Abstract
The enzyme PAM is required for activation of many peptide hormones. In adult mouse lung, immunostaining for PAM was located in Clara cells, which constitute most of the epithelial cells of the mouse bronchial/bronchiolar tree. Immunoreactivity appeared for the first time in the epithelium on gestational Day 16, being slight and mostly restricted to the apical cytoplasm. As the lung developed, the labeling became gradually stronger and extended throughout the cell. Smooth muscle of airways and blood vessels, and some parenchymal cells, probably macrophages, also showed PAM immunoreactivity. Of the two enzymatically active domains of PAM, only PHM and not PAL immunoreactivity was found at all stages studied. The early appearance of PAM in developing mouse lung, as well as its presence in a variety of tissues, probably indicates a complex role of this enzyme in pulmonary development and function.
B
The formation of an amidated peptide from its glycineextended propeptide intermediate requires an enzyme complex denoted collectively as peptidyl-glycine α-amidating mono-oxygenase (PAM) (EC 1.14.17.3) (Eipper et al. 1983). Peptidylglycine α-hydroxylating monooxygenase (PHM) mediates the first step of the PAM pathway and uses molecular oxygen to form the peptidyl-α-hydroxyglycine intermediate. Peptidyl-α-hydroxyglycine α-amidating lyase (PAL), the second enzyme of the pathway, catalyzes the conversion of the peptidyl-α-hydroxyglycine derivative into the α-ami-dated product. Both enzymes are encoded by the same gene (PAM gene), which has been cloned and appears to be highly conserved among different species (Eipper et al. 1987). The amino terminal third of the 108-kD PAM precursor contains the PHM enzyme, whereas PAL enzyme is contained in the middle third of the PAM precursor (Glauder et al. 1990; Stoffers et al. 1991; Milgram et al. 1992). The carboxy terminal third of the molecule encodes a transmembrane domain and a hydrophilic cytoplasmic tail (Eipper et al. 1987).
The amidating enzymes have been localized in endocrine glands (May et al. 1990; Braas et al. 1992), endocrine cells of pancreas and digestive tract (Braas et al. 1992; Martínez et al. 1993a,b), salivary glands (Braas et al. 1992), heart (Eipper et al. 1988; Ouafik et al. 1989; Braas et al. 1992), neurons, and glial cells (Rhodes et al. 1990; Schafer et al. 1992). Although several regulatory substances (Table 1), some of them amidated, are known to be involved in the maturation and function of mammalian lung, the presence of PAM in adult lung has been reported in only one species (human) (Saldise et al. 1996), and no studies have been made in developing lung. The aim of the present investigation was to study the distribution of PAM enzymes in developing and adult mouse lung and to determine co-localization between this enzyme and the regulatory peptides found in the mammalian pulmonary neuroendocrine system.
Antisera used to study the regulatory substances present in mouse lung
lncstar, Stillwater, MN.
Royal Postgraduate Medical School, Hammersmith Hospital, UK.
Milab, Malmö, Sweden.
Materials and Methods
Lungs of 86 Swiss mice at different points in fetal and postnatal development were studied. The mean time of gestation in this species is approximately 19 days. Lungs were obtained on gestational (E) Days 14, 15, 16, 17, 18, and 19 and on postnatal (P) Days 0, 1,2, and 6, the age at which mouse lung is considered to be mature. The lungs of pregnant females (adult lung, Pad) were also studied.
The pregnant mice were anesthetized with 12.5% urethane (1 ml/100 g). The abdomen was opened and the fetuses were rapidly removed and chilled in ice. The newborn mice were anesthetized with ice. The chests of fetuses, new born, and adult mice were opened and lungs were removed, except in the case of the small E-14 fetuses, which were immersed in toto in the fixative. All samples were fixed in Bouin's fixative for 20-24 hr and were paraffin-embedded.
Immunocytochemistry
Sections (4 μm thick) were mounted on glass slides. After dewaxing, immunocytochemical staining was performed using the avidin-biotin method (Hsu et al. 1981). Endogenous peroxidase was blocked by treatment with 3% hydrogen peroxide in methanol for 30 min and sections were hydrated through alcohols and then placed in Tris-HCl-buffered saline (TBS: Tris buffer 0.05 M, NaCl 0.5 M, pH 7.36). Nonspecific background was blocked with normal swine or rabbit serum (1:20; University of Navarra) for 30 min and the sections were incubated overnight at 4C with the primary antiserum. Table 2 shows all the PAM antisera used in this study as well as their source and optimal dilution. To reveal neuroendocrine elements, antisera against regulatory substances described in mammalian respiratory tract (Table 1) were also applied.
Antisera against PAM enzymes used in the present study
BA Eipper, Johns Hopkins University School of Medicine, Baltimore, MD.
AM Treston, National Cancer Institute, Rockville, MD.
P, polyclonal.
M, monoclonal.
Antisera raised against the same domain have been numbered consecutively to help in their identification. The peptide immunogens for PHM(3) and PHM(4) antibodies represent the same region of the domain, a highly conserved sequence close to the active site of the enzyme.
The sections were then incubated for 30 min with biotinylated swine anti-rabbit (K353; Dakopatts, Glostrup, Denmark) or rabbit anti-mouse (K354; Dakopatts) immunoglobulins and then with avidin-biotin-peroxidase complexes (K355; Dakopatts) for 30 min. After each incubation, sections were rinsed in TBS. The bound antibodies were visualized with 3-3′-diaminobenzidine tetrahydrochloride (D-5637; Sigma, St Louis, MO) in sodium acetate/acetic acid 0.1 M, pH 5.6, containing 2.5% nickel ammonium sulfate, 0.2% β-D-glucose, 0.04% ammonium chloride, and 0.001% glucose oxidase (Shu et al. 1988). The sections were counterstained with hematoxylin, dehydrated, and mounted in DPX.
Specificity Controls
Absorption controls were performed when the corresponding antigens were available. Antisera to PHM and CGRP which rendered immunolabeling were preincubated with their respective synthetic antigens at concentrations of 0.1-10 nmol of peptide per ml of optimally diluted antiserum before application to tissue sections. In both cases, preabsorption of the antisera with the peptide abolished immunoreaction.
Results
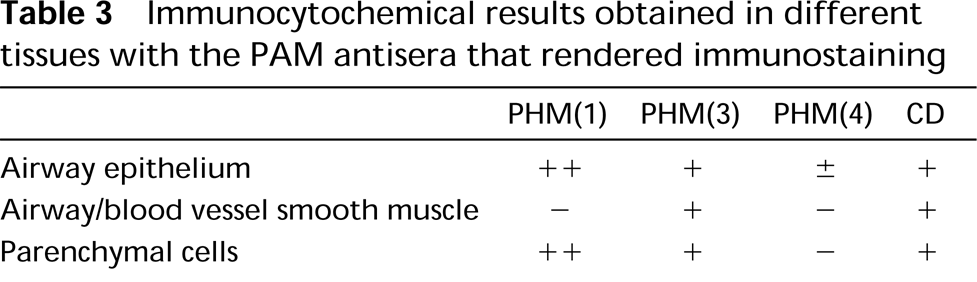
In mouse, immunoreactivity for PAM was localized in airway epithelial cells, smooth muscle of airway and blood vessel walls, and cells of pulmonary parenchyma (Table 3). Immunostaining was obtained with some of the antisera raised against PHM enzyme and the hydrophilic cytoplasmic domain (CD), although with variations in tissue localization (Figure 1). No immunoreaction was observed with the antiserum against the PAL domain.
