Abstract
Some biomedical research procedures, such as organ xenotransplantation, usually require intensive hemotherapy. Knowledge of the whole phenotype of blood donor and graft could be useful in the field of xenotransplantation. Human and simian-type categories of blood groups have been established and they can be tested by standard methods used for human blood grouping. The aim of this work was to study the incidence of non-ABO blood group systems in different species of nonhuman primates, which are employed in biomedical research. The phenotype of Rh, Lewis, Kidd, Kell, MNSs, Lutheran, P and Duffy antigens was investigated in olive baboon (n = 48), chacma baboon (n = 9), Guinea baboon (n = 14), Rhesus macaque (n = 38) and squirrel monkey (n = 30) by using commercial microtyping cards. Kell, Lutheran, Kidd and Duffy antigens have been detected in all species, Rh in squirrel monkey, MNSs in rhesus macaque and squirrel monkey, and Lewis in baboon and rhesus macaque. There were differences in frequency and haemagglutination scores between species regardless of their gender and age. The main differences were found in squirrel monkey when compared with baboons and macaques. This typing system provides a tool to assess the presence of antigens in animals used for experimental procedures, such as xenotransplantation and xenotransfusion.
Studies of blood groups in non-human primates (NHPs) began more than 80 years ago,1–3 and different approaches were taken over the time to classify the blood groups of Old World Monkeys (OWMs) (chimpanzee, gorilla, baboon and rhesus monkey) and New World Monkeys (NWMs) (spider monkey, squirrel monkey and marmoset). 4 Up to date, two categories of blood groups have been established for apes and monkeys (human-type and simian-type), and they can be tested by standard methods used for human blood grouping. 5 ABO is the most investigated blood system, 6 but special interest has been paid to Duffy antigens 7 because of their implication in malaria, which was tested by using different NHPs species as an animal model for the infection. There is also an increased interest in Lewis system because BabA and SabA adhesins of Helicobacter pylori specifically adhere to the host Leb and sialyl-Lex antigens.8,9 Furthermore, the role of Duffy and Kidd antigens as minor histocompatibility molecules has been shown in renal allotransplantation rejection. 10
Phenotyping of primate blood has several applications. Some biomedical research procedures, such as liver xenotransplantation, usually require intensive haemotherapy. In these cases it is important to know the whole phenotype of the blood donor. 11 For these research procedures, several rapid blood phenotyping systems, based on direct haemagglutination can provide a fully typed blood donor. 12
The aim of this work was to phenotype the Rh, Kell, P, Lewis, Lutheran, Kidd, MNSs and Duffy blood groups systems antigens present on RBCs surface of different species of NHPs, commonly used in biomedical research, by using commercial microtyping cards. To our knowledge, such a wide group of antigens has
Materials and Methods
Animals
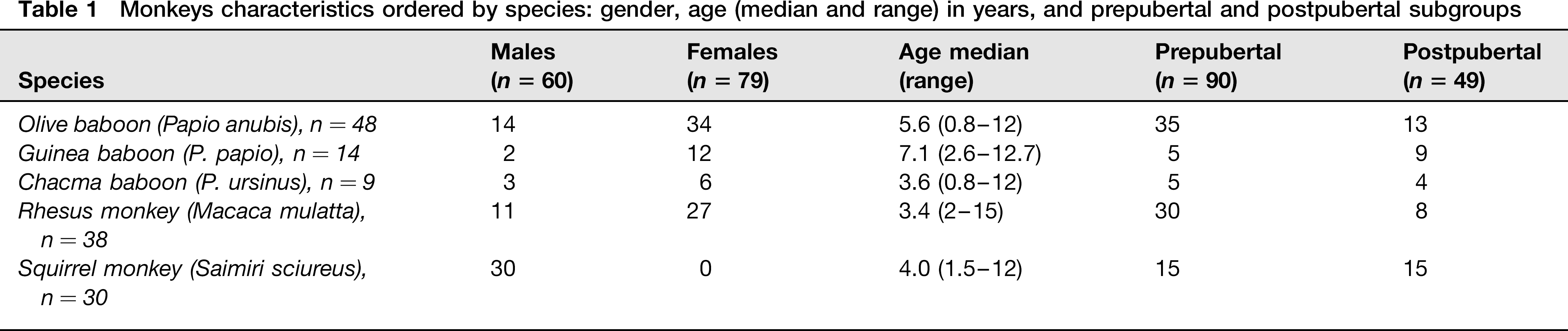
Twenty-four olive baboon (Papio anubis), 14 Guinea baboon (Papio papio), nine chacma baboon (Papio ursinus), 38 Rhesus macaques (Macaca mulatta), 30 squirrel monkey (Saimiri sciureus) from the Primatology Station of the French National Center for Scientific Research (Centre National de la Recherche Scientifique: CNRS) in Rousset (France), and 24 olive baboons from the Primatology Unit at the University of Murcia were studied. The age and gender are shown in Table 1.
