Abstract
Summary
Buprenorphine is a potent analgesic commonly used clinically in humans and rodents experiencing severe pain. However, effects of therapeutic doses on locomotor activity and the cardiovascular system have not been studied in conscious animals. The effects of buprenorphine were therefore evaluated in this study using telemetric monitoring in conscious animals. Telemetry transmitters were implanted in the peritoneal cavity of Wistar rats with a pressure catheter in the aorta and electrodes for electrocardiogram (ECG) recording subcutaneously. After a single subcutaneous administration of saline, each rat was administered single subcutaneous doses of 0.006, 0.03 or 0.15 mg/kg body weight (bw) of buprenorphine. During a 10 h period after administration, buprenorphine induced a varying dose-dependent increase in body temperature, heart rate, d
Buprenorphine is an opioid analgesic that, after parenteral administration of low doses, has approximately 24–40 times the analgesic potency of morphine in humans (Jasinski et al. 1978) and rodents (Cowan et al. 1977). It has therefore been used extensively for nearly two decades as a potent analgesic in animals experiencing severe pain, including post-surgical pain in clinical studies (Roughan & Flecknell 2002). Acute myocardial infarction (Boachie-Ansah et al. 1989) and neoplastic disease (Brema et al. 1996) are usually associated with severe pain, and it is common practice among emergency physicians to administer narcotic analgesics such as buprenorphine. Thus, physicians use buprenorphine as a current medication for a variety of patients. Moreover, the Food and Drug Administration has recently approved the use of buprenorphine for detoxification and office-based therapy for opiate addiction, particularly in individuals who are at high risk of developing acquired immunodeficiency syndrome (AIDS), hepatitis B and C and tuberculosis (Koch et al. 2006).
Buprenorphine is used in laboratory animal and human medicine because it has a long duration of action (Downing et al. 1977) and fewer cardiovascular and respiratory effects than do most other opioid analgesics for pain management (Taylor & Houlton 1984). Potential acute adverse effects of buprenorphine in humans are diffuse mild mental status changes, mild to minimal respiratory depression, small pupils and relatively normal vital signs (Sporer 2004). Moreover, it is compared with other opioids associated with a lower degree of sedation and respiratory depression (Mello et al. 1993). Studies
Because absorption of buprenorphine from the gastrointestinal tract is low, parenteral routes of administration are preferable. The recommended postoperative subcutaneous dose of buprenorphine is 0.05 mg/kg body weight (bw) (Thompson et al. 2004). A single oral dose of 0.5 mg/kg bw in rats produces analgesic effects for 6–8 h (Gades et al. 2000), but the obtained analgesic efficacy is more robust when 0.05 mg/kg bw is administered subcutaneously (Thompson et al. 2004). Absorbed buprenorphine is readily distributed in different body compartments, with a peak plasma concentration at approximately 90 min and a half-life of 4–5 h (Sporer 2004).
Telemetric monitoring of conscious laboratory animals has proven useful in the acquisition of reliable physiological data (Eisermann 1988, Brackee et al. 1995) and has many advantages over conventional data acquisition techniques in risk assessment of drugs and food constituents (Ilbäck et al. 2002a,b, 2007).
The aims of this study are to use telemetric monitoring in the evaluation of spontaneous locomotor activity, body temperature and cardiovascular effects that are caused by different doses of buprenorphine and to study whether the observed effects were dose-dependent.
Materials and methods
Animals and housing
Wistar Hannover GALASTM (HanTac:WH) female rats (aged 7 weeks) obtained from Taconic M&B, Ry, Denmark, were used. A microbiological health status was obtained from the supplier showing no infections to be declared according to FELASA (2002). During the experiment, clinical examination was performed daily and individual body weight was determined weekly. Rats were 13 weeks old at the time of surgery and 18 weeks old when the experiment was initiated. The body weight of the rats was approximately 300 g on the day the experiment began. The animals were housed in a barrier building and identified by an individual unique tattoo on the tail.
Animal room controls for temperature and relative humidity were set at 21 ± 1°C and 55 ± 5%, respectively. The light was controlled to provide 12 h of light (06:00 h to 18:00 h) and 12 h of darkness per 24 h.
The rats were kept in individual cages (Eurostandard Type III, Technoplast, Buguggiate, Italy). The cages, cage trays, food hoppers and water bottles were sanitized twice weekly. All animals were given free access to both food (RM1 [E] SQC Ltd, UK, vendor Scanbur A/S, Karlslunde, Denmark) and tap water. There was no indication that any (non-nutrient) substance was present in the diet or in the drinking water that influenced the effects of the test compound.
The animal experiments described in this paper took into account all ethical aspects of the welfare of the animals and were performed following the recommendations in the
Compound
Commercially available Temgesic stock solution in vials containing buprenorphine hydrochloride (0.3 mg/mL) was obtained from Schering-Plough, Stockholm, Sweden. The stock solution was diluted in physiological saline (NaCl) with a pH of 7.0. Dilutions of the test formulations (0.006, 0.03 and 0.15 mg/mL) were performed by the Department of Pharmacy, Active Biotech AB, Lund, Sweden. The placebo was saline (NaCl solution) with a pH of 7.0. The placebo solution and the test formulations were administered subcutaneously to achieve the three dose levels (i.e. 0.006, 0.03 and 0.15 mg/kg bw).
