Abstract
We used a novel adaptation of methods for microtubule polymerization in vitro to assess the MTOC activity of centrosomes in frozen-sectioned tissues. Remarkably, centrosomes of tissue sections retain the ability to nucleate microtubules even after several years of storage as frozen tissue blocks. Adaptations of these methods allow accurate counts of microtubules from individual cells and the quantitative estimation the MTOC activity of the intact tissue. These methods can be utilized to characterize MTOC activity in normal and diseased tissues and in particular tissues at different stages of development.
C
Since the earliest reports of visualization of micro-tubules by indirect immunofluorescence (Fuller et al. 1975; Weber et al. 1975), understanding of microtubule distribution in cultured cells has advanced significantly. More recently, investigation of microtubule dynamics in vivo has become possible through novel developments in tubulin labeling, video microscopy, and computer-enhanced imaging (see Waterman-Storer 1998 for a recent review). However, comparable methods for visualizing microtubules and assessing MTOC activity in cells within the structural context of normal tissue architecture have lagged behind those for cells grown in culture. This has been due, in part, to difficulty in visualizing microtubules within tissue sections and also to the loss of microtubule integrity during preparation of tissues for microscopy. Here we describe a simple method for identifying the location of centrosomes and for assessment of their capacity to nucleate microtubules in cells of frozen tissue sections. The procedure also provides a direct measure of the capacity for microtubule nucleation by individual cells within the tissue section. These methods are based on the ability of centrosomes to nucleate microtubules both in detergent-extracted cell models and after isolation in vitro when sufficient free tubulin and conditions appropriate to support microtubule growth are provided (McGill and Brinkley 1975; Snyder and Mcintosh 1975; Gould and Borisy 1977; Telzer and Rosenbaum 1979; Mitchison and Kirschner 1984).
Materials and Methods
Sample Procurement and Cell Culture
Human breast tissues obtained from mastectomy and lumpectomy surgeries were collected according to an Institutional Review Board-approved protocol. Data from breast adenocarcinoma specimens and normal breast samples from reduction mammoplasties are reported in this study. All tumors were invasive and were designated by staff pathologists at the time of surgery as histological Grade 3 or 4 (Mayo grading system). Specimens were obtained from patients who had undergone no chemotherapy or radiation therapy before surgery. Tissue specimens were frozen in liquid nitrogen immediately after surgery and were stored at — 70C until use. HeLa cells (Gey et al. 1952) were obtained from the American Type Culture Collection (Bethesda, MD). Cell cultures were maintained in Eagle's MEM medium supplemented with 10% fetal calf serum under 5% CO2 in air at 37C. Before use, HeLa cells were seeded onto clean, sterile glass coverslips and cultured for 48 hr.
Microtubule Nucleation on Cryosections and in Detergent Extracted Cell Models
Cryosections of unfixed human breast tissue mounted on coverslips coated with Fro-Tissuer (Electron Microscopy Sciences; Ft Washington, PA) and cultured cells were permeabilized for 10 min with chilled microtubule stabilizing buffer (MTSB: 1% Triton X-100, 10 mM Pipes, pH 7.2, 2 mM EGTA, 1 mM MgSO4) and washed several times in this buffer without detergent. Cold reaction mixture (75 μl), consisting of cytostatic factor arrested Xenopus egg extract (see below; Murray 1991), prepared as a dilution series (final extract concentration (v/v) 5-20% and tubulin concentration approximately 6-18 μM) in MTSB containing 1 mM GTP, was applied to the specimen and incubated on ice for 10 min. The coverslips were then incubated for between 3-7 min at 28C to initiate microtubule nucleation and growth. The reaction was stopped by dilution and microtubules were stabilized with 20 μM taxol (Sigma; St Louis, MO) in MTSB before fixation by immersion in — 20C methanol for 10 min. A particularly useful alternative to the use of whole tissue sections consisted of “touch” preparations, which were made by pressing a coverslip against a freshly cut 30-μm thick cryosection. Individual epithelial cells transferred out of the tissue section and stuck to the coverslip, leaving behind connective tissue elements that otherwise interfere with microtubule visualization. One or two touch preparations could be made from each 30-μm-thick tissue section. Individual cells transferred in this manner were processed for microtubule nucleation as described above.
