Abstract
As part of routine husbandry, an increasing number of laboratory mice receive nesting material in addition to standard bedding material in their cages. Nesting material improves health outcomes and physiological performance in mice that receive it. Providing usable nesting material uniformly and efficiently to various strains of mice remains a challenge. The aim of this study was to determine how bedding particle size, method of nesting material delivery, and processing of the nesting material before delivery affected nest building in mice of strong (BALB/cAnNCrl) and weak (C3H/HeNCrl) gathering abilities. Our data suggest that processing nesting material through a grinder in conjunction with bedding material, although convenient for provision of bedding with nesting material ‘built-in’, negatively affects the integrity of the nesting material and subsequent nest-building outcomes. We also found that C3H mice, previously thought to be poor nest builders, built similarly scored nests to those of BALB/c mice when provided with unprocessed nesting material. This was true even when nesting material was mixed into the bedding substrate. We also observed that when nesting material was mixed into the bedding substrate, mice of both strains would sort their bedding by particle size more often than if it were not mixed in. Our findings support the utility of the practice of distributing nesting material mixed in with bedding substrate, but not that of processing the nesting material with the bedding in order to mix them.
Provision of nesting material as a form of environmental enrichment for laboratory mice is not a new concept.1–4 Nesting material allows for the expression of naturalistic behaviors such as material gathering, material processing, and nest-building 5 which may also be used as a measure of well-being. 6 Once built, nests provide a favorable thermal environment in the cage, leading to improved health outcomes and physiological performance.7–9 A 2005 survey of 22 National Institutes of Health (NIH) animal facilities indicated that provision of nesting material was the most prevalent enrichment item for mice in the USA. 10 Furthermore, since EU regulations now mandate the provision of enrichment for all laboratory animals, 11 companies conducting business within the EU will have to institute formal enrichment programs to meet these requirements.
Enrichment programs for mice must take into account how a mouse views and interacts with the provided enrichment as well as the feasibility and cost-effectiveness of the material and its distribution to cages. 10 In a large facility, provision of nesting material to cages on an individual basis can be both time-consuming and a source of unintentional variation in the amount of material supplied. As personnel time increases, so does cost; as variation in quantity of nesting material increases, so does the likelihood that at least some cages will receive inadequate amounts. One method of circumventing these challenges would be the addition of nesting material to the bedding substrate by the bedding manufacturer. The manufacturer could add nesting material during the processing stage, which would allow for the mixing and processing with other bedding components. In the case of wood-based beddings, this would involve passing the nesting material through various wood chipping stages. With nesting material provided at the same time as bedding to each cage, benefits would include distribution uniformity of material, reduced personnel costs to the facility, and additional opportunities for gathering or foraging behavior.
Not all mice are equally skilled nest builders.2,12 C3H mice, in particular, are reported to struggle with the gathering component of nest-building.12,13 With this in mind, it becomes plausible that nesting material that has passed through the bedding chipper for integration into bedding material would be less beneficial for C3H mice and other ‘gathering challenged’ mouse strains or stocks. These reported differences in the ability of a particular mouse strain to complete a part of the nest-building behavior chain make testing an altered delivery method of nesting material allocation extremely important.
Related to the gathering and processing of nesting material are the type, size, and texture of the bedding material. Mice have been shown to prefer aspen chip bedding over corncob for both sleeping and waking hours.
14
Additionally, they prefer larger and more fibrous pieces of material rather than chips,1,15 and aspen shavings have been associated with improved breeding success compared with mice housed on chips.
16
Bags of aspen bedding, while generally uniform, will contain shavings of various sizes as well as small chunks of aspen wood. Anecdotally, we have observed mice selecting the larger pieces of aspen bedding and chunks of wood, processing them, and weaving them into the nest (Figure 1a). One study suggested that smaller flakes of bedding are less likely to be incorporated into a nest,
16
whereas larger pieces may provide mice with the opportunity to forage for preferred nest-building materials as well as allowing mice that excel at processing to engage in this behavior without hindering the nest-building of those animals that struggle with this part of their behavior.
(a) This is an example of a cage with large flake bedding and unprocessed nesting material. It illustrates the variability in flake size for the large flake treatment and how some mice incorporated the large flakes into their nests. (b) This image shows the consistency of particles in the small flake and processed nesting material treatments. The variability in flake size is much less and the nesting material consists of mostly shorter pieces of similar shape and size as the aspen flakes.