Monkeys characteristics ordered by species: gender, age (median and range) in years, and prepubertal and postpubertal subgroups
All baboons and monkeys were housed in outdoor enclosures connected to indoor sheltered housing units. The baboons in Rousset were housed in fenced parks, grouped in multi-male, multi-female colonies of about 25 individuals. The baboons in Murcia were kept in two cages measuring 5.5 × 10 × 6 (w–l–h), each of which housed a multi-male, multi-female colony of about 22 individuals, whereas the monkeys were kept in cages measuring 3 × 3 × 2.5 m, each one housing a one male, multi female colony of 15 individuals. All housing facilities were supplied with straw for bedding and stimulation of foraging behaviour. Animals were exposed to natural conditions throughout the year and water was supplied by an automatic watering system.
The squirrel monkeys were kept indoors in cages measuring 2.5 × 3 × 3 m, each one housing 15 males. The temperature and humidity were maintained at 24°C and 45%, respectively.
All primates were fed with commercial feed pellets (Harlan, KY, USA) supplemented with fresh season fruit and vegetables. None of the females were pregnant during blood sampling.
Husbandry of animals and sampling procedures were carried out in compliance with Spanish transpositions of EU welfare laws regarding laboratory animals (R.D.1201/2005) and pigs (R.D.1135/2002).
All animals were monitored once a year for herpesvirus and retrovirus by enzyme-linked immunosorbent assay, tested for tuberculosis using TST (tuberculin skin test), and subjected to prophylactic ecto and endo parasite treatment every six months. Each species was subjected to a biochemical and haematological blood test.
For statistical analysis, animals were divided into two groups: prepubertal and postpubertal. The threshold of puberty was established at age 4 for both male and female baboons, 3.5 for male squirrel monkey and 3 and 4 for female and male rhesus macaques, respectively.13–15
RBCs samples
The blood samples were obtained from sedated animals (ketamine, 10 mg/kg, intramuscularly) by femoral venipuncture, using ethylendiaminetetraacetic acid tubes, following the recommendations of the Animal Care Guide of the University of Murcia. RBCs were stored until use in a medium for RBCs preservation (CellStab; Diamed, Switzerland).
Antigens phenotyping
ID-cards, and ID-Diluents (1 and 2) (Diamed, Switzerland) were used for antigens phenotyping: IDP-RhK card for Rh system and K antigens, ‘Antigen profile I’ (IDP-I) for P, Lewis and Lutheran systems antigens, ‘Antigen profile II’ (IDP-II) for Kell and Kidd systems antigens and ‘Antigen profile III’ (IDP-III) for MNS and Duffy antigens.
For IDP-RhK, IDP-I and IDP-II, the RBCs were diluted to 5% using ID-Diluent 1, containing bromelin. After 10 minutes incubation at room temperature, 12.5 μL of RBCs suspension was added to each well of the ID-card and centrifuged 10 minutes at 85 g. For IDP-III, a 0.8% RBCs solution was prepared using ID-Diluent 2, containing a Liss-Coombs modified solution. Fifty microliters of RBCs suspension were added to each IDP-III well, and subsequently, 50 μL of the antibody for each antigen. After 10 minutes incubation at room temperature the cards were centrifuged for 10 minutes at 85 g.
A five degree scale (0–4) was used to evaluate the results. When agglutinated RBCs formed a red line on the top of the gel, the reaction was graded 4. When the agglutinates dispersed in the gel, reactions were graded from 3 to 1, depending on the level of agglutination. Finally, when the RBCs appeared packed at the bottom of the column, the reaction was considered negative (0).
