Abstract
In this study we examined if an association exists between expression of an alternatively spliced “embryonic” fibronectin isoform EIIIA (Fn-EIIIA) and α-smooth muscle actin (α-SMA) in the maturing and adult rat kidney and in two unrelated models of glomerular disease, passive accelerated anti-glomerular basement membrane (GBM) nephritis and Habu venom (HV)-induced proliferative glomerulonephritis, using immunohistochemistry and in situ hybridization. Fn-EIIIA and α-SMA proteins were abundantly expressed in mesangium and in periglomerular and peritubular interstitium of 20-day embryonic and 7-day (D-7) postnatal kidneys in regions of tubule and glomerular development. Staining was markedly reduced in these structures in maturing juvenile (D-14) kidney and was largely lost in adult kidney. Expression of Fn-EIIIA and α-SMA was reinitiated in the mesangium and the periglomerular and peritubular interstitium in both models and was also observed in glomerular crescents in anti-GBM nephritis. Increased expression of Fn-EIIIA mRNA by in situ hybridization corresponded to the localization of protein staining. Dual labeling experiments verified co-localization of Fn-EIIIA and α-SMA, showing a strong correlation of staining between location and staining intensity during kidney development, maturation, and disease. Expression of EIIIA mRNA corresponded to protein expression in developing and diseased kidneys and was lost in adult kidney. These studies show a recapitulation of the co-expression of Fn-EIIIA and α-SMA in anti-GBM disease and suggest a functional link for these two proteins.
Keywords
A
Recent interest has centered on a role for fibronectin (Fn), particularly an isoform containing an extra domain EIIIA (Fn-EIIIA), as a mediator of mesenchymal cell activation. The functions of the EIIIA domain are not known. However, its close proximity to the RGDS cell binding domain suggests that this isoform has specific functional roles (Schwarzbauer et al. 1985; Paul et al. 1986; Hynes 1990; Schwarzbauer 1991; ffrench-Constant 1995). The Fn-EIIIA variant is abundantly expressed during embryogenesis (ffrench-Constant and Hynes 1988,1989; Peters and Hynes 1996) and at the margins of healing wounds (ffrench-Constant et al. 1989; Brown et al. 1993), whereas this domain is spliced out of plasma Fn (derived from hepatocytes) and many tissue-specific cells in adult tissues (Peters et al. 1996), suggesting that it has important functions in remodeling (Paul et al. 1986; Schwarzbauer 1991; ffrench-Constant 1995). Functions for Fn-EIIIA have not been determined, but a close association with cells undergoing high rates of migration, proliferation, and differentiation suggests a role in cell activation (Schwarzbauer et al. 1985; ffrench-Constant and Hynes 1988,1989; ffrench-Constant et al. 1989; Hynes 1990; Schwarzbauer 1991; Brown et al. 1993; Barnes et al. 1994a,1995b; ffrench-Constant 1995; Peters and Hynes 1996).
Fn protein has been detected in glomeruli and the interstitium in developing kidney decreasing in intensity during maturation (Mounier et al. 1986; Peters and Hynes 1996; Peters et al. 1996). Similarly, α-SMA localizes in mesangial and peritubular structures during kidney development but is lost in adult kidney (Carey et al. 1992). Fn localizes in glomeruli and the peritubular interstitium in renal disease (Barnes et al. 1994a, 1995b; Yamamoto et al. 1994; Nickeleit et al. 1995; Alpers et al. 1996; El Nahas et al. 1996; Kliem et al. 1996), similar to areas of increased α-SMA expression during fibrosis, suggesting that these two proteins may be associated in tissues undergoing high rates of remodeling. However, the studies listed above examined localization of either Fn or α-SMA alone and did not examine if these two proteins co-localize or follow the same course of expression. Moreover, expression of Fn-EIIIA isoform in renal disease has been examined (Barnes et al. 1994a, b; Yamamoto et al. 1994; Nickeleit et al. 1995; Alonso et al. 1996), but correlations between this isoform and α-SMA expression have not been determined. Similarly, it is not known if the Fn-EIIIA isoform and α-SMA co-localize and follow the same course of expression during renal maturation. Because the Fn-EIIIA isoform may have an important role in cell activation in the kidney during nephrogenesis and remodeling, we examined the course of expression of Fn-EIIIA mRNA and the co-localization of these two proteins in embryonic, maturing, and adult kidney and in two unrelated models of renal disease characterized by mesangial cell proliferation, interstitial nephritis, and/or glomerular crescents. The results showed that Fn-EIIIA and α-SMA co-localize and follow the same course of expression during kidney development and maturation and that a recapitulation of co-expression occurs in the mesangium and interstitium during renal disease.
Materials and Methods
Embryonic, Juvenile, and Adult Kidney
To examine the course of expression of Fn-EIIIA and α-SMA during kidney maturation, kidneys were obtained from 20-day embryos just before birth (rat gestation is 22 days), 7- (D-7) and 14-day (D-14) postnatal juveniles, and 6-8-week adult Sprague-Dawley rats (Charles River; Raleigh, NC). Kidney tissue was sliced and immediately frozen in liquid nitrogen, and stored in cryogenic tubes at −70C for subsequent immunohistochemistry and in situ hybridization.
