activity, HR, RVDP and RVSP monitored by telemetry during 24 h in two different rabbits. A concomitant increase in RVSP, HR and activity was observed during the dark period corresponding to the night activity of prey animals such as rabbits.
Twenty-four-hour monitoring of right ventricular pressure (RVP), heart rate (HR) and activity by telemetry. Right ventricle systolic pressure (RVSP) and right ventricle diastolic pressure (RVDP) (a, b), HR (c, d) and activity (e, f) were monitored every 15 min for 20 s during 24 h by telemetry in two different rabbits: rabbit R46 (a, c, e) and rabbit R59 (b, d, f). RVP was expressed in mmHg, HR in beats per minute (bpm) and activity in counts per min.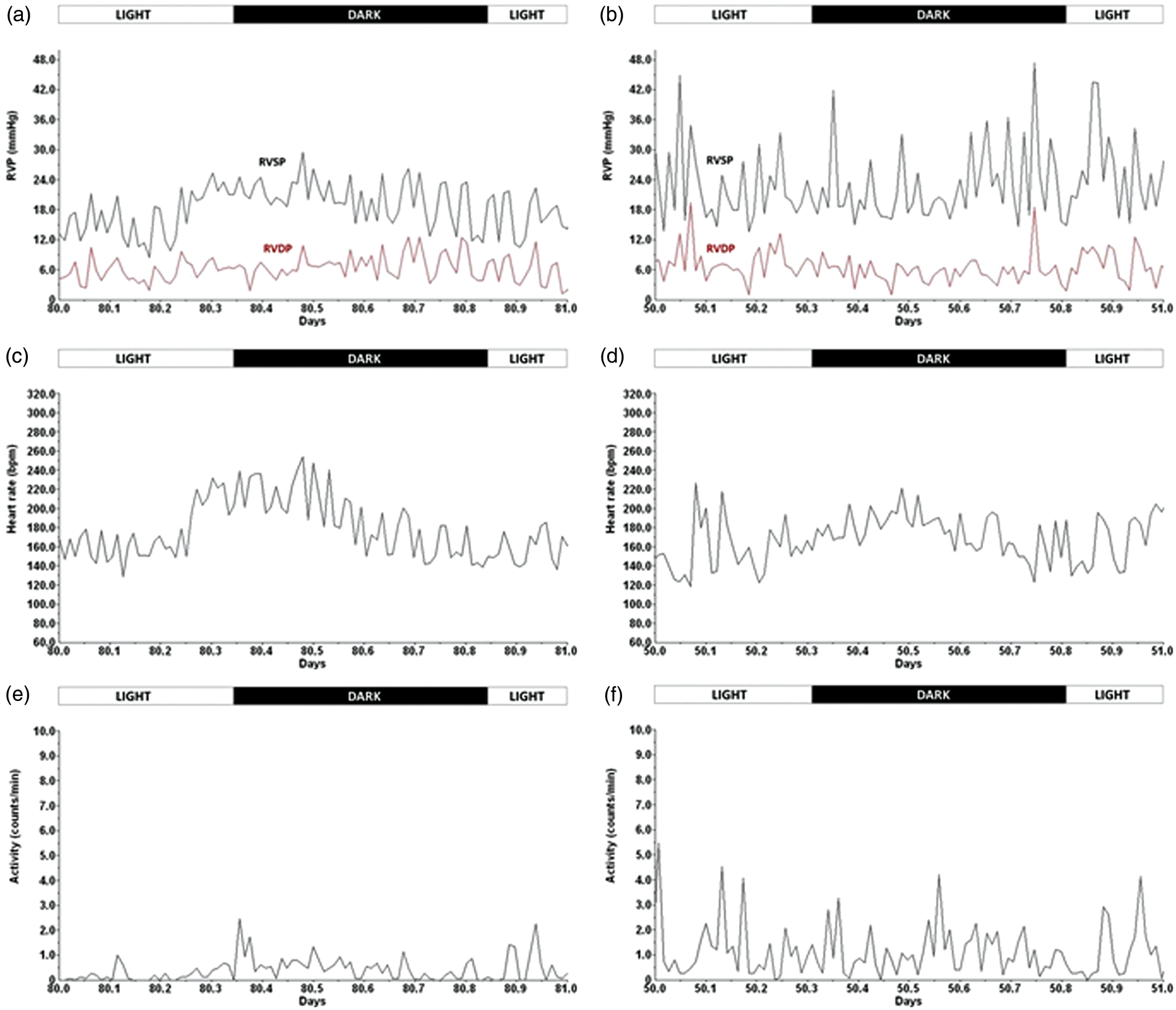
Stable RVSP and RVDP monitored by telemetry were observed in six control rabbits over an average time period of 90 ± 19 days (range: 61 to 106 days). In parallel, HR and activity were also monitored. Figure 5 shows the time curves of activity, HR, RVSP and RVDP monitored by telemetry in two representative rabbits.