Statistical analysis
Non-parametric Pearson's χ2 test was used to compare the frequencies for each antigen and phenotypes. The differences in agglutination scores between species were assessed using the Mann-Whitney's U test. Data were analyzed using SPSS v.15.0 (SPSS Inc, Chicago, USA).
Results
Rh system
Positive results were found for Rh antigen c (100%) and Rh antigen E (3.3%) phenotyping in squirrel monkeys, regardless of age and gender. Rh antigens (D, C, c, E and e) have not been found in other tested species.
Kell system
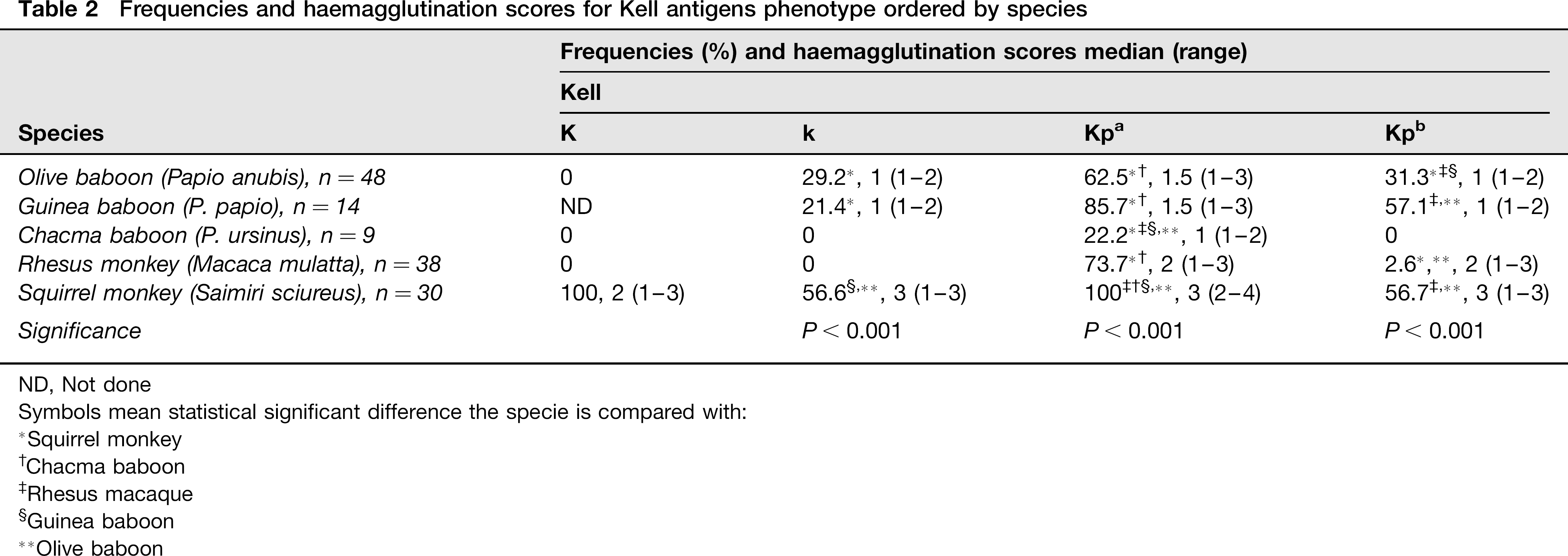
In the Kell system (K, k, Kpa, Kpb), K antigen was only detected in squirrel monkey, while k antigen was observed in olive baboon, Guinea baboon and squirrel monkey. Kpa was present in all NHPs and Kpb was typed in all NHPs except for chacma baboon. Antigens expression was not different between genders (Table 2).
Frequencies and haemagglutination scores for Kell antigens phenotype ordered by species
ND, Not done
Symbols mean statistical significant difference the specie is compared with:
Squirrel monkey
Chacma baboon
Rhesus macaque
Guinea baboon
Olive baboon
All prepubertal and 18.8% of postpubertal squirrel monkeys were positive for k and Kpb antigens (P < 0.001). For k antigen, all prepubertal and only 12.6% of postpubertal monkeys scored ≥2. Regarding Kpa, there were no differences between age groups in frequency but there were differences in agglutination score; all prepubertal and 56.7% of postpubertal scored ≥3. All prepubertal and 12.5% of postpubertal scored 3 for Kpb.