Laboratory mice build the most complex nests when provided with long paper strips, likely because these are similar to the plant material that wild mice would use to build nests. 5 Modifying the length and/or texture of the strips may decrease their usefulness as nest-building substrates since these modifications may make the strips appear or behave less like the grasses used as typical nest substrates by wild mice. Testing both processed and unprocessed nesting materials become relevant, as processing alters both the length and integrity of the strips. It should be noted that, in this case, processing refers only to nesting material that has been passed through a chipper. Autoclaving, another form of processing, has been successfully used with bedding material with few reported adverse effects. 17
The aim of this study was to determine the effect of bedding particle size, processing level of nesting material, and the method of nesting material allocation on nest-building success in mice reported to have different gathering behavior abilities. We hypothesized that bedding particle size, the level of processing needed to combine nesting material with the bedding, whether or not the nesting material was mixed in with the bedding, and strain would all have significant effects on the complexity of the nests built. We predicted that mice that were poor at gathering materials would build lower scoring nests when nesting material was mixed in with the bedding, regardless of whether the mixing occurred before or after normal bedding processing. We also predicted that nesting material that had not been processed would allow mice to build more complex nests, as well as that mice given large flake bedding would build higher scoring nests by incorporating those larger flakes into the nest. Finally, we predicted that mice with higher scoring nests would, due to increased thermal stability, either consume less food and maintain a steady body weight, or consume more food and gain more weight than those mice with lower scoring nests.
Animals, materials and methods
All work was conducted at the Charles River’s Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC)-accredited facility in Wilmington, Massachusetts, USA and approved by the Institutional Animal Care and Use Committee (IACUC, P03292009). At the start of study, animals were free of a list of common mouse infectious agents; further details may be found at http://www.criver.com/files/pdfs/rms/hmsummary.aspx. Food and water consumption and overall health status of all mice were monitored daily by trained Charles River staff, and no adverse environmental conditions or health outcomes were noted.
Factorial treatment combinations and number of replicates per combination.
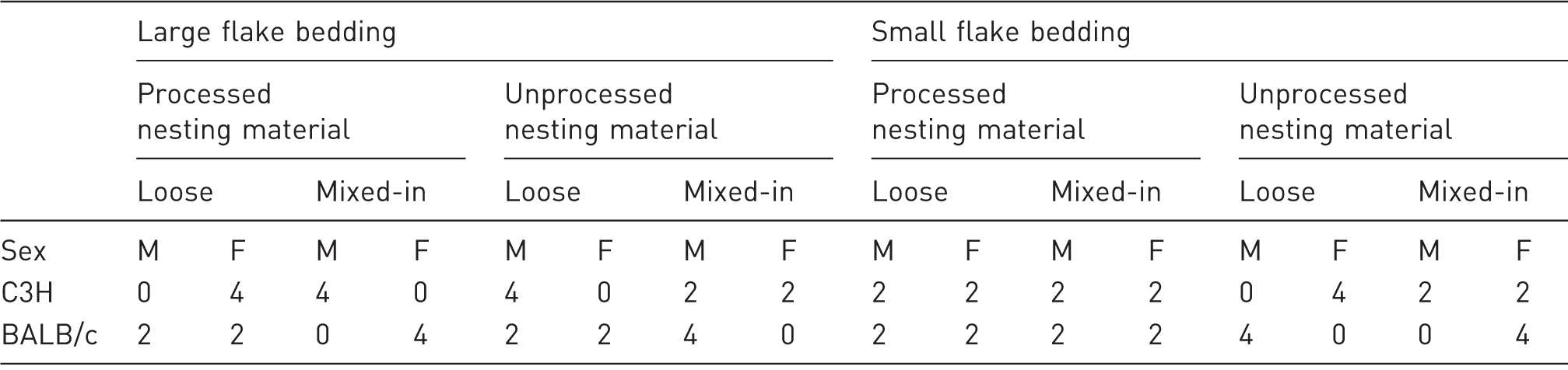
Experimental unit/replicate is cage.