Experimental procedure
Anaesthesia
The rats (
Surgical preparations
Each rat was placed on a heated bed (Kanthal, AB Ninolab, Upplands Väsby, Sweden) during surgical preparation. Under aseptic conditions, an abdominal incision of about 5–6 cm was made to the right of the midline; at this time, the intestines were retracted and the abdominal aorta exposed. The catheter of the transmitter was inserted into the vessel and sealed in place with tissue adhesive. The intestines were gently put back in place and the flap of the transmitter was secured to the abdominal wall. After the telemetry transmitters had been implanted in the peritoneal cavity with the pressure catheter in the aorta, electrodes for electrocardiogram (ECG) monitoring were placed subcutaneously on the right side of the thorax and near the left hindlimb.
After surgery, the rats were subcutaneously treated with the recommended postoperative dose (0.05 mg/kg bw) of buprenorphine (Thompson et al. 2004) and kept under continuous observation in the postoperative unit until they had regained full consciousness, at which time the animals were transferred back to their cages. Approximately four weeks after surgery, the rats entered the study. When treatment and all registrations according to the protocol were finished, the rats were administered a lethal dose of pentobarbital.
Treatment and experimental design
The rationale for selection of the dose levels (i.e. 0.006, 0.03 and 0.15 mg/kg bw) was based on the combination of clinical use and earlier studies in rats with single doses of buprenorphine (Hayes & Flecknell 1999, Sharp et al. 2003). Each rat was administered with a single subcutaneous dose; for each dose, responses in four rats were investigated. The time point for administration of buprenorphine is marked with an arrow in the figures. The rats (
Test procedure and recordings
TL11 M2-C50-PXT transmitters (Datasciences Inc, St Paul, MN, USA) were used. The transmitters were turned on one day before implantation in order to stabilize the signal and to check the zero offset value. The signals from the transmitters were received by two RLA 2000 receivers and transmitted via a Data Exchange Matrix to a Dataquest PCI card CQ 2240 in the computer. The data were interpreted by the Dataquest program ART version 2.2 Gold Version.
Program set-up and checks
Registration was made with the Dataquest acquisition program version 2.2 and run with the operative system Windows NT 4.0. The configuration file was set as follows: for blood pressure, the sampling rate was 500 Hz with a filter cut-off set at 100 Hz and full scale at 500 mmHg. The sampling rate for ECG was 1000 Hz with no filter cut-off and full scale at 10 mV.
Sampling of ECG, blood pressure, body temperature and locomotor activity