Microtubule Localization on Tissue Sections and Cell Preparations
Tissue sections and cell preparations were stained for micro-tubules using the indirect immunofluorescence technique. Specimens were rehydrated in three changes of MTSB and then treated in blocking buffer, consisting of PBS, pH 7.2, with 5% normal goat serum, 1% glycerol, 0.1% bovine serum albumin (Fraction V; Sigma), 1% fish skin gelatin, and 0.04% sodium azide, for 5 minutes at room temperature (RT). Specimens were reacted for 1 hr at RT with primary antibody consisting of a mixture of anti-α-tubulin and anti-β-tubulin monoclonal preparations (B512 and 2.1; Sigma) at 1:2000 and 1:200 dilution, respectively, in the blocking buffer. Some specimens were stained for centrosome localization using antibodies against centrin [26/14-1 (Baron et al. 1992)], a ubiquitous centrosome protein. The coverslips were then washed three times for 5 min each in PBS, treated a second time with blocking buffer for 5 min at RT, and then reacted with secondary FITC-goat-anti-mouse IgG (Cappel; Warrington, PA) or FITC-goat-anti-rabbit IgG (Cappel) at 1:800 dilution. Finally, the preparations were washed three times in PBS, mounted in Vectashield mounting medium (Vector; Burlingame, CA) containing DAPI (0.2 μg/ ml) or propidium iodide (2.5 ng/ml), observed on a Zeiss inverted IM35 or a Nikon FXA epifluorescence microscope, and recorded on film or using a Hamamatsu digital camera (C4742) and Metamorph (Universal Imaging; West Chester, PA) computer software.
Preparation of Xenopus Egg Extract
Eggs of the frog Xenopus laevis are laid arrested in metaphase II of meiosis (Murray 1991). Cytoplasmic extracts were prepared in the presence of EGTA to maintain low levels of free Ca2+ [cytostatic factor (CSF)-arrested extracts]. Such extracts have been widely used as a rich source of tubulin and MAPs, and for studies of cell cycle regulation of centrosome microtubule nucleation and microtubule dynamics in vitro (Karsenti et al. 1984; Verde et al. 1990; Murray 1991; Andersen 1998). We followed the procedures of Murray (1991) to prepare Xenopus CSF-arrested cytoplasmic extracts, which were snap-frozen in liquid nitrogen as 100 μl aliquots and stored at − 70C until use. Extracts were defrosted immediately before use. Aliquots from a given preparation yielded results that were qualitatively consistent from experiment to experiment for up to 2 months from the date of their initial preparation.
Results
Microtubule Nucleation by Centrosomes In Vitro
Figure 1 shows an “aster” of microtubules that was nucleated by the centrosome of a human breast tumor cell prepared using the touch method described above. Over 120 microtubules originate from a single focus and extend outward approximately 20-25 μm in a uniform radial array. Individual microtubules are either relatively straight or show one or more gently curved regions. Under the conditions used here [8% extract concentration (v/v), 7-min incubation time, and 28C], few free microtubules are present that do not appear to originate from the centrosome region. Unattached microtubules may originate by spontaneous assembly, or some may represent broken ends of microtubules that originate at the centrosome. Varying the concentration of cytoplasmic extract from 5 to 20% produced greater numbers of centrosome-bound microtubules until a plateau was reached. This observation indicates that the number of nucleation sites at the centrosome is finite and can be saturated. However, when cytoplasmic extract was used at high concentration, many spontaneous polymerization events occurred, which resulted in many free microtubules that were not anchored at centrosomes. Because excess free microtubules made accurate counts difficult, each experiment was carried out with a series of cytoplasmic extract dilutions (extract concentration from 5 to 20%). Only the highest concentration of extract that gave centrosome nucleation and no or very little spontaneous free microtubule assembly was used for comparative analysis. Furthermore, because cells attached to the glass coverslip may cause steric limitations to microtubule growth from centrosomes, these methods may result in an underestimation of the actual microtubule nucleation capacity. Nevertheless, for identically treated samples, statistically significant differences in microtubule nucleation capacity were observed between normal and tumor tissue and for cells at different stages of the cell cycle (see below).
