Abstract
Some experimental procedures are associated with placement of animals in wire-bottom cages. The goal of this study was to evaluate stress-related physiological parameters (heart rate [HR], body temperature [BT], locomotor activity [LA], body weight [BW] and food consumption) in rats under two housing conditions, namely in wire-bottom cages and in bedding-bottom cages. Telemetry devices were surgically implanted in male Sprague-Dawley rats. HR, BT and LA were recorded at 5 min intervals. Analysis under each housing condition was performed from 16:00 to 08:00 h of the following day (4 h light, 12 h dark). During almost all of the light phase, the HR of rats housed in wire-bottom cages remained high (371 ± 35 bpm; mean ± SD;
The use of solid-bottom cages with bedding for the housing of rodents was recommended by the
For a number of experimental situations, wire-bottom cages are considered methodologically preferable. Advantages referred to in the literature include that under fasting conditions, such cages eliminate the effects of food on metabolism and on the pharmacokinetics of an administered drug and coprophagy is prevented, eliminating exposure to drugs and drug metabolites in faeces and urine. 6 They facilitate the observation of copulatory plugs 7 and the collection of accurate data on food consumption. 8 They enhance sanitation, decreasing the exposure of staff to allergens, 8 and they eliminate contact by animals and staff with possible contaminants from bedding.4,5,9
Concerns over animal welfare in wire-bottom cages have however been raised. In fact, rodents held in wire-bottom caging are unable to behave normally in a species-specific manner (for example, nest building and foraging are impossible), and preference tests have shown that rats prefer a solid-bottom environment, especially when they are resting.8,10 However, preference testing alone, in the absence of other measurements of animal wellbeing, should not be the sole determinant for the selection of one husbandry condition over another.
Apart from welfare-related issues, housing conditions of animals might be related to physiological changes, which might be undesirable in some studies or which should, at least, be considered in the interpretation of results. It has been reported that there are no differences in terms of body weight (BW) and other physiological (i.e. haematological) parameters between animals housed in cages of both types.10–12 However, it has been demonstrated that the presence or absence of bedding affects thermoregulation and metabolic responses in rats. 13 It has also been reported that, when rats are housed in wire-bottom cages, hypoglycaemia, decreases in the size of the gastrointestinal tract and the liver, 6 painful foot lesions after animals reach a certain weight, 14 and even limb lesions and injuries can occur. Moreover, it was suggested recently that such housing conditions exacerbate the physiological effects of acute stress in rats. 15
Telemetry has been shown to be a useful tool for evaluating the impact of different housing conditions on rats.11,16,17 In fact, radiotelemetry avoids external interferences (i.e. handling) in the measurements of physiological parameters and allows continuous monitoring in unrestrained animals. 18 By this method, cardiovascular parameters have been previously evaluated in both normal and hypertensive rats housed in cages with wire mesh bottom,11,16,17 with differing results.
The goal of the present study was to evaluate potential alterations in various stress-related physiological parameters (heart rate [HR], body temperature [BT], locomotor activity [LA], BW and food consumption) in telemetry-implanted rats housed overnight in wire-bottom cages.
Materials and Methods
Animals
Male Sprague-Dawley rats, whose BWs ranged from 312 to 350 g, were used for the study. They were taken from a colony established at the animal facility at Palau Pharma SA (Barcelona, Spain) from founders that had been purchased from a commercial breeder (Harlan Laboratories Models SL, Barcelona, Spain). The animals were maintained under controlled ambient conditions (temperature, 22 ± 2°C; and relative humidity, 45–65%). The light cycle was 12/12 h: lights were turned off at 20:00 h and turned on at 08:00 h. Breeding and care of animals were undertaken in compliance with European Community Directive 86/609/CEE for the use of laboratory animals and with Autonomous Catalan law (Decret 214/1997). Experimental procedures were approved by the Animal Experimentation Ethical Committee of Palau Pharma SA.
From birth to the end of the experiments, all rats had unlimited access to water and autoclaved standard rodent diet (Harlan Teklad 2014; Harlan Laboratories Models SL).