P system
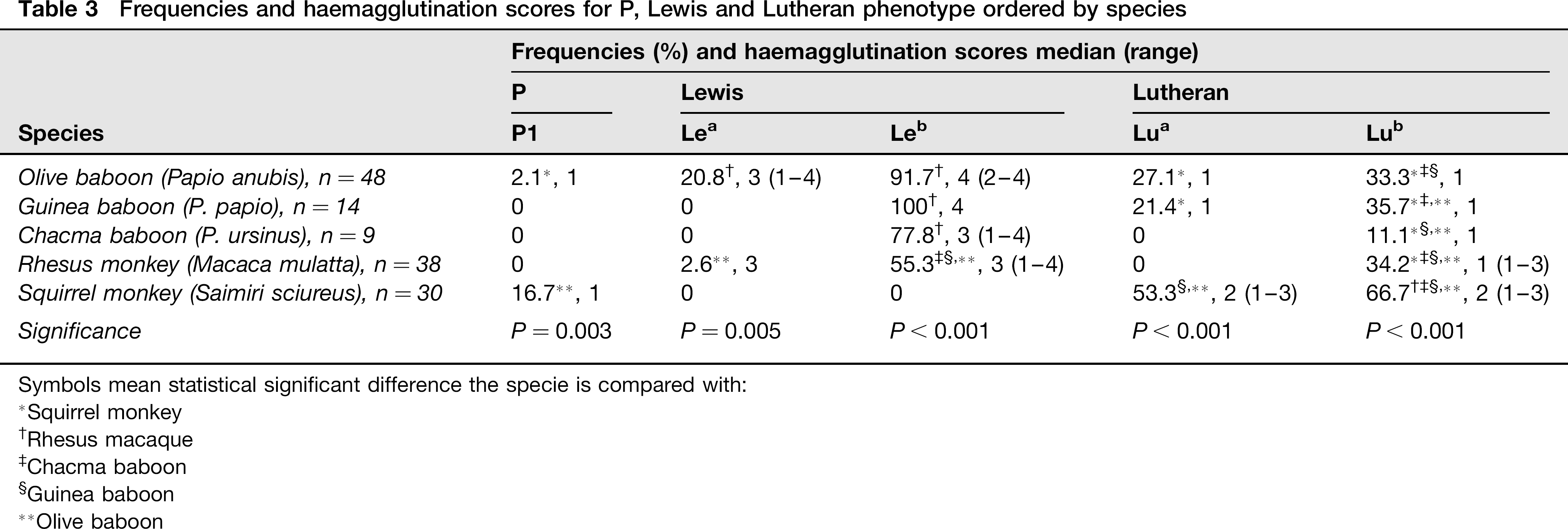
P1 antigen was found on olive baboon (2%) and squirrel monkey (17%). There were no differences in frequency or agglutination score between NHPs species. All squirrel monkeys positive for P1 antigen were prepubertal (Table 3).
Frequencies and haemagglutination scores for P, Lewis and Lutheran phenotype ordered by species
Symbols mean statistical significant difference the specie is compared with:
Squirrel monkey
Chacma baboon
Rhesus macaque
Guinea baboon
Olive baboon
Lewis system
In Lewis system antigens (Lea and Leb), Lea was found in olive baboon and rhesus macaque with differences in agglutination score (P = 0.005), and Leb was detected in all baboon species and rhesus macaque, showing a higher prevalence than Lea. The agglutination score of Leb in olive baboon and Guinea baboon was higher than in chacma baboon and rhesus macaque (P < 0.001) (Table 3).
Neither age nor gender had influence on frequencies or agglutination scores. Lewis antigens were not detected in squirrel monkeys.
Lutheran system
Lua was detected in olive baboon, Guinea baboon and squirrel monkey, and Lub was identified in all NHPs. The squirrel monkeys had higher agglutination scores for Lua than olive and Guinea baboons (P < 0.001) (Table 3).
Lua and Lub antigens were more frequent in prepubertal than in postpubertal animals (P < 0.001). The agglutination scores were also higher in younger animals (P < 0.001).
