Abstract
Polyclonal antibodies were raised against a common and a specific epitope present only in longer α1E isoforms of voltage-gated Ca2+ channels, yielding an “anti-E-com” and an “anti-E-spec” serum, respectively. The specificity of both sera was established by immunocytochemistry and immunoblotting using stably transfected HEK-293 cells or membrane proteins derived from them. Cells from the insulinoma cell line INS-1, tissue sections from cerebellum, and representative regions of gastrointestinal tract were stained immunocytochemically. INS-1 cells expressed an α1E splice variant with a longer carboxy terminus, the so-called α1Ee isoform. Similarily, in rat cerebellum, which was used as a reference system, the anti-E-spec serum stained somata and dendrites of Purkinje cells. Only faint staining was seen throughout the cerebellar granule cell layer. After prolonged incubation times, neurons of the molecular layer were stained by anti-E-com, suggesting that a shorter α1E isoform is expressed at a lower protein density. In human gastrointestinal tract, endocrine cells of the antral mucosa (stomach), small and large intestine, and islets of Langerhans were stained by the anti-E-spec serum. In addition, staining by the anti-E-spec serum was observed in Paneth cells and in the smooth muscle cell layer of the lamina muscularis mucosae. We conclude that the longer α1Ee isoform is expressed in neuroendocrine cells of the digestive system and that, in pancreas, α1Ee expression is restricted to the neuroendocrine part, the islets of Langerhans. α1E therefore appears to be a common voltage-gated Ca2+ channel linked to neuroendocrine and related systems of the body. (J Histochem Cytochem 47:981–993, 1999)
Keywords
V
Ten different α1-subunits of voltage-gated Ca2+ channels have been cloned thus far (Table 1) and are classified according to their voltage dependence of activation as high voltage- (α1S, α1C, α1D, α1F, and α1A, α1B, α1E) or low voltage-activated Ca2+ channel types (α1G, α1H, and α1I) (Hofmann et al. 1994; Perez-Reyes and Schneider 1995; De Waard et al. 1996; Bech-Hansen et al. 1998; Cribbs et al. 1998; Perez-Reyes 1998; Perez-Reyes et al. 1998; Strom et al. 1998; Lee et al. 1999).
The ion-conducting subunits of voltage-gated Ca2+ channels and related functions
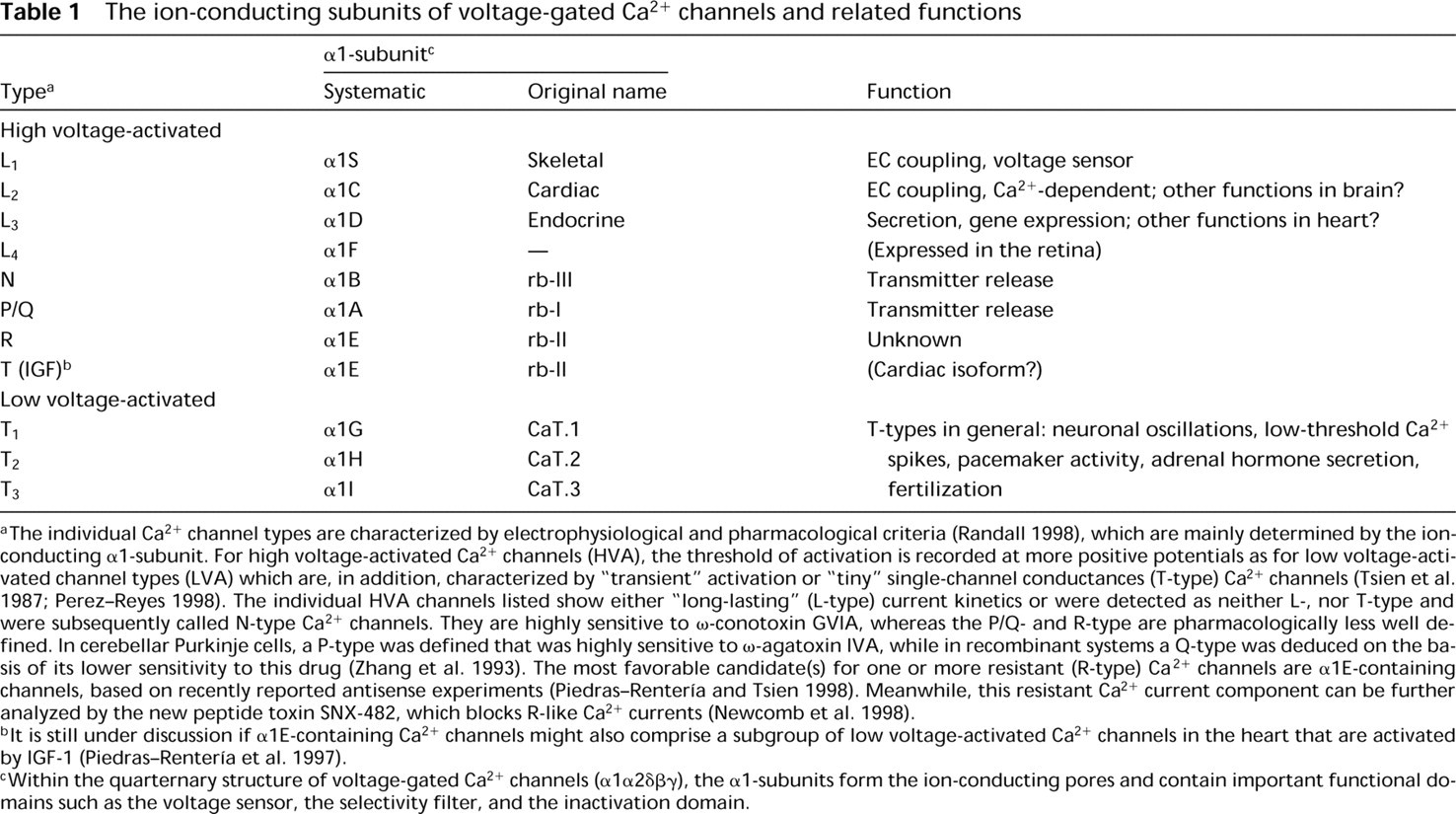
The individual Ca2+ channel types are characterized by electrophysiological and pharmacological criteria (Randall 1998), which are mainly determined by the ion-conducting α1-subunit. For high voltage-activated Ca2+ channels (HVA), the threshold of activation is recorded at more positive potentials as for low voltage-activated channel types (LVA) which are, in addition, characterized by “transient” activation or “tiny” single-channel conductances (T-type) Ca2+ channels (Tsien et al. 1987; Perez-Reyes 1998). The individual HVA channels listed show either “long-lasting” (L-type) current kinetics or were detected as neither L-, nor T-type and were subsequently called N-type Ca2+ channels. They are highly sensitive to ω-conotoxin GVIA, whereas the P/Q- and R-type are pharmacologically less well defined. In cerebellar Purkinje cells, a P-type was defined that was highly sensitive to ω-agatoxin IVA, while in recombinant systems a Q-type was deduced on the basis of its lower sensitivity to this drug (Zhang et al. 1993). The most favorable candidate(s) for one or more resistant (R-type) Ca2+ channels are α1E-containing channels, based on recently reported antisense experiments (Piedras-Rentería and Tsien 1998). Meanwhile, this resistant Ca2+ current component can be further analyzed by the new peptide toxin SNX-482, which blocks R-like Ca2+ currents (Newcomb et al. 1998).
lt is still under discussion if α1E-containing Ca2+ channels might also comprise a subgroup of low voltage-activated Ca2+ channels in the heart that are activated by IGF-1 (Piedras-Rentería et al. 1997).
