Abstract
We are in the process of assessing the response of cancer tissues to chemotherapy, evaluating, among other points, the proportion of cancer cells undergoing apop-tosis. However, the apoptotic index obtained with the original TUNEL technique was lower than that obtained by evaluation of apoptosis on H&E-stained sections. Here we describe a small modification of the TUNEL technique that significantly increases the sensitivity of the assay. In the nonmodified TUNEL technique, a digoxigenin-labeled probe is detected using a direct peroxidase-conjugated system, whereas here we report the advantage of using a streptavidin-biotin-immunoperoxidase system. This, in conjunction with pretreatment of tissue sections with proteinase K and microwave irradiation, improved the detection of apoptotic cells.
Keywords
Although uncontrolled neoplastic growth is believed to result from increased cell proliferation, a diminished rate of cell death, resulting from the failure of cells to undergo apoptosis in response to different signals, has also been implicated in tumor growth (Carson and Ribeiro 1993). Furthermore, the cytotoxic effects of most anticancer drugs produce damage to DNA, which can lead, either directly or indirectly, to induction of apoptosis in tumor cells. We are assessing the response to chemotherapeutic agents by evaluating, among other parameters, the proportion of cancer cells that are dying. These studies are being performed on tissue sections from pre- and postchemotherapy biopsy specimens obtained from cancer patients treated with induction chemotherapy (Vargas-Roig et al. 1998).
Ideally, apoptosis should be assessed by more than one method. In the small biopsies that we have been studying, apoptosis was evaluated by morphological characteristics and by the TUNEL (terminal deoxynucleotidyl transferase-mediated dUTP nick end-labeling) technique. This technique has become the most widely used histochemical method to detect apoptotic cells. However, it is somewhat disappointing because of its limited sensitivity. Labat-Moleur et al. (1998) have recently examined and reviewed important points that affect the sensitivity and specificity of the TUNEL technique: fixation (including archival paraffin-embedded specimens) and pretreatment of the tissue sections (detergents, proteinase K, microwave irradiation). The main conclusion of these authors is that improvement “does not come from retrieval reinforcement but from qualitative adaptations of retrieval techniques.”
In this study we have developed a slight modification of the detection system using the ApopTag Plus kit (Oncor, Gaithersburg, MD; S7101-KIT), which enabled us to significantly increase the sensitivity of the technique. We replaced the direct immunoperoxidase detection of digoxigenin-labeled genomic DNA with an amplification system. Briefly, tissue sections 5-6 μm thick were mounted on 3-aminopropyltrietoxysilane (Sigma Chemical; St Louis, MO)-coated slides. Thereafter, the sections were deparaffinized and rehydrated through graded alcohols to water. The sections were then incubated with 20 μg/ml of proteinase K (Promega; Madison, WI) in PBS at pH 7.4 for 15 min at room temperature (RT). Endogenous peroxidase activity was quenched by incubation with 2% H2O2 in PBS for 5 min at RT. We did not observe decreased activity of TdT when the samples were pretreated with H2O2 to block endogenous peroxidase, contrary to what has been reported by Migheli et al. (1995). The equilibration buffer and enzymatic incorporation of nucleotides labeled with digoxigenin (contained in the reaction buffer) were performed according to the manufacturer's instructions. The sections were then treated with 10% BSA for 30 min at RT (this step was important to block nonspecific protein binding, which could lead to nonspecifically labeled nuclei). The detection system was modified as follows. The antibody provided in the kit was replaced by a biotin-conjugated mouse monoclonal anti-digoxin antibody (Sigma; B-7405). This antibody was used at 1:750 dilution in PBS, pH 7.4, to incubate the tissue sections overnight at 4C in a humidified chamber. After a brief rinse with automation buffer [80 mM Tris-HCl, 20 mM Tris(hydroxymethyl)-aminomethane and 0.075% BRIJ 35 (v/v), pH 7.5] for 10 min at RT, the sections were incubated with biotinylated rabbit anti-mouse IgG (whole molecule) (Dako, Carpinteria, CA; E0354) at 1:100 dilution for 45 min at RT. The tissue sections were then washed with automation buffer for 15 min at RT and incubated with peroxidase-labeled streptavidin (streptABComplex/ HRP, Dako; K0377) at 1:100 dilution for 45 min at RT. After a brief wash, the sections were incubated for 5 min with 3,3'-diaminobenzidine tetrahydrochloride (0.5 mg/ml)/H2O2 (0.01%), which was used as chromogen substrate. Tissue sections were lightly counterstained with 0.5% methyl green or with hematoxylin to reveal nuclei, and the slides were observed with an IM35 microscope (Carl Zeiss; Oberkochen, Germany).