Induction of Anti-glomerular Basement Membrane (GBM) Nephritis
Anti-GBM Antibody. Glomeruli were isolated from Sprague-Dawley rat kidneys by differential sieving and GBM was purified according to the methods of Meezan et al. (1975). Rabbits were immunized with 100 μg GBM in complete Freund's adjuvant, followed 10 days later with an equal amount of antigen in Freund's incomplete adjuvant. The rabbits received periodic booster injections of 50-100 μ g GBM in saline
Protocol
An accelerated nephrotoxic nephritis was induced according to the methods of Lan et al. (1991). Nine rats were immunized with 5 mg rabbit IgG in Freund's complete adjuvant. Five days later the rats were divided into two groups, one challenged with 15 mg rabbit anti-GBM IgG in PBS (n = 5)
Proliferative Glomerulonephritis Induced by Habu Venom (HV)
To examine an association between Fn-EIIIA and α-SMA in lesions in a model of nonimmune glomerular disease, five rats were injected with HV as previously reported (Barnes 1989; Barnes and Abboud 1993; Barnes et al. 1994a, b, 1995b). Briefly, the rats were unilaterally nephrectomized to increase the incidence of subsequent glomerular lesions and 24 hr later they were injected with HV (Trimeresurus flavoviridis; Sigma Chemical, St Louis, MO) at a dose of 3.5 mg/kg
Immunohistochemical Localization of Fn-EIIIA and α-SMA Proteins
Localization of Fn-EIIIA and α-SMA was assessed by immunofluorescence and immunoperoxidase histochemistry using mouse monoclonal antibodies (MAbs) specific for the alternatively spliced extra domain (EIIIA) of cellular Fn (clones 3E2, Sigma and IST-9, Serotec; Harlan Bioproducts for Science, Indianapolis, IN). Mouse anti-human α-SMA MAb clone 1A4 was obtained from Sigma. Acetone-fixed frozen sections (6 μ m) were treated as previously described (Barnes 1989; Barnes and Abboud 1993; Barnes et al. 1994b). Sections were incubated with nonimmune IgG of the same species as the second antibody to block nonspecific binding, then with primary antibody followed by FITC- or biotinlabeled second antibodies [rat anti-mouse IgM MAb (Sigma) or donkey anti-mouse IgG MAb (Chemicon International; Temecula, CA)]. Second antibodies were adsorbed with IgGs of a variety of species (other than the primary) to avoid crossreactivity with endogenous rat IgG or to exogenous rabbit IgG administered to elicit anti-GBM nephritis. Sections employing the avidin-biotin complex (ABC; Vector Laboratories, Burlingame, CA) technique for immunoperoxidase were incubated with 0.6% hydrogen peroxide in methanol to block nonspecific peroxidase activity and 0.01% avidin, 0.001% biotin to block endogenous biotin activity. All incubations of primary and second antibody were for 30 min with three washes with PBS containing 0.1% bovine serum albumin (BSA), 5 min each between steps. Controls consisted of nonimmune mouse IgM, IgG, or PBS-BSA in place of primary antibody, followed by detection procedures as outlined above.
Dual Label Immunohistochemistry
To verify co-localization of EIIIA and α-SMA, dual label immunohistochemistry was employed utilizing two separate fluorescence tags. Tissue sections of representative kidneys of D-7 juveniles, anti-GBM, and 72-hr HV experiments were incubated with anti-Fn-EIIIA antibody followed by an affinity-purified FITC-labeled donkey anti-mouse IgG adsorbed with IgG derived from multiple species for dual labeling (Chemicon International). α-SMA was detected by direct immunostaining utilizing a Cy3-labeled mouse MAb (Sigma). Sections were washed with PBS-BSA between all antibody incubations. In addition, sections were incubated with normal nonimmune mouse IgG immediately before Cy3-anti-α-SMA to prevent potential binding of this labeled primary to the localized FITC-labeled second anti-mouse IgG antibody. Sections were viewed and photographed using an Olympus Research microscope equipped for epifluorescence using excitation and bandpass filters optimal for either FITC or Cy3. Sections incubated with anti Fn-EIIIA and FITC-second antibody viewed with the Cy3 filter set and Cy3-anti-α-SMA viewed with the FITC filter set were negative, indicating efficient barrier filtration of cross-illumination from the opposing fluorochromes.
In Situ Hybridization
Synthesis of riboprobe, tissue preparation, in situ hybridization and autoradiography were identical to methods used previously (Barnes et al. 1994a,1995a, b). Briefly, cDNA probes containing a 160-
Preparation of Riboprobes
Linearized cDNA was transcribed in vitro using a Riboprobe system II kit (Promega; Madison, WI) according to the manufacturer's instructions. Either SP6 or T7 RNA polymerase and [35 S]-uridine-5'-(a-thio)-triphosphate (1300 Ci/mMol; New England Nuclear, Boston, MA) were included in the reaction mixture to generate [35 S]-labeled antisense and sense riboprobes. The reaction mixture was incubated for 60 min at 40C, and then the DNA template was removed by digestion with 0.5 U RNase-free DNase, followed by removal of unincorporated nucleotides by phenol-chloroform extraction and ethanol precipitation. RNA probes (activity approximately 4 × 106 CPM/μl) were stored at — 70C and used within 3 days.