Rats were maintained in polycarbonate cages (basal surface area: 500 cm2) with wood bedding (Lignocel® type S 8/15; Harlan Laboratories Models SL) prior to experiments.
Surgical implantation of telemetric devices
Ten animals were anaesthetized with an intraperitoneal injection of a mixture of ketamine (75 mg/kg) and xylazine (5 mg/kg). Analgesia was also administered with buprenorphine (0.05 mg/kg subcutaneously) after the surgery and then at 8 h intervals for three consecutive days. A radiotelemetry transmitter (TL11M2-C50-XT; Data Sciences International, St Paul, MN, USA) was surgically implanted in the peritoneal cavity of each rat. The surgical procedure was performed as previously described by Kramer
During the first three days after surgery, the rats were individually housed and the surgical wound was checked daily and disinfected with iodine povidone. On the fourth day, each implanted rat was placed together with a non-telemetrically implanted companion, of the same sex and age, in a bedding-floor cage, and these pairs of animals were maintained under the environmental conditions described above.
Animals were weighed daily during the first week after surgery to evaluate their recovery, and they were allowed to recover for a minimum of 15 days before experimental data were recorded. The electrocardiogram (ECG) of each animal was recorded with the telemetry system to assess the correct placement of electrodes (lead II position). One of the rats was excluded from the experiment at this time because the telemetry implant failed to provide data. Three of the remaining nine animals were discarded for measurements of HR because the electric signal did not match the standard rat ECG. However, all nine animals were used for measurements of LA and BT, since these two parameters were independent of the ECG recording.
Telemetric recording of data
A commercially available telemetry system (Data Sciences International) including receivers (RLA1000), telemetry transmitters (TL11M2-C50-PXT), a Data Exchange Matrix (BCM 100), an Ambient Pressure Reference (APR-1) and Dataquest™ ART™ version 2.3 software were used for recordings of ECG, HR, BT and LA parameters.
We programmed the acquisition software to record data at 5 min intervals, and the duration of each recording segment was 10 s.
HR was measured in beats per minute (bpm) and BT in degrees centigrade (°C). The Data Exchange Matrix provided the activity counts. As an animal moved in its cage, the telemetry signal transmitted to the receiver antennae varied in strength. When the signal strength changed by a specified amount, an activity count of one was generated as a measure of LA. The number of counts generated depended on both distance moved and the speed of movements. The activity data provided a strictly relative measure of LA and were not in any way related to absolute measurements of distance moved or spatial position. The system reported a value of six counts per minute for a single activity count within a 10 s sampling period.
Design of the study
The nine rats with telemetric devices, together with their non-telemetrically implanted companions, were consecutively housed in bedding-bottom cages and in wire-bottom cages. The time period analysed for each housing condition extended from 16:00 to 08:00 h the next day, as follows.
All cages were placed on top of the corresponding telemetry receivers. Recording of HR, LA and BT started at 16:30 h.
During this experimental procedure, the animals were always handled by the same caretaker.
Statistical analysis
For the parameters recorded by telemetry, means were calculated at each point of recording. Comparisons of means were made by Student's
The relationship between BT and HR was determined by Pearson correlation analysis.
Statistical analyses were performed with EPI-INFO, version 6.04 (Centers for Disease Control and Prevention, Atlanta, GA, USA), and Stat Plus 2007 (AnalystSoft, http://www.analystsoft.com/es/).
Results
None of the environmental parameters of the animal room (temperature, humidity, light cycle and air cycles per hour) varied along the three days of the experiment.