The data were stored on a hard disk in accordance with the parameters entered in the configuration files. Refractory for diastolic and systolic blood pressure and heart rate was 0.2. Sampling duration for ECG, heart rate, and systolic and diastolic blood pressure was set to 10 s. Data were sampled once per minute. Locomotor activity was registered through horizontal movement of the transmitter, i.e. each movement results in a count.
Data evaluation and statistical procedures
Values of systolic and diastolic blood pressure, heart rate, body temperature and locomotor activity were imported to Microsoft Excel, and from these data diurnal mean values and correlation were calculated. Statistical evaluation of the effects of buprenorphine on body temperature, heart rate, systolic and diastolic blood pressure and QT time were performed between 11:00 h and 15:00 h and between 05:00 h and 08:00 h. All ECG recordings were investigated visually for signs of arrhythmia or changes in the shape of the ECG complexes. For the d
Results
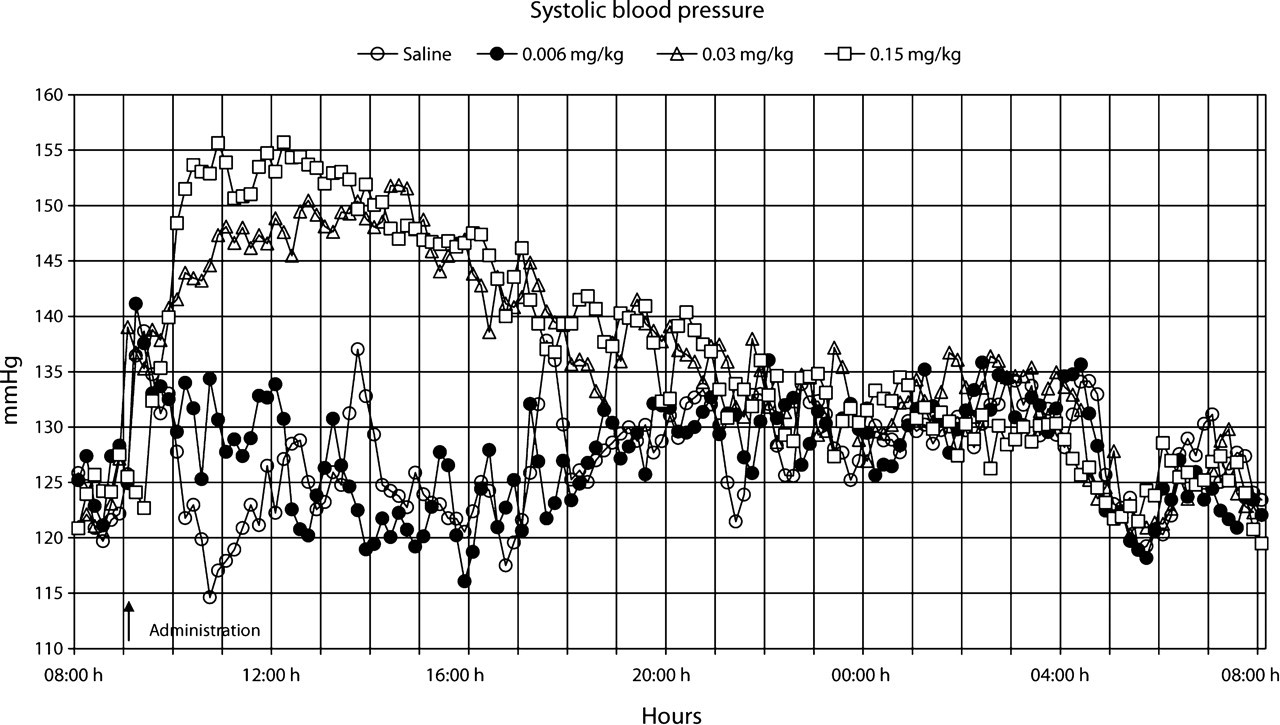
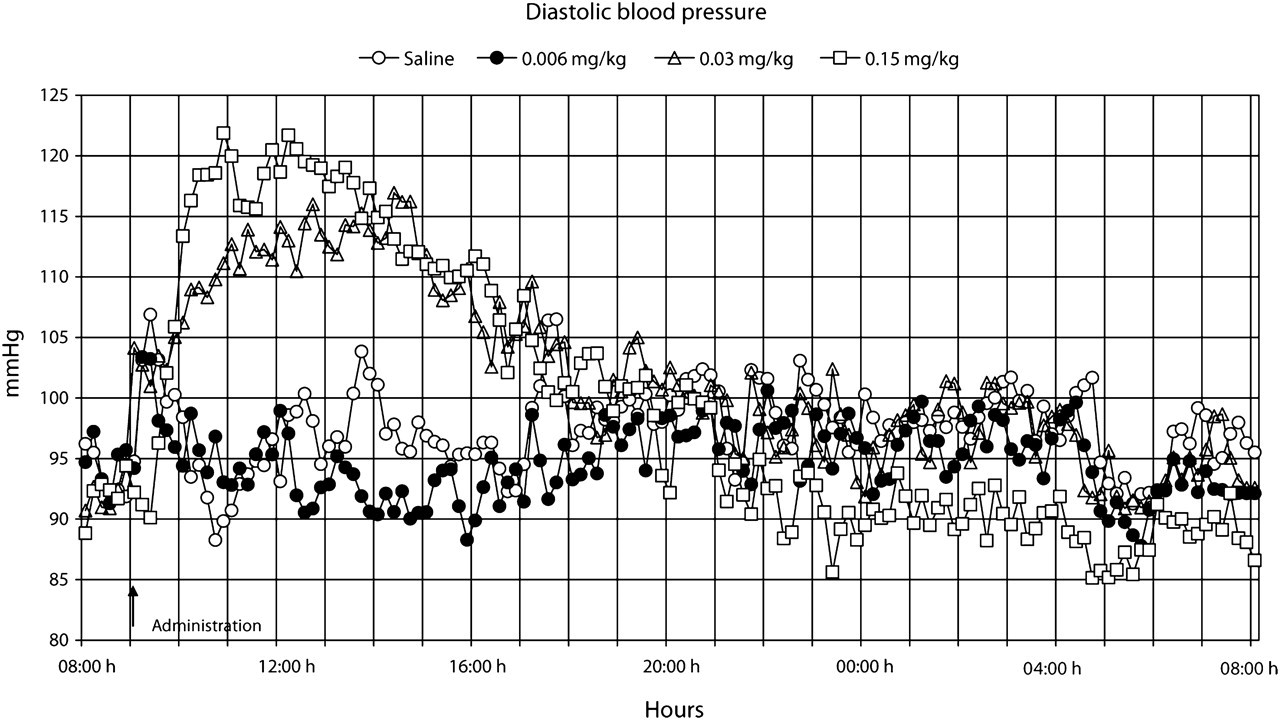
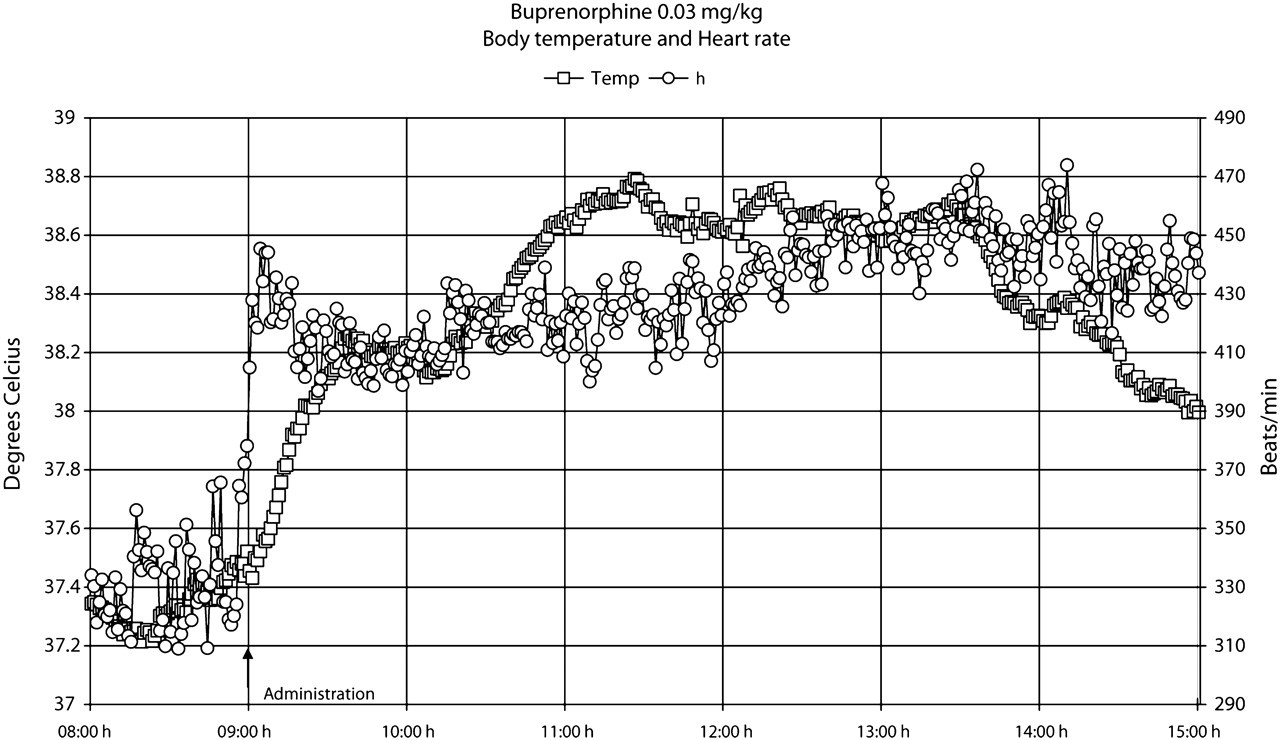
Our observations indicated that the rats did not develop any visual signs of adverse effects during or after treatment with the presently used single doses (0.006, 0.03 or 0.15 mg/kg bw) of buprenorphine administered subcutaneously. The data points shown in Figures 1–7 are mean values of 10 registrations, whereas in Figures 8 and 9 all data points are shown.