Tissue Preparation
Frozen sections (6 μm) were cut and collected on aminosilane-glutaraldehyde-treated slides, then fixed for 20 min in 4% paraformaldehyde in 0.01 M PBS, pH 7.4. The sections were washed twice in PBS, dehydrated through a graded series of ethanols, air-dried, and used immediately for in situ hybridization.
Tissue Hybridization
In situ hybridization procedures were performed as previously described, involving prehybridization, hybridization, and removal of nonspecifically bound probe. Prehybridization steps included treatment with 0.2 N HCl, proteinase K (1 μ g/ml), and acetic anhydride to block background and enhance probe penetration. Twenty-five μ l of hybridization mixture containing 50% formamide, 10% dextran sulfate, 10 mM dithiothreitol, 0.1 M Tris-HCl, pH 7.5, 0.1 M NaPO4, 0.3 M NaCl, 50 mM EDTA, 1 × Denhardt's solution, 0.2 mg/ml yeast tRNA, and 2 × 105 cpm of 35 S-labeled riboprobe was applied to each section and covered with a siliconized coverslip. Hybridizations with EIIIA probes were performed in a sealed humid chamber for 18 hr at 50C. α-SMA probes were very sticky, possibly due to a high content of G-C (75%) in the first half of the strand, and were hybridized at 58C. Excess probe was removed by washing slides in TE buffer, treatment with RNase A to decrease nonspecific background activity, and rinsing in 2 × SSC. Sections were dehydrated in graded ethanols, air-dried, and immersed in the dark in Kodak NTB-2 photographic emulsion (Eastman-Kodak; Rochester, NY). After air-drying the sections were exposed for 2-3 weeks at 4C. The emulsion was developed and sections were stained with hematoxylin and eosin for subsequent bright- and darkfield microscopic analysis.
Results
Embryonic, Juvenile, and Adult Kidney
Expression of Fn-EIIIA and α-SMA is very similar in the renal parenchyma in the late embryo and early developing kidney, showing a nearly parallel localization of these two proteins in the glomerular mesangium and peritubular interstitium. Interstitial expression of both proteins also showed a parallel reduction of expression in the maturing and adult kidney, but glo-merular α-SMA expression appeared to be preferentially reduced relative to Fn-EIIIA at the D-14 timepoint and beyond. Maturation of the metanephric parenchyma occurs in an outward direction from the interior towards the outer aspect of the cortex, with newly developing structures in the most peripheral aspect of the kidney and more mature structures deeper within the cortex. The pattern of expression of Fn-EIIIA and α-SMA protein followed this course, showing strongest intensity of staining in the peritubular and periglomerular interstitial mesenchymal cells and in the glomerular mesangium (Figures 1A and 1B) in developing cortex in 20-day embryos and in the outermost aspects of the cortex in D-7 kidneys (Figures 1C and 1D). Staining of Fn-EIIIA and α-SMA in more mature structures in the inner aspects of the cortex in D-7 kidneys showed less intensity of staining (Figures 1C, 1D, 2A, and 2B). Staining for both Fn-EIIIA and α-SMA in D-14 kidneys was substantially reduced and showed weak but evenly distributed staining of peritubular and periglomerular structures throughout the cortex (Figures 1E and 1F). Glomeruli expressed Fn-EIIIA and α-SMA in D-14 kidneys. However, staining intensity of α-SMA diminished, particularly in more mature glomeruli, towards the inner cortex. In adult kidneys, staining for Fn-EIIIA (Figure 1G) was lost in the peritubular and periglomerular interstitium, but the glomerular mesangium retained weak staining. Expression of α-SMA (Figure 1H) was entirely lost in the peritubular interstitium and glomerular mesangium throughout the kidney cortex in adult cortex. Dual labeling experiments verified a close correlation of Fn-EIIIA and α-SMA staining in peritubular and glomerular structures in maturing (D-7) kidney (Figures 2A and 2B). However, renal parenchyma destined to become arterial and arteriolar structures in embryonic tissue, as well as arteries and arterioles in maturing and adult kidney, showed a departure from the parallel staining pattern and stained intensely for α-SMA but weakly for Fn-EIIIA (Figures 1C–1H).
Anti-GBM Nephritis
Anti-GBM nephritis was characterized by a mild mesangial proliferative glomerulonephritis, glomerular crescents, and focal areas of interstitial nephritis characterized by tubular atrophy and interstitial expansion. Expression of Fn-EIIIA and α-SMA protein was evident in all areas of disease and showed a distribution similar to that described for developing kidney, with strong staining in the periglomerular and peritubular interstitium and the mesangium (Figures 2C, 2D, and 3A–3D). Glomerular crescents also stained strongly for Fn-EIIIA and α-SMA (Figures 3A and 3B). Co-localization of Fn-EIIIA and α-SMA staining was verified by dual label immunofluorescence microscopy, showing a correlation between location, staining intensity, and severity of lesions in peritubular interstitium (Figures 2C and 2D) and glomeruli.
