Abstract
Earlier in vitro studies suggest opposing roles of laminin and fibronectin in regulation of differentiated properties of vascular smooth muscle cells. To find out if this may also be the case in vivo, we used immunoelectron microscopy to study the distribution of these proteins during formation of intimal thickening after arterial injury. In parallel, cell structure and content of smooth muscle α-actin was analyzed. The results indicate that the cells in the normal media are in a contractile phenotype with abundant α-actin filaments and an incomplete basement membrane. Within 1 week after endothelial denudation, most cells in the innermost layer of the media convert into a synthetic phenotype, as judged by loss of actin filaments, construction of a large secretory apparatus, and destruction of the basement membrane. Some of these cells migrate through fenestrae in the internal elastic lamina and invade a fibronectin-rich network deposited on its luminal surface. Within another few weeks a thick neointima forms, newly produced matrix components replace the strands of fibronectin, and a basement membrane reappears. Simultaneously, the cells resume a contractile phenotype, recognized by disappearance of secretory organelles and restoration of α-actin filaments. These findings support the notion that laminin and other basement membrane components promote the expression of a differentiated smooth muscle phenotype, whereas fibronectin stimulates the cells to adopt a proliferative and secretory phenotype.
Keywords
S
Earlier studies in our laboratory have indicated that a substrate of fibronectin promotes transition of arterial SMCs from a contractile to a synthetic phenotype during the first few days in primary culture, whereas substrates of the basement membrane proteins laminin and collagen Type IV hold the cells back in a contractile phenotype (Hedin et al. 1988, 1989). Recently, similar results have been reported by other groups (Hayward et al. 1995; Li et al. 1994; Yamamoto et al. 1993) It is believed that these effects are mediated via integrin receptors, but the signaling mechanisms remain to be elucidated (Thyberg 1996). Distinct isoforms of fibronectin and laminin are expressed in the arterial media during fetal and adult life, and a shift from adult to fetal variants of these proteins takes place during the formation of atherosclerotic and experimentally induced lesions (Glukhova et al. 1989, 1990, 1993). On the other hand, no high-resolution analyses of the distribution of fibronectin and laminin in the normal arterial media and in different types of intimal lesions are available. Therefore, it is difficult to judge the ability of these molecules to interact directly with the SMCs to influence their phenotype.
In a recent study, we used electron microscopic and stereological techniques to describe the formation of neointimal thickenings in the rat carotid artery after balloon injury. Within 1 week after removal of the endothelium, the cells in the innermost part of the media took on a synthetic phenotype, as revealed by loss of myofilaments and outgrowth of an extensive endoplasmic reticulum and a large Golgi complex. Later, some of these cells migrated into the intima via fine openings in the internal elastic lamina. Here, they began to divide and secrete ECM components, and within another week a thick neointima had formed. Subsequently, the majority of the cells eventually resumed a contractile phenotype with a cytoplasm largely occupied by myofilaments (Thyberg et al. 1995). In the present investigation, the same experimental model was used in combination with immunogold staining to study how the distribution of laminin and fibronectin is changed in connection with the phenotypic modulation of the SMCs. The results are in good agreement with the hypothesis that basement membrane components help to maintain the cells in a differentiated phenotype, whereas fibronectin stimulates them to revert to a proliferative and secretory phenotype (Thyberg 1996).
Materials and Methods
Arterial Injury
Four- to 5-month-old male Sprague-Dawley rats were used (400–450 g). The left carotid artery was denuded of endothelium with a Fogarty 2F embolectomy catheter as previously described (Thyberg et al. 1995; Clowes et al. 1983). Briefly, the catheter was inserted through the external carotid artery, advanced to the aortic arch, inflated with air, and gently withdrawn (repeated three times). The catheter was then removed, the external carotid artery ligated, and the wound closed. After 1, 2, or 5 weeks, the carotid arteries were fixed in a mixture of 2% formaldehyde and 0.1% glutaraldehyde in PBS, pH 7.3, by retrograde perfusion via the abdominal aorta for 15–20 min at a pressure of 80 cm water. The arteries were removed, cut into rings with a razor blade, and left in fresh fixative for another 1–2 hr (the right carotids were used as controls).
Electron Microscopy
After repeated rinsing in PBS, the tissue was dehydrated in graded ethanol (70, 95, and 100%) and embedded in LR White (London Resin Company; Reading, UK). For the latter purpose, the specimens were first incubated in a mixture of equal parts ethanol and LR White (v/v) for 30 min and then left in pure resin for 12–15 hr at 4C. After two additional incubations in LR White (30 min each), the carotid rings were put into gelatin capsules completely filled with resin and tightly capped. The capsules were placed in a uv polymerization unit (Agar Scientific; Stansted, UK) for 12–15 hr. Thin sections were cut with diamond knives on an LKB Ultrotome IV, picked up on nickel grids coated with a carbon-stabilized formvar film, stained as described below, and finally examined in a JEOL EM 100CX operated at 60 kV.
Antibodies
As primary antibodies, mouse anti-smooth muscle α-actin (N-terminal synthetic decapeptide; no. 2547; Sigma Chemical, St Louis, MO), rabbit anti-laminin (Engelbreth-Holm-Swarm mouse sarcoma; Sigma no. 9393), and rabbit anti-fibronectin (human plasma; Sigma no. 3648) were used. These products react with human as well as animal tissues and show a high degree of antigen specificity. As secondary antibodies, goat anti-mouse IgG (Sigma no. 7652) and goat anti-rabbit IgG (Sigma no. 7402) conjugated to 10-nm colloidal gold particles were used. Control stainings without primary antibodies and with nonimmune IgG fractions were negative.