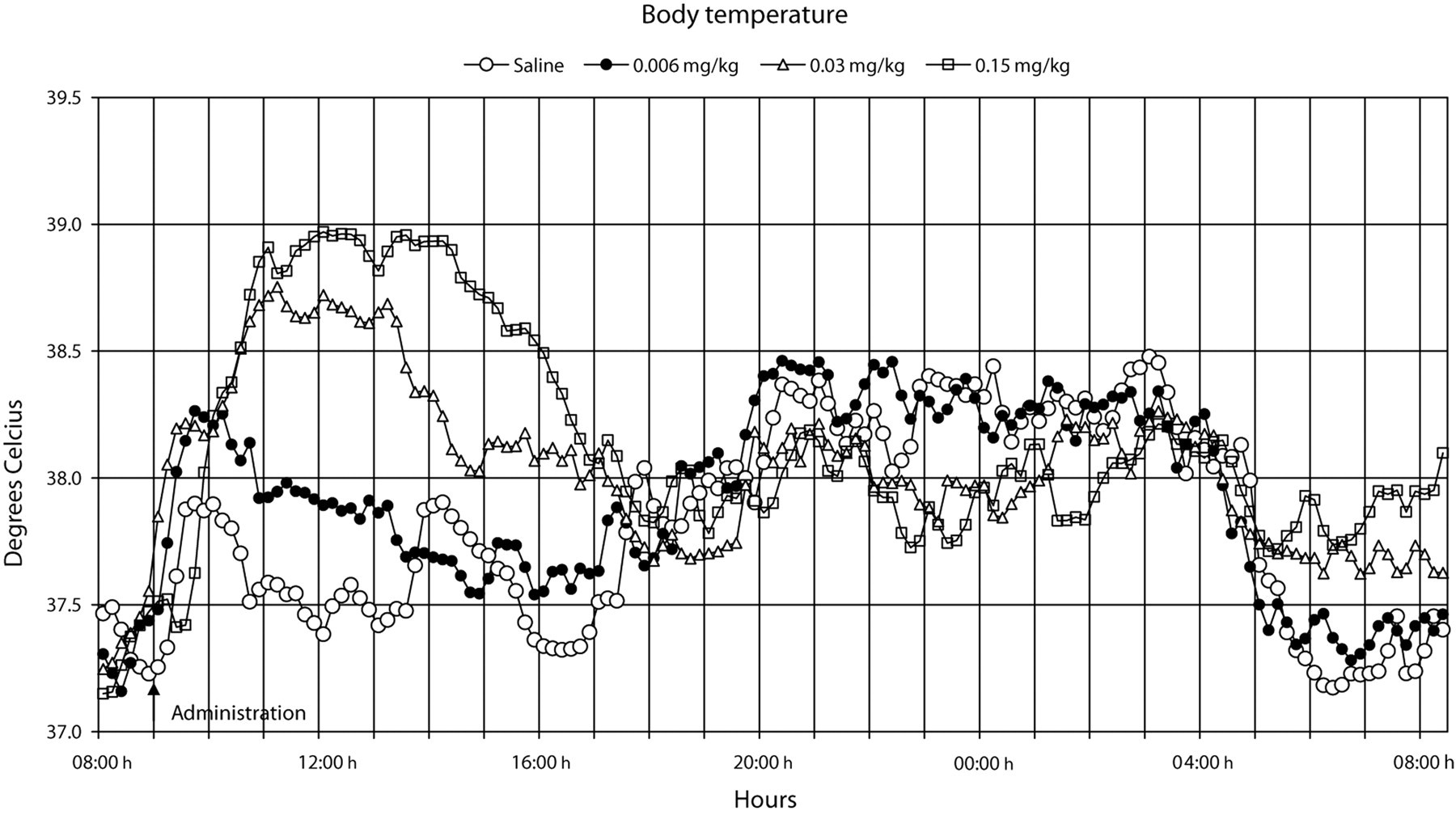
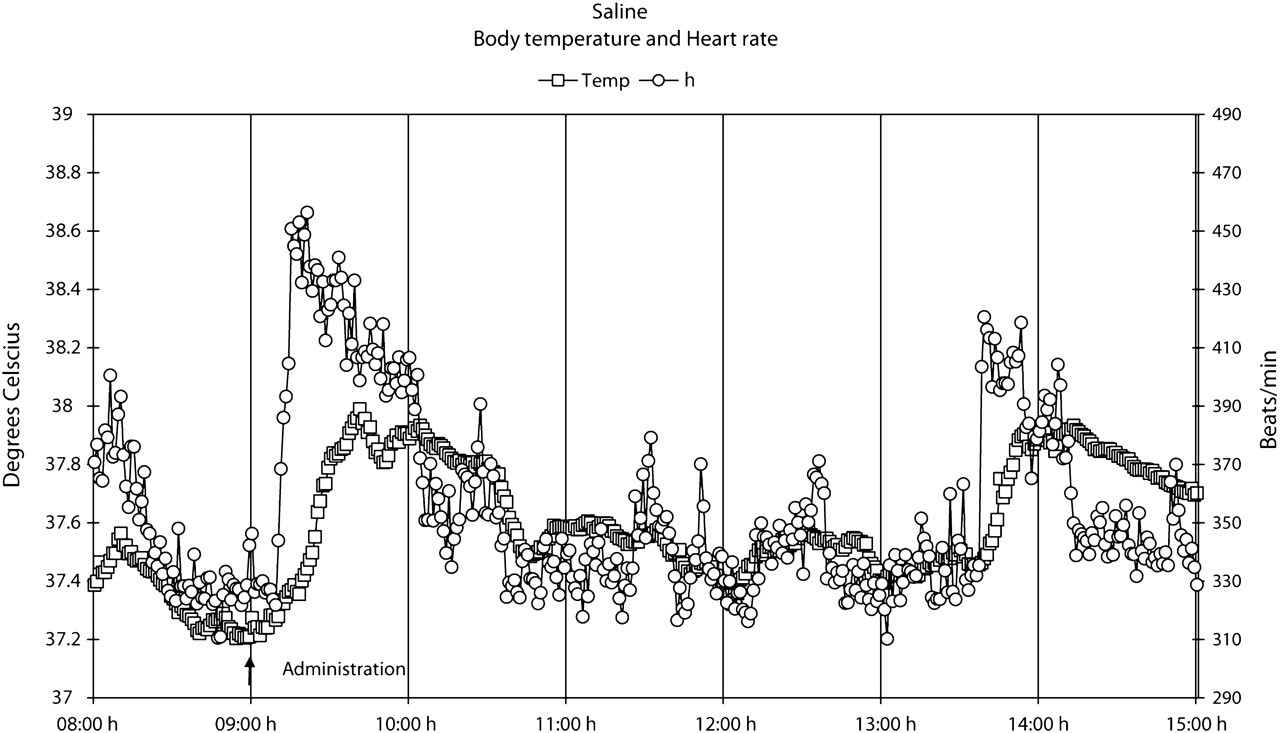
The administration procedure, as shown after the administration of the saline solution, rapidly induced an increase in heart rate, body temperature, locomotor activity and systolic blood pressure, all of which lasted for approximately 1–2 h (Figures 1–9). Buprenorphine was found to affect, to various degrees, body temperature, spontaneous locomotor activity, heart rate, d
All doses of buprenorphine, when compared with saline, induced a dose-dependent increase in body temperature, both early and late periods after administration (
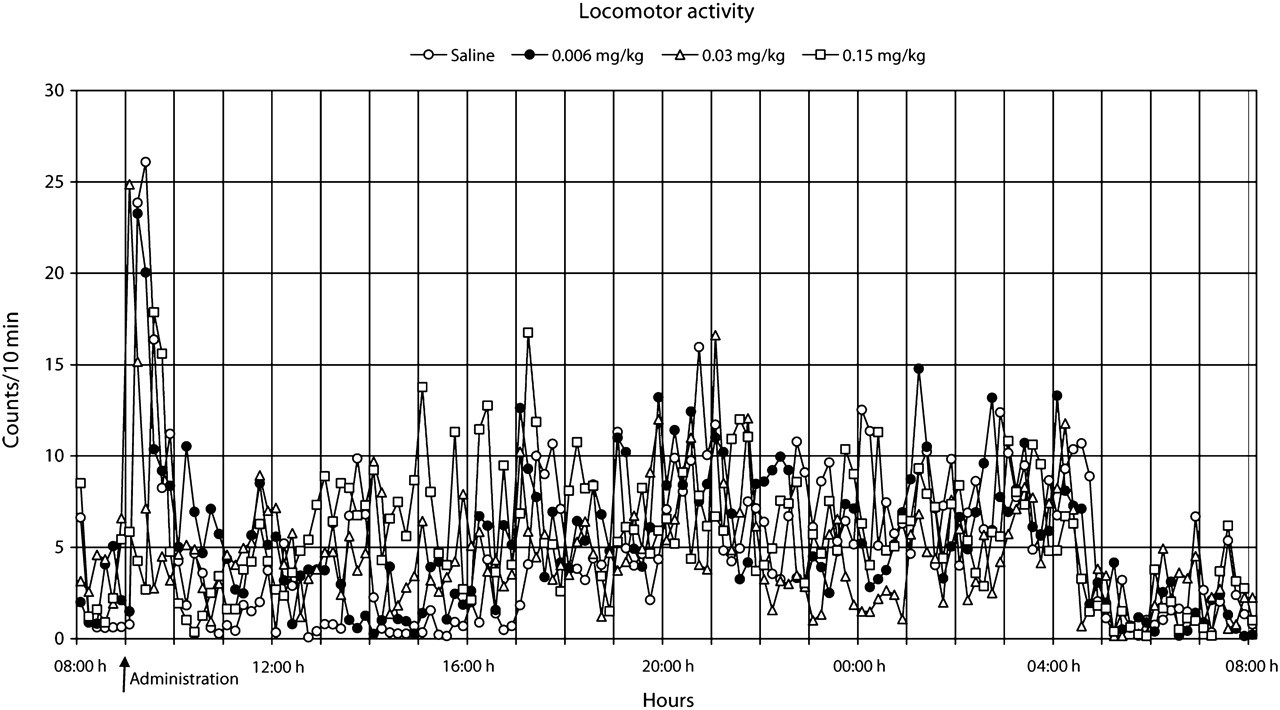
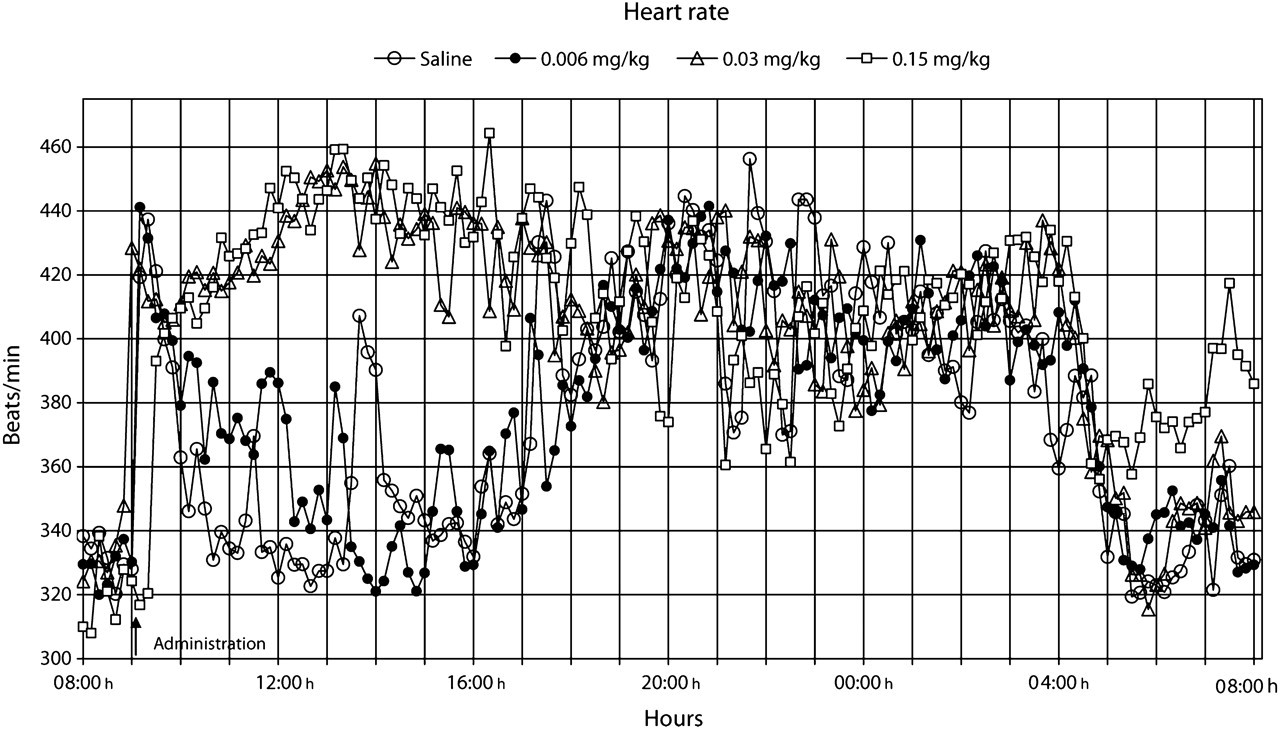
The heart rate normalized within 2 h after the administration of saline and the low dose of buprenorphine (Figure 3). However, a pronounced but similar increase (
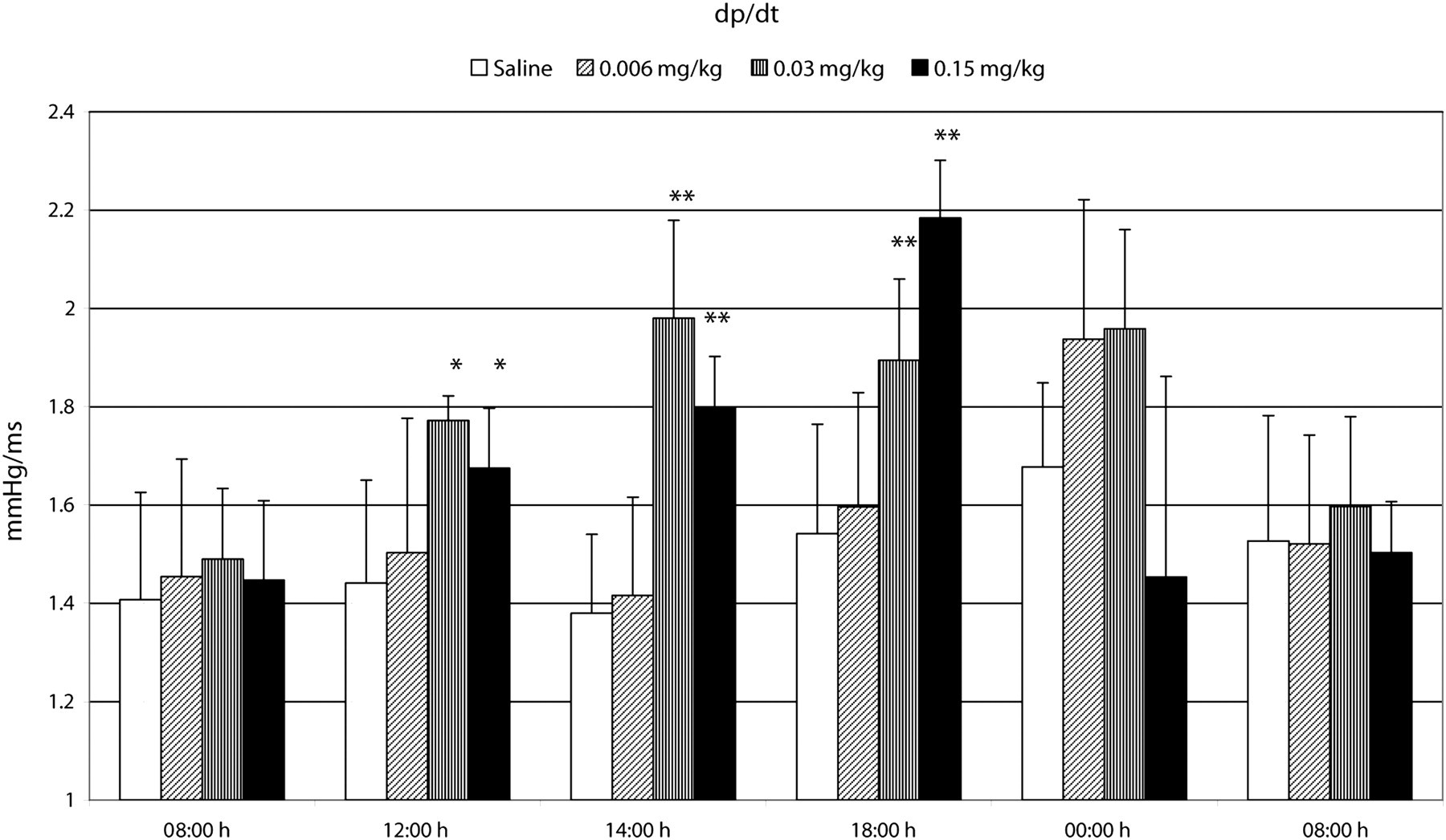
At the medium and high doses of buprenorphine, a substantial increase in d
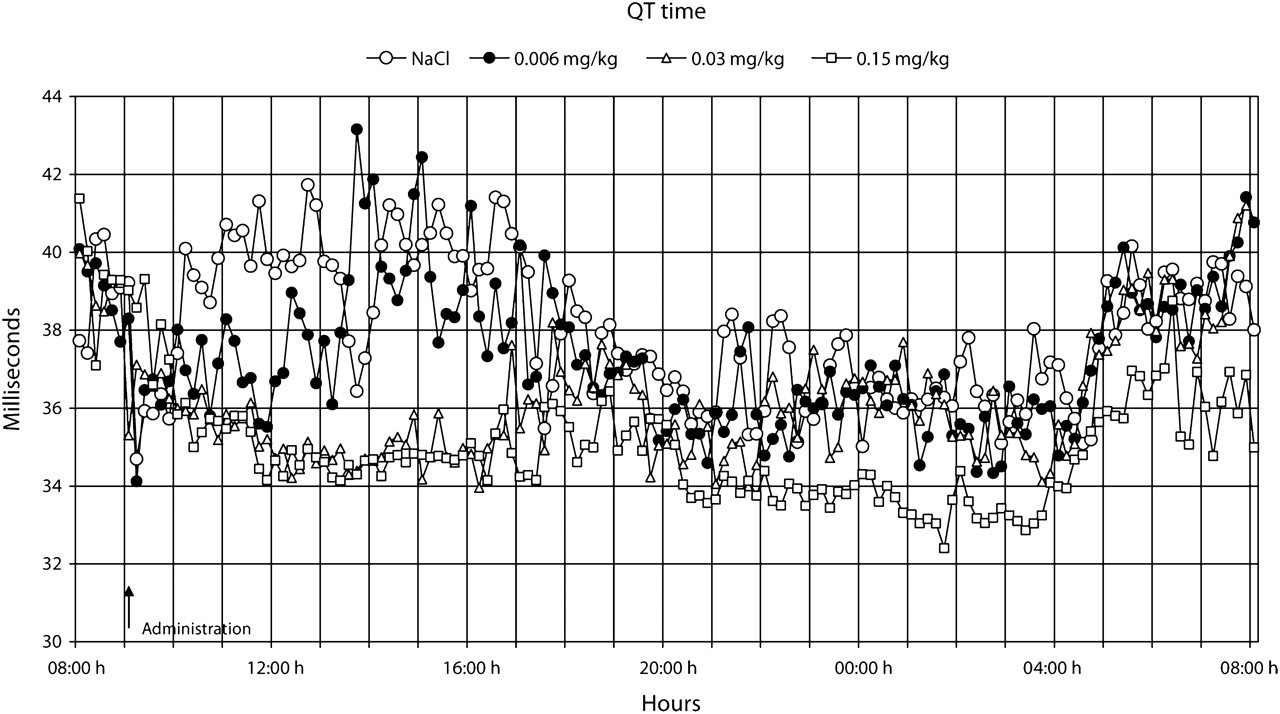
The QT time showed a dose-dependent decrease (
The results also showed that the increase in heart rate preceded the increase in body temperature, both after saline and buprenorphine administration (Figures 8 and 9). Moreover, during the 24 h following buprenorphine administration, increases and decreases in body temperature occurred even though no changes were observed in heart rate (Figure 9).
Discussion