HV-induced Glomerulonephritis
Administration of HV results in an accelerated proliferative glomerulonephritis characterized by mesangiolysis, development of microaneurysms, and resulting in mesangial proliferative lesions by 72 hr after injection (Barnes 1989; Barnes and Abboud 1993; Barnes et al. 1994b,1995a). In all kidneys studied, micronodules stained intensely for Fn-EIIIA and α-SMA (Figures 2E, 2F, 3E, and 3F). Periglomerular and peritubular structures stained weakly for Fn-EIIIA and α-SMA (Figures 2E, 2F, 3E, and 3F). Dual label immunofluorescence verified a co-localization of both proteins in all lesions examined (Figures 2E and 2F).
In Situ Hybridization
Expression of Fn-EIIIA and α-SMA mRNA corresponded to the localization of their respective proteins in embryonic, maturing, adult, and diseased kidneys (Figure 4). Message for Fn-EIIIA was observed in the interstitial mesenchyme and glomeruli, primarily in the deep cortex of 20-day embryos (Figure 4A). It became most concentrated in these same structures in the outer developing cortex of D-7 kidneys (Figure 4B) and was virtually lost in 2-week (Figure 4C) and adult rats. Similarly, Fn-EIIIA mRNA was detected in glomerular crescents and mesangium (Figure 4D) and in periglomerular and peritubular interstitium in areas of interstitial nephritis in anti-GBM rats (Figure 4E). Glomerular micronodules in kidneys from rats with HV-induced nephritis (Figure 4F) and, to a lesser extent, periglomerular and peritubular structures also showed enhanced expression of Fn-EIIIA mRNA. Sense controls showed negligible background staining.
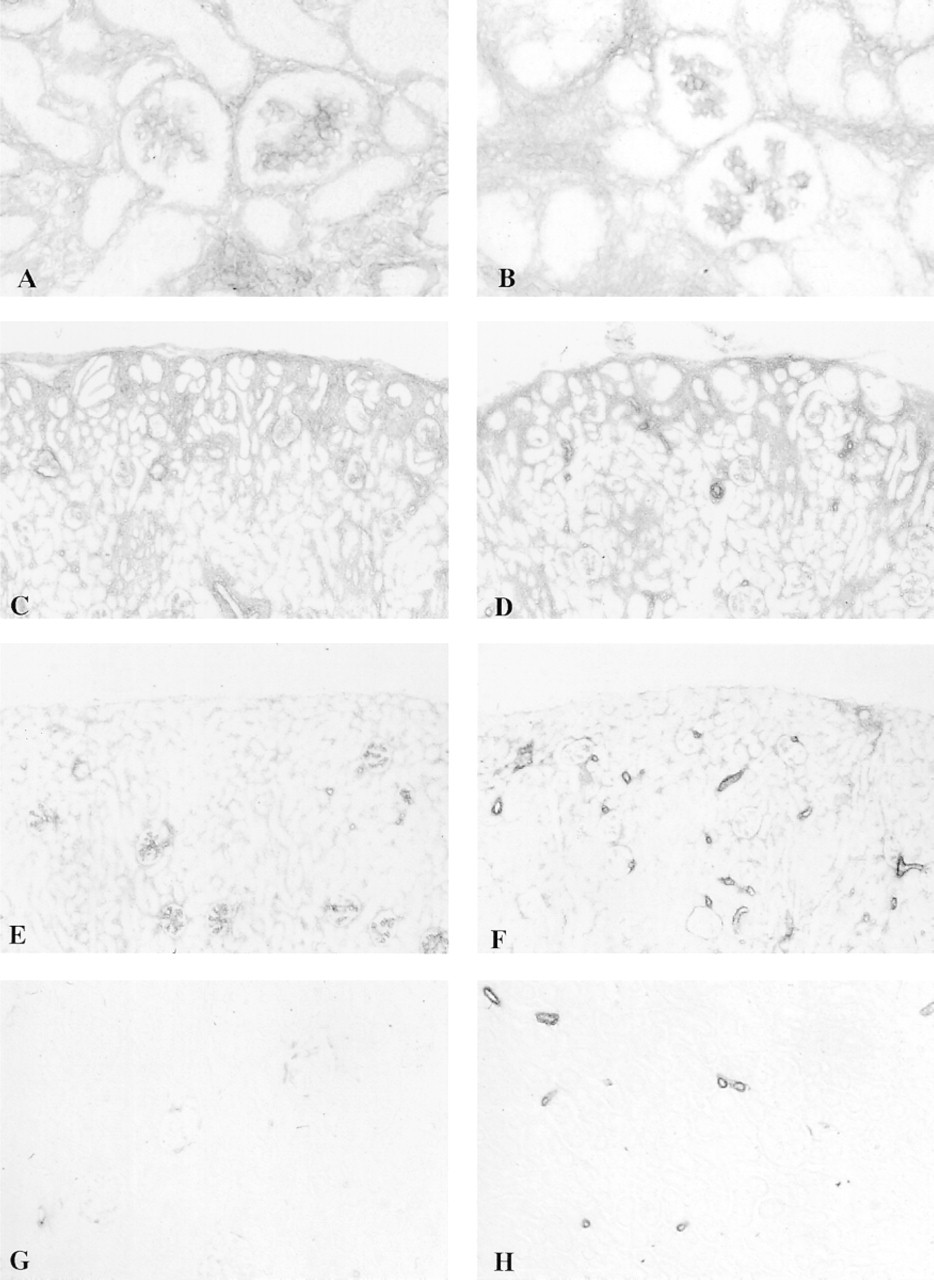
Immunoperoxidase localization of Fn-EIIIA (
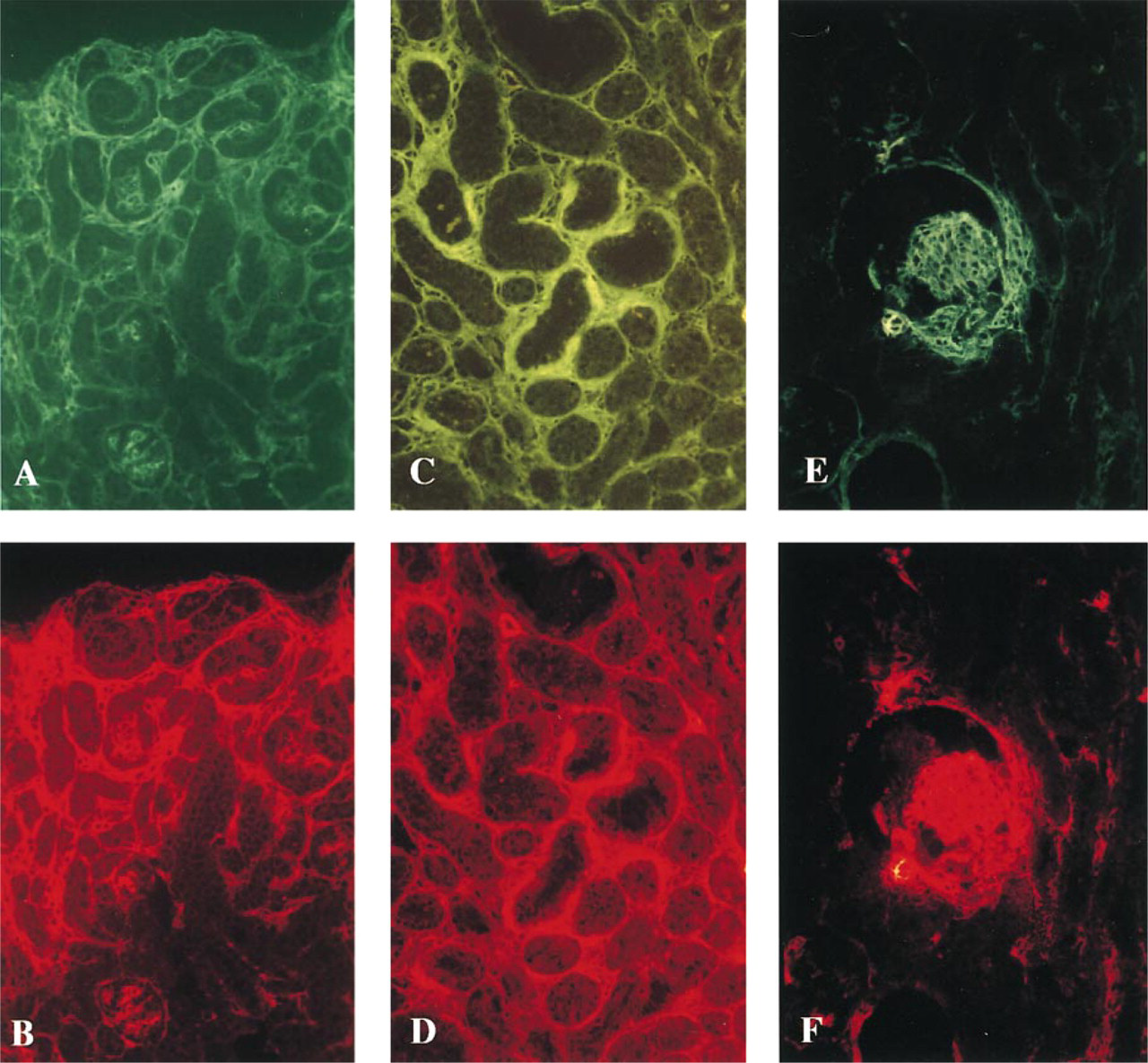