Mice were acquired from Charles River (Raleigh, NC, USA) at eight weeks of age. Immediately after transport, they were housed in disposable individually ventilated cages (Innovive, San Diego, CA, USA; 37.3 [L] × 23.4 [W] × 14.0 [H] cm), evenly distributed and balanced across the rack. Mice were allocated to treatment combinations and rack position using the random integer set generator found at random.org. Food (5L79; LabDiet, St Louis, MO, USA) and ultrafiltered hyperchlorinated water (via water bottle) were provided ad libitum. The light cycle was 12:12 light:dark (on at 06:30 h, off at 18:30 h), humidity was maintained between 30 and 70%, air changes per hour (ACH) on the individually-ventilated rack were 50 ACH, and temperature ranged between 19 and 22℃. Room level lighting was maintained according to the Guide recommendations at less than 325 lux. 18 Animals were euthanized using a custom-built machine that added 100% CO2 to room air at a volume of 20% chamber displacement per minute and a 5 min hold time after chamber levels reached (calculated) 100%. Mice remained in their home cage for this process. Large flake bedding consisted of kiln-dried aspen shavings processed once through a chipper at the bedding supplier (NEPCO, Warrensburg, NY, USA). Small flake bedding consisted of kiln-dried aspen shavings processed twice through the chipper. Processed nesting material (EnviroDri; Shepherd Specialty Papers, Watertown, TN, USA) was passed through the chipper; unprocessed nesting material was used as is from the manufacturer (Figures 1a and 1b). ‘Mixed’ was nesting material uniformly mixed into the aspen bedding, while ‘loose’ was nesting material provided in a pile at one end of the cage. To control for amount of bedding and amount of nesting material provided in the cages, we measured three 75 g samples of processed nesting material + bedding and three 75 g samples of unprocessed nesting material + bedding, as supplied by the manufacturer. We then separated the nesting material from the bedding, and found a mean amount of nesting material in those samples of 7 g. Based on these observations, the loose treatment cages were provided with 68 g of aspen bedding and 7 g of nesting material.