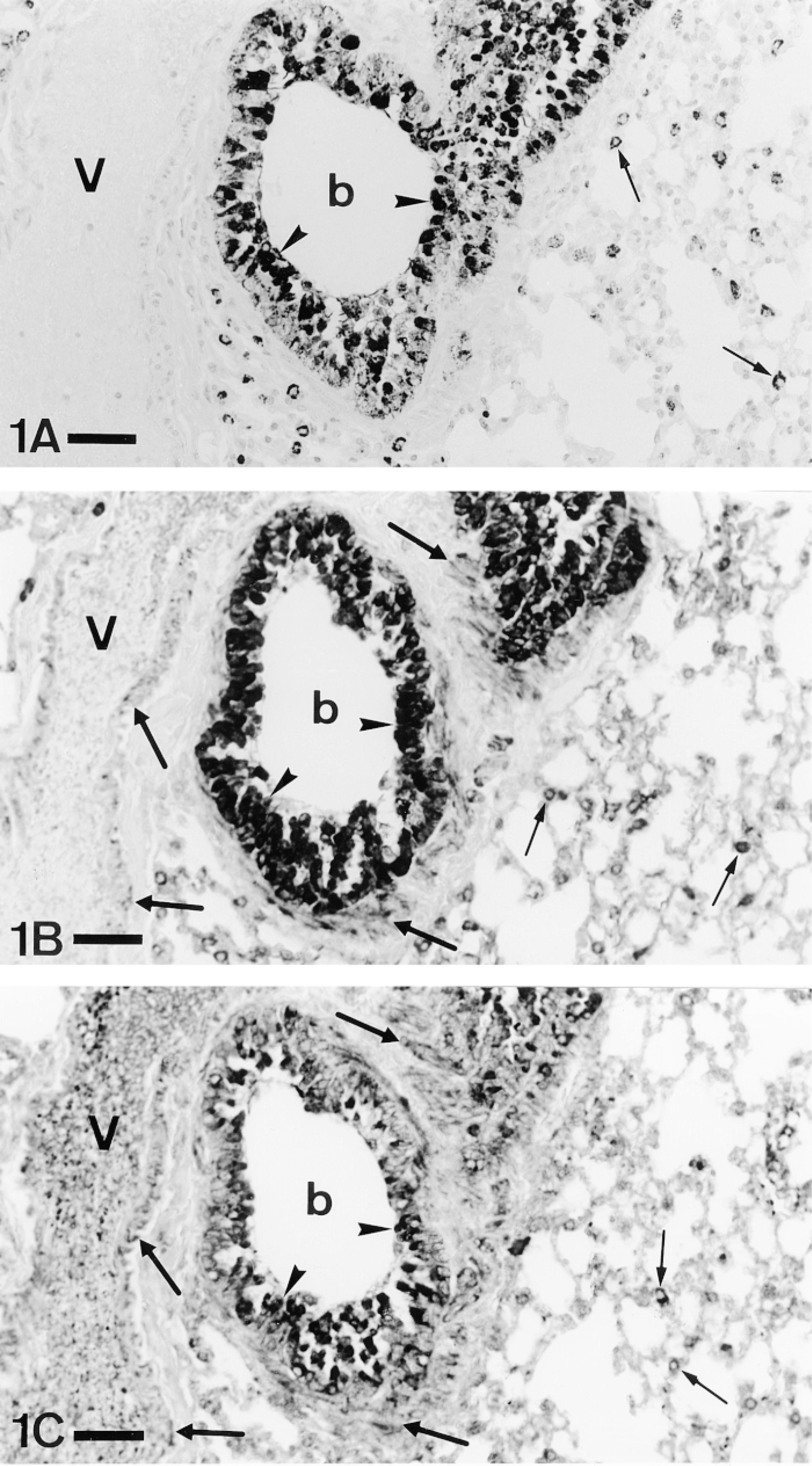
Three serial sections of mouse lung stained with PHM(1) (A), PHM(3) (B), and CD (C) antisera. The three of them stain epithelial (arrowheads) and parenchymal cells (thin arrows). In addition, the PHM(3) and CD antisera label smooth muscle (thick arrows) of both bronchioles (b) and blood vessels (V). P-ad. Bars = 40 μm.
Immunocytochemical results obtained in different tissues with the PAM antisera that rendered immunostaining
In adults, most of the epithelial cells of both bronchi (Figure 2) and bronchioles (Figure 3) showed immunoreactivity for PAM, with the proportion of positive epithelial cells higher in distal than in proximal airways.
(A) Panoramic view of a bronchus (B) in the hilus of mouse lung. Most of the epithelial cells show immunoreactivity for PAM. C, cartilage. Bar = 100 μm. (B) Detail of the rectangle in A. Positive immunostaining for PAM (arrows) can be observed throughout the cytoplasm of many epithelial cells. ABC, PHM(1) antiserum. P-ad. Bar = 20 μm.