Dual label immunofluorescence verifying co-localization of Fn-EIIIA (
Probes used for the detection of α-SMA mRNA were sticky (see Materials and Methods) and had a high degree of background staining, overshadowing the detail of peritubular expression of α-SMA message. However, specific message was detected above background in areas of high cellular activity, such as the outer aspect of the kidney cortex in D-7 postnatal kidney (Figure 4G), glomerular crescents (Figure 4H), and in HV-induced glomerular micronodules (Figure 4I), identical to areas of abundant expression of Fn-EIIIA mRNA and their respective proteins, as described above.
Discussion
These studies largely show a co-expression of Fn-EIIIA and α-SMA in embryonic and maturing rat kidneys and a recapitulation of expression in two unrelated models of renal disease. Expression of Fn-EIIIA and α-SMA was negligible or absent in adult renal parenchyma. However, a spatial and temporal association between these proteins was evident at sites of high cellular activity and activation during kidney development. Both proteins were expressed in the peritubular interstitium and glomerular mesangium, with staining intensity following the course of cortical development and a parallel reduction in expression during maturation. Fn-EIIIA mRNA was confined to the interstitium, in contrast to developing and maturing tubules, indicating that this Fn isoform is not synthesized by the tubular epithelium and is not a component of mature tubular basement membrane. Instead, Fn-EIIIA may be expressed by mesangial cells and myofibroblasts or their precursors and as a provisional matrix for developing mesenchymal structures, and may be required for capillary growth and glomerulogenesis.