Immunostaining
The grids were rinsed in water, placed on a droplet of PBS with 0.25% bovine serum albumin (PBS/BSA) for 5 min to block unspecific binding sites, transferred to primary antibodies diluted in PBS/BSA, and incubated for 2–3 hr in a humid atmosphere. The grids were then rinsed repeatedly with PBS, placed on a droplet of PBS/BSA for 5 min, transferred to secondary antibodies diluted in PBS, and incubated for 1–2 hr. After renewed rinsing with PBS, the sections were postfixed with 2% glutaraldehyde in PBS and then rinsed again, first with PBS and then with water. Finally, contrast staining was made with aqueous uranyl acetate for 30 min followed by alkaline lead citrate for 30 sec.
Results
Smooth Muscle α-Actin as Phenotypic Marker During Neointima Formation
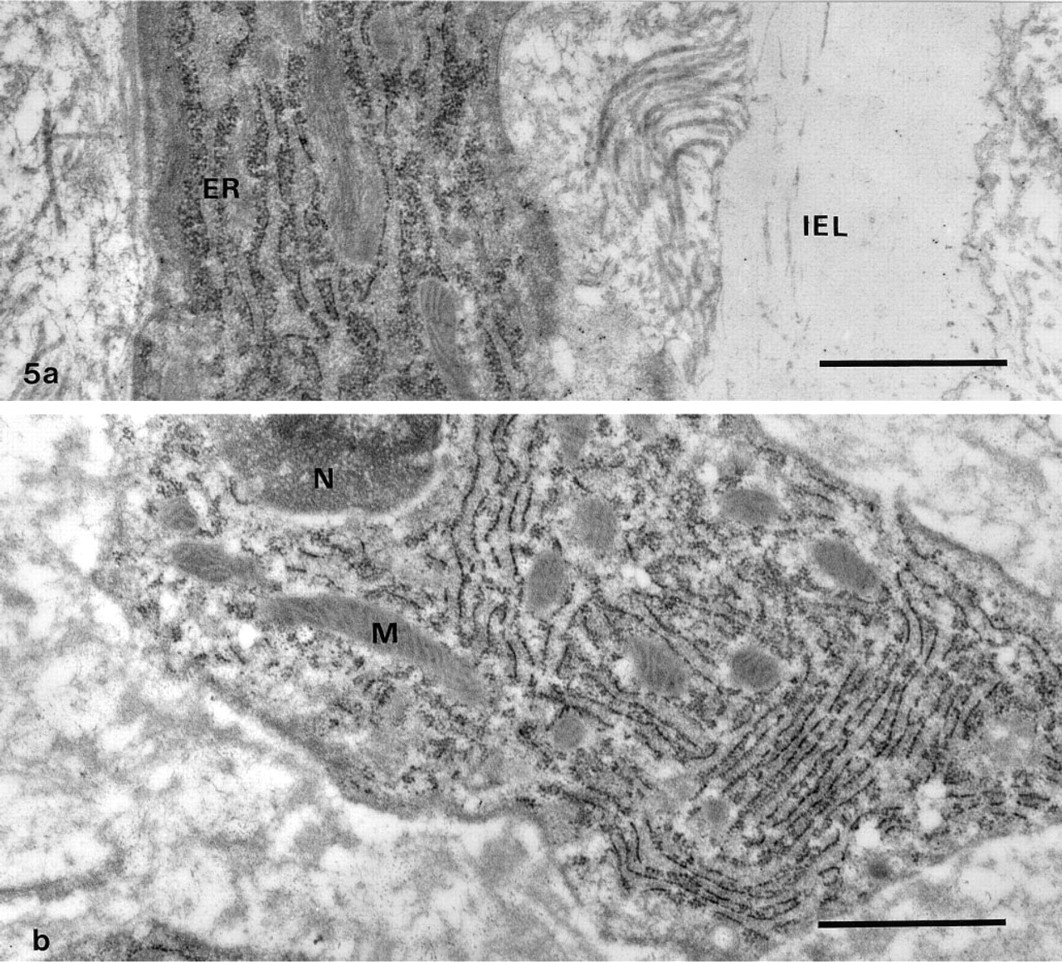
The fine structural characteristics of the vessels and the changes occurring after balloon injury were the same as described previously (Thyberg et al. 1995). In the controls, the SMCs in the media were in a contractile phenotype, with a cytoplasm occupied by filaments stained for smooth muscle α-actin (Figure 1a). The majority of the SMCs in the media of the injured arteries likewise retained a differentiated phenotype as judged by these criteria (Figure 1b). However, 1 and 2 weeks after the operation, most of the SMCs in the inner layer of the media were modified and showed a reduced content of filaments positive for smooth muscle α-actin (Table 1; cf. Figure 5a). Such cells were also observed migrating from the media to the intima via small holes in the internal elastic lamina. In the 1- and 2-week-old neointima, a further modification of the SMCs was evident, with a prominent endoplasmic reticulum and Golgi complex, few filaments, and only a weak reactivity for smooth muscle α-actin (Figure 2). Five weeks after the operation, the secretory organelles had again become smaller and most of the cytoplasm was instead filled with filaments stained for smooth muscle α-actin (Figure 3; Table 1).