Microtubule Nucleation by Centrosomes of Tissue Sections
Figures 2A-2E illustrate centrosome location and microtubule nucleation on frozen-sectioned normal tissue from human breast specimens. Centrosomes were localized using antibodies directed against centrin, a protein found in centrosomes of all eukaryotic cells (Salisbury et al. 1986; Salisbury 1995). Figure 2A illustrates centrosome staining (FITC, green) in epithelial cells of a normal breast duct. The breast duct is lined with stratified epithelium; the inner continuous cell layer of the duct consists of cuboidal epithelial cells and the outer discontinuous layer consists of myoepithelial cells. Each epithelial cell has a single centrosome that lies at the apical-most region of the cell and above the nucleus (propidium iodide, red). Several basal or myoepithelial cells show centrosomes that are oriented either towards the duct lumen or towards the basement membrane side of the cell. Figures 2B and 2C illustrate the microtubule-nucleating capacity of normal breast duct epithelial cells. Frozen-sectioned tissue was incubated in cytoplasmic extract as described above. Figure 2B shows microtubule nucleation in a normal breast duct, including both cross-sectional and tangential views of a portion of its lining epithelium at low magnification. Each cell has a bright apical aster of microtubules. Figure 2C shows a micro-tubule aster nucleated by a centrosome located in the apical-most region of a cell lining a normal duct. This centrosome nucleated 12-13 microtubules that extend out into the duct lumen or inward over the cell nucleus. This microtubule aster clearly demonstrates both the location and activity of the centrosome. Figures 2D and 2E show microtubule asters nucleated by fibroblasts located in the dense connective tissue beyond the area of a lobule of a normal breast specimen. These centrosomes nucleated four and 12-13 microtubules, respectively. Not all cells in a frozen-sectioned specimen of normal tissue show the capacity to nucleate microtubule asters. This may be due to the absence of a centrosome in the plane of the section, centrosome loss during preparation, low inherent microtubule-nucleation capacity, or inactivation of some centrosomes during processing or storage.