Heart rate (n = 6)
When animals were moved to fresh cages (bedding-bottom or wire-bottom cages), the mean HR of rats in both groups, as measured by telemetry, ranged from 400 to 410 bpm. Then, the HR of animals that had been transferred to the bedding-bottom cages started to decrease, reaching values of approximately 320–340 bpm, which were maintained until the end of the light cycle (Figure 1). The HR of rats that had been moved to wire-bottom cages remained consistently higher (371 ± 35 bpm; mean ± SD) during almost the entire light phase, being significantly different from the HR of rats that had been moved to the bedding-bottom cages (340 ± 29 bpm;
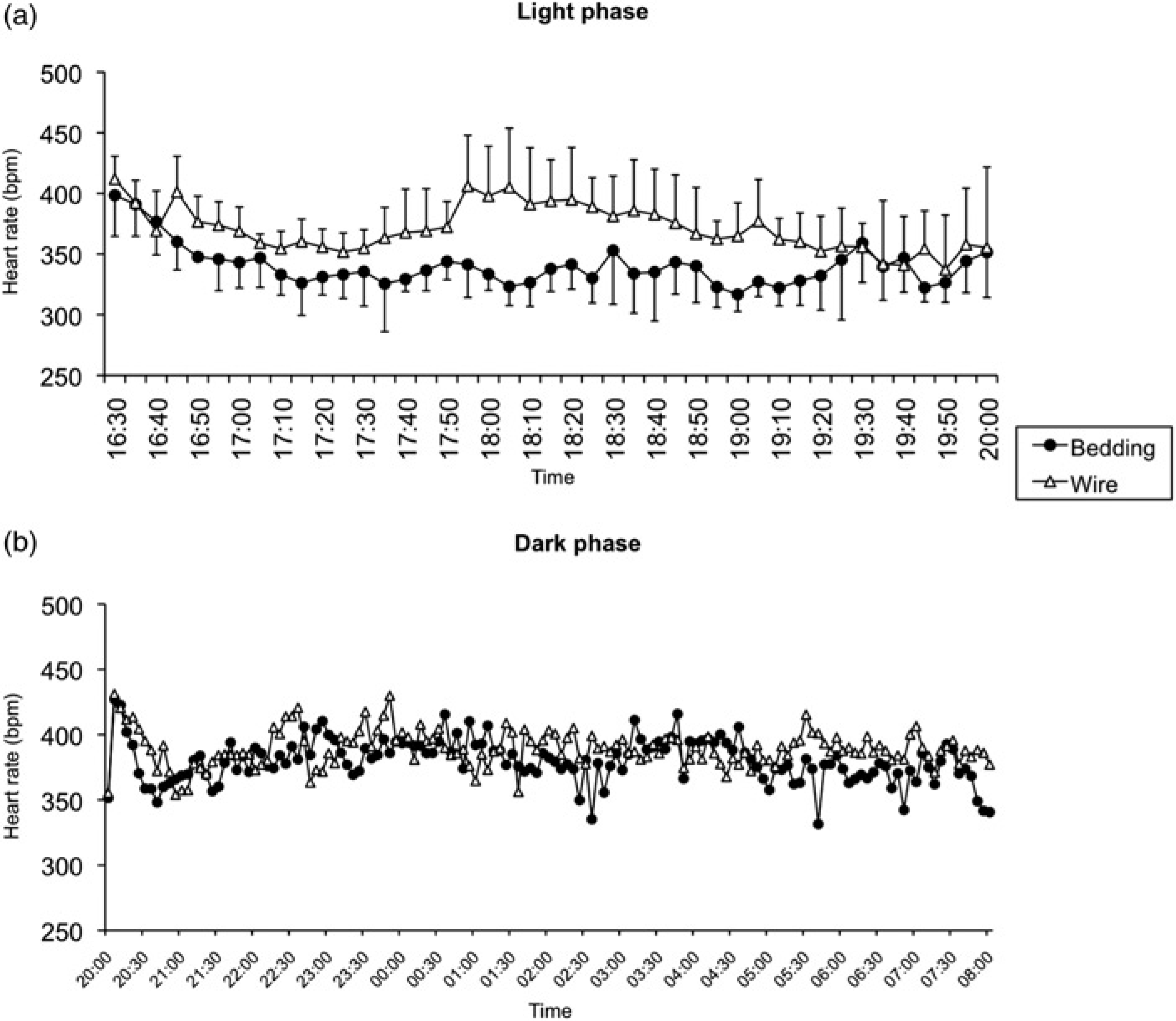
Graphic representation of the mean (with SD) heart rates, recorded during the light phase (4 hours; a) and dark phase (12 hours; b), of rats housed in wire-bottom and in bedding-bottom cages (
At the beginning of the dark phase, after the lights had been turned off, there was a sudden increase in HR in both groups for the first 5 min. The HR increased from 352 ± 37 to 427 ± 34 bpm in rats in bedding-bottom cages and from 355 ± 66 to 431 ± 2 bpm in rats in wire-bottom cages. Then the HR of all animals started to decrease. The rats in bedding-bottom cages seemed to recover more rapidly than the rats in wire-bottom cages. At the 30th minute of the dark phase, the former rats had a mean HR of 359 ± 23 bpm, whereas the latter had a mean HR of 395 ± 8 bpm. At the 40th minute, the mean values were 348 ± 23 and 372 ± 13 bpm, respectively. However, the differences at the two time points were not statistically significant. During most of the dark period, the HR of all rats was similar.