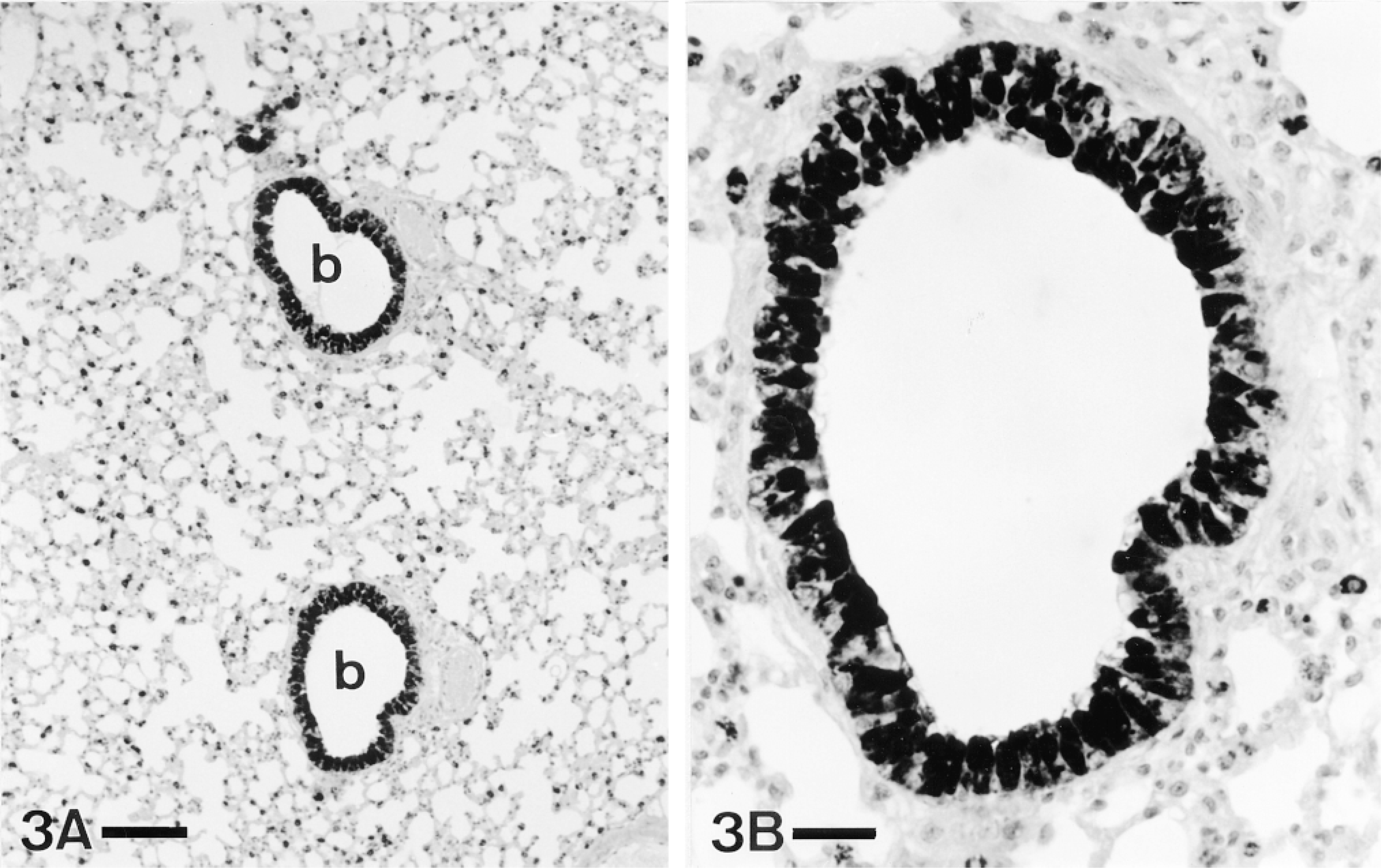
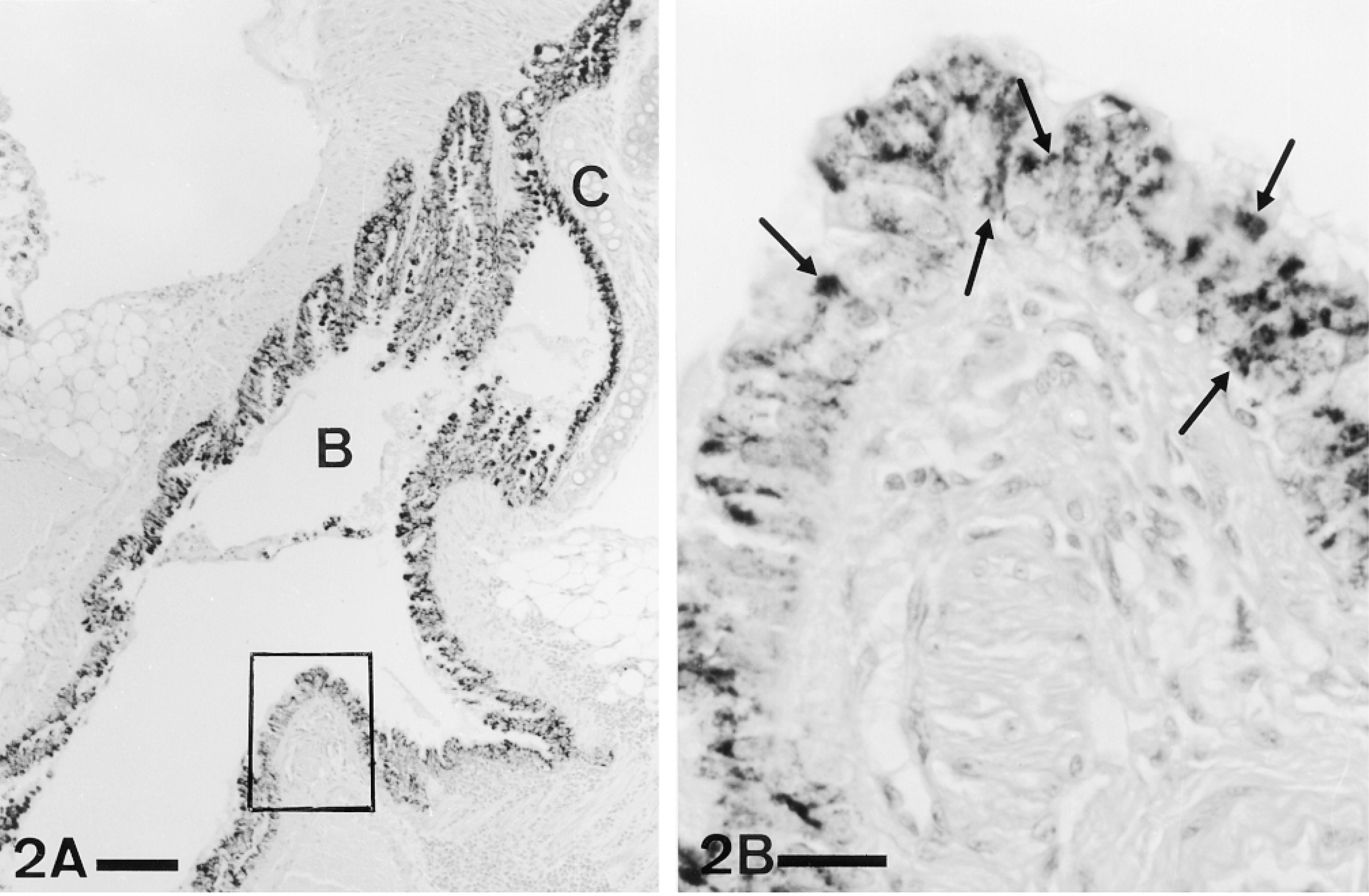
(A) Low-power micrograph showing terminal bronchiole (b) displaying immunoreactivity for PAM. Bar = 100 μm. (B) Detail of one of these bronchioles (bottom). Most of the epithelial cells are intensely stained. ABC, PHM(1) antiserum. P-ad. Bar = 25 μm.
The identification of the PAM-immunostained cells was not always evident in paraffin sections. Both Clara and ciliated cells could be recognized in the sections immunostained for PAM on the basis of morphological criteria. Clara cells, clearly identifiable because of their characteristic dome-shaped apex, were intensely stained both in bronchi (Figure 4A) and bronchioles (Figure 4B). On the contrary, when their identification was certain (Figure 4A) the ciliated cells were seen to be negative.