Kidd system
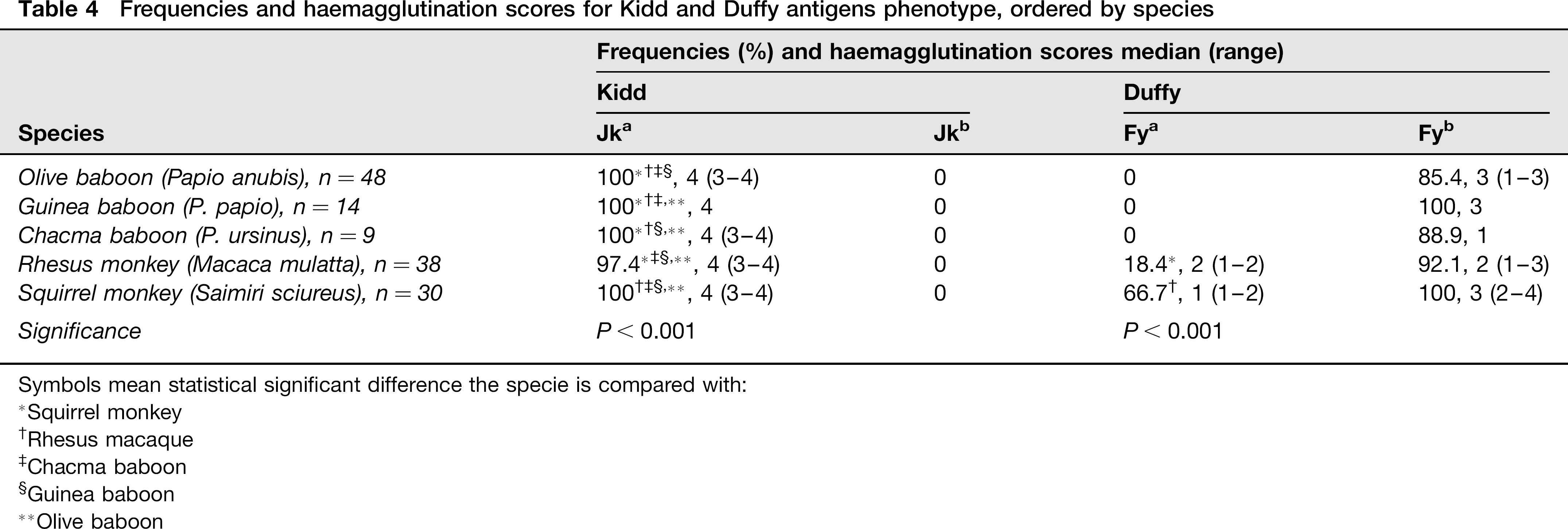
In the Kidd system (Jka and Jkb), Jka was found in all NHPs however, Jkb was not detected. There were no differences regarding age or gender (Table 4).
Frequencies and haemagglutination scores for Kidd and Duffy antigens phenotype, ordered by species
Symbols mean statistical significant difference the specie is compared with:
Squirrel monkey
Chacma baboon
Rhesus macaque
Guinea baboon
Olive baboon
Duffy system
In the Duffy system (Fya and Fyb), the Fya antigen was found in rhesus macaque and in squirrel monkey. Fyb was found in all NHPs with high prevalence (>80%). There were no differences in frequency due to age or gender. However, agglutination score of both antigens was higher in postpubertal than in prepubertal animals (Table 4).
MNSs system
Regarding the MNSs system (M, N, S and ŝ), M and N antigens were not found in NHPs. The S antigen was detected in 5% of rhesus monkeys and in 90% of squirrel monkeys (P < 0.001). The ŝ antigen was found only in one rhesus macaque. There was no influence of age but postpubertal animals had higher scores than prepubertal ones (Table 5).