For positive control, we used paraffin sections from involuting prostates of castrated rats (n =2). In this tissue apoptosis is well characterized and easy to detect on H&E-stained sections. We also evaluated the apoptotic index (AI) in tumor cells from postchemotherapy biopsies from breast (n = 2) and cervical (n = 2) cancer patients. In each case, a total of eight slides were examined and the AI was obtained in 1000 cells from each slide. Table 1 shows the AI obtained on H&E-stained sections and on serial sections treated with the TUNEL technique. The best results were obtained when the samples were subjected to proteolytic digestion with proteinase K (20 μg/ml) for 15 min at RT followed by microwave irradiation with 0.01 M citrate buffer, pH 3 (Labat-Moleur et al. 1998), and using the modified detection system described above. Figure 1 shows representative sections stained with the standard and the modified TUNEL technique. The sensitivity of this protocol allowed use of the reaction buffer from the commercial kit at one third of the concentration suggested by the manufacturer. Finally, we should mention that, in the involuting prostate, a number of apparently nonapoptotic nuclei (preapoptotic?) (18%) were stained with the modified TUNEL protocol. In the cancer biopsies these “preapoptotic” nuclei were detected more infrequently (8%). This may be because these samples were taken 21 days after chemotherapy, whereas the prostate tissue was obtained soon (3 days) after castration.
Examples of the apoptotic indices obtained on H&E-stained sections and on tissue sections processed with the different TUNEL protocols a
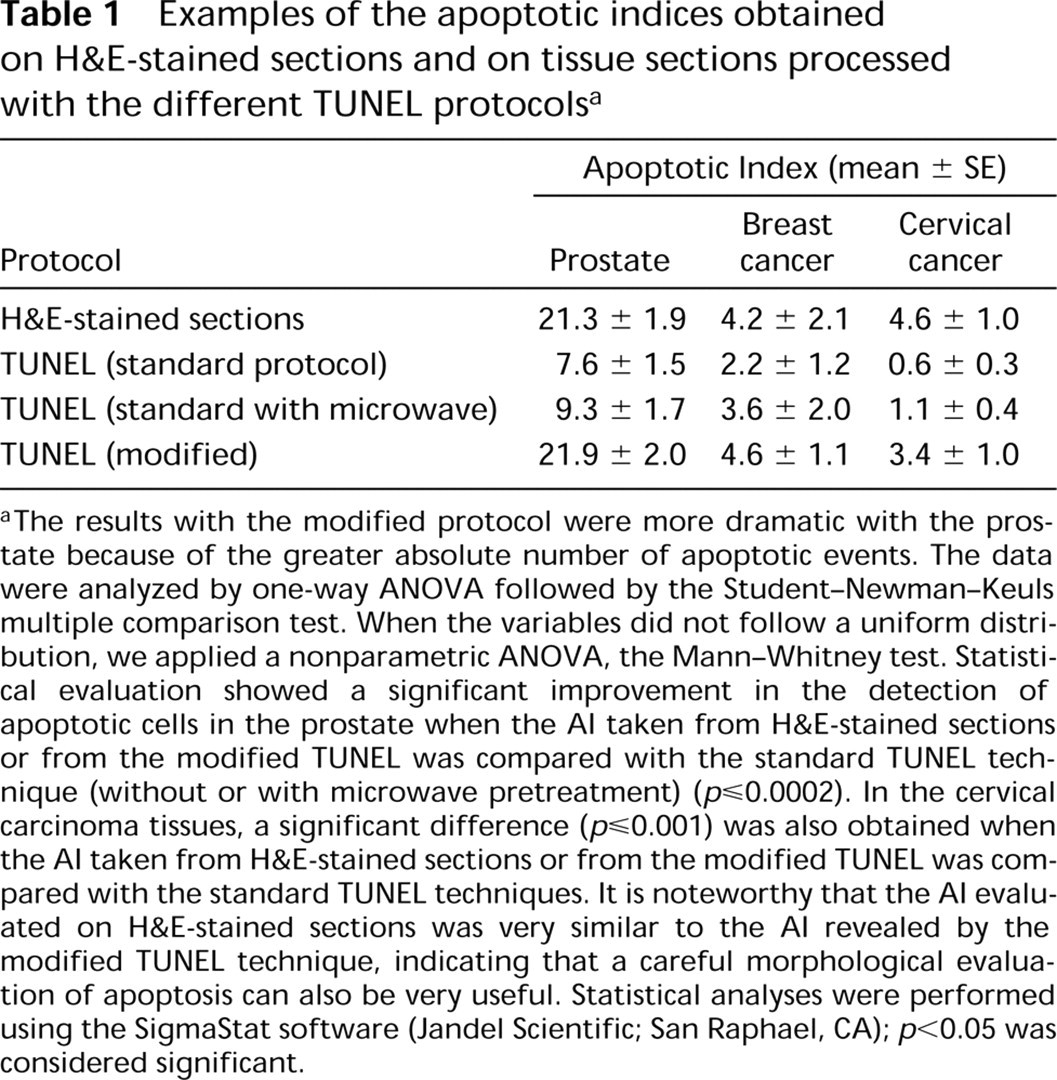
aThe results with the modified protocol were more dramatic with the prostate because of the greater absolute number of apoptotic events. The data were analyzed by one-way ANOVA followed by the Student-Newman-Keuls multiple comparison test. When the variables did not follow a uniform distribution, we applied a nonparametric ANOVA, the Mann-Whitney test. Statistical evaluation showed a significant improvement in the detection of apoptotic cells in the prostate when the AI taken from H&E-stained sections or from the modified TUNEL was compared with the standard TUNEL technique (without or with microwave pretreatment) (p≤0.0002). In the cervical carcinoma tissues, a significant difference (p≤0.001) was also obtained when the AI taken from H&E-stained sections or from the modified TUNEL was compared with the standard TUNEL techniques. It is noteworthy that the AI evaluated on H&E-stained sections was very similar to the AI revealed by the modified TUNEL technique, indicating that a careful morphological evaluation of apoptosis can also be very useful. Statistical analyses were performed using the SigmaStat software (Jandel Scientific; San Raphael, CA); p<0.05 was considered significant.