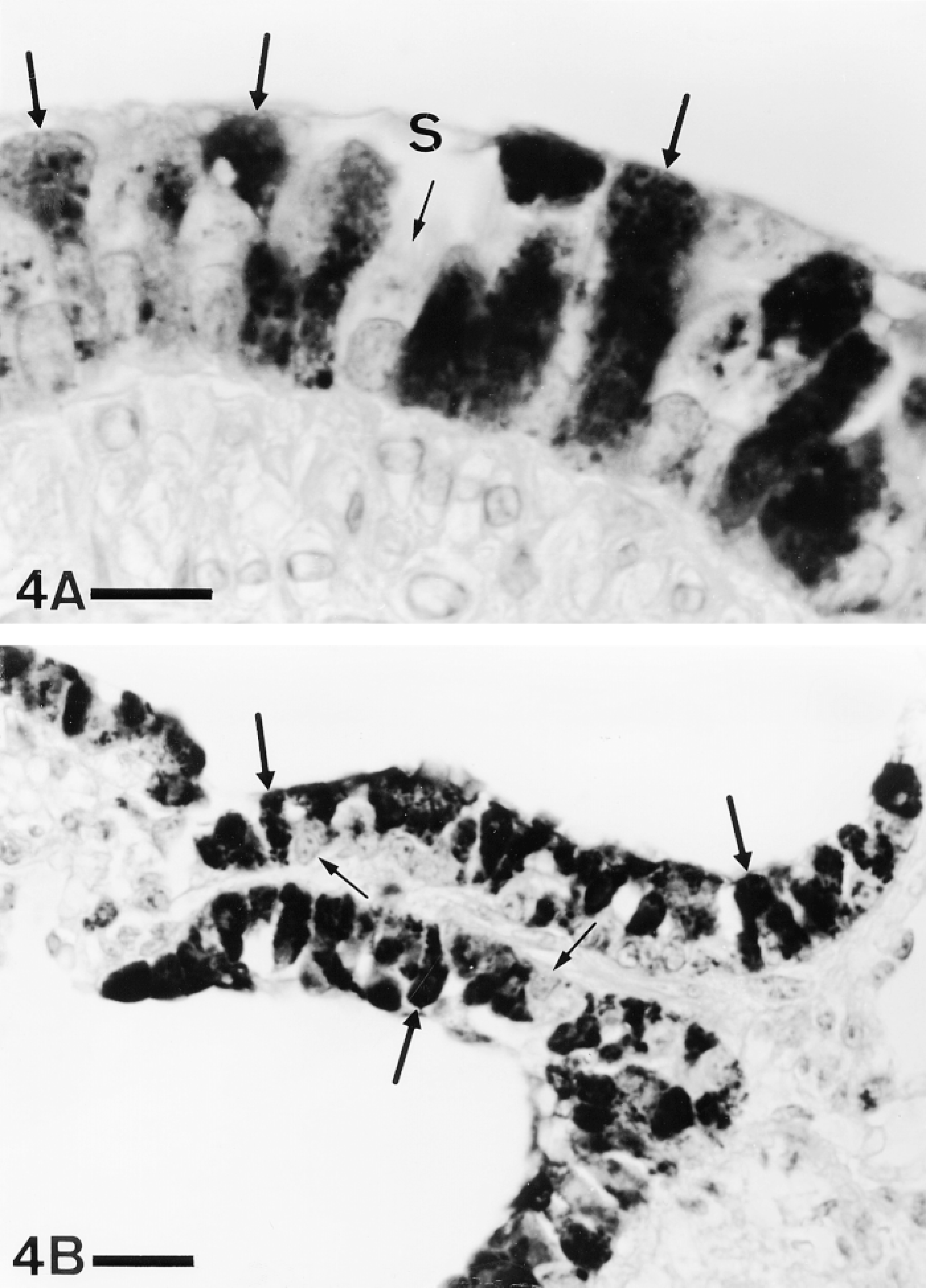
(A) Bronchial epithelium. Clara cells (thick arrows) are intensely immunoreactive for PAM, whereas ciliated cells (thin arrow) are negative. S, surface fluid. Bar = 10 μm. (B) Bronchiolar epithelium. Clara cells (thick arrows) are also immunoreactive, whereas other cells (thin arrows) remain unstained. ABC, PHM(1) antiserum. P-ad. Bar = 20 μm.
The identification of neuroendocrine cells required, on the contrary, immunocytochemical techniques using antibodies against regulatory substances (Table 1). Only CGRP-positive cells were found in mouse lung in our study (Figures 5A and 5C). Using serial reversedface sections, we observed a total lack of co-localization between CGRP, which is generally considered an amidated peptide, and PAM immunoreactivities (Figure 5) with the antisera used (Table 2). Both CGRP-positive isolated endocrine cells and neuroepithelial bodies (Figures 5A and 5C) lacked PAM immunostaining throughout development (Figure 5B) and in adulthood (Figure 5D).
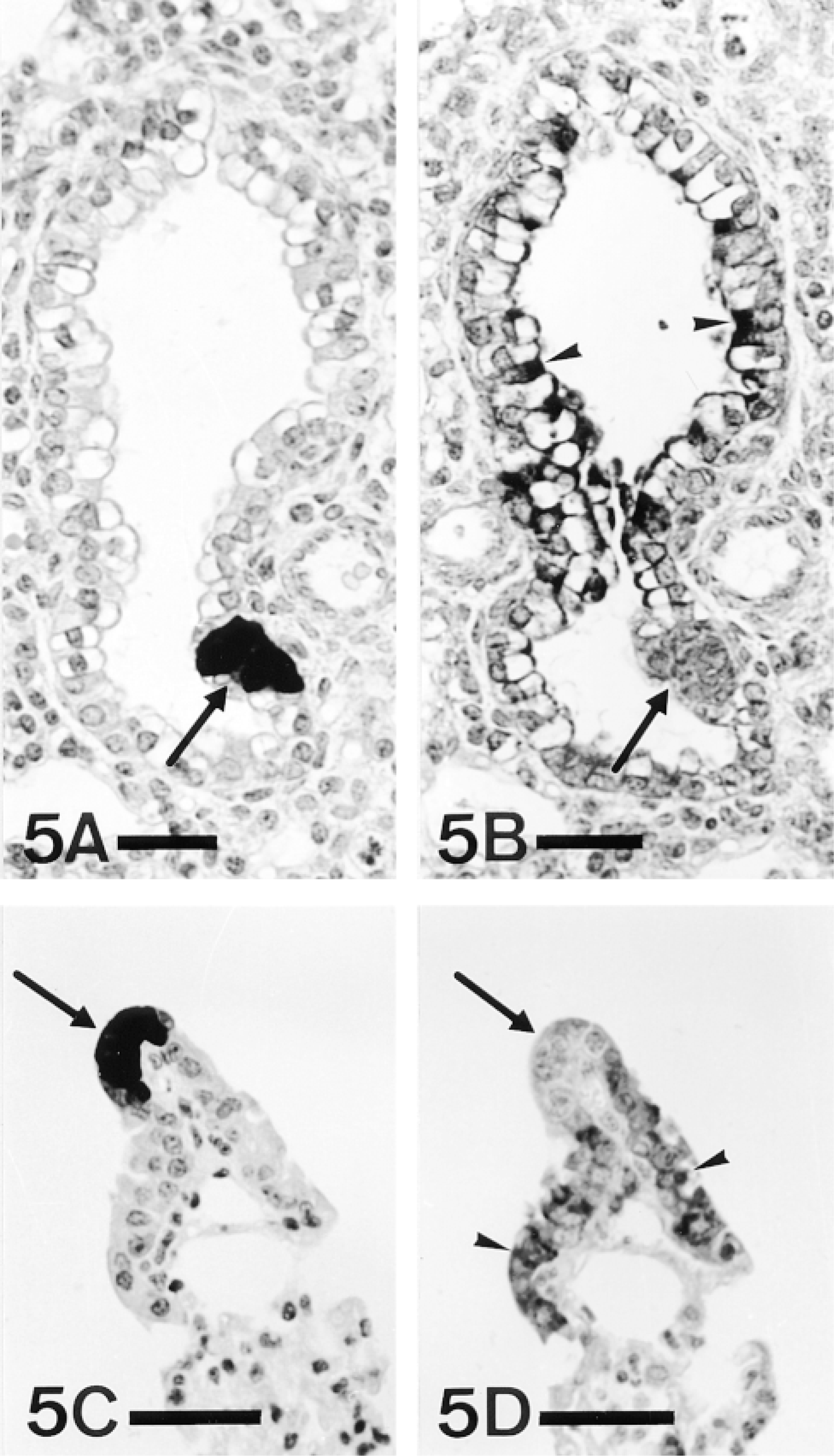
Serial paraffin sections stained with antibodies against CGRP (A,C) and PAM (B,D). Neuroepithelial bodies (arrows) positive for CGRP (A,C) lack PAM immunoreactivity (B,D). Arrowheads, PAM immunostaining. ABC; B,D, PHM(3) antiserum. A,B, E-19; C,D, P-ad. Bars: A,B = 25 μm; C,D = 20 μm.
In developing lungs, PAM immunoreactivity was first detected in airway epithelial cells at E-16 (Figures 6A and 6B). At this gestational age, the immunostaining, although slight, was observed in most bronchial epithelial cells and intrapulmonary epithelial tubules. Immunolabeling was mainly located in the apical region of epithelial cells and showed a granular aspect (Figure 6B). A lesser number of positive granules could also be observed scattered in the rest of the cytoplasm.