Within the quarternary structure of voltage-gated Ca2+ channels (α1α2δβγ), the α1-subunits form the ion-conducting pores and contain important functional domains such as the voltage sensor, the selectivity filter, and the inactivation domain.
High voltage-activated Ca2+ channels can be further subdivided as a dihydropyridine-sensitive L-type subfamily containing α1S, α1C, and α1D Ca2+ channels (Table 1). The recently identified retinal α1F subunit is structurally related to the L-type subfamily but not yet functionally characterized in detail (Bech-Hansen et al. 1998; Strom et al. 1998).
The non-L-type subfamily of high voltage-activated Ca2+ channels consists of α1A-, α1B-, and α1E-subunits which are pharmacologically divergent. Two of them, α1A and α1B, have similar physiological functions. They are both involved in hormonal modulation of presynaptic Ca2+ currents (Davalli et al. 1996) and in insulin secretion (Ligon et al. 1998). In addition, a tight association with synaptic vesicle proteins has been described for α1A and α1B (Sheng et al. 1996; Rettig et al. 1996, 1997; Leveque et al. 1998). All three non-L-type α1-subunits, α1A, α1B, and α1E, have been intensively investigated for their G-protein-dependent modulation (as summarized in Schneider et al. 1997; Mehrke et al. 1997; Dolphin 1998; Zamponi and Snutch 1998).
The physiological function of α1E-containing voltage-gated Ca2+ channels is not yet well understood. The α1E-subunit has been related to the resistant (R)-type Ca2+ channels from cerebellar granule cells (Forti et al. 1994; Randall and Tsien 1997) based on the inhibition of resistant-type current by α1E antisense oligonucleotides (Piedras-Rentería and Tsien 1998). The same antisense approach has led to the suggestion that an IGF-I-inducible form of cardiac T-type Ca2+ currents may be carried by a specific α1E isoform (Piedras-Renteria et al. 1997; Chu and Best 1998). In recent immunohistochemical investigations, the neuronal localization of α1E has been analyzed in detail by using antibodies raised against common regions of individual α1E-subunits (Volsen et al. 1995; Yokoyama et al. 1995).
Neurons and endocrine cells that are responsible for the production of polypeptide hormones are developmentally and functionally related to each other (Le Douarin and Fontaine-Perus 1991). The endocrine cells of the gut epithelium and of the endocrine gland associated with the digestive tract (pancreatic islet cells) are regarded as the largest endocrine organ of the body (Pearse 1974), showing a functional relationship with neural cells. More than 30 endocrine cells/mm2 can be found in the pancreas and duodenum (Bloom and Polak 1978).
A new isoform of α1E was isolated from rat pancreas and human kidney by amplifying cDNA fragments of α1E (Vajna et al. 1998). We proposed the name “α1Ee” for this splice variant according to the guidelines of nomenclature for voltage-gated Ca2+ channels (Birnbaumer et al. 1994). In the present study we used a pair of antibodies designed to discriminate among three longer and three shorter α1E isoforms.
The cDNA fragments of longer isoforms of α1E have been detected recently in mouse, rat, and human cerebellum (Pereverzev et al. 1998; Vajna et al. 1998).
Our recent RT-PCR results from isolated islets of Langerhans (Vajna et al. 1998) indicate that a longer α1E isoform is expressed in several peripheral organs. This led to the hypothesis that other endocrine systems might contain α1E. Therefore, we investigated the expression of the neuronal α1E calcium channel isoforms in the central nervous system and in selected areas of the human gut: the antrum of the stomach, the small and large intestine, and the islets of Langerhans isolated from pancreas. In addition, the rat insulinoma cell line INS-1 was included both in the immunohistochemical investigation and electrophysiological studies to establish the physiological function of α1E in this cell line and in related tissues.
Materials and Methods
Materials
The ECL detection kit for immunoblotting was purchased from Amersham (Braunschweig, Germany) and collagenase P from Boehringer Mannheim (Germany). The monoclonal antibodies (MAbs) against human chromogranin A and human insulin were purchased from Camon (Wiesbaden, Germany) and BioGenex (DCS; Hamburg, Germany), respectively. The polyclonal antibody against human somatostatin was a kind gift from BioGenex. All reagents were of standard biochemical quality.
Cell Lines and Tissues
All procedures used in the present study that involved tissues from animals and humans were performed in accordance with the regulations governing their use in scientific research.
Aliquots of the INS-1 cell line were a gift from Dr. S. Ullrich with the kind permission of Dr. C.B. Wollheim (Geneva). The INS-1 cells are grown in RPMI 1640 medium, 10% heat-inactivated fetal calf serum, 50 μM 2-mercaptoethanol, 1 mM Na-pyruvate, 10 mM Hepes, pH 7.2-7.5, 100 IU/ml penicillin, and 100 μg/ml streptomycin.
Stable HEK-293 cell lines expressing human Ca2+ channel α1E-subunits (Schneider et al. 1994) were created by transfecting either the full-length wild-type α1Ed cDNA or a deletion mutant of α1Ed (β1Ed-CDEL) lacking a 129-bp insert of the 3′-end (Mehrke et al. 1997; Pereverzev et al. 1998).
Antibody Production
Two different peptides were used for the immunization of rabbits. Peptide Nast-195 is homologous to a sequence close to the pore region in Domain I and therefore corresponds to a common sequence in all published α1E isoforms. Peptide Nast-197 is part of the 43-amino-acid (aa) extension cloned within the fetal brain α1Ed isoform (Schneider et al. 1994; Pereverzev et al. 1998), which is also present in two other longer α1E isoforms, α1Ee and α1Ef (Vajna et al. 1998). The peptides Nast-195, SGILEGFDPPHPCGVQGC (aa 256-273 in the loop IS5 to pore region; GenBank L27745) and Nast-197, GIYLPSDTQEHAG[C] (aa 1981–1993 in the car-boxy terminus of the human α1Ed isoform) were synthesized by the solid-phase method (Merrifield 1986). At the carboxy terminus, peptide Nast-197 contains one additional cystein residue for coupling to a matrix as shown in brackets. Immunization of the rabbits with the hemocyanin-coupled peptides was performed by Dr. W. Nastainczyk (Medicinal Biochemistry, Homburg) as described (Pereverzev et al. 1998).