Mouse body weight and food usage were measured at the beginning of the study and weekly during cage change for two weeks. Nests were scored using a 0–5 scale.
5
Briefly, a score of 1: manipulated material but no evidence of a central nest site; 2: a flat nest; 3: a cup nest; 4: an incomplete dome; 5: a complete and enclosed dome. Nests were scored (during daylight hours at 13:00 h) on days 1, 2, 3, and 6 over the two weeks. Therefore, each cage was scored eight times over the course of the two-week study period. At the time of nest scoring, sorting was also documented (Yes/No). Sorting was defined as non-uniform distribution of aspen bedding substrate or nesting material, with either nesting material or larger aspen flakes concentrated in a particular area of the cage (Figure 2). Nesting material and bedding were replaced during cage change (which occurred weekly). Nest scores were averaged over a single week. Sorting is reported as the total number of times sorting was observed at the time of nest scoring ( A cage displaying the sorted bedding substrates, with smaller aspen flakes on the left side of the cage and larger flakes on the right.
Data were analyzed using JMP statistical software (JMP®, Version 10; SAS Institute Inc, Cary, NC, USA). A full factorial general linear model (GLM) analysis of the following factors: strain, nesting material processing level, bedding material size, and allocation of nesting material was conducted for nest score, food usage, body weight change, and presence or absence of sorting behavior. Strain, bedding type, allocation of nesting material, and processing level of nesting material were nested within cage. Cage average body weight was included as a covariate when analyzing food usage. Nest scores were added as a covariate for analyzing food usage but were not significant, and so were removed from the model
Results
Effects that are not reported were not significant for that analysis.
Nest score
A significant three-way interaction between strain, bedding type, and processing (GLM, Mean nest scores and the interaction between processing level, strain, and bedding flake size. Asterisk indicates significant differences (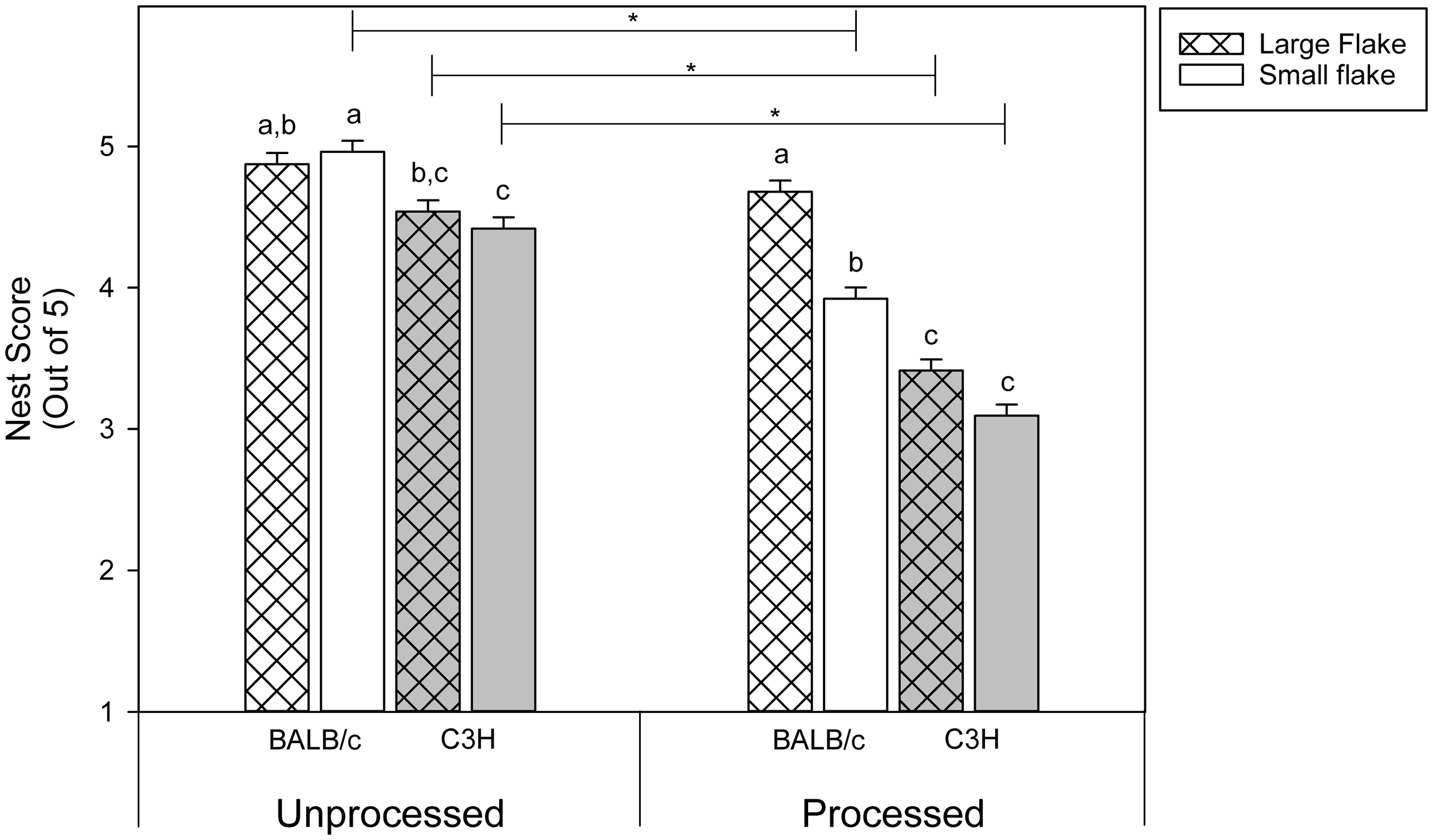
Food use