Body temperature (n = 9)
When telemetry recording was started at 16:30 h, a statistically significant difference in BT was observed between rats in bedding-bottom and rats in wire-bottom cages (38.6 ± 0.3 versus 38.3 ± 0.2°C, respectively;
At the beginning of the dark phase, BT was similar under the two housing conditions (approximately 37.5°C) and, when the lights were turned off, there was a sudden increase in BT in all rats, with BT rising to 38.2–38.3°C Afterwards, although changes in BT again exhibited similar patterns, there were several intervals during which the BT of the rats in wire-bottom cages was significantly lower than that of the rats in bedding-bottom cages (approximately 2, 5, and 8 h after the beginning of the dark phase; Figure 2).
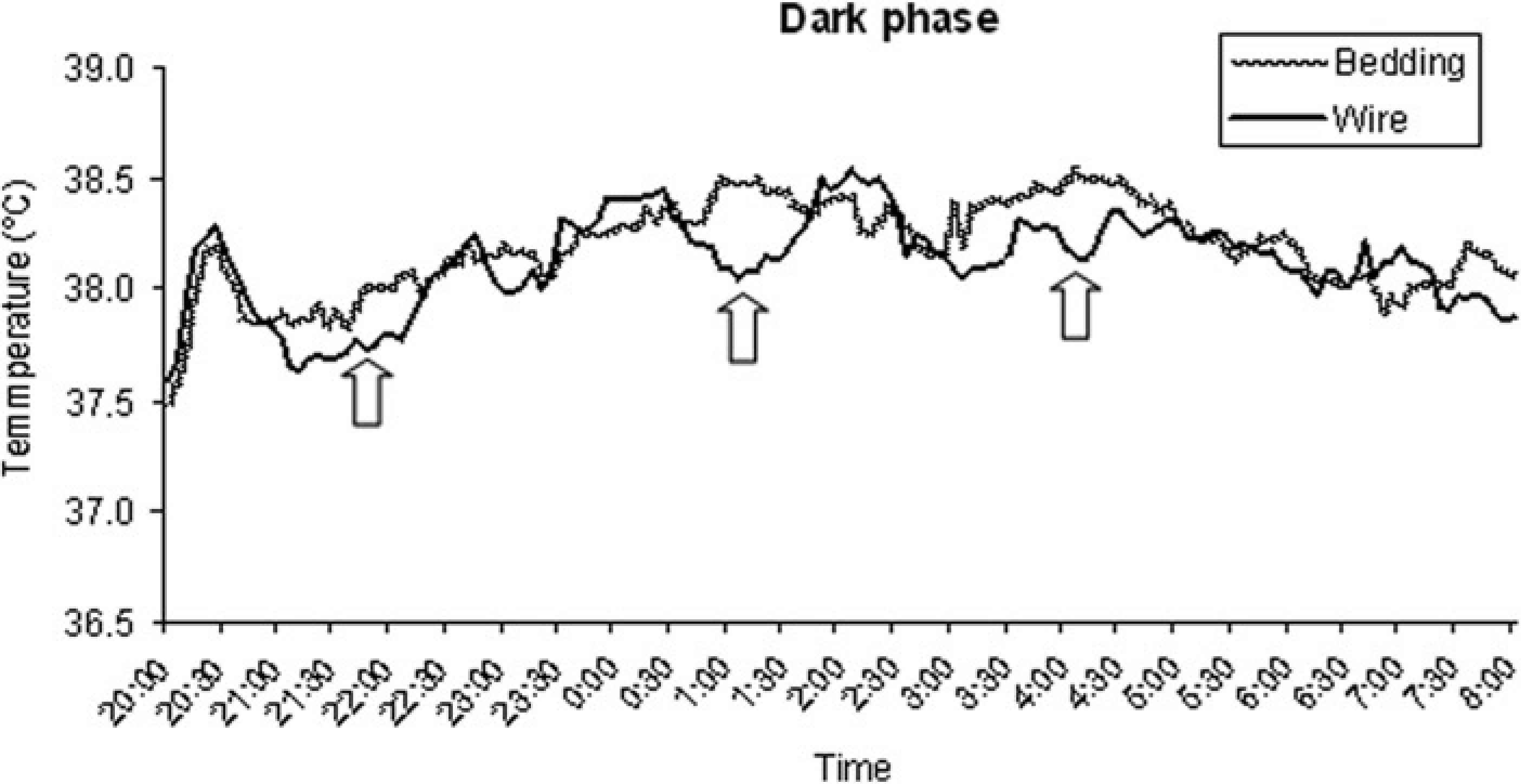
Graphic representation of mean body temperatures, recorded during the dark phase, of rats housed in wire-bottom and in bedding-bottom cages (
Statistical analysis of the relationship between HR and BT revealed a negative correlation for rats housed in wire-bottom cages during the dark phase (
Locomotor activity (n = 9)
Since there was a high degree of dispersion of LA data, we analysed this parameter statistically using a qualitative score (movement/absence of movement), as follows: counts equal to zero were assumed to be equivalent to the absence of movement, and values greater than zero were defined as movement. For the comparison of LA, all the samples (i.e. all the readings recorded by the telemetry system) were taken into account.