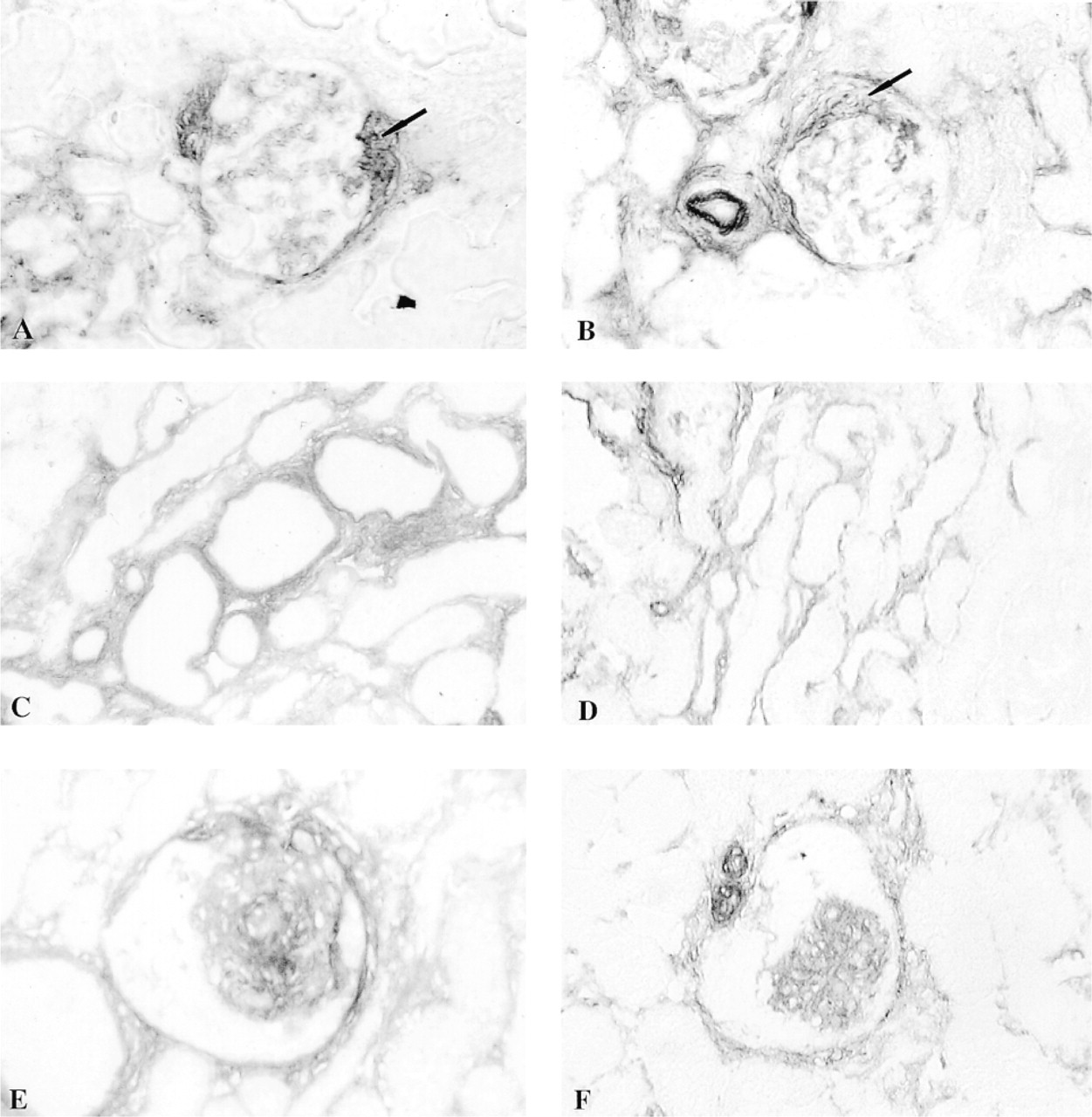
Immunoperoxidase localization of Fn-EIIIA (
A recapitulation of Fn-EIIIA and α-SMA expression was observed during renal disease, with a coexpression of these proteins in the periglomerular and peritubular interstitium (by myofibroblasts) and in glomerular mesangium, similar to the areas of expression described during kidney development and maturation. A co-localization of Fn-EIIIA and α-SMA was also observed in glomerular crescents in anti-GBM nephritis. The cell types in crescents that may express α-SMA have not been identified but are believed to be myofibroblasts (Atkins et al. 1996) or transdifferentiated epithelial cells (Ng et al. 1998). The close association and course of dual expression of Fn-EIIIA and α-SMA in discrete tissue structures in three different conditions of cellular remodeling (nephrogenesis, proliferative glomerulonephritis, and interstitial disease) suggest that these two proteins are tightly linked and share common functional roles required for remodeling.