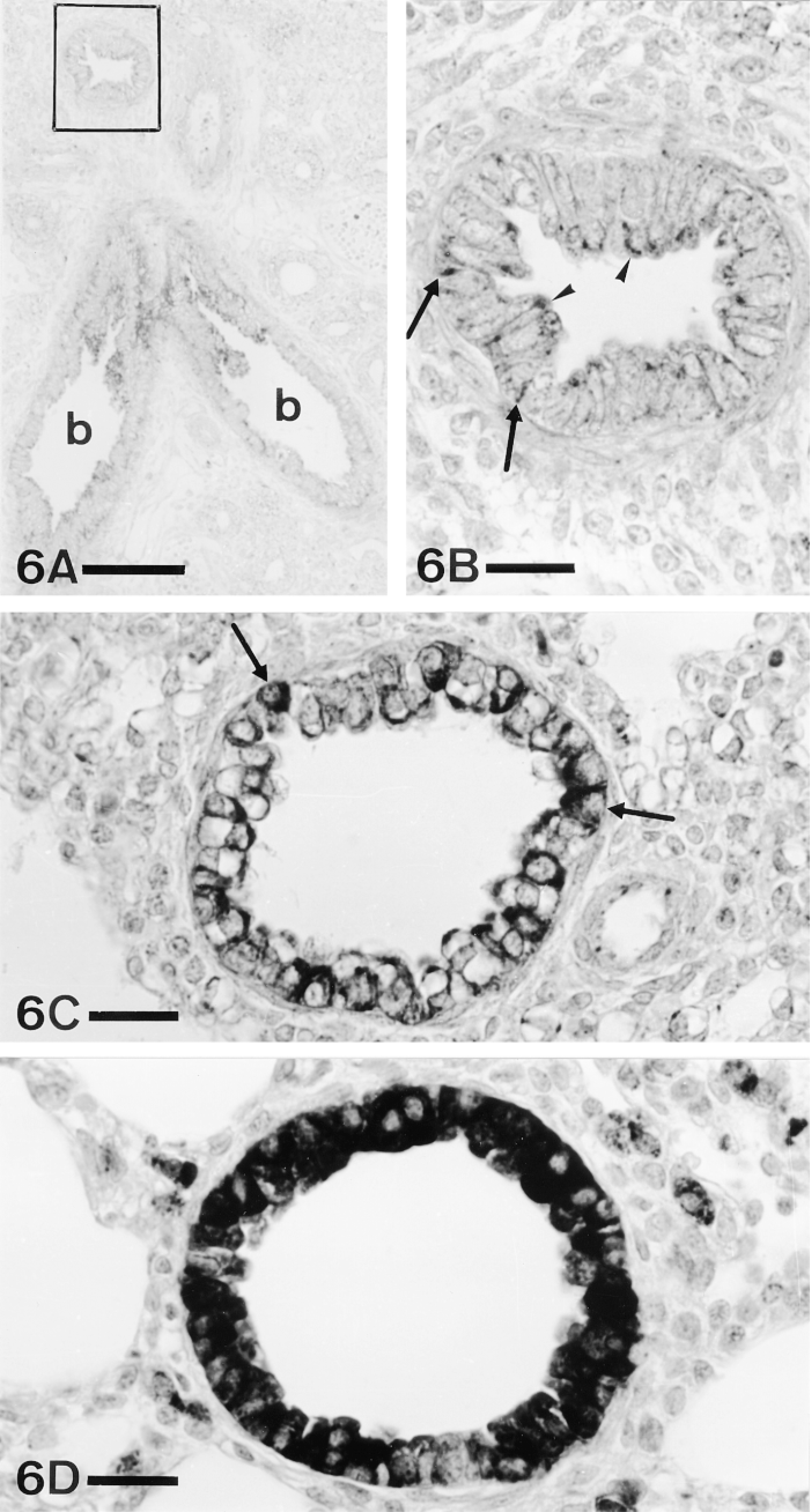
Appearance and evolution of PAM immunoreactivity in bronchiolar epithelium. (A) Panoramic view of an E-16 lung. The bronchiolar epithelium (b) shows immunoreactivity for PAM. Bar = 100 μm. (B) Detail of the rectangle in A. The immunoreactivity is mainly located in the apical region of the epithelium (arrowheads), although some staining in the basal region (arrows) can be observed. (C) E-18. Some epithelial cells (arrows) show immunostaining throughout the whole cytoplasm. (D) P-6. Most of the bronchiolar epithelial cells show the entire cytoplasm intensely stained. ABC, PHM(3) antiserum. Bars = 20 μm.
As development continued, the immunolabeling in creased and extended towards the base of epithelial cells (Figure 6). At E-18 and E-19 (Figure 6C), some epithelial cells showed the whole cytoplasm immunostained, whereas in others the immunoreactivity was still restricted to the apical region. After birth, almost all the positive epithelial cells showed immunostaining throughout the cytoplasm (Figure 6D). This developmental pattern was similar in the epithelial cells of bronchi and bronchioles.
Although this study focused on epithelial cells, other tissues also exhibited immunostaining for the PAM enzyme.
Most of the smooth muscle cells of the bronchi (Figures 7A and 7B) and bronchioles (Figure 7C) were PAM-immunoreactive. In blood vessels (Figure 7D), variable numbers of positive cells, usually less frequent than in airways, were observed. As in the case of the epithelium, only some of the antisera against the PHM domain and the cytoplasmic tail rendered labeling. However, not all the antisera that stained epithelial cells were positive in smooth muscle (Table 3; Figure 7E).