During the light period, 387 samples were evaluated for each housing condition. When the rats were housed in bedding-bottom cages, we recorded an LA index of 59% (229 movement counts and 387 total counts). This value was higher than that observed in the rats housed in wire-bottom cages, which was 52% (200 movement counts over 387 total counts), and the difference was statistically significant (
During the dark phase (Figure 3), 1305 samples were evaluated. The animals housed in bedding-bottom cages had an LA index of 81% (1053 movement counts and 1305 total counts). There was a significant difference between this value and the value for rats in wire-bottom cages, namely 71% (923 activity counts and 1305 total counts;
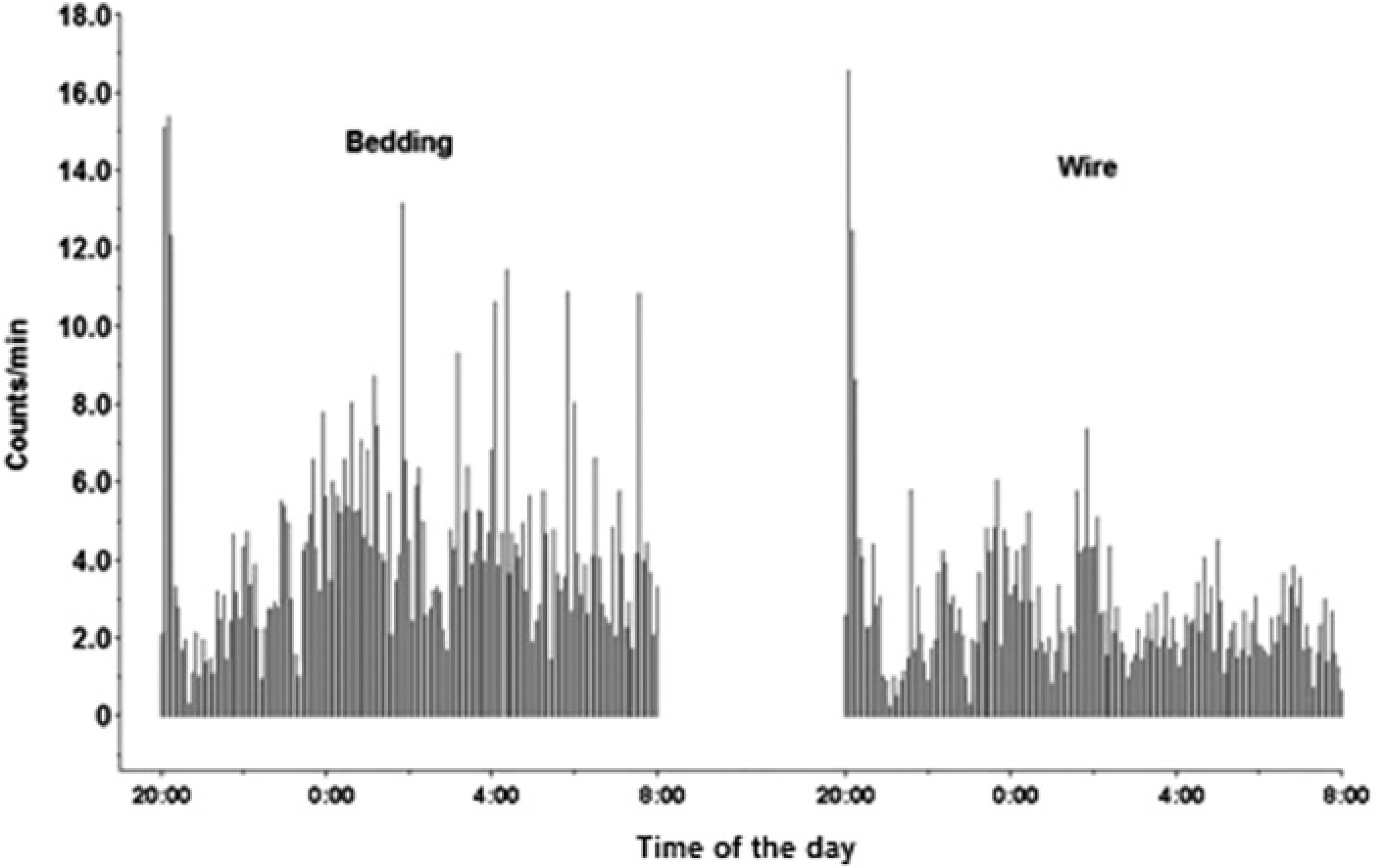
Graphic representation of the locomotor activity recorded during the dark phase in rats housed in wire-bottom and in bedding-bottom cages (
Body weight (n = 9)
The animals housed in bedding-bottom cages had a weight gain of 12 ± 2 g, which corresponded to an increase of 3.5% in BW. By contrast, when rats were housed in wire-bottom cages, they did not gain and, even, lost weight. The mean decrease in BW was 2 ± 3 g, which corresponded to a decrease of 0.6% in BW (Figure 4). There was a significant difference between the two groups (
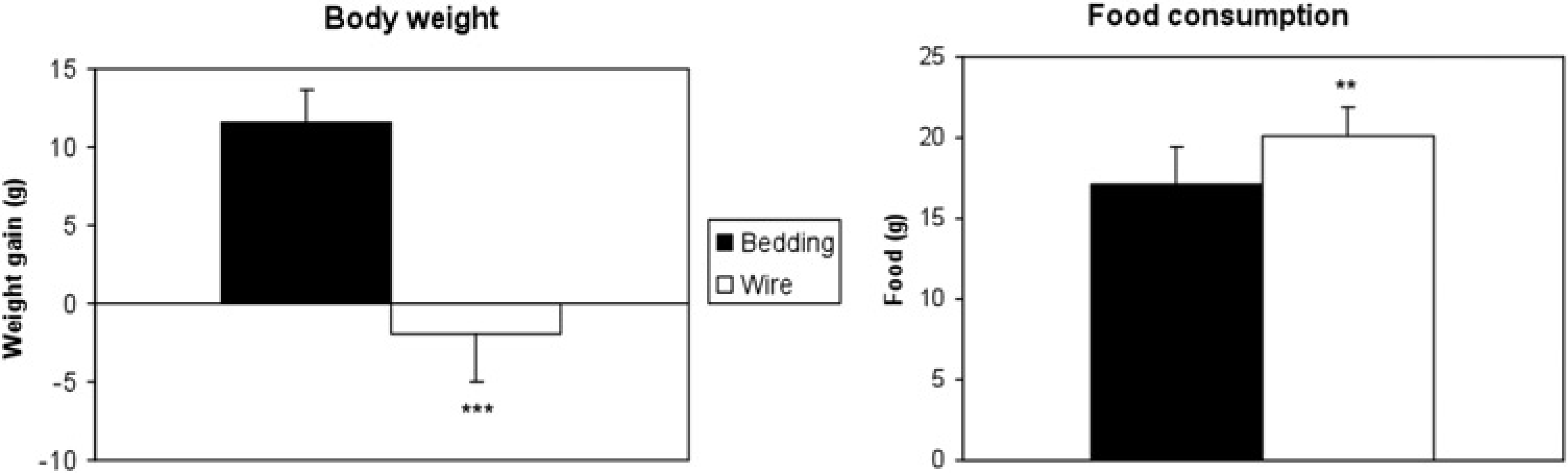
Histograms showing the means of the changes in body weight and of the food consumption (including pellet spillage) of rats housed in wire-bottom and in bedding-bottom cages (
Food consumption (n = 9)