Frequencies and haemagglutination scores for MNSs antigens phenotype ordered by species
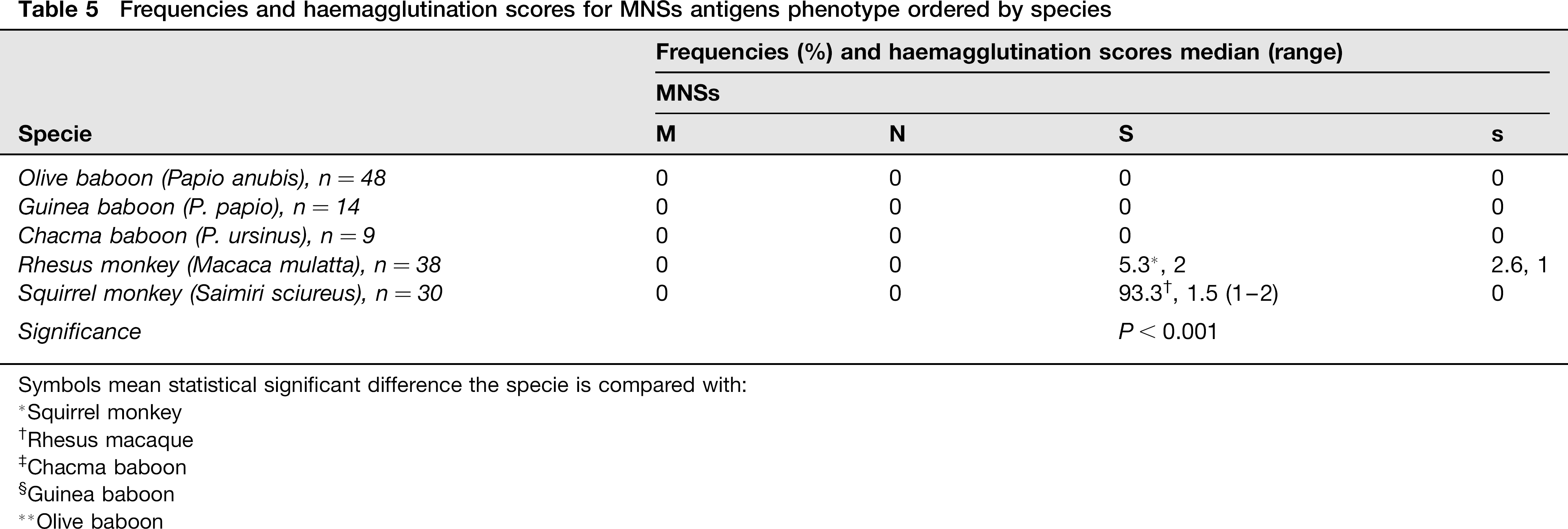
Symbols mean statistical significant difference the specie is compared with:
Squirrel monkey
Chacma baboon
Rhesus macaque
Guinea baboon
Olive baboon
Comparison between OWM and NWM
Frequency of most of the tested antigens showed a significant difference in species comparisons. Kpb, k, Lea and Lua were absent in chacma baboons, while these antigens were found in all the other baboons (except for Lea in Guinea baboon). The most noticeable difference between baboon subspecies and rhesus macaques was the absence of Lua and the presence of S and ŝ antigens in rhesus samples. The main differences between OWMs and squirrel monkey were the presence of Rh antigens (c and E) and the absence of Lewis’ antigens in squirrel monkey.
Discussion
Reagents prepared from some species’ blood would crossreact to a greater or lesser degree with RBCs from related species. In fact, blood grouping sera prepared for testing human blood groups have been used on monkeys for long time.1–3,16–19
Although Rh-polypeptide genes are present in all mammals, 20 none of the tested RBCs samples from OWMs in this study were positive for Rh human antigens, as it has been previously reported.20–22 NWMs species (tamarins and marmosets) previously tested, failed to show Rh antigens;20,21 however, with our method, E and c antigens were found in squirrel monkeys.
Lewis antigens are histocompatibility antigens, and anti-Le antibodies can cause organ rejection, as has been recently shown in kidney transplantation. 23 Therefore, any research involving transplantation or xenotransplantation with NHPs should include typing for these antigens. Our results show that Lea was present in olive baboon and rhesus macaque, and Leb in all OWMs. Lewis phenotype frequency was similar to those previously recorded. 24 Downing et al. 25 tested different chacma baboons and found all specimens to be apparently devoid of Lea activity but very strong in Leb activity. None of the chacma baboons in this study have shown the presence of Lea antigen, confirming previously reported data. The fucosyltransferase encoding genes for rhesus macaques and squirrel monkey were sequenced in 2002, 26 and homologous sequences were found for rhesus macaques but not for squirrel monkeys, meaning that functional alpha 3/4-fucosyltransferases could be present in OWMs and absent in NWMs. This may justify the absence of Le antigens in all the squirrel monkeys tested in this study.