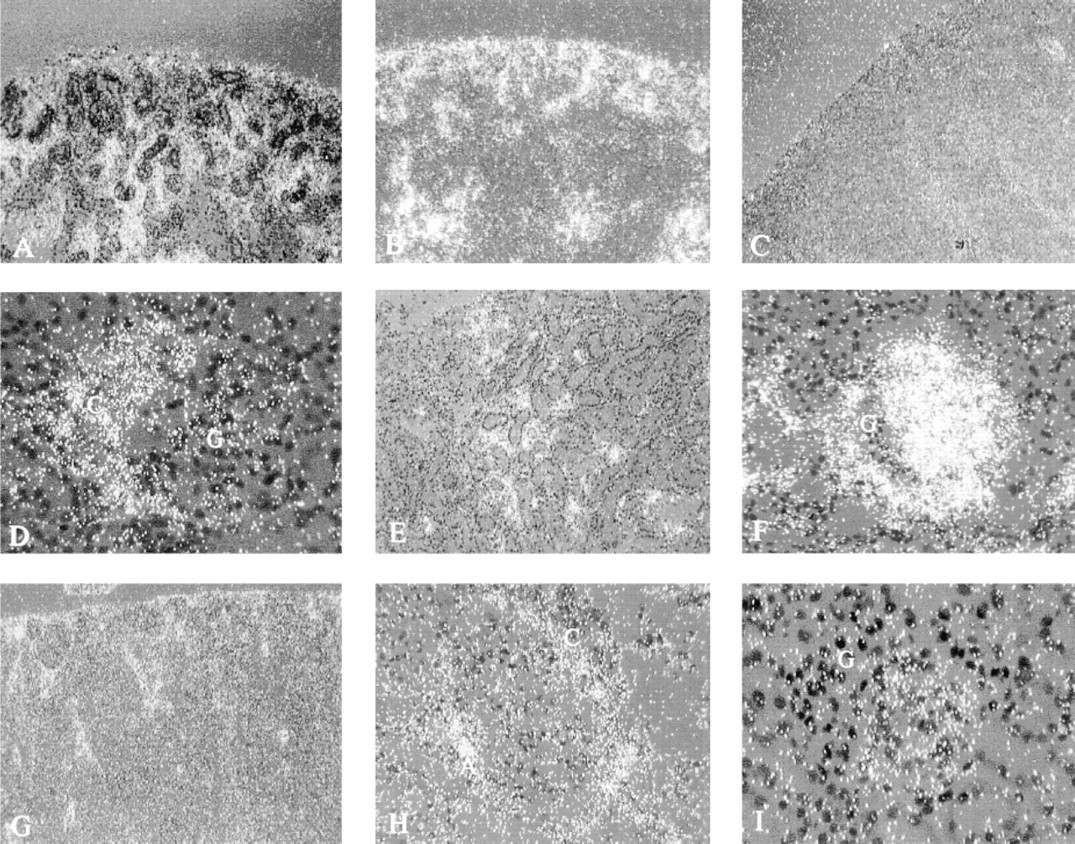
Darkfield localization of Fn-EIIIA mRNA (bright grains) by in situ hybridization in embryonic (
Activation of various mesenchymal cells is associated with a switch to an α-SMA-positive phenotype. Recent findings indicate that several cell types, such as liver fat-storing cells (Ito cells), breast stromal cells, fibroblasts, brain pericytes, and glomerular mesangial cells, do not express α-SMA in normal adult tissue or in primary culture. However, prolonged culture or exposure to growth factors activates these cells and elicits the expression of this cytoskeletal protein (Sappino et al. 1990; Johnson et al. 1991,1992; Elger et al. 1993; Schmitt-Graff et al. 1994; Serini et al. 1998). In addition, maintenance of Ito or mesangial cells on a surface that mimics normal basement membrane (i.e., Matrigel, an extract of Englebroth-Holm-Swarm tumor) maintains cellular quiescence and downregulation of α-SMA expression, suggesting that extracellular matrix interactions are important in cell activation (Rockey et al. 1992; DeLuca et al. 1993). Such cellmatrix interactions are supported by the observations that an Fn-EIIIA-enriched substratum induces fibroblast stress fiber formation and activation of a focal adhesion kinase (p125FAK), an important transmembrane signal transduction protein believed to be involved in cell activation (Xia and Culp 1995; Schlaepfer and Hunter 1996). Moreover, a role for Fn-EIIIA in cell activation has been reported in which Ito cells in normal liver and primary culture are α-SMA-negative but can be stimulated to express α-SMA when plated on Fn-EIIIA-fusion protein or endothelial cell-derived Fn-EIIIA, an interaction that could be inhibited by blocking with specific antibody to the EIIIA domain (Jarnagin et al. 1994). Interestingly, Ito cells during liver injury, fibroblasts in fibrotic lung disease, palmar fibromatosis, wound healing, interstitial nephritis, and mesangial cells during glomerulonephritis are converted from a resting cell phenotype to a myofibroblast-like cell during injury, and many of these mesenchymal cells are also associated with Fn-EIIIA during disease (Hynes 1990; Sappino et al. 1990; Kuhn and McDonald 1991; Barnes et al. 1994a,1995b; Jarnagin et al. 1994; Schmitt-Graff et al. 1994; Yamamoto et al. 1994,1996; Berndt et al. 1995; Nickeleit et al. 1995; Alpers et al. 1996; Kliem et al. 1996; Tang et al. 1997; Serini et al. 1998). This study shows for the first time that Fn-EIIIA and α-SMA are temporally and spatially associated in kidney maturation and disease.