During a 10 h period after administration of buprenorphine, the analgesic was found to cause, to various degrees, a dose-dependent increase in body temperature, heart rate, d
The administered doses of buprenorphine in this study were well tolerated and toxic/pharmacological effects were minor in comparison with those effects reported to be associated with a high intake of buprenorphine in which severe adverse effects and response patterns develop, including respiratory depression and death (Gueye et al. 2001). It was intriguing to note how well spontaneous locomotor activity, body temperature, heart rate and blood pressure changes followed each other in the saline-treated rats.
Buprenorphine seems to be a valuable agent for pain relief in a wide range of animal species when used in an appropriate manner (Roughan & Flecknell 2002). After buprenorphine administration, plasma concentration peaks at approximately 90 min with a half-life of 4–5 h (Sporer 2004). A single high dose of 0.5 mg/kg administered subcutaneously to rats produces analgesic effects over a 6–8 h period (Gades et al. 2000). Symptoms of buprenorphine in clinically treated patients persist from 12 to 24 h (Pickworth et al. 1993, Sporer 2004). This pattern corresponded well to the duration (i.e. approximately 10 h) of the presently observed treatment-related effects.
A new finding in this study was that buprenorphine induced dose-dependent hyperthermia in the rats. This effect was probably not immune-mediated because buprenorphine has not been shown to affect immune system functions (Gomez-Flores & Weber 2000). In addition, buprenorphine has been shown to increase the general activity of rats in some studies (Gades et al. 2000, Roughan & Flecknell 2002) but not in others (Hansen et al. 2002). In the present study, there was only a tendency of minor increase in spontaneous locomotor activity. Thus, the reason for the concomitant increase in body temperature and heart rate could not be explained by increased spontaneous locomotor activity. It is evident that the increase in heart rate immediately after buprenorphine administration precedes the increase in body temperature. These early effects, however, are probably related to a stress effect associated with the administration procedure. It was noteworthy that in the 24 h following buprenorphine administration increases and decreases in body temperature occurred even though no changes were observed in heart rate. Moreover, the medium and high doses of buprenorphine induced a similar response in heart rate but a dose-dependent increase in body temperature. It is therefore tempting to suggest that buprenorphine, in addition to possible cardiovascular effects, directly influences regulatory mechanisms of body temperature.