Figures 2F-2H illustrate centrosome staining and microtubule nucleation capacity on frozen-sectioned tissue of human breast adenocarcinoma specimens. Centrosomes were stained using indirect immunofluorescence for centrin (Figure 2F). Unlike normal tissue, tumor cells show unusually large and irregular masses of centrin staining, with many cells displaying more than one concentrated region of centrin localization. A previous study of 35 high-grade tumors demonstrated that centrosomes of human breast adenocarcinoma cells characteristically display abnormal structure, aberrant protein phosphorylation, and supernumerary centrioles (Lingle et al. 1998). Figures 2G-2H demonstrate that breast tumor cells show unusually high microtubule nucleation capacity, with individual cells generating large numbers of microtubules that initiate from multiple foci and from the general region of the nuclear envelope. Furthermore, most tumor cells in a given section show nucleation capacity, whereas normal tissue cells show less frequent nucleation. Because of the high cell density seen in tumors, the microtubule arrays generated in the nucleation assay often overlap with one another, making accurate counts of microtubule difficult. Nevertheless, it is clear from comparison of normal and tumor specimens that tumor cells show more frequent nucleation of larger microtubule numbers than do cells of normal tissues (see below).
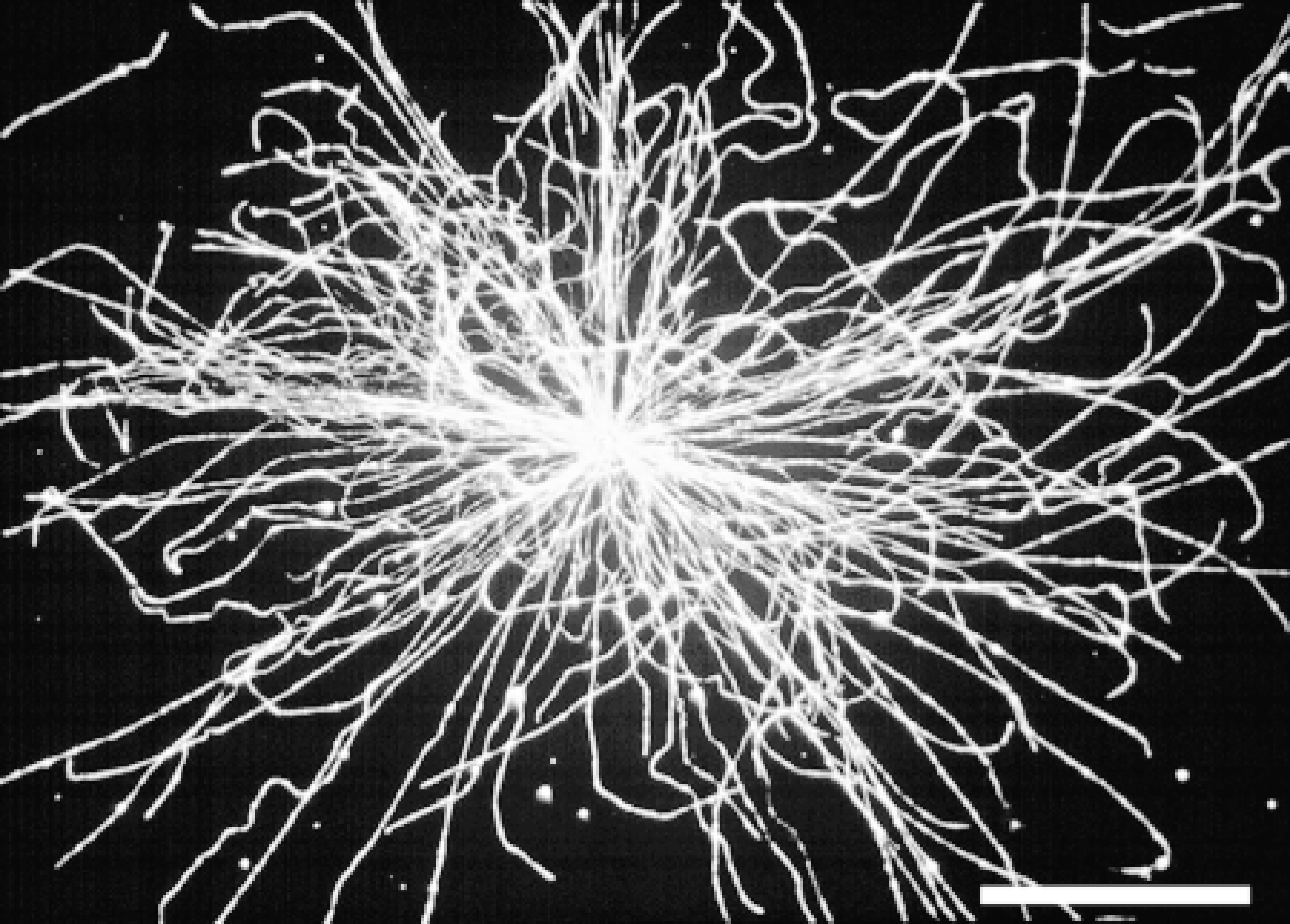
Microtubule nucleation activity by a centrosome from a cell of a human breast tumor touch preparation. Bar = 10 μm.
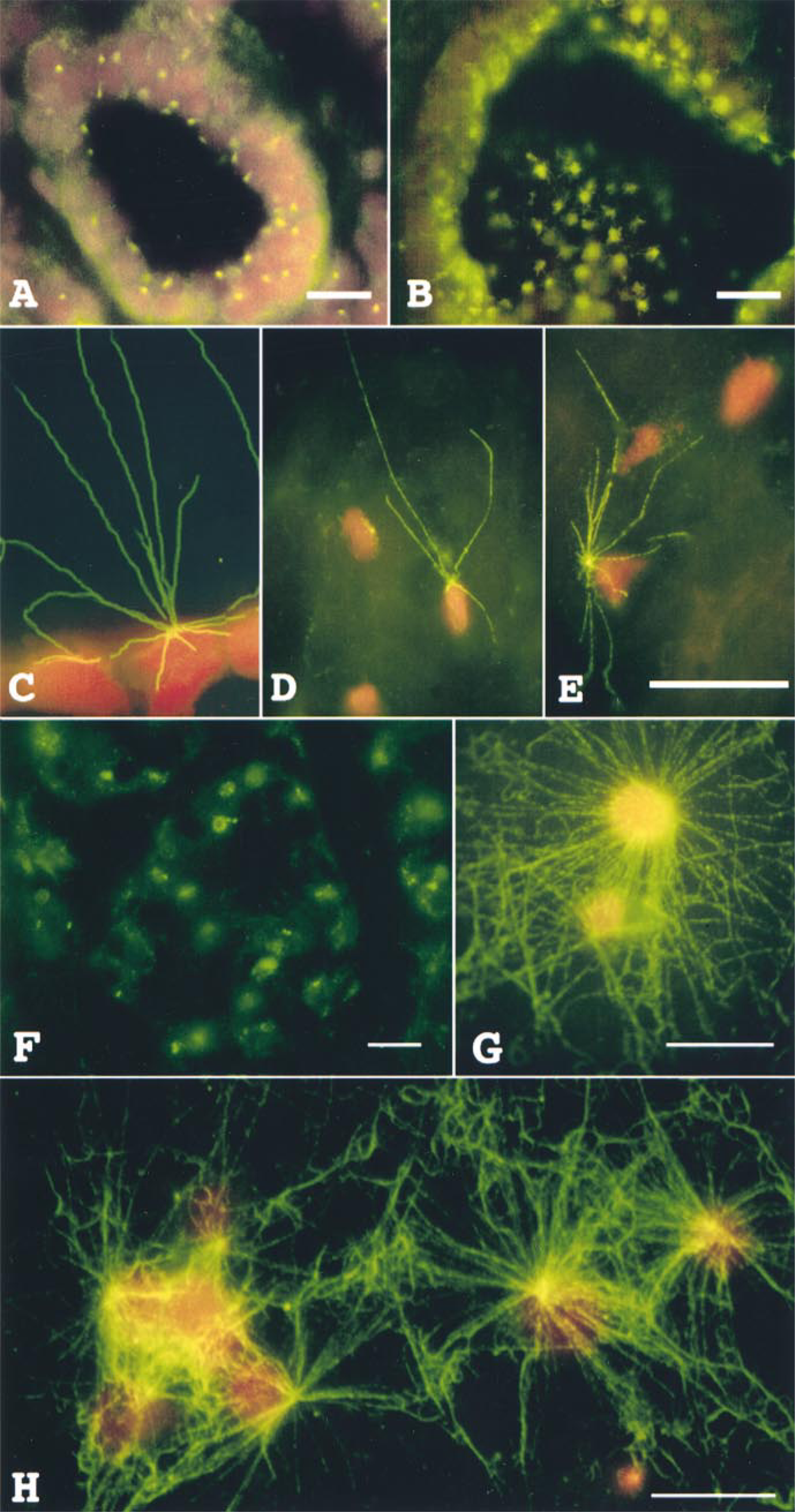