Study monitoring of right ventricular pressure (RVP), heart rate (HR) and activity by telemetry. Right ventricle systolic pressure (RVSP) and right ventricle diastolic pressure (RVDP) (a, b), HR (c, d) and activity (e, f) were monitored every 15 min for 20 s during respectively 100 and 120 days by telemetry in two different rabbits: rabbit R46 (a, c, e) and rabbit R59 (b, d, f). RVP, HR and activity parameters were averaged for each 24 h period and plotted. RVP was expressed in mmHg, HR in beats per minute (bpm) and activity in counts per min.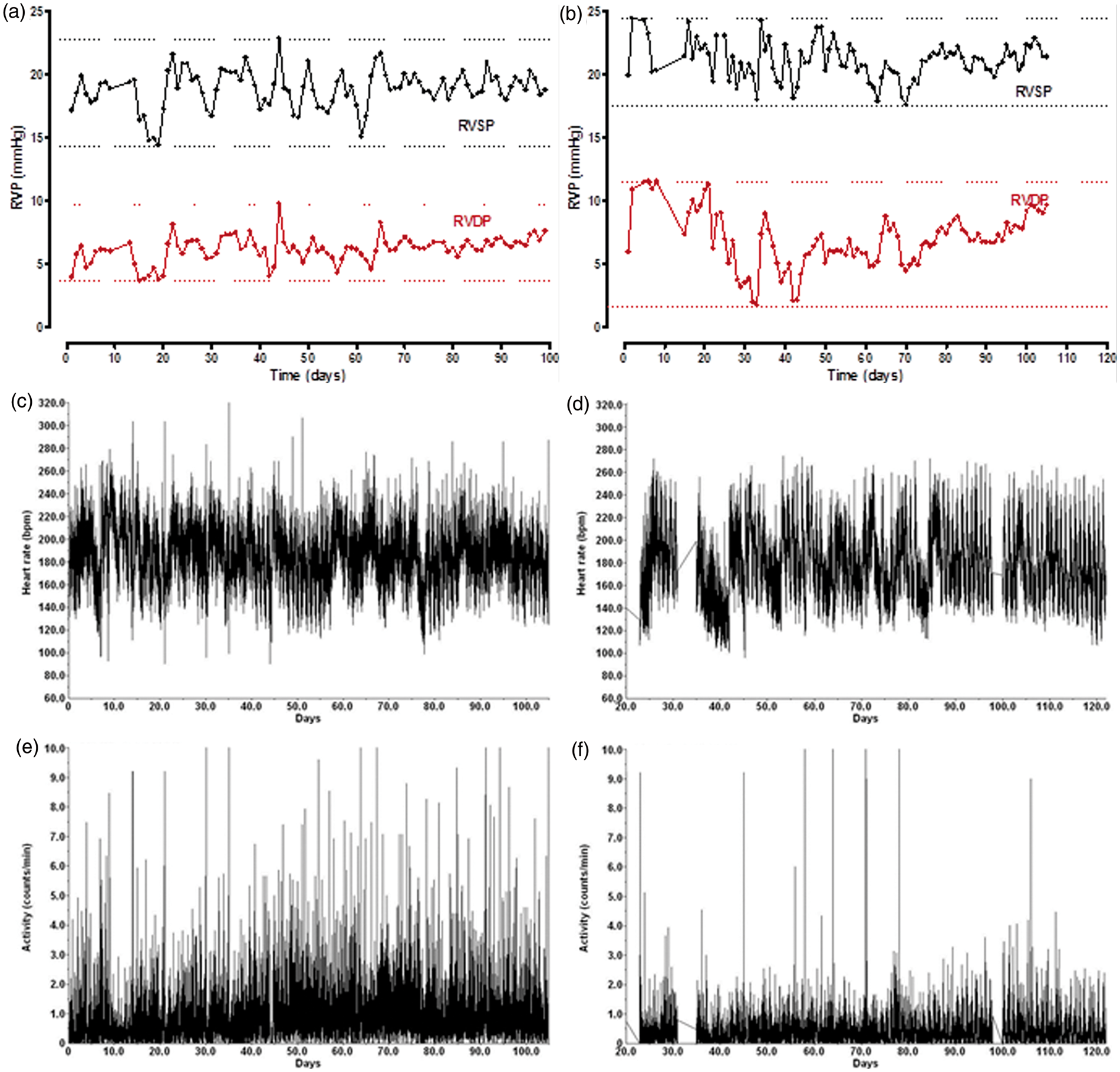
Autopsy
As previously described, autopsy revealed only local fibrosis of the right ventricle near the insertion point of the catheter; no rupture or spontaneous opening of the diaphragm was observed in any animal and the liver was often partially fused with the healed wound of the diaphragm.