The presence of Duffy antigens is not surprising since all NHPs are susceptible to Plasmodium vivax infection which uses the Fyb antigen as receptor. The encoding region for Fy antigens has also been sequenced in different NHPs and other mammal species (such as pigs, rabbits and mice).6,27 Expression of Fya and Fyb antigens on NHPs RBCs has been previously analyzed by using human polyclonal and monoclonal antibodies. As the results showed that Fya is only restricted to human, 28 it has been proposed that FY × A gene expression is too scarce in NHPs, and its protein may not be expressed. 6 We have phenotyped the Duffy antigens in squirrel monkeys. To our knowledge this is the first report taking into account the age of the animals. However, the tested animals were colony bred, so the difference in antigen expression could be caused by inbreeding. This question needs to be further investigated.
The MNSs antigens have been studied in different apes and monkeys shortly after they were discovered.16–19 None of the macaques in our study were found positive for M antigen, although it has been previously described in Barbary macaque. 29 Interestingly, the S antigen expression in older squirrel monkey was stronger than in the young ones. The same effect was assessed for Lutheran or Kell antigens. This phenomenon could be caused by inbreeding or by an increase in the antigenic expression through the life of the animals.
Blood phenotyping is essential to many research procedures such as xenotransplantation and allotransplantation. The alpha-gal antigen is particularly important in these procedures, although there are some carbohydrates antigens that could also act in xenorejection. 30 However, Zhu et al.31–33 showed that alpha-galactosidase treatment of porcine RBCs does not prevent binding to human natural antibodies, consistent with the existence of nonalphaGal xenoantigens on porcine RBCs.31,32 Similarly, MacLaren et al. 34 showed that the removal of antialphaGal antibody from human serum reduces, but does not eliminate haemagglutination. Thus, blood phenotyping of primates is relevant in xenotransplantation compatibility research with pigs as donors.
To date, the main interest has been focused on ABO compatibility, as well as on allotransplantation, but the other blood group systems should not be forgotten, and careful attention should be paid in order to assess the compatibility between donor and recipient. In many transplant studies using primates as recipients and pigs as donors, these antigens remain an unknown and untested variable. 35 According to our findings; there are several blood antigens on primates that could act as haemagglutinins. Even more, a severe mismatch between the blood antigens between donor and recipient could trigger rejection in xenotransplantation as has been previously demonstrated in kidney allotransplantation. 23
Transfusion support is also an important aspect of some experimental procedures in transplantation studies involving primates. The immune response to these transfused blood components may affect the outcome of these experiments and should be considered when designing transplant studies. 35
The use of microtyping cards in primates may have some limitations. Firstly, these cards include reagents derived from human sera, that may contain antispecies panagglutinins or anti-alpha-gal antibodies resulting in falsepositives, as it has been previously demonstrated. 36 However, as primate's antigens tested in our studies with human-based reagents (Lua, Lub, K, k, Kpa, Kpb, S, s, Fya and Fyb) appeared to give either positive or negative agglutination results the false-positive results can be discarded. Secondly, it has been previously described that the use of monoclonal antibodies for human blood antigens testing may fail to recognize the blood groups antigens in different species. 37 On the other hand, our studies showed that only anti-M, N and Jkb antibodies did not react with blood antigens from any of the tested animals. Additionally, protein alignment of human and known NHP blood group antigens, performed using protein basic local alignment search tool (BLAST), showed that human antigens are well conserved among primate species.
Moreover, protein BLAST search carried out at the National Center for Biotechnology Information (NCBI) database showed no similarities between the primate blood antigens and non-related proteins.
In conclusion, ID-cards offer a quick, easy (to perform and to interpret) and cheap method for the classification of anti-non-ABO blood groups based on the principle of haemagglutination. This test procedure could be very useful in the foundation of a blood bank for research purposes.
Footnotes
Acknowledgements
We are grateful to the staff of the Primatology Station of the French National Center for Scientific Research (CNRS) at Rousset, especially to Dr Guy Dubreuil and Dr Mourad Mekaouche for their assistance with the sampling in France and to the staff of the Primatology Unit at the University of Murcia. This work has been partially supported by a grant from The Seneca Foundation (Spain) for the sampling period in France.