The sequences of the synthetic peptides were compared with the aa sequences in GenBank by using the program blastp 2.0.4 (Altschul et al. 1997). Only the α1E fragments cloned from mammalian species were picked up and shown to be similar or identical to the synthetic peptide sequences. The peptide sequence of Nast-195 is identical to the rat, mouse, and all published human sequences, whereas the rabbit α1E shows a single amino acid substitution of valine for isoleucine. For the peptide Nast-197, both the rat and the mouse sequence show one aa difference with an additional proline instead of a serine residue compared to the human α1E sequence. However, no differences were observed during immunostaining comparing human and rat cerebellum or pancreas. Both sera do not stain the α1G- or the α1H-subunit in membranes from stably transfected HEK-293 cell lines (unpublished results). Therefore, the anti-E-com serum (anti-Nast-195) is used to detect all known α1E isoforms and the anti-E-spec serum (anti-Nast-197) detects the longer α1Ed, α1Ee, and α1Ef or structurally related isoforms containing a longer carboxy terminus (Pereverzev et al. 1998). For the detection of rat α1E isoforms in the cerebellum, both sera were purified by adsorption to liver powder to reduce any unspecific staining. Rat liver (6 g) was homogenized in cold PBS, pH 7.4 (6 ml), and proteins were precipitated by adding a fourfold volume of acetone. After 30 min on ice, the unsoluble proteins were separated by centrifugation for 10 min at 10,000 x g. The precipitated proteins were washed once in 5 ml ice-cold acetone by rehomogenization and precipitated by centrifugation as before. The protein pellet was spread on filter paper and, after drying, homogenized to a powder, which was stored in aliquots at −20C. The peptide-specific antisera (0.5 ml of 1:50 diluted crude sera) were incubated with 20 mg of liver powder for 1 hr at 37C and centrifuged for 10 min at 2000 x g. The supernatant was sterile-filtered through 0.2-μm filters and used for immunohistochemistry of rat cerebellar sections.
Preparation of Paraffin-processed Tissue
Female Wistar rats weighing 180-250 g were anesthetized with CO2, decapitated, and the organs rapidly removed. The individual tissues were dissected into smaller blocks and fixed by immersion in 10% neutral buffered formalin at room temperature (RT) overnight. Tissue sections of human gastric antrum, small and large intestine (cecum), and pancreas were obtained from surgically resected specimens. All tissues, including rat cerebellum, were fixed in 10% buffered formalin and embedded in paraffin. Immunohistochemistry was performed on 4-μm deparaffinized serial tissue sections that were mounted on silane-coated slides and dried at 50C before staining.
Western Blot Analysis and Immunocytochemical Staining of Cell Lines and Tissues
The sensitivity and specificity of the anti-α1E sera were tested on immunoblots of microsomal membrane proteins from stably transfected HEK-293 cells. These cells were transfected either with the full-length human α1Ed (Schneider et al. 1994) or with a truncated α1Ed-CDEL lacking a 129-bp insertion of the carboxy terminus, which carries the region of the epitope for anti-E-spec (Pereverzev et al. 1998). Microsomal membrane proteins from HEK-293 cells were prepared according to Wei et al. (1995) as specified in Pereverzev et al. (1998) and aliquots were stored at −80C. The separation of membrane proteins by polyacrylamide gel electrophoresis and immunoblotting were performed using standard protocols as reported (Pereverzev et al. 1998). Protein concentrations were determined with the BCA method (Pierce; Rockford, IL).
In addition, the specificity of the sera was tested by immunostaining untransfected and stably transfected HEK-293 cells expressing human α1E (Schneider et al. 1994). After fixation of the cells in 10% buffered formalin for 20 min at RT, cells were washed in PBS. For detection of α1E, the cells were treated with a standard streptavidin-biotin-horseradish peroxidase technique according to the instructions of the manufacturer (BioGenex). The primary antibody was diluted 1:500 or 1:1000 in PBS containing 2% BSA.
For immunohistochemistry, the polyclonal antibodies anti-E-com and anti-E-spec were used at a working dilution of 1:20 (in several tissues as indicated), 1:50 (cerebellum), or 1:200 (large intestine). Before immunostaining, sections underwent a microwave-based heat-induced epitope retrieval (HIER) treatment in a solution of 10 mM citrate buffer, pH 6.0. Endogenous peroxidase activity was quenched by exposure to a solution of 0.3% hydrogen peroxide in methanol for 20 min at 37C. After washing with PBS, the sections were immunostained by the streptavidin-biotin-horseradish peroxidase technique according to the instructions of the manufacturer (Super Sensitive detection system from BioGenex). 3,3′Diaminobenzidine (DAB) was used as chromogen with and without copper sulfate enhancement. Two negative control procedures were performed. Either the primary antibodies were replaced by the preimmune serum or the serum was preincubated with an excess of the peptide (20 μM) used for immunization. Sections from a multitissue block were used to verify appropriate positive and negative reactions.
In some experiments the purified anti-E serum directed against an α1E fusion protein was used as a positive control (Volsen et al. 1995).
Electrophysiological Recordings
Electrophysiological recordings from INS-1 cells were done in the whole-cell configuration of the patch-clamp technique at RT (20-22C) with an Axopatch 200 patch-clamp amplifier (Axon Instruments; Burlingame, CA). The bath solution contained (in mM) 15 BaCl2, 130 N-methyl-glucamine, 10 HEPES, 5 KCl, pH adjusted with HCl to 7.4. The patch electrodes had resistances of 2-4 MΩ when filled with an electrode solution composed of (in mM) 130 CsCl, 5 oxalacetate, 5 creatine, 5 pyruvate, 1 EGTA, 10 HEPES/CsOH (pH 7.4). The cells were placed on a 12-mm-wide round coverslip in a recording chamber and were continuously superfused at a rate of 2-5 ml/min driven by gravity from several reservoirs. The membrane currents were corrected for leakage and capacitive currents on line using a P/4 subpulse protocol (ISO2; MFK, Germany). Capacitance of a cell was estimated by means of ramp depolarizations. Currents were filtered at 1 kHz and digitized at 5 kHz. Currents were measured either immediately after breakthrough or at 1 or 2 min later after establishment of the whole-cell configuration of the patch-clamp technique.