A statistically significant difference was found between the two housing conditions. The total consumption of food, including spillage, was significantly larger when rats were housed in wire-bottom cages (20.2 ± 1.8 g) than in bedding-bottom cages (17.2 ± 2.3 g;
Discussion
Previous studies, designed to compare the effects on rats of solid- and wire-bottom caging, have focused on a variety of parameters, with two main objectives, namely to determine whether the use of grids for housing animals affects the wellbeing of animals, and to examine the reproducibility of results of toxicological studies under different housing conditions.
It has been reported that solid-bottom cages do not appear to introduce any significant bias during the routine clinical–pathological analyses that are commonly required in toxicological studies (complete blood count, serum chemistry, urinalysis, urine levels of creatinine and corticosterone, etc. 12 ). In addition, it has been demonstrated that the ability of technicians to detect clinical signs during toxicity tests is not reduced when rats are housed in solid-bottom cages with bedding, as distinct from wire-bottom cages. 21 Thus, some prejudices have been refuted, contributing to a move towards solid-bottom cages for rodents in recent toxicological studies.
It is widely accepted that solid-bottom caging, with bedding, provides an environment that is more suitable for the biological activities of rats than wire-bottom caging. However, it is necessary to define, by accurate measurement, the negative effects of wire-bottom housing on rat physiology. A previous study demonstrated that rats under the two housing conditions differed in their corticosterone profiles in response to acute restraint, suggesting that housing in wire-bottom cages might exacerbate the physiological effects of stress. 15
The study of HR changes in wire-bottom cages has generated a great interest, since this parameter may hint at stress.22,23 In fact, the detection of cardiovascular alterations by radiotelemetry has been the objective of previous studies with rats housed in different types of cages,11,16,17 although the results have been different. Rock
In the present study, we examined whether the housing of rats in wire-bottom cages overnight might alter parameters related to acute stress, such as HR, BT and LA, which might be expected to interfere with the results of some studies. As in previous works,11,16,17 we used telemetry-implanted animals to circumvent the stress related to the manipulation that is normally required for conventional measurements of BT and HR. However, we focused the analysis on the first hours after the change of housing, since this interval time was not deeply analysed in those previous studies, and it has a high interest in procedures that include a short-term stay of the animals over a grid floor. In addition, since the placement of animals in wire-bottom cages is frequently associated with fasting (to avoid the ingestion of bedding material and faeces), we monitored animals during the time period that is usual for such procedures (from 16:00 to 08:00 h of the following day). However, we did not deprive the rats of food in order to avoid any stress that was unrelated to cage flooring.