Analysis of food use showed a significant interaction between week and strain (GLM, Weekly food use and the interaction between week and strain. Asterisk indicates significant differences (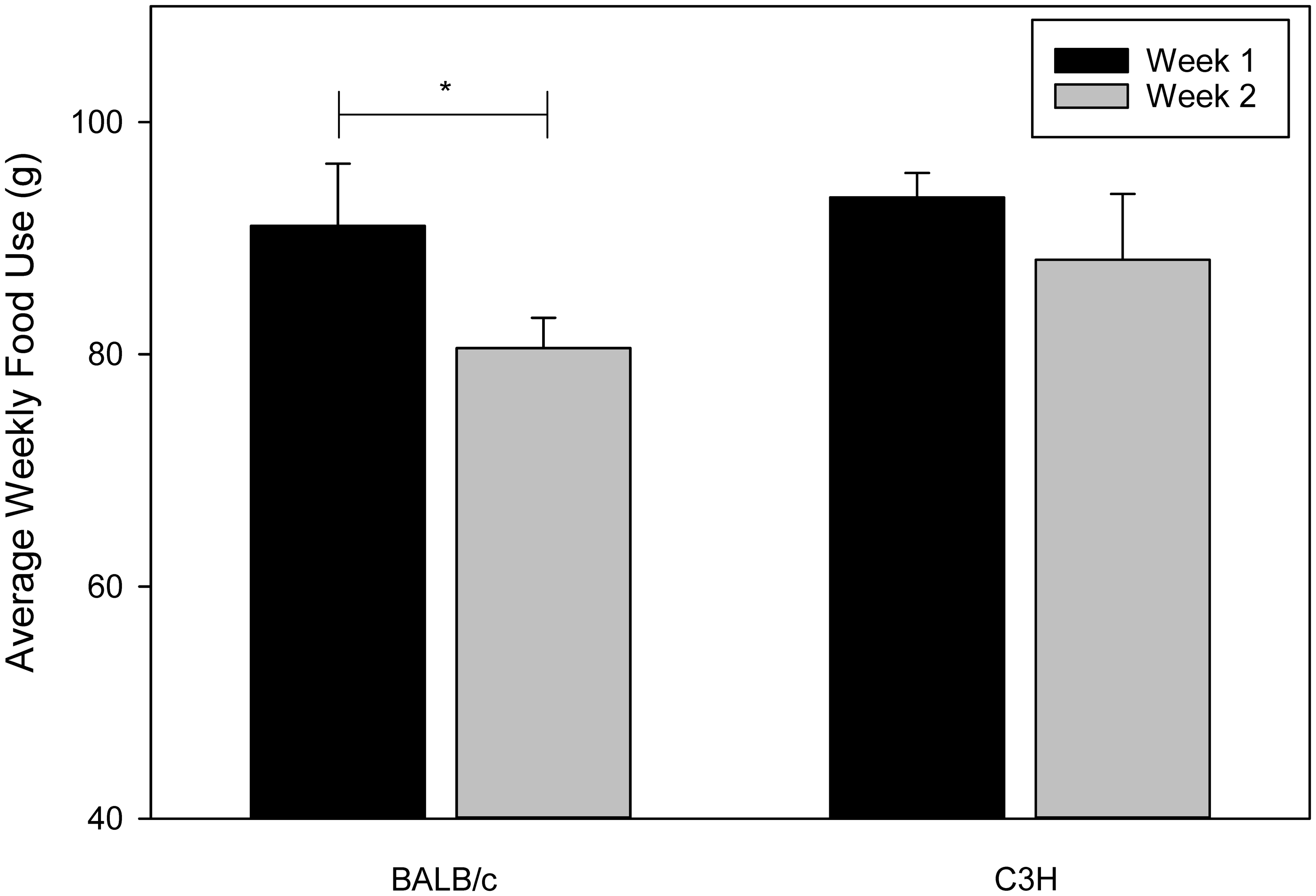
Body weight change
A highly significant main effect of week (GLM,
There were no differences in weight gained by C3H mice regardless of the combinations of nesting treatments (test slices, Average weekly body weight changes in BALB/c mice only and the interaction between bedding flake size, nesting material processing, and nesting material allocation. There were no differences found within the processing treatments for BALB/c mice. There were no differences in levels within the C3H mice therefore these are not graphed, for simplicity. Asterisk indicates significant differences (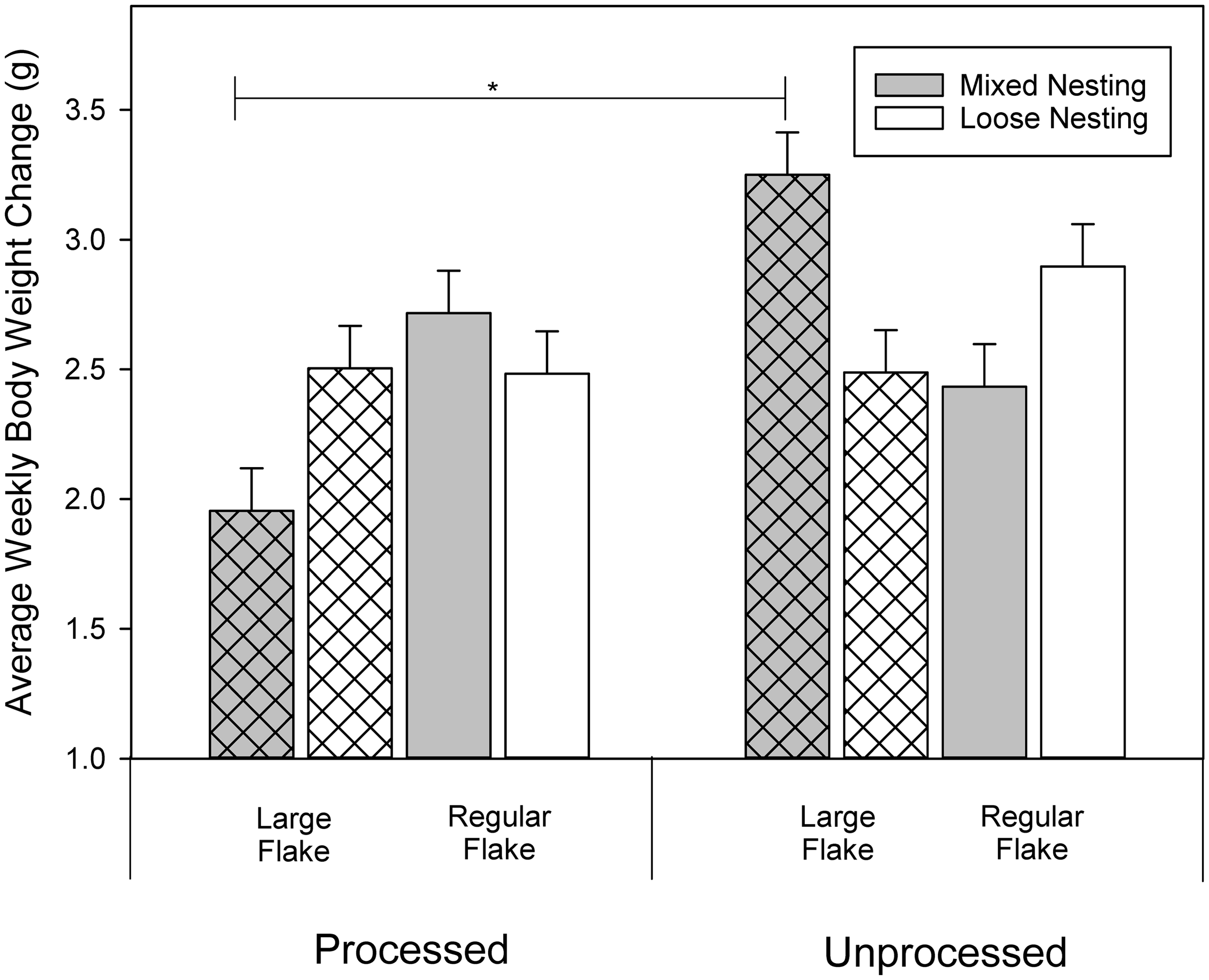
Sorting
Strain had a significant main effect on sorting (GLM, Number of observations of sorting made (out of a total of 4) and the interaction between bedding flake size, nesting material processing, and nesting material allocation. Asterisk indicates significant differences (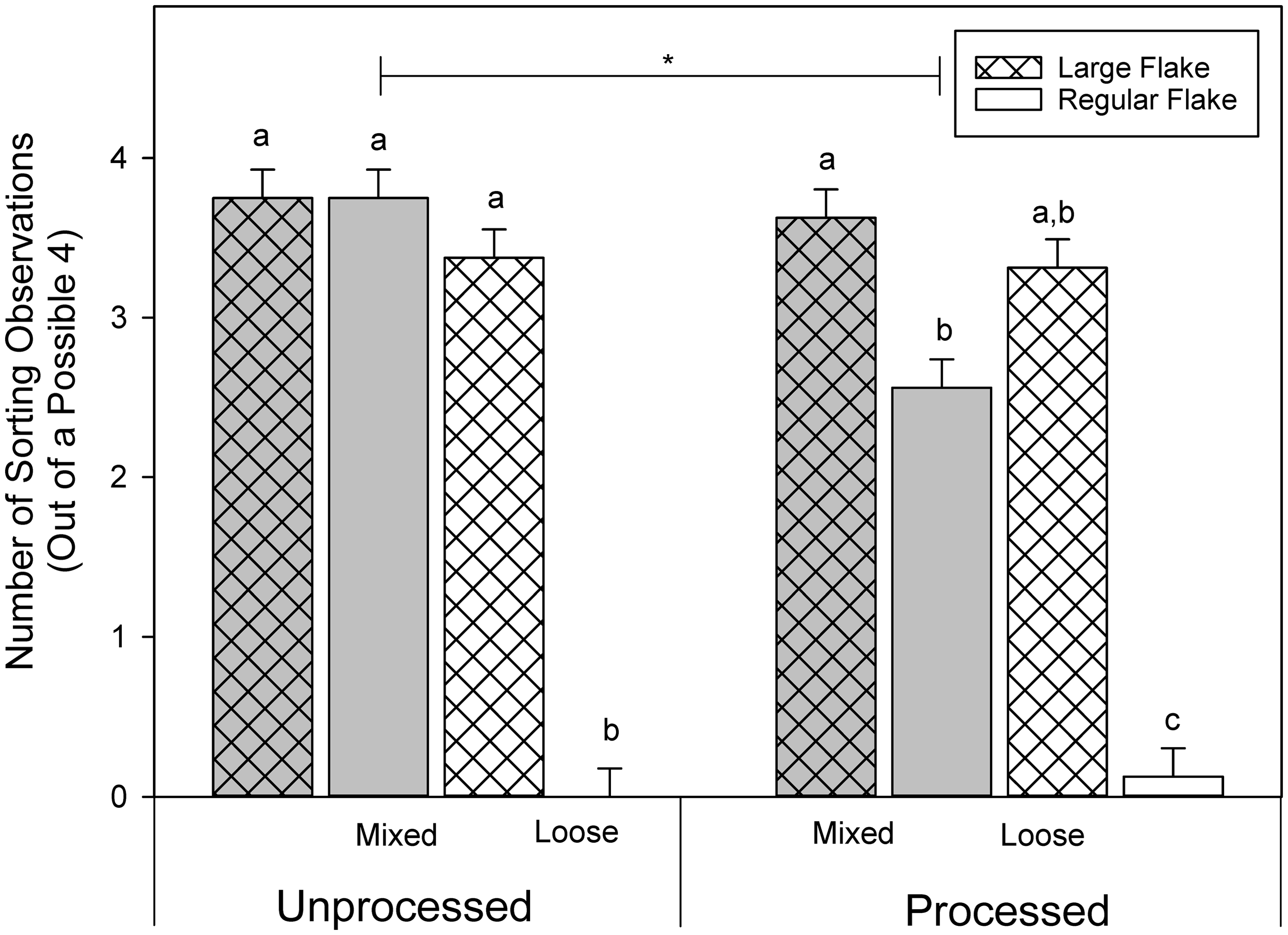
Mice were observed to be sorting with the same frequency regardless of processing or delivery when housed on large flake bedding. When housed on regular flake bedding and given loose nesting material, whether processed or not, mice sorted less than those housed on large flake bedding. Mixing in processed nesting material also significantly reduced sorting for mice with regular bedding (
Discussion
Our prediction that mice given unprocessed nesting material would build higher scoring nests was borne out, except for BALB/c mice with large flake bedding which had similar nest scores regardless of the processing level. Perhaps BALB/c mice are better at incorporating large bedding flakes into their nests, which allows them to compensate for the compromised infrastructure produced in processed nesting material. When passed through a chipper strips of nesting material are substantially shortened which presumably complicates the process of weaving them into a nest. Additionally, BALB/c mice are reported to be excellent nest builders 8 and this may be reflected in their ability to compensate for shorter pieces of nesting material.