Results
T-type and Non-L-type Currents in the Insulinoma Cell Line INS-1
The INS-1 cell line is a favored model system for insulin secretion (Asfari et al. 1992) and expresses high-threshold (L-type, dihydropyridine-sensitive) voltage-gated Ca2+ channels (Kennedy et al. 1996), which are well known to trigger insulin secretion. In addition, the INS-1 cell line contains mRNA from different non-L-type (dihydropyridine-insensitive) Ca2+ channels. The neuronal Ca2+ channel α1E-subunit was detected in insulinoma cell lines and islets of Langerhans by RTPCR using specific oligonucleotide primer pairs (Vajna et al. 1998). Using degenerated primer pairs during PCR, no α1E cDNA fragments were amplified (Horváth et al. 1998; Plant et al. 1998). On the basis of these conflicting RT-PCR results, we wanted to determine if substantial amounts of dihydropyridine-insensitive Ca2+ currents could be observed in this cell line. Whole-cell Ca2+ currents in INS-1 cells were recorded in the presence of 2 μM (±)isradipine, a concentration, that does not reduce currents induced by the stable expression of human α1Ed Ca2+ channel subunit in HEK-293 cells (unpublished results). Low and high voltage-activated Ca2+ currents, not related to L-type currents, are clearly detected in INS-1 cells, as shown by a representative example (Figure 1A). With Ba2+ as charge carrier, fast-activating transient inward currents were observed, which are later superimposed by high voltage-activated Ca2+ currents that arise during run-up (Figure 1B). INS-1 cells clearly express dihydropyridine-insensitive voltage-gated Ca2+ currents which might contain resistant components related to α1E.
Specificity and Sensitivity of the Sera
Two α1E-specific sera were used for immunostaining of α1E Ca2+ channels. The anti-E-com serum has been designed to recognize all cloned mammalian α1E-isoforms, while the anti-E-spec serum is directed against part of the 43 amino acid long insert III (Pereverzev et al. 1998), present only in the carboxy terminus of the longer α1E isoforms, α1Ed, α1Ee, and α1Ef (Vajna et al. 1998). In Western blots, both sera specifically recognized a high molecular weight protein of the predicted size using membranes from full-length α1E expressing HEK-293 cells (Nakashima et al. 1998; Pereverzev et al. 1998). Only the anti-E-com serum detected a positive polypeptide of the predicted size when membranes from truncated α1Ed-CDEL were analyzed lacking the peptide epitope (peptide Nast-197) used for raising antibodies (Pereverzev et al. 1998).
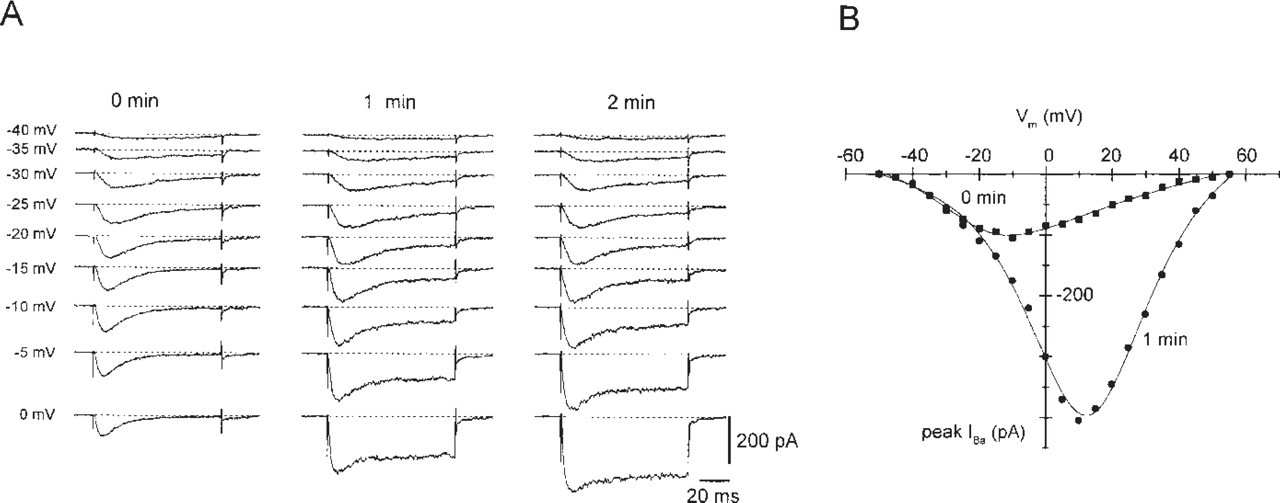
Dihydropyridine-insensitive low voltage- and high voltage-activated Ca2+ currents in the rat insulinoma cell line INS-1. Inward currents were recorded in the presence of 2 μM (±) isradipine. (A) Time-dependent changes of IBa recorded in INS-1 cells. Currents were recorded in response to 100 msec test pulses from -90 mV to the indicated potentials directly after establishing the whole-cell configuration of the patch-clamp technique (0 min), after 1 and 2 min. In the absence of Na+ ions and in the presence of 15 mM Ba2+ as charge carrier, the Ba2+ inward currents show a marked run-up. The dihydropyridine-insensitive voltage-gated Ba2+ inward currents are up to threefold increased similar to a relief of G-protein-mediated downmodulation as reported for chromaffin cells (Carabelli et al. 1998). (B) Current voltage-relation of peak IBa recorded in INS-1 cells. The amplitude of peak Ba2+ inward currents (IBa) was recorded immediately after establishing the whole-cell configuration (0 min) or 1 min later and was plotted against the test potential.
The untransfected HEK-293 cells were negative for the peptide antigens, whereas the α1Ed-transfected HEK-293 cells reacted strongly positive with the anti-E-spec serum (Figure 2A). In the insulinoma cell line INS-1, the positive staining using anti-E-spec was blocked by preincubation of the serum with the peptide Nast-197, which was used originally for immunization of the rabbits (Figure 2B). However, positive staining could be observed with the anti-E-spec serum (Figure 2C), which corresponds to recent RT-PCR results (Vajna et al. 1998).
Localization of Longer α1E Isoforms in Rat Cerebellum
In human cerebellar Purkinje cells, expression of α1E has been detected by use of common anti-α1E antibodies (Volsen et al. 1995). The level of α1E transcripts detected by in situ hybridization was reported to be below the limits of detection in rat cerebellar Purkinje cells (Kamphuis and Hendriksen 1998). To explain these conflicting results, the original immuno-histochemical approach was extended and the immunolocalization of α1E isoforms in rat cerebellum was investigated in the present study.
Longer α1E isoforms were detected in the somata and axons of rat Purkinje cells using the anti-E-spec serum (Figure 2D). Similar results were obtained with the common antibody (Figure 2E). When the incubation time with the anti-E-com serum was prolonged, additional positive neurons were detected in the molecular layer of rat cerebellum (Figure 2F). There was no staining when preimmune serum was used instead of primary antibody (not shown) or when the serum was preincubated with the corresponding peptide (Figure 2G). Hence, the immunolocalization of α1E shows that longer α1E isoforms are expressed in rat cerebellum in a pattern similiar to that reported for human cerebellum (Volsen et al. 1995). In rat, neurons scattered throughout the molecular layer express the shorter α1E isoforms at lower protein density. The identification of a shorter α1E isoform in addition to the predominant longer one corresponds to faint but consistent staining in rat cerebellum when transcripts were detected by a riboprobe for the shorter α1E isoform (Kamphuis and Hendriksen 1998).