(A-E) Centrosome position and microtubule-nucleation capacity in frozen-sectioned normal human breast tissue. (A) Centrosome localization in a human breast duct revealed by immunostaining for centrin and FITC-secondary antibody. Each epithelial cell lining the duct shows an FITC-labeled apical centrosome. (B,C) Microtubule-nucle-ation capacity of breast duct epithelial cells from normal tissue shown at low (B) and high (C) magnification. (D,E) Microtubule-nucleation capacity of breast connective tissue fibroblasts from normal tissue. Nuclei are stained with propidium iodide. (F-H) Centrosome staining and microtubule nucleation capacity in tissue sections of human breast tumors. (F) Centrosome localization in a human breast adenocarcinoma revealed by immunostaining for centrin and FITC-secondary antibody. Each tumor cell shows unusually large and irregular centrosome staining. (G-H) Microtubule-nucleation capacity of breast adenocarcinoma cells from tumor tissue shown at high magnification. Nuclei are stained with propidium iodide except in F, where no nuclear stain was used. Bars = 10 μm.
Microtubule Nucleation by Centrosomes of Touch Preparations
To more accurately quantify microtubule nucleation capacity, we have exploited the nucleation assay using touch preparations of normal and tumor tissue. Figure 3 shows individual normal breast (Figures 3A and 3B) and breast tumor (Figures 3C and 3D) cell touch preparations that were employed in the microtubule nucleation assay as described in Materials and Methods. The low cell density and lack of interference from extracellular matrix material in touch preparations allows accurate counts of microtubules to be made. Touch preparations made from normal breast tissue (Figures 3A and 3B) show a single distinctly focused microtubule aster of less than 30 microtubules for each cell. On the other hand, breast tumor touch preparations (Figures 3C and 3D) show large numbers of microtubules that emanate from one or more centrosomes. These observations corroborated our findings on frozen whole-mounts of sectioned tissue. Both touch preparations and sections from normal tissue consistently nucleated fewer microtubules, which were focused at a single centrosome, and tumor preparations nucleated many more microtubules, often emanating from multiple foci in individual cells. Table 1 shows quantitative analysis of these differences.