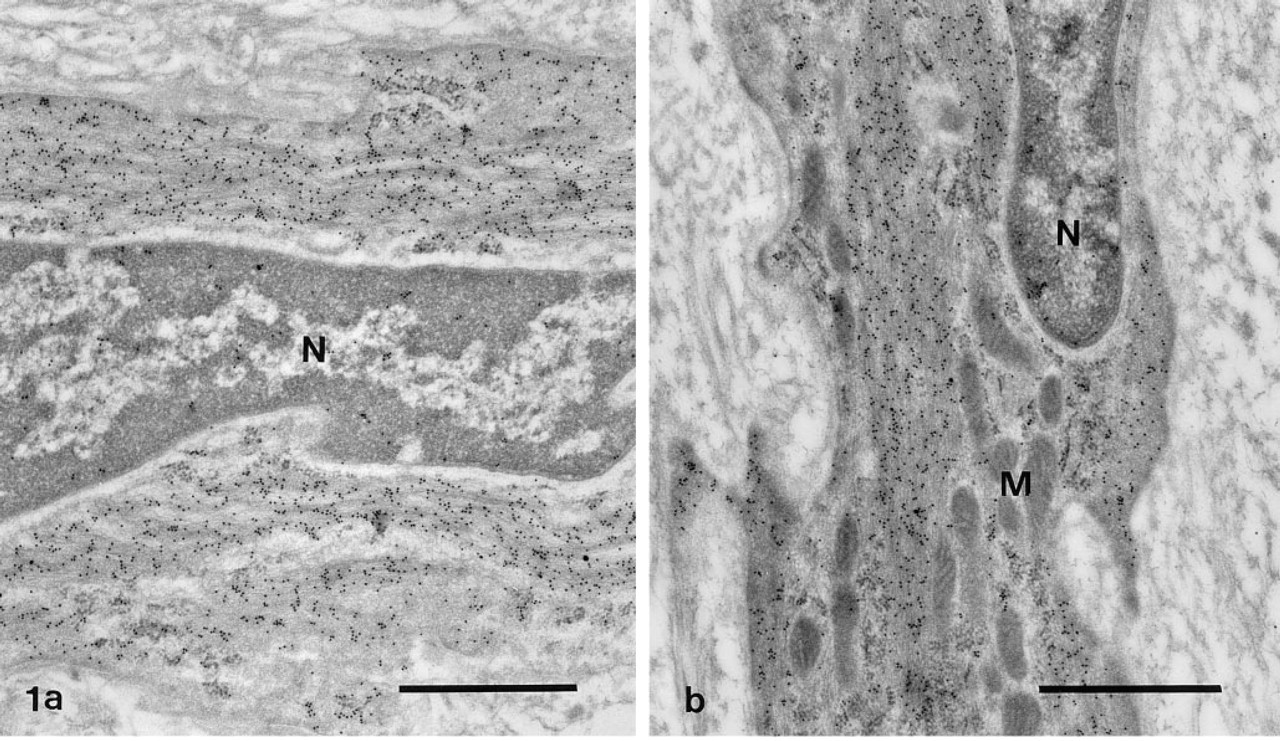
Immunogold staining for smooth muscle α-actin in the media of two carotid arteries fixed either without
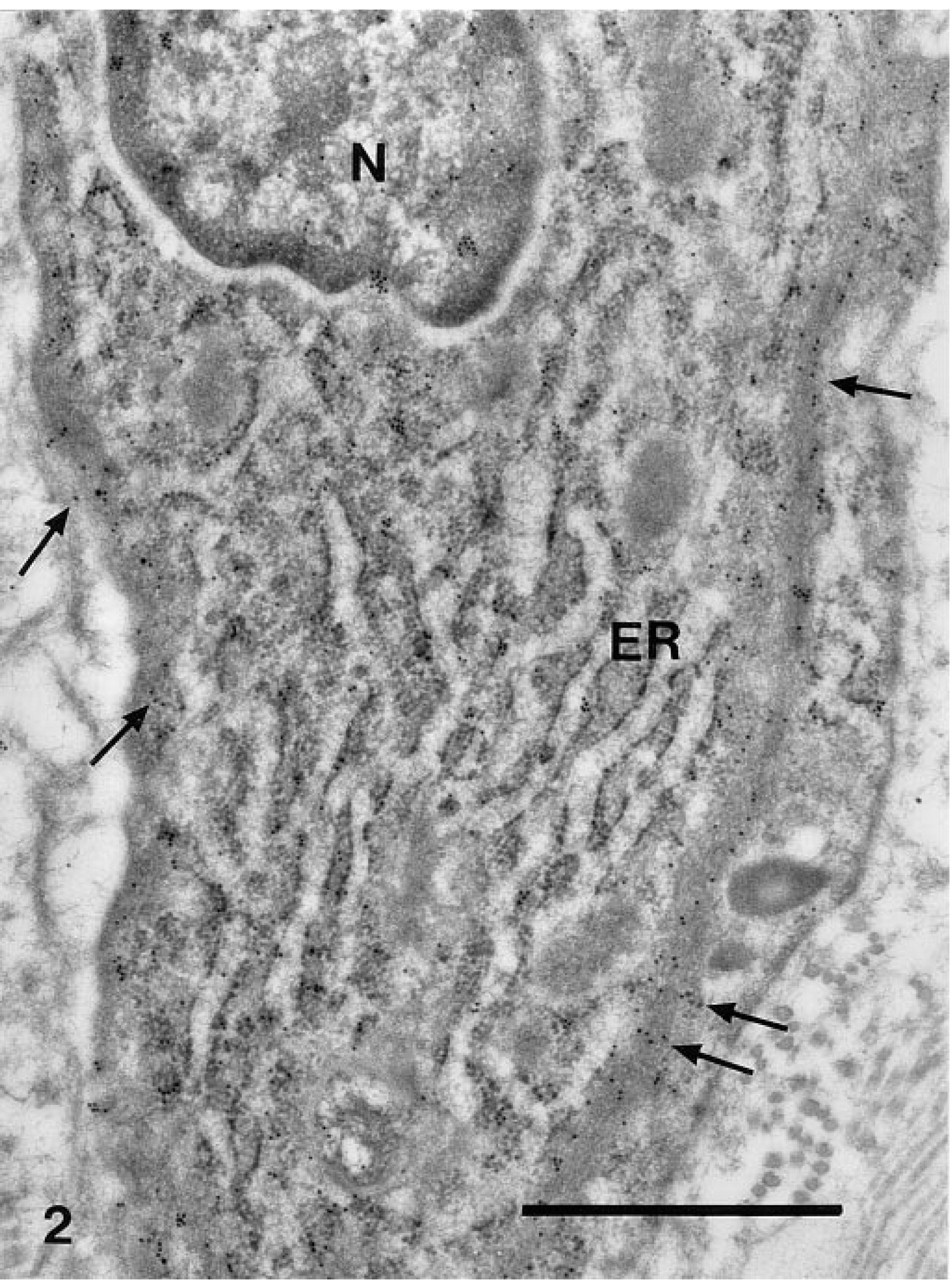
Immunogold staining for smooth muscle α-actin in the neointima of a carotid artery fixed 2 weeks after balloon injury. The SMC is in a synthetic phenotype, with an extensive endoplasmic reticulum (ER) and only a weak reaction for smooth muscle α-actin in the sub-plasmalemmal region (arrows). N, nucleus. Bar = 1 μm.