Detection of α1E in the Gastrointestinal System
After RT-PCR screening of neuroblastoma cell lines, insulinoma cell lines, and cell lines of the pituitary gland, major expression of an α1Ee isoform was observed in the insulinoma cell lines and subsequently in RNA from the islets of Langerhans (Vajna et al. 1998). In our present report, serial sections of human pancreas were stained with an antibody against chromogranin A (Figure 2H), the polyclonal serum directed against the longer α1E isoforms (Figure 2I), and an MAb against human insulin (Figure 2K). No staining was observed in the negative control when the primary antibody was replaced with preimmune serum (Figure 3I). When antibodies directed against chromogranin A, insulin, and α1E were used, no staining was seen in the exocrine pancreas. In serial sections (Figures 2H–2K), the pattern of positive cells in the endocrine pancreas stained by anti-E-spec closely resembled that of chromogranin A used as the marker for neuroendocrine cells. Similar results were obtained with the anti-E-com serum and the purified anti-E serum against an α1E fusion protein, which was recently used for immunohistochemical characterization of α1E in the cerebellum (Volsen et al. 1995). In the islets of Langerhans there are more anti-E-spec-positive cells compared to the number of cells positive for anti-insulin, suggesting that not only B- but also A-cells, and presumably other endocrine cells, express the longer α1E isoform.
The immunohistochemical results from the islets of Langerhans led to the hypothesis that the longer α1E isoform may also be expressed in other (neuro)endocrine cells. We used human stomach (antrum mucosa), small intestine, and large intestine (cecum) because the gastrointestinal tract contains more than 20 distinctive endocrine cell types characterized on the basis of their hormonal content (Lechago and Shah 1998), and most of them are localized in the epithelium. A small number of gastrointestinal neuroendocrine cells are also observed in the lamina propria without attachment to the overlying epithelium (Lechago and Shah 1998). In all these experiments, chromogranin A was used as a marker for neuroendocrine cells.
In the gastric antral mucosa, the immunohistochemical staining pattern and the morphology of chromogranin A-positive cells (Figure 3A) approximately correspond to the cells positively stained with anti-E-spec antibodies (Figure 3B). Anti-gastrin antibodies were used as markers for the G-cells and showed a distribution pattern similar to that seen with anti-E-spec (Figure 3C).
In the mucosa of the small intestine, few dispersed cells were detected by the chromogranin A antibody. These cells were usually located within the epithelium or sometimes within the lamina propria (Figure 3D). A similar picture of scattered positively stained cells was observed with the serum specific for the longer α1E isoforms (Figures 3E and 3F). With the anti-somatostatin antibody, smaller numbers of cells were positive (Figure 3G) compared to the anti-E-spec serum (Figure 3H). Anti-E-spec serum detected groups of positive cells at the bottom of the crypts and single positive cells in other regions (Figure 3H). No staining was detected when the anti-E-spec serum was replaced by preimmune serum or when preincubated with the peptide (see Figures 3I and 3K for negative controls of pancreas and colon, respectively).
In conclusion, anti-E-spec-positive cells are dispersed within the antrum of the stomach, the small intestine, and the large intestine, and appear to co-localize with the chromogranin A-positive neuroendocrine cells. In addition, α1E is also expressed in the Paneth cells and the lamina muscularis mucosae, and therefore may be related to lysozyme secretion or other functions.
Discussion
Transcripts of the α1E voltage-gated Ca2+ channels are detected in rat, rabbit, mouse, and human brain and in other organs from different species (Williams et al. 1994; Marubio et al. 1996; Vajna et al. 1998). In mammalian brain, more than four different isoforms can be predicted on the basis of cloned (Niidome et al. 1992; Soong et al. 1993; Schneider et al. 1994; Williams et al. 1994) and PCR-amplified α1E cDNA fragments (Marubio et al. 1996; Pereverzev et al. 1998; Vajna et al. 1998; Kamphuis and Hendriksen 1998). In cerebellum, three different longer α1E isoforms are identified by RT-PCR, which have been described as α1Ed, α1Ee, and α1Ef. The α1Ee isoform is also expressed in rat and human pancreas and kidney (Vajna et al. 1998; Schramm et al. in press). In the present study, the RT-PCR experiments are extended by measuring Ca2+ inward currents and by immunostaining. The detection of dihydropyridine-insensitive Ca2+ inward currents leads to the suggestion that the INS-1 cell line should be a valuable tool for future investigations of the physiological function of α1E. Specific antagonists of α1E, which react not only in vitro but also in vivo (for SNX-482 see Piedras-Rentería and Tsien 1998), are rare. Therefore, other techniques must be substituted for the classical pharmacological tools.
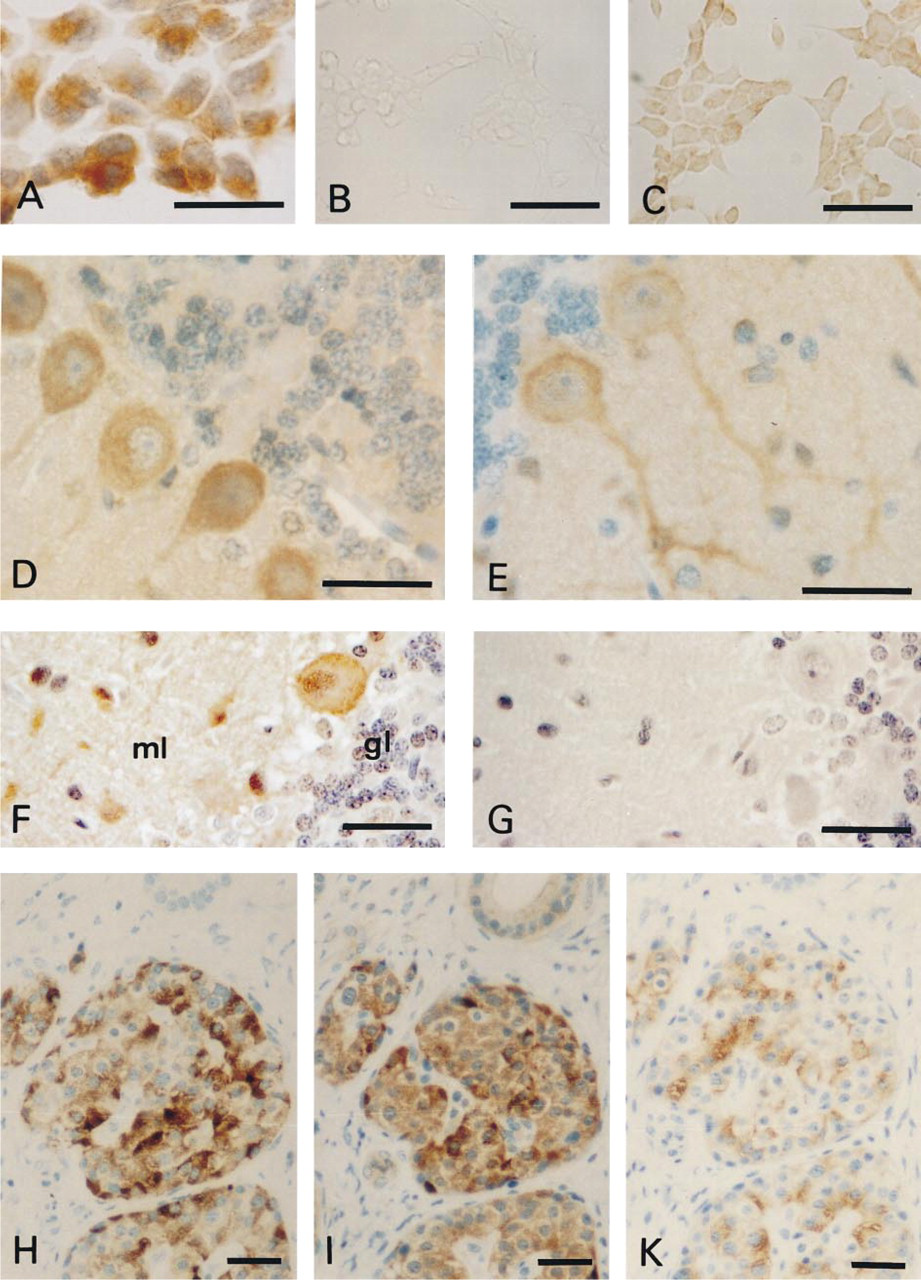