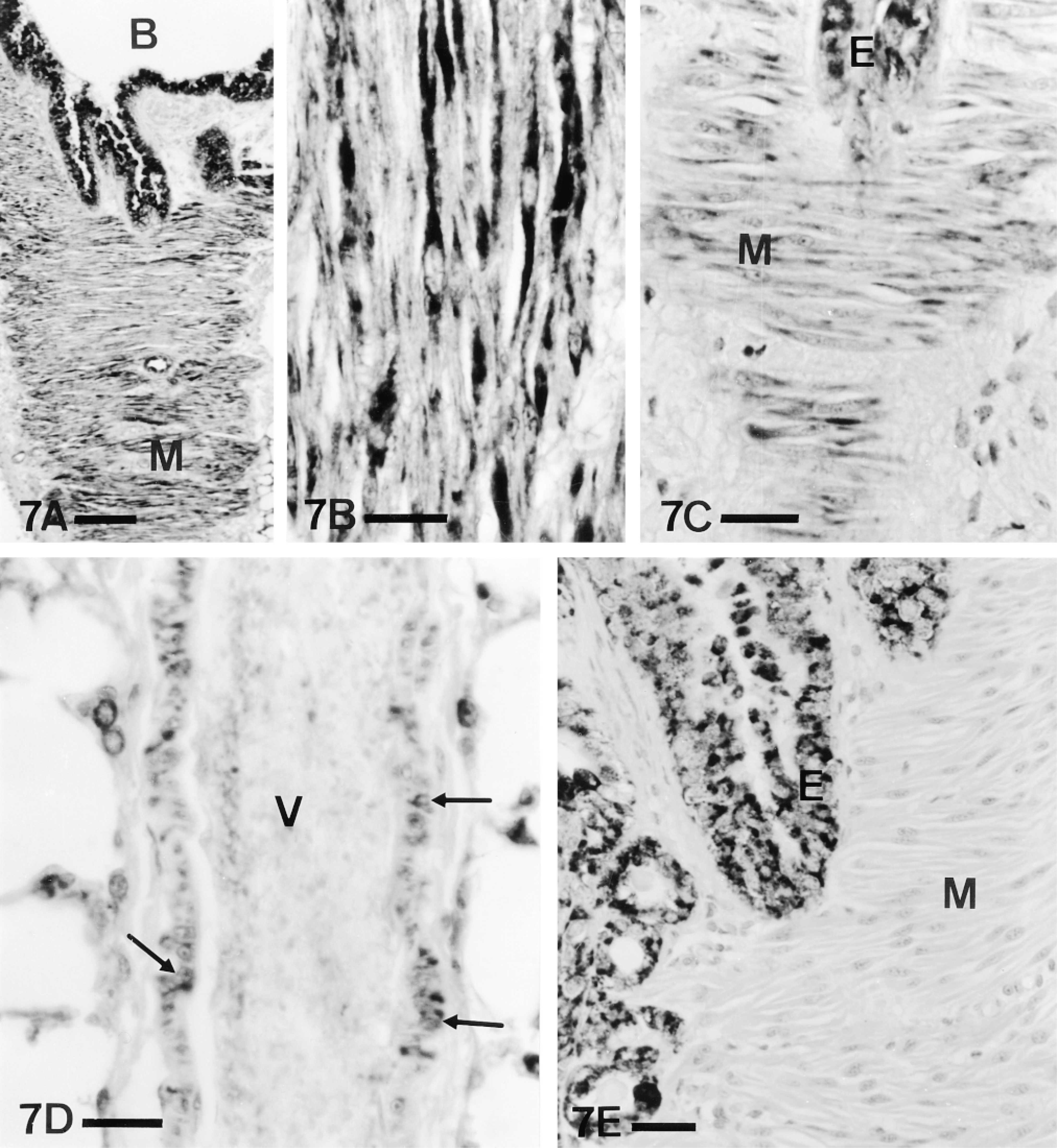
PAM immunoreactivity in smooth muscle of airways (A-C,E) and blood vessels (D). (A) Panoramic view of bronchial wall, tangentially sectioned. In addition to the epithelium, PAM immunostaining can be observed in the smooth muscle (M). B, bronchial lumen. An identical pattern of immunostaining with PHM(3) antiserum is shown in Figure 9A. Bar = 100 μm. (B) Detail of the bronchial smooth muscle, longitudinally sectioned. Many myocytes show PAM immunoreactivity. (C) A similar tangential section of a bronchiole. Bronchiolar smooth muscle cells (M) may also show PAM immunoreactivity. E, epithelium. (D) Pulmonary blood vessel (V) showing PAM immunoreactivity in some cross-sectioned smooth muscle cells (arrows). Bars = 20 μm. (E) Tangentially sectioned bronchial wall immunostained with the PHM(1) antiserum. Whereas epithelium (E) shows immunoreactivity, smooth muscle (M) remains unstained. ABC; A,B, CD antiserum; C,D, PHM(3) antiserum. P-ad. Bar = 25 μm.
In adult lungs, cells in the pulmonary parenchyma were also positive for PAM (Figures 8A and 8B), the immunoreactivity extending throughout the cytoplasm. Neither fetal nor P-0 to P-2 lungs showed positive parenchymal cells. At P-6, some positive parenchymal cells were observed (Figures 8C and 8D), but in lesser numbers and less intensely stained than in adults.
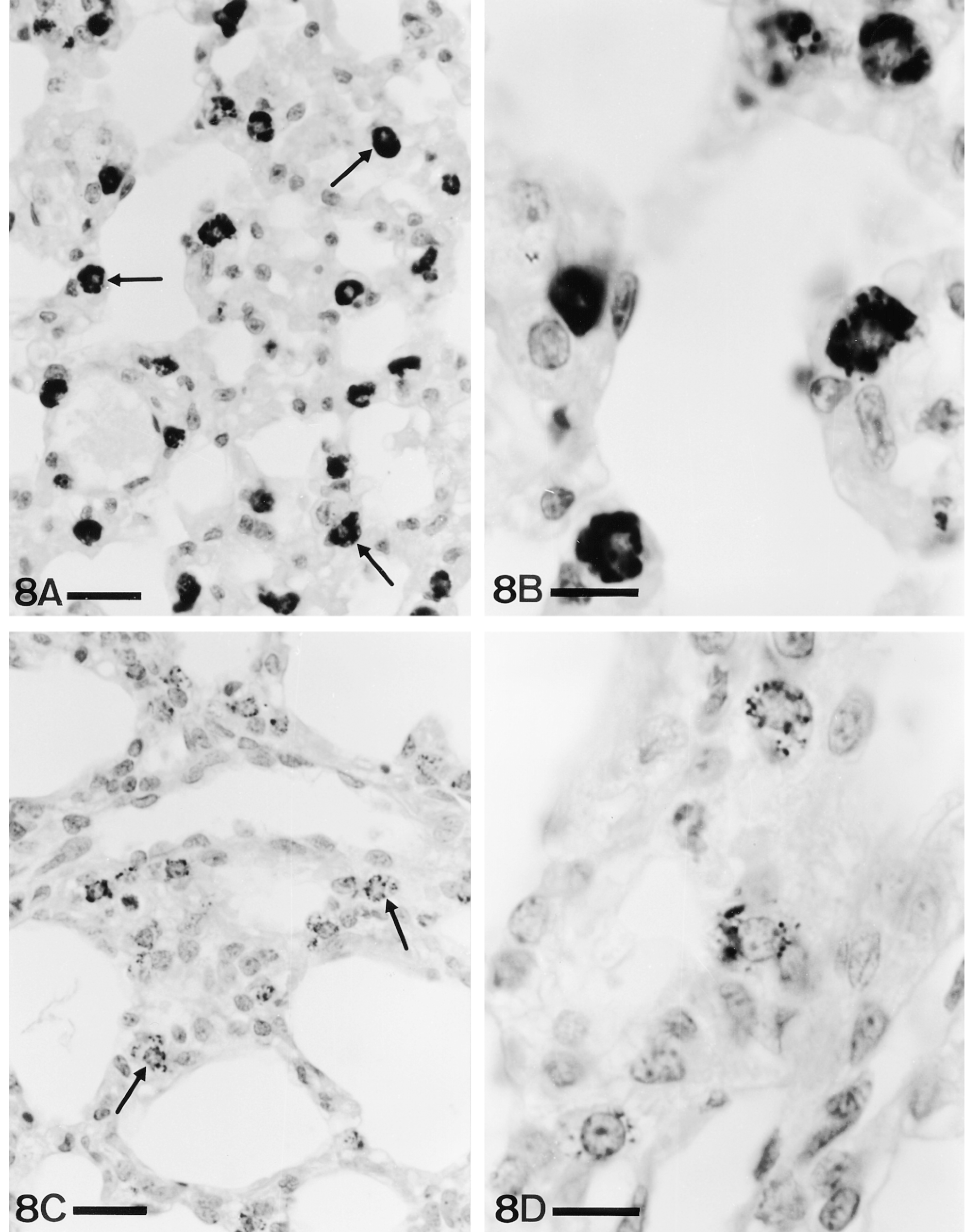
(A) P-ad. Frequent parenchymal cells (arrows), showing a variety of shapes, display immunoreactivity for PAM. Bar = 20 μm. (B) Detail of some of these cells. Bar = 10 μm. (C) P-6. Immunoreactivity in parenchymal cells (arrows) appears at this stage. Note the lesser numbers of positive cells and amount of immunoreactivity compared with adult lungs. Bar = 20 μm. (D) Detail of P-6 parenchymal cells. ABC, PHM(1) antiserum. Bar = 10 μm.
In the three cell types—epithelium, smooth muscle, and parenchymal cells—preabsorption of the antiserum with its antigen abolished immunolabeling (Figure 9).