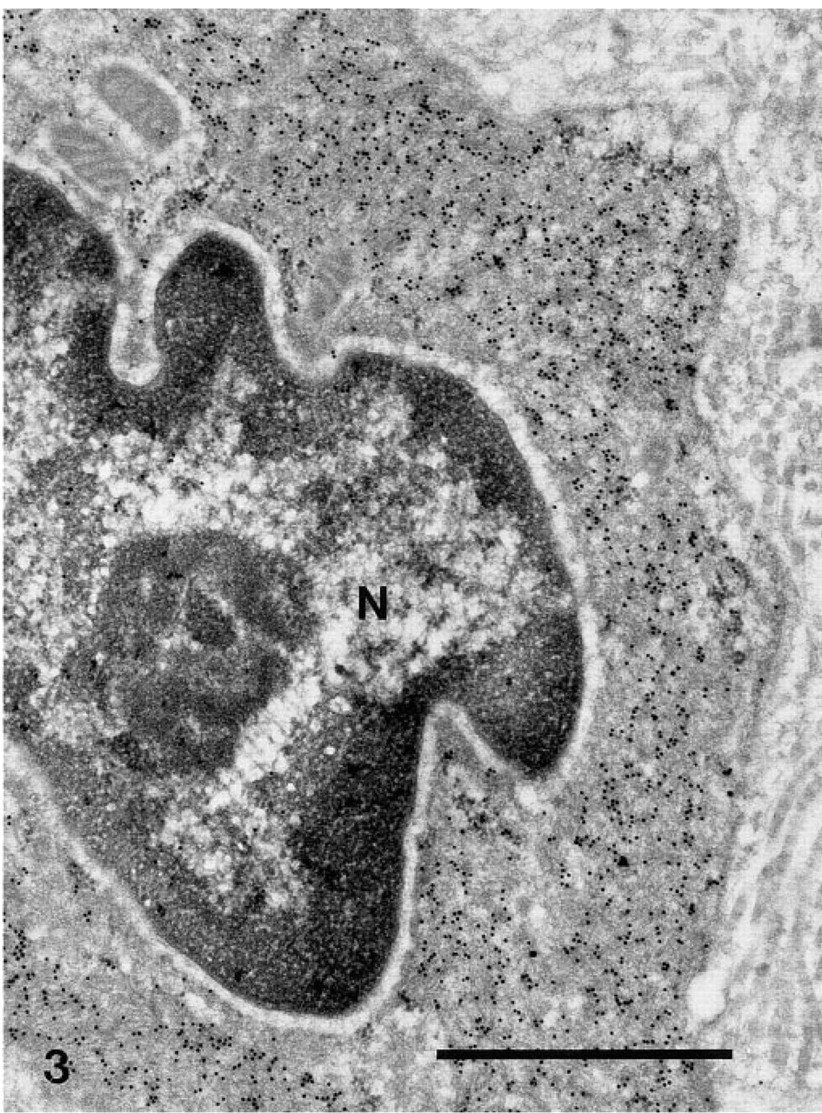
Immunogold staining for smooth muscle α-actin in the neointima of a carotid artery fixed 5 weeks after balloon injury. The SMC is in a contractile phenotype, with a cytoplasm filled by filaments positive for smooth muscle α-actin. N, nucleus. Bar = 1 μm.
Overall phenotypic properties of medial and neointimal SMCs in carotid arteries at different times after balloon injury a
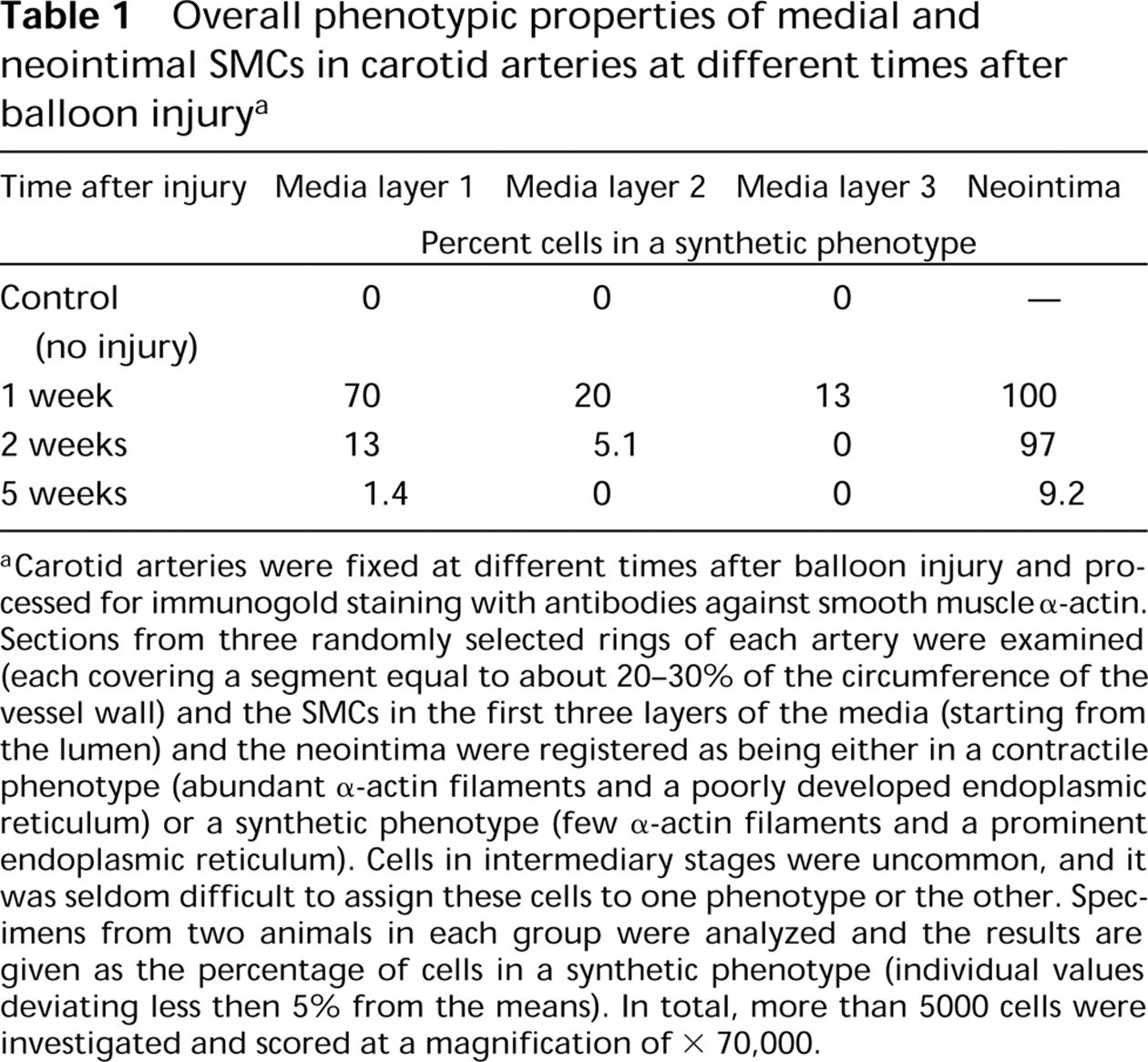
aCarotid arteries were fixed at different times after balloon injury and processed for immunogold staining with antibodies against smooth muscle α-actin. Sections from three randomly selected rings of each artery were examined (each covering a segment equal to about 20–30% of the circumference of the vessel wall) and the SMCs in the first three layers of the media (starting from the lumen) and the neointima were registered as being either in a contractile phenotype (abundant α-actin filaments and a poorly developed endoplasmic reticulum) or a synthetic phenotype (few α-actin filaments and a prominent endoplasmic reticulum). Cells in intermediary stages were uncommon, and it was seldom difficult to assign these cells to one phenotype