It is apparent from the literature that buprenorphine can either directly or indirectly affect the respiratory and cardiovascular system (Boachie-Ansah et al. 1989, Gueye et al. 2001). Buprenorphine has been reported to cause a decrease in heart rate, systolic and diastolic blood pressure, and stroke volume in anaesthetized dogs (Martinez et al. 1997). However, the effects of therapeutic doses of buprenorphine on these parameters have not been studied in conscious animals. We found that buprenorphine caused an early and dose-dependent increase in heart rate, d
Our study showed a clear dose-dependent increase in body temperature and associated heart rate responses. Although atrial fibrillation and ventricular tachycardia have been associated with prolongation of P-wave and QRS complex duration (Guidera & Steinberg, 1993, Lander et al. 1993), buprenorphine reduced the QT time in the present study. Moreover, no arrhythmias or effects on the ECG complex were observed at visual inspection.
Although only four animals per dose were used, the telemetry model in the conscious, unrestrained rat seems to be sensitive and demonstrate high predictive power in the evaluation of toxicological/pharmacological effects induced by buprenorphine. These results in the rat indicate that a single high dose of buprenorphine (0.15 mg/kg bw) may induce long-lasting effects, though this does not seem to involve changes in the ECG complex.
Footnotes
Acknowledgement
This study was supported by grants from the Faculty of Medicine, Uppsala University, Uppsala, Sweden.