More surprising, however, was the finding that the interaction between strain and nesting material delivery was not significant for nest scores. C3H mice built similarly scoring nests with unprocessed nesting material regardless of the material delivery method. This contradicts observations in other studies where C3H mice were found to gather less material than other mouse strains, and their latency to collect material was slower.3,12 Rock et al. noted, however, that with repeated trials C3H mice improved their material gathering abilities; perhaps the prolonged exposure to the mixed material with no time limit compensated for the differences in gathering speed. The overall lower nest scores of C3H mice seem to be driven by the dramatically reduced scores with processed nesting material, so it would appear that the altered form of the nesting material has a greater effect than how it is delivered.
The majority of nesting research tests either nest scores as a whole5,8,9,12 or gathering abilities,2,3,13 and there has been few to no studies describing the actual process of nest construction, which involves a series of complex behaviors including foraging, gathering, sorting, processing, weaving, and digging. Although some observations may indicate that C3H mice are ‘poor’ gatherers, and therefore ‘should’ have lower nest scores, those conclusions are not supported by this study. If gathering were the weak spot in the process for C3H mice, we should have seen lower nest scores for C3H mice with mixed-in nesting material as compared to those with loose material – instead there were no differences based on nesting material allocation. We would argue that this gathering behavior is quite similar, if not indistinguishable from, foraging behavior. While foraging is typically thought of as being exclusively about searching out and consuming or collecting food items, there are multiple references to it in a non-food context. Sociologically and psychologically, people may forage for information 20 and use internally generated cognitive solutions. 