or the other. Specimens from two animals in each group were analyzed and the results are given as the percentage of cells in a synthetic phenotype (individual values deviating less then 5% from the means). In total, more than 5000 cells were investigated and scored at a magnification of × 70,000.
Changes in Distribution of Laminin During Neointima Formation
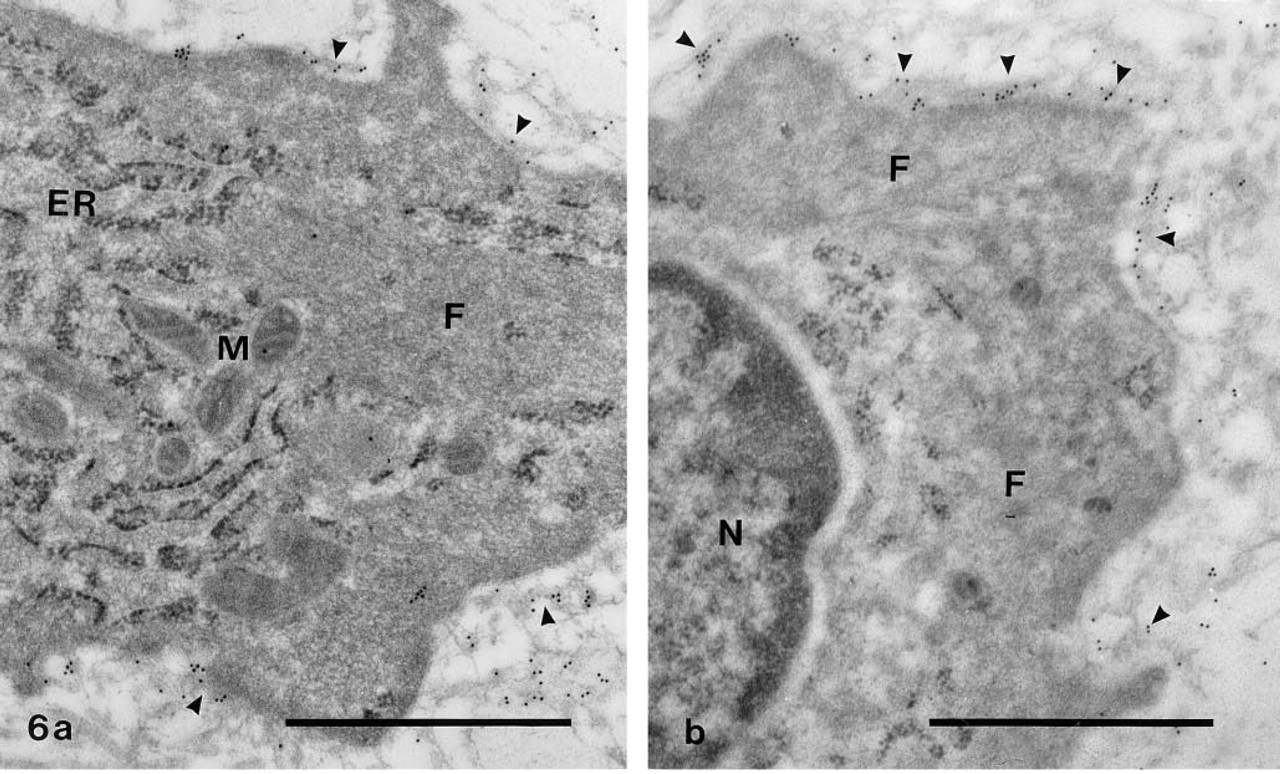
In the media of both the control and the injured carotid arteries, an incomplete basement membrane with a positive staining for laminin surrounded the SMCs (Figures 4a and 4b). On the other hand, no or only weak staining for laminin occurred around the phenotypically modified SMCs in the inner layer of the media and in the neointima 1 and 2 weeks after the operation (Figures 5a and 5b). In connection with the redifferentiation of the SMCs in the neointima, laminin was again found both underneath the regenerating endothelial cells (recognized by their thin shape and the presence of Weibel-Palade bodies) and around the SMCs themselves (Figures 6a and 6b). At this time, faint laminin staining was also seen in the endoplasmic reticulum and Golgi complex of the endothelial cells and SMCs.