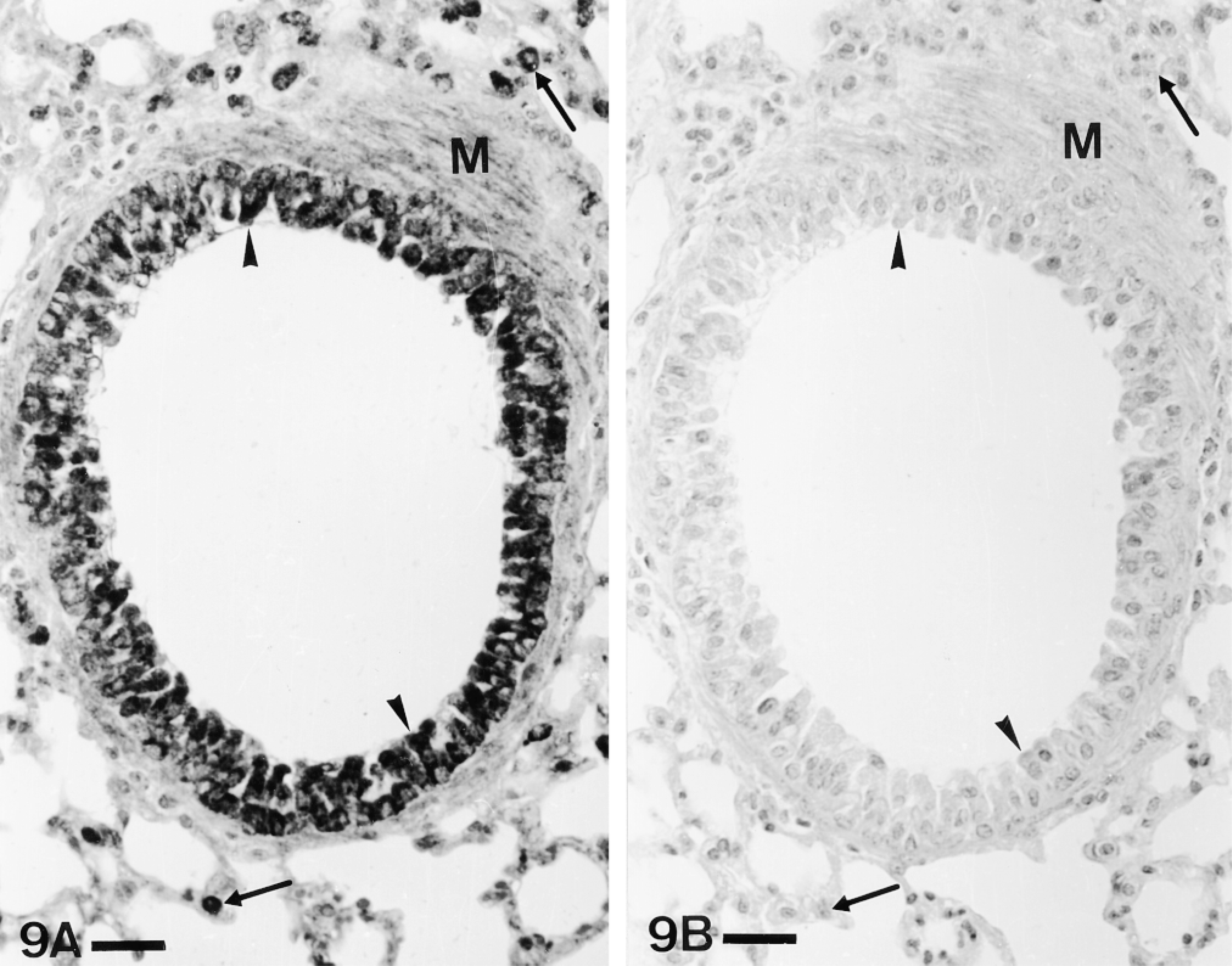
Absorption controls. The immunoreactivity for PAM (A) disappears when the antiserum is preabsorbed with the corresponding antigen (B) in the three immunoreactive tissues found in mouse lung: epithelium (arrowheads), smooth muscle (M), and parenchymal cells (arrows). ABC, PHM(3) antiserum. P-ad. Bars = 25 μm.
Discussion
This study shows the distribution of the amidating enzyme PAM in developing and adult mouse lung. PAM immunoreactivity was localized in diverse cell types: epithelial cells and smooth muscle (both in fetal and postnatal lungs) and pulmonary parenchymal cells, probably macrophages (only in postnatal lungs). Epithelium was the first tissue in which immunostaining was detected, on E-16 fetuses.
In the respiratory tract, PAM immunoreactivity has been reported only in human lung (Saldise et al. 1996). PAM immunostaining was found in airway epithelium, smooth muscle, and alveolar macrophages, as it occurs in the mouse lung but, in addition, endothelium, neurons, Schwann cells, and chondrocytes were also immunostained with some of the antisera used in the present study.
Immunoreaction was obtained in the mouse lung with antisera raised against the PHM domain and the hydrophilic cytoplasmic tail but not with the PAL antiserum. These results are, again, only partially coincident with those in human lung, in which, in addition to PHM immunoreactivity, PAL immunostaining has also been detected (Saldise et al. 1996) using the same antiserum used in this study.
According to immunocytochemical studies on the presence of PAM in mammalian tissues, three patterns of PAM labeling have been found: immunoreactivity to both PHM and PAL, or immunostaining only for PHM or PAL. Because PHM and PAL are derived from the same mRNA precursor, it would be expected to find both enzymes in the same cells, i.e., the first pattern of staining seems more logical. This pattern has been found in several organs, including normal human lung and some lung epithelial tumors (Saldise et al. 1996), but not in mouse lung (only PHM).
Considering that mouse and bovine PAL2 sequences are identical, the lack of PAL immunoreactivity in mouse lung is an intriguing result. The lack of PAL immunoreactivity has also been reported in most human lung epithelial tumors (Saldise et al. 1996) and in other mammalian cells, such as glucagon/GLP-1-containing endocrine cells of rat colon (Martinez et al. 1993a). Because it has been reported that PHM is able to complete the amidation process under certain conditions (Katopodis et al. 1990), a similar process might occur in pulmonary cells lacking PAL immunoreactivity. We must consider that tissue-specific expression of alternative mRNA splice forms and posttranslational proteolysis have also been extensively demonstrated, showing the existence of different PAM isoforms, some of them lacking the PAL domain (Stoffers et al. 1989,1991; Eipper et al. 1992a; Milgram et al. 1992). Such diversity of isoforms could explain some of the differences in the distribution pattern of PHM and PAL immunoreactivities. More difficult to explain is the presence of immunostaining for both PHM and CD domains in mouse lung, because an alternative mRNA splice form containing both PAM and CD domains but lacking the PAL domain has never been described. Therefore, the absence of PAL immunostaining in mouse lung could more reasonably be due to the low sensitivity of the PAL antibody used in the present study. This and other possibilities may be adressed using in situ hybridization and generation of new antibodies.
Our immunocytochemical results indicate that Clara cells of both bronchi and bronchioles of the mouse respiratory tract are immunoreactive for PAM, whereas CGRP-positive endocrine cells and the ciliated cells—at least most of them—are negative. In a similar manner, Clara cells of human lung were shown to be positive and GRP endocrine cells were negative. On the contrary, the many ciliated cells present in human airways were immunoreactive for PAM (Saldise et al. 1996).
Endocrine Cells
The presence of PAM enzymes has been described in endocrine organs producing amidated peptides (Birnbaum et al. 1989; Markosian et al. 1989; Braas et al. 1992), but little is known about the presence of these enzymes in the diffuse neuroendocrine system (Martinez et al. 1993a,b).