21 Ecologically, social insects,22,23 beavers, 24 and woodrats 25 have all been documented to forage for nest-building items. These authors have, apparently successfully, applied foraging theory to non-food acquisition in all of these studies. This is unsurprising, considering that in many cases the differences between foraging for food and foraging for nesting materials are discernable only by the item(s) acquired and the name allocated to it (foraging versus gathering).
Interestingly, while foraging enrichment is commonplace in the zoological world,26–28 and not uncommon in the research primate world, 29 it has been infrequently studied in laboratory mice. It may be plausible that one reason the laboratory animal community has not spent much time focusing on foraging enrichment for mice is the Guide’s requirement of placing feeders ‘to minimize contamination with urine and feces’. 18 Anecdotally, investigators and animal care staff raise concerns that mice provided with novel foods will then preferentially consume those foods rather than their normal lab diet. With these potential obstacles and lack of solid information about the provision of food foraging enrichment to mice, perhaps we can still provide an opportunity for mice to forage, but for nesting material instead. This would eliminate the concerns about diet changes and food contamination, as well as providing an additional opportunity for mice to control their thermal environment when they utilize nesting materials.
We know from previous work that food use is linked to thermoregulation through the use of nesting material, and that mice with nesting material consume less food than those without. 9 This led us to expect that mice with lower nest scores would have higher food use. However, nest score was not a significant effect in our analysis of food use. Additionally, only BALB/c mice experienced any changes in food use, and they ate less in week 2. While we cannot say for certain, it is possible that this is a reflection of the generally higher nest scores that BALB/c mice had and their subsequent improved thermoregulation. This may not have been seen until week 2, because during week 1 they were busy growing and gaining weight. As weight gain slowed in week 2 it is plausible that food consumption decreased along with it.