Other alternatively spliced isoforms, such as EIIIB, may also have functional roles in embryogenesis and disease (Schwarzbauer 1991; Nickeleit et al. 1995; Peters et al. 1996; Peters and Hynes 1996). Expression of Fn-EIIIA and Fn-EIIIB shows widespread co-distribution in embryonic tissues. However, it shows divergent tissue staining patterns in the adult mouse, suggesting variable functions for alternatively spliced Fn isoforms (Peters et al. 1996; Peters and Hynes 1996). Our previous studies showed a preferential enhancement of Fn-EIIIA mRNA and protein compared to Fn-EIIIB in late HV-induced glomerular lesions at a time when α-SMA was abundant (Barnes et al. 1995b), suggesting that a functional link between these two proteins is unique for the alternatively spliced EIIIA domain.
A functional role for Fn-EIIIA in cell activation and a switch to an α-SMA phenotype have not yet been defined. Remodeling during wound repair or after injury involves cellular behaviors including cell migration, proliferation, and synthesis of extracellular matrix, all of which Fn has been shown to influence (Hynes 1990; Schwarzbauer 1991; ffrench-Constant 1995). Clues to the function of Fn-EIIIA in cell activation can be found in the observation that expression of α-SMA has been related to mesangial cell proliferation (Johnson et al. 1991; Elger et al. 1993) and hypertrophy (Glass et al. 1997) in vitro. Indeed, expression of α-SMA parallels mesangial and Ito cell proliferation and fibrogenesis (Johnson et al. 1991; Alpers et al. 1992,1996; Johnson et al. 1992; Barnes et al. 1994a, 1995b; Jarnagin et al. 1994; Boukhalfa et al. 1996; Tuchweber et al. 1996). Mesangial cells in culture also require their own synthesis of Fn during migration and hillock formation (Glass et al. 1996). Mesangial cells in culture synthesize a matrix abundant in Fn-EIIIA (personal observations), unlike normal adult mesangial cells in vivo (Barnes et al. 1994a, 1995b; Peters et al. 1996; and this report). Therefore, Fn-EIIIA may also have some effect on mesangial cell migration.
We have previously characterized a model of proliferative glomerulonephritis, induced by HV, by a distinct temporal course involving mesangial cell migration, proliferation, and extracellular matrix synthesis (Barnes 1989; Barnes and Abboud 1993; Barnes et al. 1994a,1995a, b). Our previous studies suggest that migrating mesangial cells do not require their own synthesis of Fn-EIIIA but may rely on exogenous sources of Fn isoforms derived from platelets and macrophages (Barnes et al. 1994a,1995b), and agree with our in vitro studies (Barnes and Hevey 1991) showing a potent migratory mesangial cell response to platelet Fn, which is abundant in EIIIA (Paul et al. 1986; Peters et al. 1995). A switch to an α-SMA phenotype appeared to be related to mesangial cell synthesis of Fn-EIIIA and coincided with expression of α-SMA, proliferation, and matrix synthesis, suggesting that autocrine synthesis of Fn-EIIIA by mesangial cells has specific functions during the course of glomerular remodeling. Interestingly, Fn-EIIIA was expressed in early HV-induced glomerular lesions before mesangial cell expression of α-SMA (Barnes et al. 1994a); similar to a recent report by Serini et al. (1998) that ED-A (EIIIA) deposition precedes and then parallels α-SMA expression by fibroblasts during granulation tissue evolution in wound healing.
Serini et al. (1998) also showed that a functional ED-A domain is mandatory for α-SMA induction by transforming growth factor-β1 (TGF-β1). TGF-β1 differentially regulates the expression of Fn-EIIIA in fibroblasts (Borsi et al. 1990) and induces expression of α-SMA in a variety of mesenchymal cells in culture (Desmouliere et al. 1993; Serini et al. 1998). Moreover, TGF-β1 is frequently associated with a switch of fibroblast to a myofibroblast phenotype in liver, lung, and kidney disease (Sappino et al. 1990; Milani et al. 1991; Yamamoto et al. 1994; Zhang et al. 1995), and all three proteins frequently co-localize near myofibroblasts in these disease settings. Therefore, regulation of Fn-EIIIA synthesis by TGF-β1 may provide a functional link for cell activation and α-SMA expression.
These studies show that Fn-EIIIA largely follows a parallel co-expression with α-SMA in several settings of remodeling, including embryonic, maturing, and diseased kidney. An exact role for alternatively spliced Fn-EIIIA in glomerular cell activation (expression of α-SMA) and cell function (migration, proliferation, matrix synthesis, and hypertrophy) has not been determined and remains the focus of current research.
Footnotes
Acknowledgments
Supported by NIH grant DK38758 from the National Institutes of Health (NIDDK), by the Office of Research and Development, Medical Research Service, Department of Veterans Affairs, and by the Southern Arizona Foundation.
We thank Dr George Henderson (Department of Medicine, UTHSCSA) for providing embryonic kidneys for this project. We also thank Drs Richard Hynes, and Gabriel Gabbiani for their generous gifts of cDNA probes to detect Fn-EIIIA and α-SMA mRNA, respectively.