The α1E-subunit in cell lines and cerebellar neurons. Immunocytochemical detection of α1E in stably transfected HEK-293 cells and in INS-1 cells (A-C). HEK-293 cells stably transfected with human α1E (A) were stained by a biotin-streptavidin-horseradish peroxidase technique with DAB as chromogen and hematoxylin as counterstain. INS-1 cells were stained by the same biotin-streptavidin-horseradish peroxidase technique without any counterstain. The primary antibody anti-E-spec was diluted 1:1000. No staining was detected when the primary antibody was replaced by preimmune serum (not shown). INS-1 cells: The serum (anti-E-spec) was diluted 1:500 in the presence (B) or absence (C) of its antigenic peptide, 20 μM peptide Nast-197 to inhibit specific binding of the anti-E-spec serum. (D-G) Detection of α1E isoforms in rat cerebellum. Tissue sections (4 μm) from rat cerebellum were stained by the biotin-streptavidin-horseradish peroxidase technique with DAB as the chromogen and hematoxylin as counterstain. The sera anti-E-spec (D) and anti-E-com (E) were diluted 1:50 after purification with liver powder. The anti-E-com (F) and peptide-pretreated anti-E-com serum (G) were used at 1:100 dilution. The peptide was used during the preincubation at a final concentration of 20 μM. In rat cerebellum, the longer α1E isoforms were detected in Purkinje cells and the shorter in the molecular layer (ml). Less if any staining is seen in the granule cell layer (gl). (H-K) Serial sections of human pancreas (see also Figure 31). Staining is detected in the islets of Langerhans of the pancreas. The pattern of positive cells in the islets after staining with anti-chromogranin A antibody (H) is similar to anti-E-spec, diluted 1:20 (I). Smaller numbers of cells are stained by anti-insulin (K), which is known to stain only the B-cells in the islets. No staining is detected when preimmune serum is used or when the anti-E-spec serum was incubated with 20 μM peptide Nast-197 (I). Bar = 40 μm.
In our study, the major focus was directed towards the expression pattern of shorter and longer α1E isoforms by using two peptide-directed antibodies. The polyclonal serum anti-E-com was designed to detect all known isoforms, and the anti-E-spec was designed to detect the longer α1E isoforms α1Ed, α1Ee, and α1Ef. Rat cerebellum was investigated because it represents the site of major α1E expression.
The number of peripheral isoforms had been deduced from PCR fragments of each of two regions, the II III loop and the carboxy terminus. The estimated structure was confirmed by sequencing subcloned cDNA fragments (Vajna et al. 1998). Neuroendocrine cells of the digestive system were analyzed because they apparently represent a system in which only one isoform of α1E is expressed. If different isoforms of α1E are related to different functions, then the expression of a single isoform in neuroendocrine cells of the digestive system may help to deduce putative function(s) of this α1Ee isoform.
Expression of α1E Isoforms in the Cerebellum
In human, rat, and rabbit cerebellum, the expression of α1E has been investigated in the past by in situ hybridization (Soong et al. 1993; Wakamori et al. 1994; Williams et al. 1994; Volsen et al. 1995) by antibodies raised against a fragment of the II III loop (aa 958-1098 of human α1E; see Volsen et al. 1995) or by peptide-directed antibodies derived from two highly divergent regions in the intracellular II-III loop of rat α1E (Yokoyama et al. 1995). Recently, α1E was found by an antisense riboprobe in the cell bodies of cerebellar Purkinje cells and in the granule cell layer (Volsen et al. 1995). At the protein level, α1E was found to be expressed in Purkinje cells, especially in the cell body extending into the dendritic tree. Axons of Purkinje cells were also stained, but a lower amount of α1E was detected in the molecular layer (Volsen et al. 1995).
The expression of α1E in Purkinje cells and the granule layer of the cerebellum has been confirmed with peptide-directed antibodies (Yokoyama et al. 1995), whereas two other groups using in situ hybridization techniques either failed to detect α1E transcripts in Purkinje cells (Kamphuis and Hendriksen 1998) or found only low densities of α1E transcripts in the granular layer and single Purkinje cells (Wakamori et al. 1994). This may indicate that the mRNA level of α1E is close to the limits of detection, whereas the protein density of α1E is sufficient to be detected by antibodies.
By use of common (anti-E-com) and isoform-specific (anti-E-spec) sera in our present study, the expression of longer α1E isoforms was shown in rat cerebellar Purkinje cells, confirming recent reports of α1E expression in human cerebellum (Volsen et al. 1995). In addition, anti-E-com serum stained the shorter α1E isoforms in the molecular layer. Our data confirm the results of Volsen et al. (1995) with regard to the localization of α1E in cerebellum. In addition, we detected positive staining in the molecular layer, which is attributed to a minor expression of shorter isoforms in cerebellum, as can be deduced from a minor amplification of shorter α1E cDNA by RT-PCR (Vajna et al. 1998).
Immunohistochemical staining of neurons in the granule cell layer was only occasionally noted. Distinct staining of a few neurons in the granule cell layer has been recently reported with another set of peptide specific antibodies (Yokoyama et al. 1995). The staining of only a few neurons in the granule cell layer may be important when granule cells are investigated in primary culture in which they express R-type Ca2+ channels (Tottene et al. 1996; Randall and Tsien 1997; Schramm et al. in press). Preliminary data point to a specific distribution of α1E in the neurites of cerebellar granule cells in primary culture (Bertaggia et al. 1998). Our data lead to the conclusion that the longer α1E isoform is weakly expressed in the granule cell layer.