11
In most animals, the transmitter device catheter was correctly implanted in the right ventricle (Figure 6a) and in a few of them fibrotic tissue and/or fibrin and thrombotic material were observed around the tip of the catheter (Figure 6b). As mentioned above, thrombosis formation at the tip of the catheter was observed in some animals (Figures 6c and 6d).
Rabbit heart implanted with telemetry device at autopsy time. (a) Insertion region of the transmitter catheter into the right ventricle. (b) Inserted transmitter catheter within the right ventricle.(c, d) Presence of fibrin and thrombotic material at the tip of the catheter of the telemetry transmitter could be observed.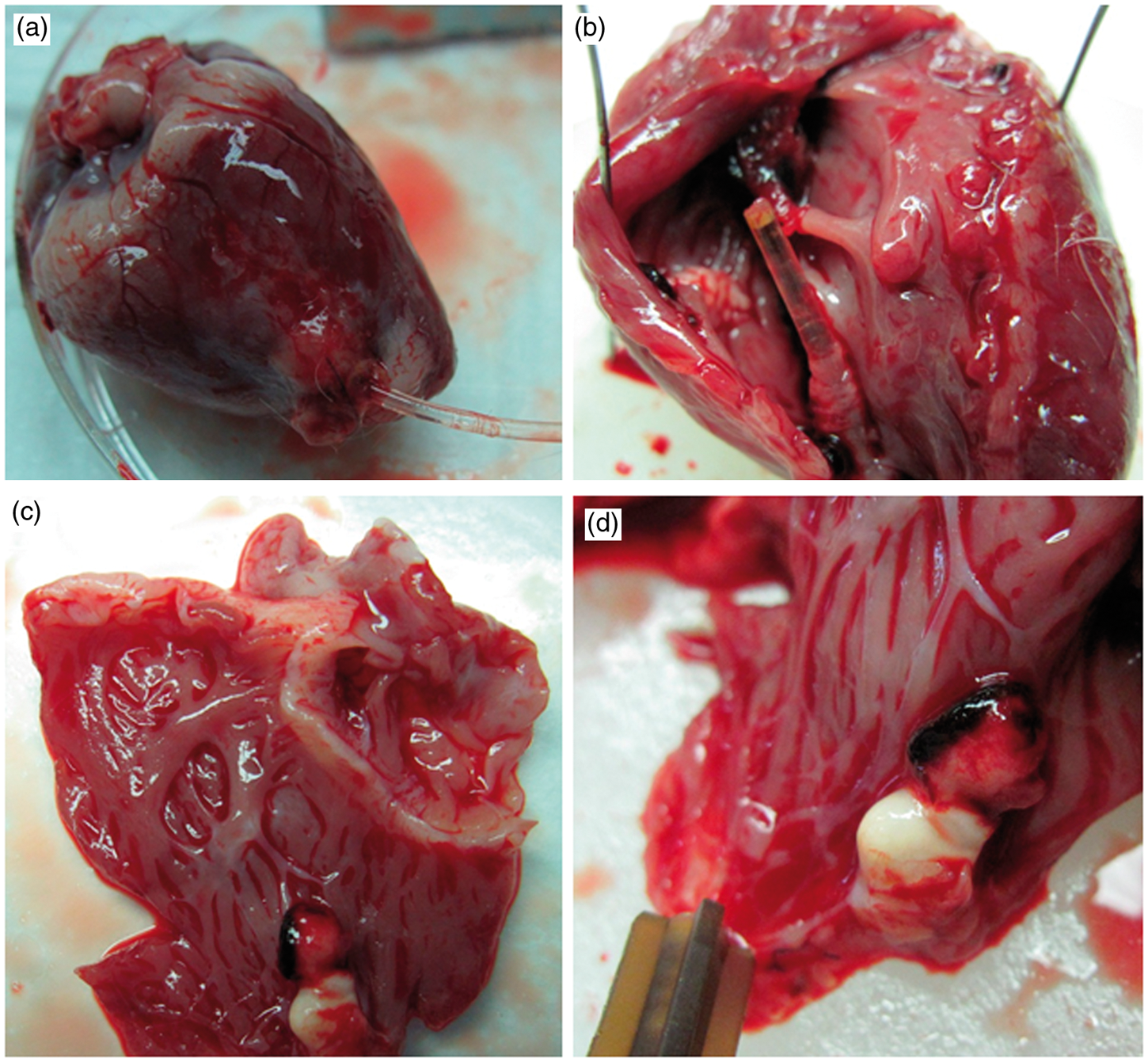
Discussion
In the present study, we have shown that monitoring RVP using implantable radiotelemetry is feasible in rabbits. To our knowledge, this is the first report of RVP measurement by telemetry in rabbits, using a trans-diaphragm approach.