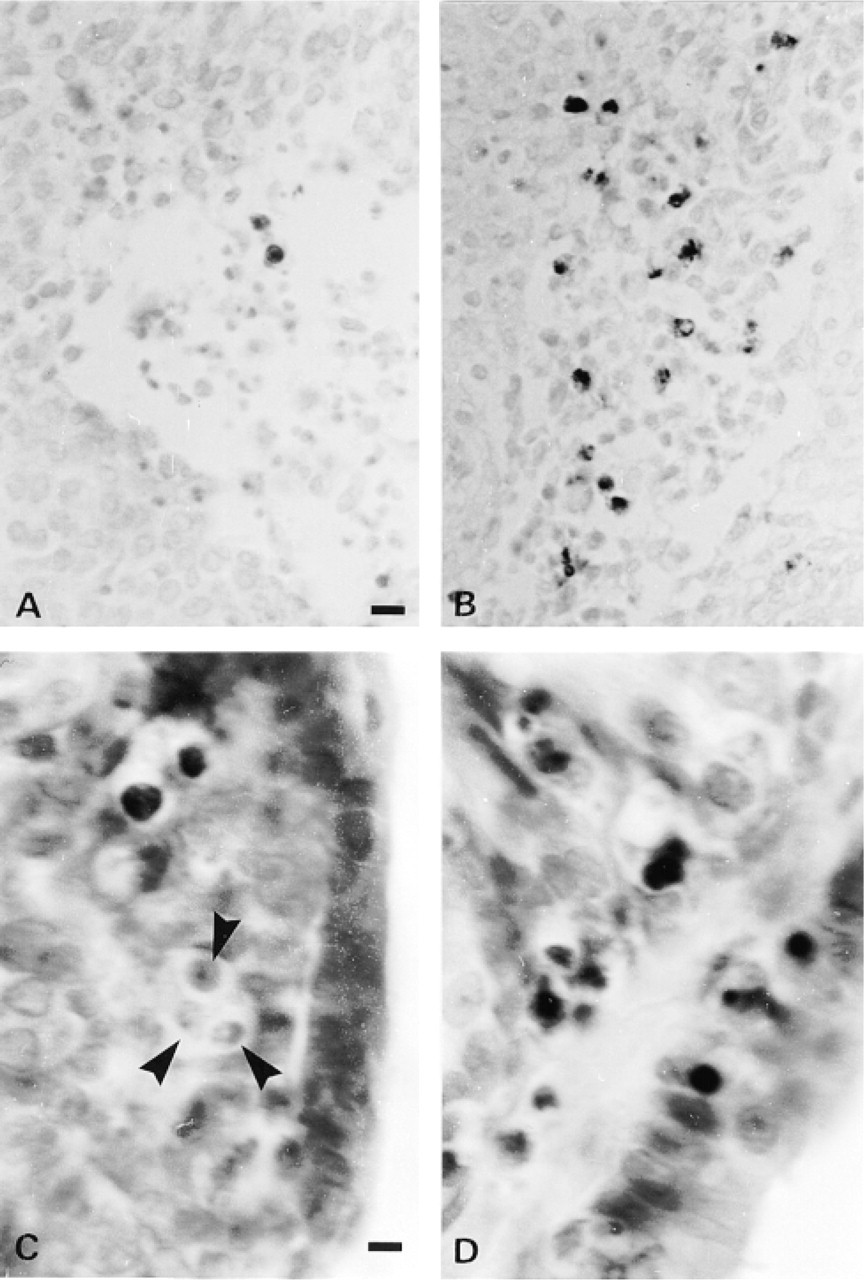
Detection of apoptotic cells (dark spots) using the standard TUNEL technique (A and C) vs the modified TUNEL technique (B and D). A and B are tissue sections from a cervical carcinoma. Methyl green counterstaining. Bar = 20 μm. C and D are tissue sections from an involuting prostate from a castrated rat. Note a cluster of apoptotic cells (arrowheads) not stained with the standard TUNEL assay (C). Light hematoxylin counterstaining. Bar = 7 μm.
Footnotes
Acknowledgments
Supported by a grant from the Agencia Nacional de Promoción Científica y Tecnológica of Argentina (BID802/OC-AR PID:05-00000–00766).
We wish to thank Dr Andrei Laszlo (Washington University School of Medicine, St Louis, MO) for editing the manuscript.