When animals were moved to fresh cages (bedding or wire-bottom) and telemetry was initiated, the mean HR of the two groups ranged from 400 to 410 bpm. This rate was probably higher than basal rates (not measured) as a consequence of handling and exploratory behaviour in a new environment. Then, the HR of rats that had been transferred to bedding-bottom cages soon decreased to 320–340 bpm, whereas HR of rats in wire-bottom cages remained elevated (Figure 1a).
At the beginning of the dark phase, there was a sudden increase in HR in all animals (Figure 1b). The decrease in and stabilization of HR in rats housed in wire-bottom cages were delayed compared with those of rats in bedding-bottom cages, in spite of the lower LA of the former (Figure 3). Moreover, in these animals, there was no decrease in HR in parallel with a decrease in LA, at the end of the dark phase, similar to that found in rats housed in bedding-bottom cages.
In summary, when animals were housed in bedding-bottom cages, the HR followed a predictable pattern with respect to the circadian cycle 24 and exhibited a predictable response to stimuli (i.e. turning off of the lights), with rapid recovery. However, when rats were housed in wire-bottom cages, the circadian pattern of HR was altered and the recovery of HR after a stimulus was retarded. In addition, these rats had a higher HR in spite of lower LA (Figure 3), which might implicate stress as the cause of the change in the pattern of HR. On the other hand, since rats in cages without bedding might have difficulties in thermoregulatory processes, the high HR together with low LA might be a consequence of this thermoregulatory stress.
Further studies are needed to clarify if the reaction that we observed is transient and a sign of acute stress, or if it persists as long as animals are in wire-bottom cages. As mentioned, previous studies in telemetry-implanted rats have shown different results about the influence of the cage type on HR after an acclimatization period.11,16
When telemetry recording was started, a statistically significant difference in BT was observed between rats in bedding-bottom and rats in wire-bottom cages. The animals had been handled and moved 30 min before the start of telemetric recording, and this might explain the initial difference. For most part of the time period analysed, BT was similar under the two housing conditions. However, when rats were in the wire-bottom cages, there were wider fluctuations during the dark phase (Figure 2). Peaks of BT matched increases in HR, although, to our surprise, we found a statistically significant negative correlation between the two variables when rats were housed in wire-bottom cages, specifically during the dark period. This result suggests a possible change in thermoregulatory processes, but multiple factors might have been responsible for this phenomenon (for example, the lower LA of the animals). Specific studies should be performed to examine this observation in greater detail.
As mentioned above, LA decreased dramatically when animals were housed in wire-bottom cages, in particular during the dark phase (Figure 3). Despite this lesser LA, these animals consume much more during the night than when they were housed in bedding-bottom cages (Figure 4). Other authors reported a similar result, 11 suggesting that this phenomenon might have been due to greater LA of animals in wire-bottom cages. Their hypothesis is not supported by the present study.
The LA of animals in wire-bottom cages might mainly focus on manipulation and consumption of food, since these animals could not perform other normal activities such as digging and foraging. In addition, as suggested by another author, 10 the increase in food consumption might have been due to greater wastage of food in the wire-bottom cages, with pellets falling through the gaps in the wire grid. In other words, not all the food that ‘disappeared’ might have been eaten, a possibility supported by the fact that animals in wire-bottom cages experienced weight loss (Figure 4). However, it is likely that this weight loss was a short-term effect since a previous study found no difference in BW between animals housed under the two conditions for 28 days. 12
As noted above, Rock
In conclusion, our results show that housing of rats, overnight, in wire-bottom cages leads to immediate alterations in some parameters that may hint at stress, such as HR. Our findings provide important objective information that can be used to support the choice of housing conditions for rats used in biomedical research.
Footnotes
Acknowledgement
The authors thank Eric Rieux (Data Sciences International) for his invaluable assistance during the course of this study.