Microtubule Nucleation by Centrosomes of Cell Models
The distribution of endogenous microtubules seen in an interphase HeLa cell is shown in Figure 3E, and microtubule nucleation by centrosomes of detergent-extracted interphase, prophase, and mitotic HeLa cell models is shown in Figures 3F-3H, respectively. Microtubules course through the cytoplasm and do not necessarily focus at a single juxtanuclear centrosome in interphase HeLa cells, although many individual microtubules converge near the nucleus (Figure 3E). Although this microtubule distribution is characteristic for many HeLa cells in interphase, mitotic cells exhibit distinct foci of microtubules at the spindle poles (not shown). Nevertheless, when interphase cells are extracted with detergent and then assayed for microtubule-nucleation capacity, most microtubules nucle ate and grow from a single juxtanuclear centrosome (Figure 3F). In the example shown here, approximately 20 microtubules originate at the centrosome and extend outward beyond the original margins of the cell boundary. Individual microtubules are quite straight, and their overall distribution is not comparable to that seen in interphase cells that were otherwise untreated (compare the distribution of microtubules in Figure 3E to those in Figure 3F). This observation has implications for the dynamics and redistribution of microtubules in vivo (see below). The microtubule-nucleating capacity of centrosomes has been estimated to be approximately fivefold greater in mitotic compared to interphase cells (Kuriyama and Borisy 1981). After incubation with cytoplasmic extract, HeLa cell models that were in mid-prophase at the time of detergent extraction show two distinct asters of nucleated microtubules (Figure 3G). Although the distribution of microtubules nucleated in the prophase cell models is qualitatively similar to that for microtubules in vivo, there are important distinctions. First, individual microtubules are longer (and generally straighter), extending beyond the cell boundary in the models. Second, there are typically many more microtubules in vivo than in the cell models after microtubule nucleation. For the prophase cell shown here, over 70 microtubules extend from the two nascent spindle poles, corresponding to 3.5 times the number of microtubules nucleated by a typical interphase cell model centrosome (compare Figures 3F and 3G; see Table 1). HeLa cells that were in mitosis at the time of detergent extraction show even greater numbers of microtubules nucleated at the spindle poles (Figure 3H). Therefore, when the centrosome duplicates to give rise to two spindle poles, each spindle pole also acquires a greater capacity to nucleate microtubules than that seen for individual centrosomes of interphase cells. Furthermore, the nucleation capacity seen in cell models does not explain the larger number of microtubules seen in typical interphase or prophase cells in vivo. It is possible that in vivo nucleation sites are utilized for multiple rounds of microtubule initiation and growth, whereas the in vitro conditions used here result in only one or a few rounds of microtubule nucleation.
Discussion
Late in the nineteenth century, Van Beneden recognized that the position of the centrosome relative to the nucleus defines a structural “cell axis” that indicates the overall functional polarity of the cell (see Wilson 1925). More recently, it has become clear that cell and tissue architecture are a consequence of complex interactions involving a dynamic interplay among elements of the cytoskeleton and their relationship to cell junctions and the extracellular matrix. The microtubule-based aspect of the cytoskeleton plays a major role in these processes through the polar nature of individual microtubules and the fact that microtubule morphogenesis is determined by the activity of a discrete, characteristically positioned centrosome. Visualization of the activity of centrosomes and of cytoplasmic microtubules is routine for cells grown in culture, where conditions can be adjusted to yield optimal cell density and rapid fixation, and where cells can adhere to the coverslip in a manner that is suitable for high-quality immunofluorescence microscopy. In general, tissue sections lack these features, and for this reason microtubule localization in tissue preparations is often unsatisfying. Here, a novel assay is presented that circumvents some of these problems. The methods developed in this study are useful for identifying the position of the cell's major microtubule organizing center (MTOC) and its activity and for revealing differences in microtubule nucleation capacity between normal and diseased tissue and for cells at different cell cycle stages. The results of this study show that centrosomes are robust organelles, retaining the capacity for microtubule nucleation even after prolonged storage as frozen specimens and cryosectioning. Centrosomes of cells in tissue sections can be localized and their MTOC activity can be assessed quantitatively on the basis of microtubule-nucleation capacity, using the methods presented here. Cells from normal breast tissue characteristically show a single MTOC that typically nucleates fewer than 30 microtubules, whereas breast tumor cells are characterized by multiple MTOCs that nucleate a larger number of microtubules. Identification of sites for microtubule nucleation in frozen-sectioned tissue both indicates the location of potential sites of active microtubule nucleation and serves as a quantitative measure of microtubule-nucleation capacity for the intact tissue. Furthermore, for accurate quantitative assessment of the number of microtubules nucleated by cells of normal and disease tissue, touch preparations were evaluated as the method of choice for specimen preparation.