Expression Patterns of the α1E Ca2+ Channels and the Marker for Neuroendocrine Cells, Chromogranin A, Are Similar in Pancreas and in Cells of the Endocrine Digestive System
Neurons and neuroendocrine cells possess specific organelles for the secretion of peptide hormones, neuropeptides, and neurotransmitters. These signaling molecules are stored either in the small, transparent synaptic vesicles of neurons and their counterparts in endocrine cells or in the so-called large, dense-core vesicles of endocrine cells and their counterparts in neurons (Huttner et al. 1991; Gainer and Chin 1998). Most neuroendocrine granules contain, in addition to their specific peptide hormones or neuropeptides, one or more of the chromogranin/secretogranin peptides (Portela-Gomes et al. 1997), which led to their use as markers for neuroendocrine cells. The validity of anti-chromogranin A antibodies as markers for various endocrine cell types in different parts of the human gastrointestinal tract has recently been shown by use of double- and triple-immunofluorescence staining of the same section (Portela-Gomes et al. 1997). According to this report, chromogranin A is co-localized with gastrin and serotonin in endocrine cells of the antrum ventriculi, as well as with cholecystokinin, secretin, and enteroglucagon in endocrine cells of the villi, crypts, and Brunner's glands in duodenum.
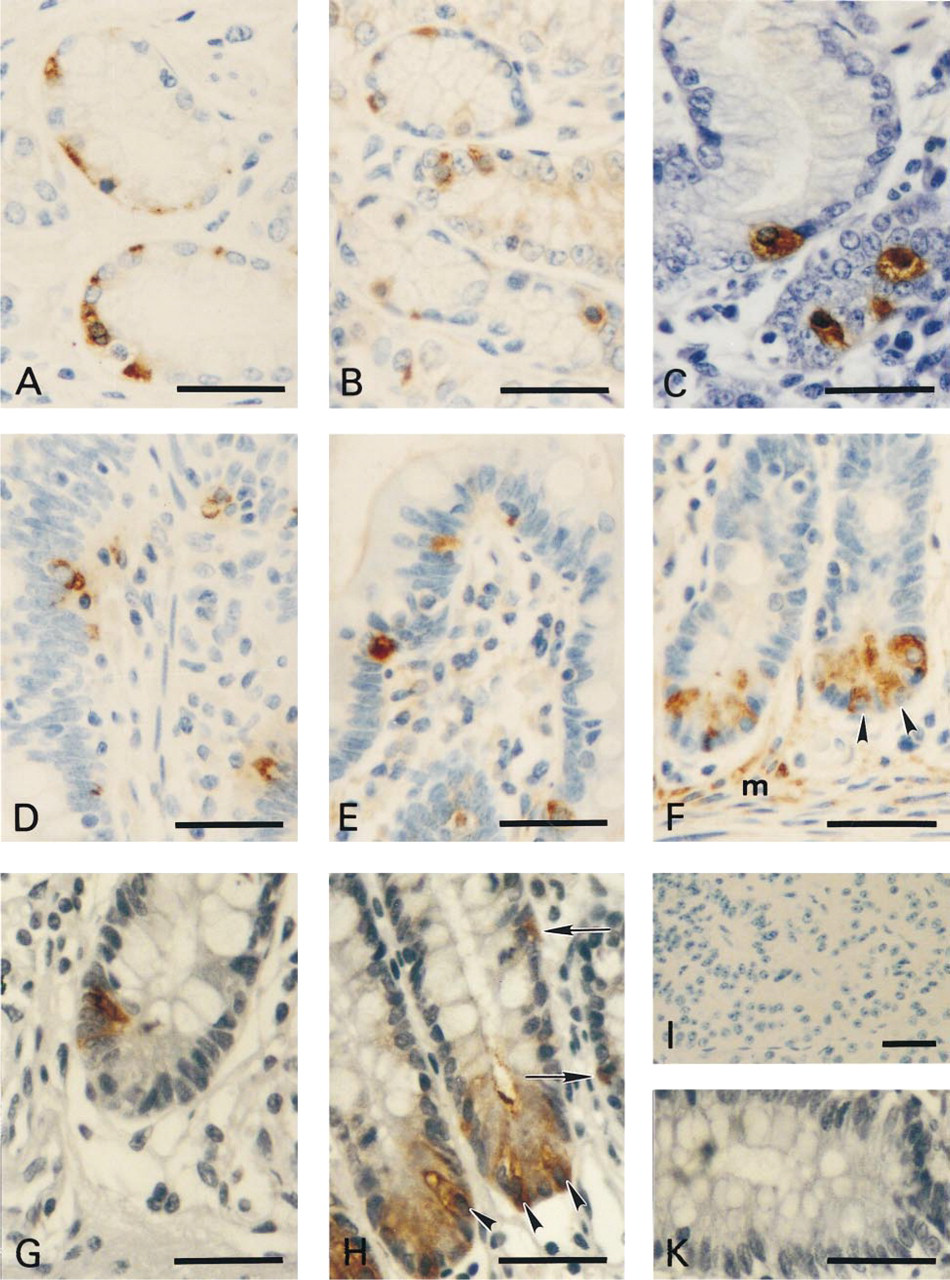
Immunohistochemical localization of α1E in the endocrine digestive system. Immunohistochemical localization of α1E in the antrum of stomach (B), small intestine (E,F), and large intestine (H). Anti-chromogranin A was used as a marker for neuroendocrine cells in antrum ventriculi (A) and small intestine (D). In addition, an anti-gastrin antibody was used to demonstrate G-cells in the antral mucosa (C). In large intestine (cecum), anti-somatostatin (G) stained a smaller number of cells compared to anti-serotonin antibodies (not shown). Positive staining with anti-E-spec was detected not only in endocrine cells (H, arrows) but also in Paneth cells (arrowheads in F,H) as well as punctate staining in the lamina muscularis mucosa (F, m). No staining was observed when the anti-E-spec serum was preincubated with 20 μM peptide Nast-197 (K). (I) The negative control for immunohistochemical staining of the pancreas from Figure 2 is shown. Bars = 40 μm.
In selected tissues from the human gastrointestinal tract, a similar, almost identical localization of the longer α1E isoform and chromogranin A has been observed by examination of serial tissue sections of human pancreas, antrum ventriculi, and small and large intestine (cecum). In islets of Langerhans, the number of α1E-positive cells is almost equal to the number of chromogranin A-positive cells but exceeds the number of insulin-positive cells. Therefore, it can be concluded that the longer α1E isoform, probably α1Ee, is co-localized with endocrine cells of the pancreas and with endocrine cells in the gut. This relation of α1E to cells of the neuroendocrine system points to a putative function of α1E-containing Ca2+ channels during secretion of neurohormones and neuropeptides.
Additional staining of Paneth cells and of distinct locations in the lamina muscularis mucosae points to similar or additional functions of α1E that might be related to lysozyme secretion in the gut and to neurotransmitter secretion at the presynaptic nerve terminals (Wu et al. 1999). Together with two other non-L-type subunits, α1A and α1B, the α1E-subunit has been detected in single calyx-type synapses by immunostaining. Therefore, the positive staining detected in the lamina muscularis mucosae might be related to nerve endings rather than to smooth muscle cells.