In mouse lung, although CGRP is an amidated peptide, endocrine CGRP-positive cells have been found to be negative for PAM with the antisera used in all stages studied. Because PAM has been demonstrated in endocrine cells producing amidated peptides (Martinez et al. 1993a,b; Steel et al. 1994), this is an unexpected finding. Similar results have been reported in human fetal lung (canalicular period, spontaneous miscarriage), in which GRP-positive cells lacked PAM immunoreactivity (Saldise et al. 1996). The lack of co-localization of PAM and amidated peptides has also been previously reported in endocrine cells of other organs, such as human pancreas (Martínez et al. 1993b) and rat small intestine (Martínez et al. 1993a). In rat stomach, only a subset of gastrin cells were also positive for PAM antisera (Martínez et al. 1993a), suggesting that only the more mature cells contain the amidating enzymes. Some possibilities have been suggested to explain the lack of PAM immunoreactivity in amidated peptide-producing cells, which could apply to mouse lung endocrine cells. The presence of an amidating enzyme different from PAM has been suggested (Martinez et al. 1993a,b), but all attempts to identify other amidating enzymes have been unsuccessful (Martinez and Treston 1996). Another possibility could be the existence of tissue-specific isoforms of PAM that may not be detected by the antibodies used (Braas et al. 1989). A third possibility includes different functions for amidated and nonamidated forms of peptide hormones (Seva et al. 1994).
Clara Cells
The presence of amidating enzymes in Clara cells, which constitute most of the epithelial cells of mouse airways, is difficult to explain because the existence of amidated factors in these cells is as yet unknown. Perhaps the presence of PAM in them may be an indicator of the production of amidated peptides, still unknown, by this cell type. A possible explanation for the presence of PAM immunoreactivity in airway epithelial cells is provided by the fact that mammalian epithelial cells produce endothelin (ET) (Giaid et al. 1990; Rozengurt et al. 1990; Springall et al. 1991). Although ET itself is not amidated, the presence of PAM enzymes could be related to the amidation of other peptides derived from the ET precursor, which contain predicted amidation sites (Cuttitta 1993). The biological actions of these peptides remain unknown. In addition, immunoreactivity for adrenomedullin, another amidated peptide, has recently been described in human airway epithelium (Martínez et al. 1995). These authors indicated that distribution of adrenomedullin was coincident with that of PAM in human lung (Saldise et al. 1993,1996). Finally, it has been suggested that enzymes required for processing peptide hormone precursors, such as PAM, may be a common feature of cells containing secretory granules, even though they may not produce any endogenous prohormone substrate (Dickinson et al. 1993). Further biochemical and cytochemical studies are needed to clarify this point.
Classically, Clara cells have been described as exocrine cells secreting towards the lumen of airways. During mouse lung fetal development, immunoreactivity for PAM appears first in the apical cytoplasmic region of Clara cells. Later, the labeling is observed throughout the cell but remains especially evident in the apical region. Similarly, PAM immunoreactivity in adult human airways has been described in the apical region of epithelial cells, and the possible secretion of amidating enzymes to airway lumen has been suggested (Saldise et al. 1996). Some other observations support this hypothesis. First, PAM has also been reported in lung exocrine glands (Saldise et al. 1996), in salivary glands (von Zastrow et al. 1986; Braas et al. 1992), and in saliva (von Zastrow et al. 1986; Kato et al. 1992). Furthermore, PHM and PAL enzymatic activities can be found in human bronchoalveolar lavage fluid (Scott et al. 1993), and cultured cells expressing PAM secrete this enzyme (Maltese and Eipper 1992).
On the other hand, the presence of PAM immunoreactivity in the base of the Clara cells could be explained as merely a consequence of physical expansion of the increasing amount of PAM, which would be exported later to the apex of the cell. However, we must consider that the existence of basal PAM could also reflect other specific functions taking place in the basal cytoplasm.
Smooth Muscle
PAM immunostaining was clearly observed in smooth muscle cells in neonatal lungs (P-0). The fact that not all the PAM antisera that stained epithelial cells in mouse lung gave immunoreaction in smooth muscle suggests the presence of different isoforms in both cell types. The presence of PAM in smooth muscle cells has been previously reported in hypophysial arteries (May and Braas 1991) and in airways and blood vessels of human lung (Saldise et al. 1996). The presence of PAM in these cells suggests the production of amidated regulatory peptides that could be involved, through an auto- and/or a paracrine mechanism, in the control of broncho- and vasomotor tone. In fact, ET (Giaid et al. 1993; Seldeslagh and Lauweryns 1993a,b) and adrenomedullin (Martínez et al. 1995) have also been found in pulmonary smooth muscle cells.
Parenchymal Cells
PAM-immunoreactive cells present in the pulmonary parenchyma of mouse are suggested to be macrophages because they were absent in fetal animals, appearing only in postnatal lungs (P-6). In addition, the localization of PAM in macrophages, although alveolar, has also been reported in human lung (Saldise et al. 1996). The presence of PAM in these cells could be related to the reported production of IGF-I (Room et al. 1988), adrenomedullin (Martinez et al. 1995), or ET (Ehrenreich et al. 1990). Similar to ET, IGF-I is not an amidated peptide but its precursor contains a biologically active amidated peptide (Siegfried et al. 1992).
Development
To our knowledge, this is the first description of the appearance and evolution of PAM immunoreactivity in developing lung. In mouse lung, PAM immunoreactivity in epithelial cells appeared in the fetal period (E-16), rose while development continued, and reached the strongest immunostaining in mature lungs (from P-6 onwards). During development, the early apical immunolabeling of the epithelium, probably in relation to the amidation of secretory products, extended towards the base of the cells. As indicated, PAM in the basal cytoplasm could also be involved in the processing of hypothetical peptides secreted into the basal extracellular space.
It is difficult to date the appearance of PAM immunoreactivity in smooth muscle cells, because in mouse fetuses mesenchymal cells gradually differentiate into smooth muscle cells.
Little is known about the presence of PAM in developing organs. To our knowledge, only rat and sheep pancreas (Ouafik et al. 1987; Scharfmann et al. 1988; Maltese et al. 1989; Zhou and Thorn 1990; Kapuscinski and Shulkes 1995) and rat heart (Ouafik et al. 1989) have been studied in relation to the presence of the PAM enzyme during fetal and/or neonatal stages. In both organs, peaks of the enzyme at different points in development and a decrease of PAM in the adult period have been described. On the contrary, in mouse lung, as already stated, the strongest PAM immunostaining was found in adult lungs. These results indicate a different pattern of PAM expression in developing organs, which supports the existence of a tissue-specific regulation of PAM expression during development (Maltese et al. 1989; Ouafik et al. 1989).
The early appearance of PAM in mouse developing lung, as well as its presence in a variety of fetal and adult tissues, probably indicates a complex role of this enzyme in pulmonary function.
Footnotes
Acknowledgements
Supported by the Spanish Ministry of Education and Science (DGCYT project no. PB93-0711) and the University of Navarra (PIUNA), and by a grant from the Departamento de Educación y Cultura del Gobierno de Navarra, Spain (LG).
We would like to thank Dr B.A. Eipper (Johns Hopkins University School of Medicine; Baltimore, MD) for some of the antisera against PAM and for her advice and review of the manuscript, and Prof J.M. Polak (Hammersmith Hospital; London, UK) for the antisera against regulatory substances. We thank Dr M.A. Burrell and Dr L.M. Montuenga for critically reading the manuscript, and I. Ordoqui and A. Urbiola for technical assistance.