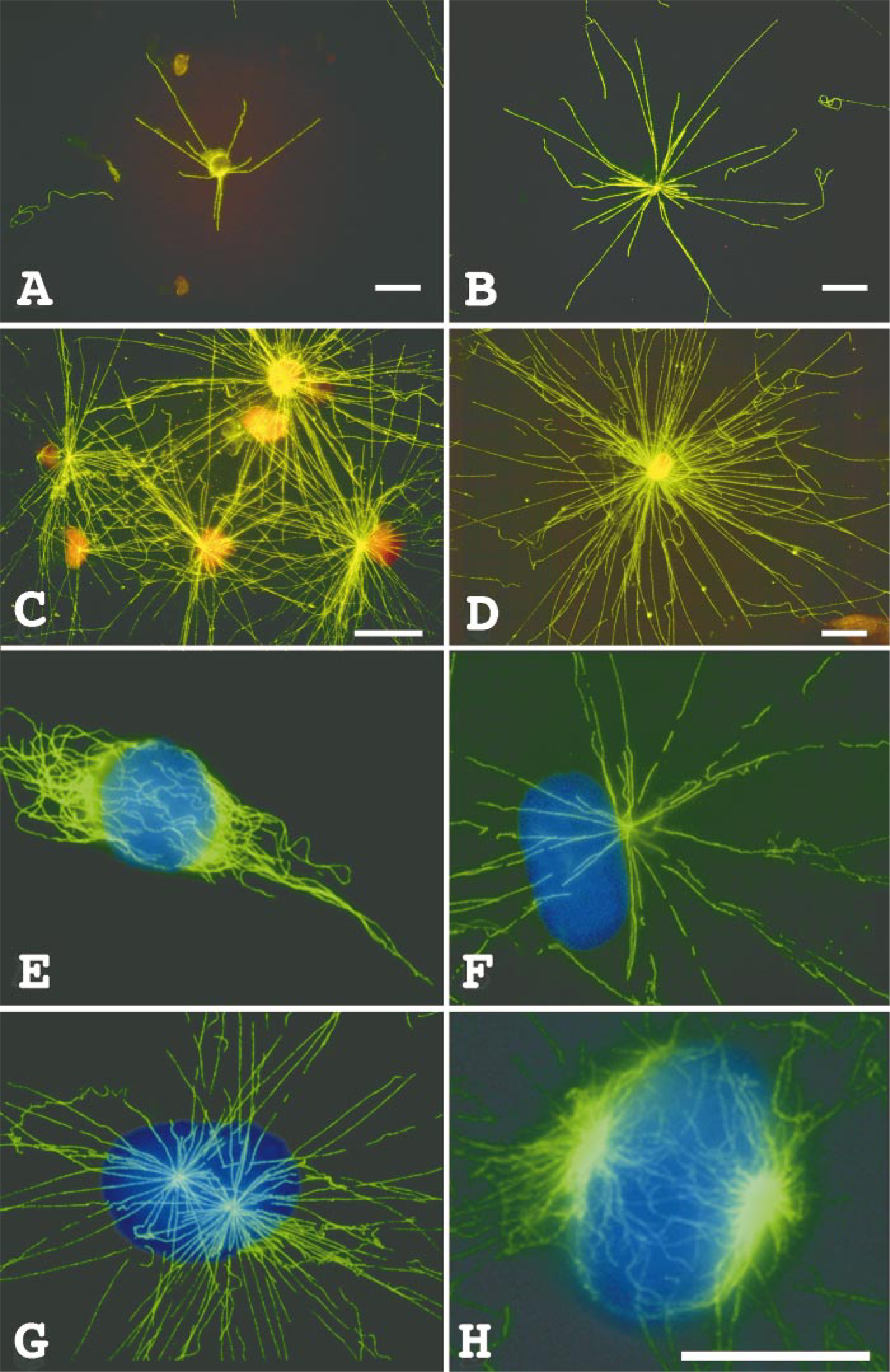
(A-D) Microtubule nucleation in touch preparations of normal and tumor tissue. (A,B) Microtubule nucleation in touch preparations from sections of normal breast tissue. (C,D) Microtubule nucleation in touch preparations from sections of breast tumor tissue. Nuclei in A-D are stained with propidium iodide. (E-H) Endogenous HeLa microtubule complex and microtubule nucleation in detergent-extracted HeLa cell models. (E) Distribution of microtubules in an interphase HeLa cell. (F-H) Microtubule nucleation by the centrosome and spindle poles of detergent-extracted interphase (F), prophase (G), and mitotic (H) HeLa cells. Nuclei in E-H are stained with DAPI. Bars = 10 μm.