Voltage-gated Ca2+ Channels and Secretion
The biological processes during neurosecretion and transmitter release include biosynthesis and processing of the propeptide or transmitter, axonal transport of the signal molecules, and Ca2+-dependent secretion from the nerve terminal. The basic mechanisms underlying secretion are assumed to be universal, from yeast to mammalian neurons (Bennet and Scheller 1993), and the molecular machinery might be conserved in neuronal transmitter release and endocrine hormone release (for review see Gainer and Chin 1998).
Ca2+-dependent insulin release has been related to several voltage-gated Ca2+ channel types. The dihydropyridine-sensitive α1D and α1C Ca2+ channels initiate major Ca2+ influx (Keahey et al. 1989; Safayhi et al. 1997). However, ω-conotoxin GVIA, a blocker of α1B/N-type Ca2+ channels (Table 1), does not contribute to the insulin secretion of human B-cells (Davalli et al. 1996), leaving about 20% of the glucose-induced insulin release resistant to blockers of L- and N-type voltage-gated Ca2+ channels.
Recently, ω-agatoxin IVA (500 nM), a blocker of α1A/P- and Q-type Ca2+ channels (Table 1), has been shown to inhibit a portion of insulin secretion (Ligon et al. 1998). It blocked almost all, but not completely the dihydropyridine-resistant insulin release. Keeping in mind that ω-agatoxin IVA also inhibits recombinant rat α1E Ca2+ channels up to 75%, with an apparent KD of 51 nM (Stephens et al. 1997), the possibility arises that part of the dihydropyridine-resistent Ca2+ currents may be carried by α1E-type Ca2+ channels and that part of the dihydropyridine-resistant insulin release may be guided by α1E Ca2+ channels. During synaptic transmission, Ca2+ influx through P/Q-(α1A), N- (α1B), and R-type Ca2+ channels (α1E) triggers the release of neurotransmitter at a calyx-type terminal in the rat medial nucleus of the trapezoid body with different efficacy (Wu et al. 1999). Two of these Ca2+ channels are believed to interact directly with proteins of the synaptic vesicle docking and fusion machinery, such as syntaxin and SNAP-25 (Rettig et al. 1996). The interacting regions of the α1A- and α1B-subunits have been localized to two adjacent segments of the intracellular loop connecting Domains II and III. This so-called synaptic protein interaction (synprint) site has been compared with the structurally related α1E-subunit (Figure 4A). It is noteworthy that the Insert I corresponds, in part, to a duplication of the adjacent downstream arginine-rich sequence and is not redundant in the peripheral isoform α1Ee. Overall, this region downstream of Insert I is very divergent in the three structurally related α1-subunits. The low sequence homology of Insert I to α1A or α1B, on the one hand, and, on the other hand, to the sequence homology of the arginine-rich fragment unique in α1E to the peptide recently identified for functional interaction between an L-type Ca2+ channel and the ryanodine receptor (Figure 4B; El-Hayek and Ikemoto 1998) leads to the conclusion that α1E may be involved in a different mechanism during excitation-secretion compared to α1A and α1B. The identification of in vivo interaction partners would help us to understand the function of α1E in neuroendocrine cells.
The number of identified Ca2+ channels involved in secretion is increasing, which raises the following question: Why does the endocrine cell need so many similar Ca2+ channels in parallel (Armstrong 1998)? A possible answer to this question with respect to α1E is that the α1E Ca2+ channels could add an easily modulated Ca2+ channel that is regulated during signaling processes by stimulating and inhibiting cascades as they are realized during secretion (Sachs et al. 1997). The α1E-subunit is modulated in vitro by protein kinase C (Zamponi et al. 1997) and somatostatin (Mehrke et al. 1997). Therefore, it is possible that α1E might be an important candidate for Ca2+-mediated secretion in addition to L-type Ca2+ channels, and might be modulated by the intercrossing pathways of protein kinase C and somatostatin. The electrophysiological properties of recently identified carcinoid cells of the human gut (Glassmeier et al. 1997) resemble those of a somatostatin-sensitive, voltage-dependent α1E Ca2+ channel. In the future, the insulinoma cell line INS-1 or other cell lines that secrete peptide hormones will be used as model systems for investigation of α1E-specific toxins and their effects on hormone secretion.

Comparison of a Ca2+ channel/synaptic vesicle protein-interaction domain, the so-called synprint region, from α1A and α1B with the homologous region of α1E. (A) Alignment of non-L-type voltage-gated Ca2+ channels (fragment of the II-III loop) containing part of the synprint site (α1A, bold and underlined; α1B, latin and underlined), and insert I of α1E (∗∗∗). The aligned sequences correspond to the human α1E-subunit (= α1E) with the accession number L27745, the human α1B-subunit (= α1B), number M94172, and the human α1A-subunit (= α1A), number X99897. The isoform-specific insert I of α1E (Pereverzev et al. 1998) represents a duplication of a 19-amino-acid arginine-rich region adjacent to insert I (see alignment in B). Note that insert I is located in a region of the II-III loop, which is structurally more divergent than the flanking sequences involved in the interaction with synaptic vesicle proteins. Especially in the upstream region, the three sequences are very similar, marked by identical positions in the “consensus” sequences. Bold and underlined: the II-III loop interaction site with synaptic vesicle proteins (= synprint site), taken from (Rettig et al. 1996). Latin and underlined: the synprint site of rat α1B is deduced from the corresponding rat sequence as mentioned in Yokoyama et al. (1997). (B) Alignment of Insert I from human α1E (aa 749-aa 767) with the adjacent sequence of α1E (aa 768-aa 786) and an arginine-rich fragment in rabbit α1S (aa 679-aa 696). Insert I is not present in cDNA fragments from cerebellum (isoform α1Ee), pancreas, and kidney (Vajna et al. 1998). Insert I shows high structural homology to the adjacent downstream region of α1E (aa 768-786). The most effective minimal peptide, which is interfering with skeletal muscle excitation-contraction coupling (El-Hayek and Ikemoto 1998), has a similar arginine-rich region that is directly compared with the sequence of insert I. In the 19-amino-acid region, five amino acids are different between the two human α1E fragments, and two of them correspond to conservative substitutions.
Footnotes
Acknowledgments
Supported by the Köln Fortune Program/Faculty of Medicine, University of Cologne (to MW and TS) and by the Center of Molecular Medicine Cologne/Zentrum für Molekularbiologische Medizin Köln (Bundesministerium für Bildung, Wissenschaft, Forschung und Technologie, Förder-kennzeichen 01 KS 9502, to TS and JH).
We thank Dr W. Nastainczyk (Universität des Saarlandes, Homburg) for the synthesis of peptides and antibodies, Ms M. Chludek (Pathologie/Leverkusen) for her permanent help during preparation of serial sections, and Ms R. Clemens, Ms S. Schulze, and Ms U. Tampier for technical assistance.