Radiotelemetry has been previously described as the technique of choice for monitoring physiological functions in awake and freely moving laboratory animals, while minimizing stress artifacts. 13 The possibility offered by radiotelemetry of recording parameters in conscious and untethered animals rules out various artifacts and drawbacks generated by drugs and/or gas anesthesia such as respiratory and cardiovascular depression. Radiotelemetry has also allowed the investigation of circadian blood pressure variability 14,15 or circadian changes in body temperature. 16,17 Accordingly, we also observed changes in RVP, HR and activity of rabbits related to light exposure, with an increase in RVP, HR and activity during the dark cycle, corresponding to the typical nocturnal activity of prey animals. Telemetry also displays a great flexibility by allowing continuous monitoring, which is useful at the moment of the implantation of the catheter into a blood vessel or into the right ventricle. Sampled monitoring during 10–20 s every 15−20 min performed in the present study allows long-term measurement, which is crucial when establishing chronic animal models.
The overall perioperative mortality rate of 32% was due to euthanasia performed because of ventilation supply problems, surgical errors or respiratory failure, excessive bleeding during the surgery procedure and extrusion of the transmitter body. These types of problems were experienced in the early part of the learning process and rapidly disappeared with learning and experience (see Table 1).
Implantable telemetry has been widely used for measuring blood pressure in mice, rats and rabbits for both acute and chronic purposes 13,18 –20 and has consequently become an alternative technique to catheterization. More recently, new surgical techniques have been developed for measuring pressure within the left ventricle in rabbits. 12 In 1996, Hess et al. were the first to describe the use of implantable telemetry to continuously monitor RVP and PAP in conscious rats. 6 In line with this, the authors have initiated a series of publications further describing the use of telemetry to monitor RVP or PAP in various models of pulmonary hypertension mainly in mice and rats, 7 –10 whereas it has thus far never been described in rabbits. In the present study, we show that the use of implantable telemetry is perfectly feasible in rabbits for monitoring RVP, HR and activity. Significant improvement in peroperative survival confirmed the learning curve of the implantation procedure. Moreover, the occurrence of only two spontaneous deaths clearly confirmed the safety of the implantation technique. Moreover, we were able to monitor the parameters mentioned above for more than three months, indicating that implantable telemetry is an interesting alternative to other techniques such as MRI or Doppler echocardiography when developing chronic models of pulmonary arterial hypertension (PAH). Thus far, most of the surgical techniques described for inserting the telemetry catheter within the right ventricle or the pulmonary artery imply an extremely invasive thoracic approach, including abdominal cavity exposition and thoracotomy. Another technique whereby the sensing catheter is inserted into the jugular vein and forwarded to the right ventricle has also been described in rats 8 and mice. 7 In 2008, Handoko et al. described a new surgical technique for implanting the telemetry catheter using a trans-diaphragm approach that is less traumatic for the animals. This technique allows better orientation for introducing the catheter within the right ventricle and especially for further forwarding it into the pulmonary artery, providing a stronger anchorage of the device, which results in a significantly better recovery following surgery. 11 Although left ventricular pressure monitoring has been performed in rabbits using the common trans-thoracic approach, 12 we have chosen to adapt the trans-diaphragm approach previously described 11 in rats for monitoring RVP in rabbits.
Finally, the use of telemetry to continuously measure RVP in rabbits can fulfill most of the 3 R (Replacement, Refinement, Reduction) issues. Concerning refinement, the use of telemetry allows non-invasive and continuous recording of hemodynamic data in freely moving animals, limiting invasive surgery to the implantation of the radiotelemetry system; and consequently avoiding repeated right heart catheterization, which implies anesthesia, jugular vein exposition and technically challenging vascular sutures. The use of telemetry is an appropriate system for reducing the number of animals per study since each animal can be its own control by allowing continuous measurement before and after any intervention.
To conclude, we have demonstrated in the present study that implantable radiotelemetry methodology can be safely used to perform continuous and long-term monitoring of RVP in freely moving conscious rabbits.
Footnotes
Acknowledgements
This work was supported by research grants from the ‘Fonds voor Wetenschappelijk Onderzoek Vlaanderen’ (FWO-Flanders KN. 1.5.187.07) and from Pfizer. MD is holder of the Actelion chair for Pulmonary Hypertension and of the GSK chair for Research and Education in pulmonary vascular pathology at KU Leuven. The authors would like to thank Solenn Caradec and Eric Rieux from Data Sciences International (St Paul, MN, USA) for their expert advice, Inge Schalij (Department of Pulmonology, VU University Medical Center, Amsterdam, The Netherlands) for her technical expertise and assistance, as well as Erna Dewil, Luc Hendrickx and Anna Vandenwaeyenberg from the staff of the animal facility at KU Leuven.