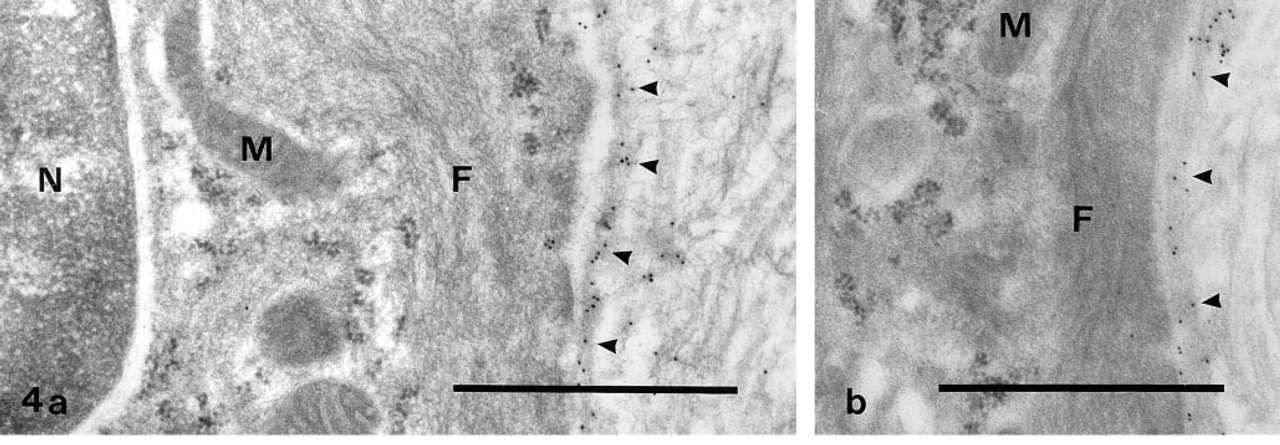
Immunogold staining for laminin in the inner part of a carotid artery fixed 1 week after balloon injury. Two phenotypically modified SMCs rich in endoplasmic reticulum (ER) are shown, one located in the first layer of the media
Changes in Distribution of Fibronectin During Neointima Formation
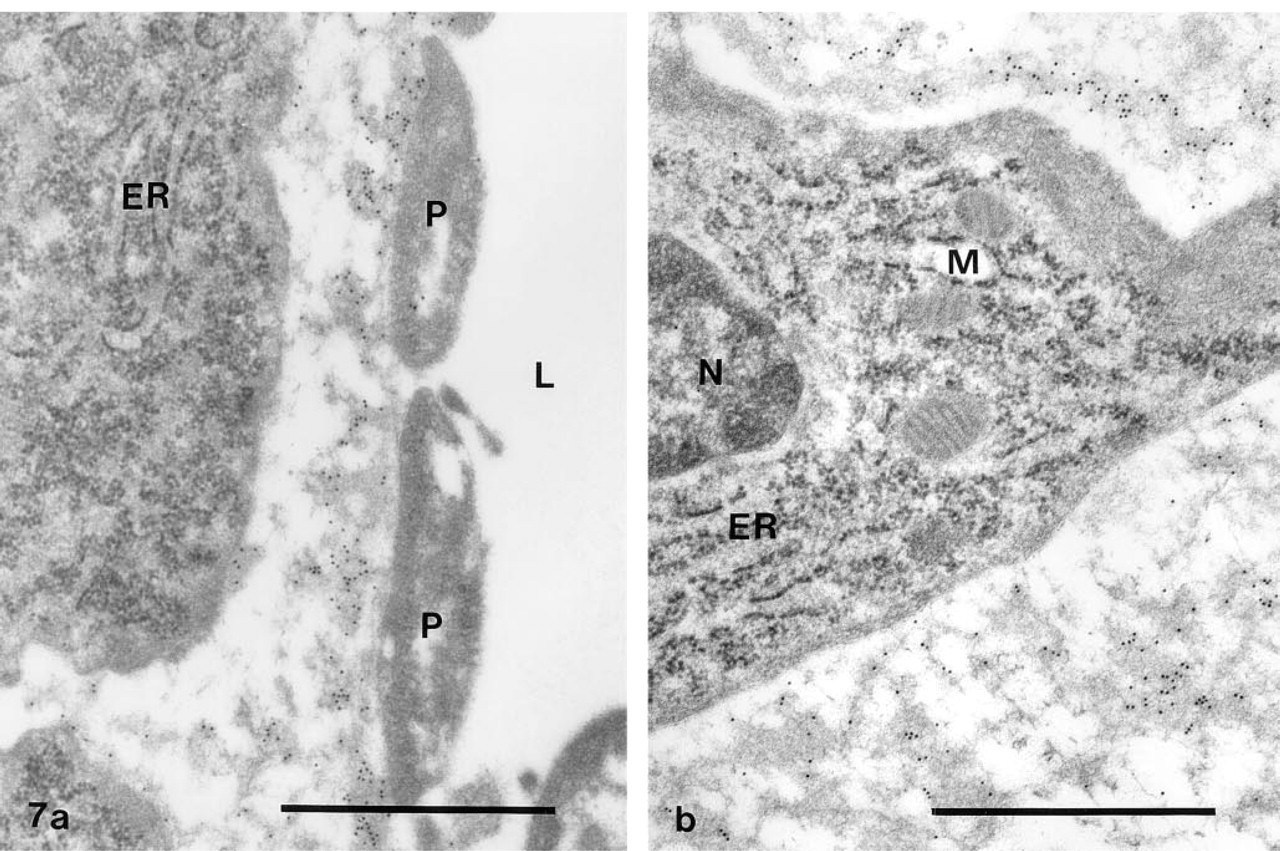
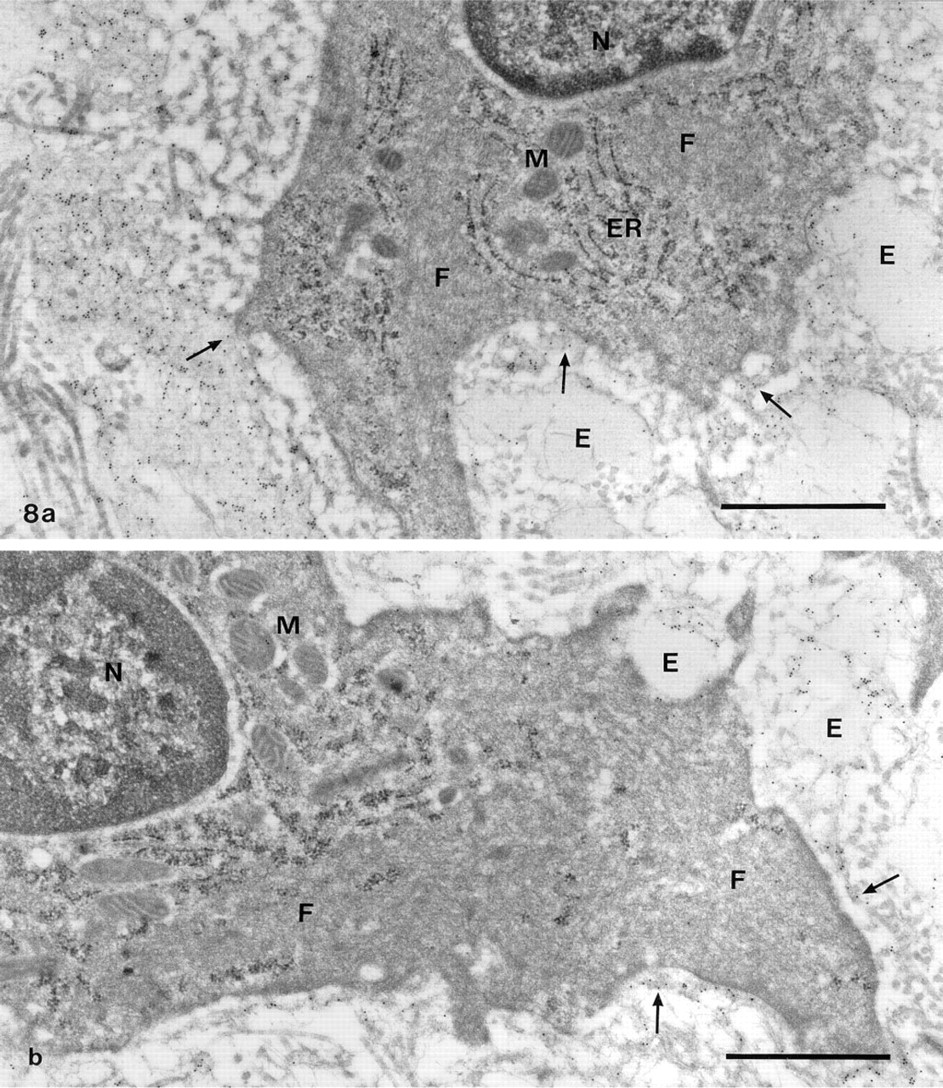
All layers of the control vessels (intima, media, and adventitia) showed only weak staining for fibronectin. In the injured vessels, a distinct reaction was obtained in the neointima and the most superficial parts of the media, close to the fenestrae in the internal elastic lamina. The other parts of the media and the adventitia remained weakly stained. One week after the operation, a thin layer of degranulated platelets typically covered the luminal surface of the vessels. Under this layer, a fine meshwork with conspicuous staining for fibronectin was found (Figure 7a). It was of variable overall dimensions and was laid down before SMCs had started to migrate into the intima. Accordingly, the first SMCs that appeared here were covered by fibronectin, whereas only little stainable material occurred between them and the internal elastic lamina. As the neointima grew in thickness, the platelets disappeared from the inner vessel surface and were replaced by SMCs that penetrated the fibronectin meshwork. The cells in the young neointima were thus surrounded by a loosely arranged matrix dominated by fibrillar strands with a positive reaction for fibronectin (Figure 7b). Two and 5 weeks after the operation, the neointima had grown further in size and was made up of multiple layers of SMCs encircled by a dense matrix of collagen fibrils and elastic fibers. At the same time, a successive reduction in the amount of fibronectin in the pericellular matrix was noted (Figures 8a and 8b).