No a priori predications regarding sorting behavior were made; it was a behavior that we observed early in the study and subsequently included in our data collection. BALB/c mice sorted more than C3H mice, which may be a reflection of previously observed differences in their gathering techniques. BALB/c mice have been reported to gather large amounts of nesting material, 2 while C3H mice have been described as sleeping where the nesting material was placed, rather than gathering and building their own nests.3,12 This increased interaction with the bedding substrate, while gathering nesting material, by BALB/c mice may have acted as a form of behavioral priming for sorting behavior. This could also be a behavior that is improved or reinforced with practice, as has been previously proposed with gathering/foraging, 12 since all mice sorted more in the second week of testing. Clues as to the motivation for this behavior may be due to the heterogeneity of bedding particle size. Mice on large flake bedding sorted with the same frequency, no matter the level of processing or delivery method of nesting material. This suggests that large flake bedding, and its variety of flake sizes, may be a stimulus for eliciting sorting behavior. If, as we are beginning to suspect, sorting is a refinement of gathering behavior, this would then be a logical progression – as a mouse looks for appropriate nesting material, it would be more efficient to collect desired (larger, manipulable) materials in one location, separate from the undesired (smaller) items. Mice on regular flake bedding with loose nesting material rarely sorted, while those with mixed-in nesting material sorted more, particularly when the nesting material was unprocessed. It would seem that, in the absence of large flake bedding, mice do not sort unless primed to do so by having to forage in their bedding for nesting material. This is a highly relevant observation because by using the typical practice of supplying nesting material in a pile in the corner of the cage we may be missing an opportunity to provide further enrichment for animals, particularly if they are easily ‘primed’ to perform this sorting behavior. However, mice with processed material may not have perceived it as nesting material, due to its shortened length and altered form. Previous work has shown that in C57BL/6 mice, at least, biological relevance is key to eliciting nesting behavior, 5 and this may also be the case for sorting behavior.
It should be noted that there are already multiple products in the market that supply nesting material mixed in with the bedding product. Different bedding materials (cob, chip, or paper) may elicit this sorting behavior as well, though it seems unlikely as the variety of particle sizes in these alternative bedding materials is not as diverse as with aspen flake bedding, leaving mice with little basis for sorting. Additionally, mice have been shown to strongly prefer aspen bedding over cob bedding 14 so this may also negatively influence any attempted sorting behavior while on cob bedding. With aspen bedding, female mice have shown a preference for shavings over chips, and larger pieces over smaller ones, 15 which further supports the idea that bedding particle size is relevant to mice.
This raises the question of whether sorting is a behavior that we should be trying to elicit in laboratory mice. In the wild mice sort materials before incorporating them into their nests. 30 Using the Fraser et al. welfare framework, 31 in which the ability to perform natural behaviors is an integral component of welfare, this would appear to be a positive behavior. However, not all natural behaviors are necessarily desirable. While many may equate natural with positive welfare, some natural behaviors (such as aggression and cannibalism) are not desirable; and though sorting behavior is obviously not a maladaptive behavior, it should not be assumed that it is necessarily a beneficial behavior.
With that caveat in mind, the possible welfare benefit from encouraging sorting behavior is intriguing. Could providing opportunities to sort lessen abnormal behaviors commonly seen in mice? Are there physiological impacts to sorting behavior of which we are unaware? Investigation of these questions could provide some comparatively simple and cost-effective ways for enriching laboratory mice and improving welfare.
Conclusions
Providing mice with the opportunity to build nests improves their well-being by giving them the power to control their exposure to cold stress.7,32 Additionally, the provision of larger flake bedding may act as a stimulus, or even outlet, for naturalistic foraging and sorting behavior in mice. Large flake bedding contributed to higher scoring nests in almost all cases, so it warrants consideration on that basis alone, especially when they are provided at little to no additional costs. While combining nesting with aspen bedding material in the processing plant may be the easiest solution to allocating these materials simultaneously, the processing required to do so does not appear to support nest-building in the strains studied and with the bedding used.
Footnotes
Acknowledgements
The authors would like to acknowledge Geomaris Maldonado, Marie Heyer, and Yesenia Vargas for their excellent animal care.
Declaration of conflicting interests and Funding
The author(s) declared the following potential conflicts of interest and funding with respect to the research, authorship, and/or publication of this article: All research was conducted at and funded by Charles River Laboratories. Bedding material was donated by NEPCO (Warrensburg, NY, USA).