Microtubule nucleation by centrosomes in touch preparations and in detergent-extracted HeLa cell models a
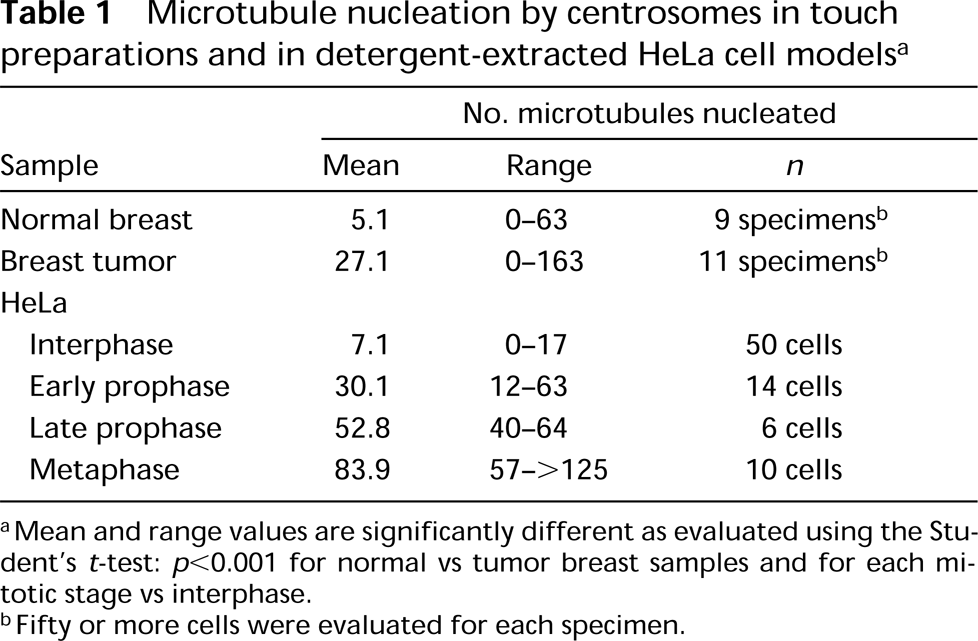
Mean and range values are significantly different as evaluated using the Student's t-test: p < 0.001 for normal vs tumor breast samples and for each mitotic stage vs interphase.
Fifty or more cells were evaluated for each specimen.
Microtubules typically originate and remain anchored at the centrosome. However, in some cells individual microtubules may not appear to be associated with the centrosome (McNiven and Porter 1988; Meads and Schroer 1995; Vorobjev et al. 1997). For example, axonal microtubules may originate at the centrosome of the neuron cell body and may subsequently be released or severed from their centrosome attachment and translocated to distant regions in axonal processes (Baas and Joshi 1992; Joshi and Baas 1993; Yu et al. 1993; Ahmad and Baas 1995). Microtubule nucleation and release from the centrosome are not restricted to neuronal cells (McBeath and Fujiwara 1990; Vale 1991; Karsenti 1993; Hartman et al. 1998). Observations made in this study suggest that these features are common to centrosomes in general. For example, the number of microtubules nucleated by centrosomes on tissue sections and in detergent-extracted cell models is significantly less than the number of microtubules observed in vivo. This suggests that microtubule-nucleation sites in living cells are used repeatedly and release polymerized microtubules, which may either remain centrosome-associated or redistribute to other regions of the cytoplasm. In addition to the spatial control of centrosome-directed nucleation, the assembly of microtubules is also subject to physiological regulation. This activity is based, in part, on the availability of active nucleation sites involving Γ-tubulin and associated proteins (Oakley et al. 1990; Joshi et al. 1992; Félix et al. 1994; Stearns and Kirschner 1994), on regulation by cell cycle specific kinases (Riabowol et al. 1989; Buendia et al. 1991; Gotoh et al. 1991; Wheatley et al. 1997; Hinchcliffe et al. 1998) and by developmental factors (Hewitson et al. 1997a,b; Holy and Schatten 1997). The methods presented here rely on the use of Xenopus egg extract to support microtubule nucleation on tissue sections and touch preparations. These extracts are rich in tubulin and other components that may affect microtubule nucleation. The use of egg extracts in these assays has several advantages. First, egg extract preparation is rapid and simple, and extract can be aliquotted and stored frozen to provide material for multiple assays with consistent results. Second, comparison of differences in microtubule nucleation by cells at different cell cycle stages demonstrates that assessed activities reflect the capacity of the centrosomes being evaluated to nucleate microtubules. Finally, our observations demonstrate that disease states may also affect the microtubule nucleation capacity of centrosomes. We have previously shown that centrosomes of breast tumor cells have unusually high levels of phosphorylated centrosome proteins, particularly centrin, and contain excessively large centrosomes and supernumerary centrioles (Lingle et al. 1998). These features are characteristic of centrosomes of other tumor types as well (Doxsey 1998; Pihan et al. 1998). Here we present a simple method for assessing the location and activity of microtubule nucleation sites in frozen-sectioned tissues. The utility of these methods is confirmed in touch preparations of tissue sections and in detergent-extracted cell models. These methods should be generally applicable to the study of centrosome position and activity in sections of tissues at different developmental states and in a variety of disease processes.
Footnotes
Acknowledgements
Supported by the National Cancer Institute of the NIH CA 72836, Race for the Cure/Twin Cities, Breast Cancer Research Foundation, Fraternal Order of Eagles Cancer Research Fund, Sitt Foundation, and the Mayo Foundation.