Discussion
Immunoelectron microscopy was used to study the formation of a neointima in rat carotid arteries after endothelial denudation with a balloon catheter. Special interest was paid to the change in differentiated properties of the SMCs and the composition of their pericellular matrix. For this purpose, stainings were made with antibodies against the cytoskeletal marker protein smooth muscle α-actin, the basement membrane protein laminin, and the plasma/ECM protein fibronectin. The results indicate that the SMCs in the normal arterial media are in a contractile phenotype with a cytoplasm filled with α-actin filaments. After 1 week, most of the SMCs in the inner medial layer had converted into a synthetic phenotype and showed a markedly reduced content of α-actin filaments as well as an enlarged endoplasmic reticulum and Golgi complex. Some of these cells were seen to migrate into the intima via holes in the internal elastic lamina, whereas cells in a contractile phenotype were never observed to do so. On the basis of these findings, we conclude that the cells that give rise to the neointima are derived from phenotypically modified SMCs in the inner part of the media. The observation that smooth muscle α-actin is lost before the SMCs migrate into the intima is in accordance with the proposal that an essential function of this actin isoform is to prevent cell motility (Rønnov-Jessen and Petersen 1996).
After the passage through the internal elastic lamina, the modification of the SMCs was completed by an additional loss of α-actin and a further growth in size of the secretory organelles. Hence, the proliferating and matrix-producing cells in the neointima were in a synthetic phenotype. In a similar manner, cultured SMCs typically show an inverse relation between the expression of α-actin and the rate of cell growth (Thyberg 1996). However, as the neointima reached its final size, the SMCs resumed a contractile phenotype, with a cytoplasm dominated by α-actin filaments. Biochemical analyses have likewise revealed a reversible change in cytoskeletal profile during the formation of neointimal thickenings in the rat carotid artery (Kocher et al. 1991). Together, these findings illustrate the ability of the SMCs to undergo a bidirectional shift in phenotype after vascular injury.
Staining for laminin showed that the SMCs in the control arteries and most of the cells in the media of the injured arteries were enclosed by an incomplete basement membrane. In contrast, no distinct reaction was found around the phenotypically modified SMCs in the inner medial layer and the developing neointima of the latter vessels. However, the ability to produce laminin was retained and a basement membrane subsequently re-formed around the redifferentiating SMCs. These observations corroborate the notion that laminin and other basement membrane components promote the differentiation of vascular SMCs (Thyberg 1996). They may do so by a receptor-mediated interaction and generation of signals that affect cell behavior. Another possibility is that they make up a physical boundary that prevents molecules with a dedifferentiating effect to contact the cells. The apparent defects in the basement membrane would, however, limit the efficiency of such a function. Although the details in the signaling system remain to be clarified, we therefore suggest that the basement membrane mainly influences the SMCs via a direct interaction.
Only weak staining for fibronectin was detected in the media. However, earlier light microscopic studies have demonstrated that this protein is present in the rat arterial media and that an alternative form containing extra Domain A appears in the neointima after balloon injury (Wolf et al. 1994; Glukhova et al. 1989). Because the antibodies used react with plasma as well as cellular fibronectin (our unpublished observations), the weak reaction in the media could possibly be due to decreased accessibility of the antigen after processing for electron microscopy. On the contrary, a strong staining was obtained in the neointima, especially in the early stages of its development (i.e., even before SMCs had moved through the internal elastic lamina). Accumulation of fibronectin in the intima was likewise noted after endotoxin-induced endothelial injury (Kang and Williams 1991). These findings agree with the idea that the endothelium makes up a major barrier for passage of macromolecules from the blood into the vessel wall and that the internal elastic lamina prevents passage of material into the media after endothelial denudation (Penn et al. 1994). They also confirm the notion that the superficial SMCs of the neointima do not form a permeability barrier to macromolecules (Clowes et al. 1978). Fibronectin and other blood products may therefore be deposited on the subendothelial matrix after balloon injury and then continue to infiltrate the neointima as long as the endothelium is lacking.
The concentration gradient created in this way could be an important factor behind the observed change in SMC phenotype as well as the subsequent migration of cells into the intima. In support of this idea, it has been shown that freshly isolated SMCs adhere to a substrate of fibronectin and in a few days of serum-free culture convert from a contractile to a synthetic phenotype (Thyberg 1996; Hedin et al. 1988, 1989). Moreover, it is known that fibronectin acts as a substrate for cell migration both during embryonic development and in adult life (Ruoslahti 1988). With regard to SMCs, earlier in vitro work has revealed that they migrate over a layer of fibronectin using at least two types of integrin receptors (Clyman et al. 1992). These experiments suggest that a substrate of fibronectin may be sufficient to promote both the shift in phenotype and the migration of vascular SMCs and that other exogenous macromolecules are not required in these processes. Like laminin, fibronectin exerts its effect by interaction with integrin receptors, and this not only produces a physical linkage to the cells but also generates signals that adjust their behavior (Schwartz et al. 1995). To understand why laminin and fibronectin affect the SMCs differently, the signaling systems activated by their respective receptors have to be disclosed.
The SMCs of the growing neointima secrete ECM components and, with increasing time, an increasing fractional volume of the tissue is occupied by a collagen- and elastin-rich matrix (Thyberg et al. 1995). Therefore, the cells in the early neointima were mainly separated by a fibronectin-containing fibrillar network (probably including both plasma fibronectin and fibronectin produced by the SMCs themselves). Later, progressively larger numbers of collagen fibrils and elastic fibers were laid down around the cells, and finally a laminin-containing basement membrane reappeared. In parallel, the strands of fibronectin were displaced from the immediate vicinity of the cells. This change in composition of the pericellular matrix may be one important factor behind the return of the cells to a more differentiated state in the maturing neointima. However, the SMCs produce many other molecules that may be involved in this autocrine or paracrine control of their phenotype, including glycoproteins and proteoglycans of the extracellular matrix (Nikkari et al. 1994; Giachelli et al. 1993; Hedin et al. 1991). In addition, the regenerating endothelial cell layer is essential in this context. Because of this complexity, much further work is needed to obtain an integrated view of the regulatory system that determines the phenotypic properties of the SMCs under normal and pathological conditions.
Footnotes
Acknowledgements
Supported by the Swedish Medical Research Council, the Swedish Heart Lung Foundation, the King Gustaf V 80th Birthday Fund, the Loo and Hans Osterman Fund, and the Karolinska Institute.
