Abstract
Endothelins (ET) are a family of regulatory peptides displaying, among other abilities, potent constrictor actions. We studied the perinatal time course expression and distribution of ET in the mouse airway epithelium. In fetal mouse, ET-immunoreactivity (IR) appeared earlier (gestational Day 18) in the epithelium of upper (bronchi and large bronchioles) than in lower airways, being scarce and mainly located in the apical cytoplasm. As the lung developed, ET-IR became gradually stronger and extended throughout the cell in both bronchi and bronchioles. ET-IR was found in most airway epithelial cells. Clara cells were positive for ET, whereas ciliated and endocrine cells were not. In adult lungs, part of the myocytes and parenchymal cells also showed ET-IR. In both developing and adult mouse lungs, the cell distribution of ET-IR in the epithelium is compatible with apical and/or basal secretion. The presence of ET in mouse airway epithelium during the perinatal period may indicate a role for ET as a growth factor in lung development and its involvement in control of lung ventilation at birth.
E
In the perinatal lung, a role for ET in the control of pulmonary vascular tone has been suggested because both vasoconstrictor (Ivy et al. 1994; Perreault and Baribeau 1995) and vasodilator (Pinheiro and Malik 1993; Wong et al. 1993; Perreault and Baribeau 1995) actions have been reported. Studies in ET-1 knockout mice revealed that ET-1 is essential in the physiological control of ventilation during the neonatal period (Kuwaki et al. 1996; Kurihara et al. 1994). However, knowledge about the histological localization of ET during lung development is still fragmentary. The cellular localization of ET has been studied in a systematic manner only in two species (human and rat), rendering different results: endocrine cells in human (Giaid et al. 1991) and epithelial cells of the peripheral bronchial tree in rat (Katsura et al. 1993). In mouse, only a few studies exist on the presence of ET in the postnatal period and, again, the results are not coincident. In neonatal mice (1 day to 1 week), ET-IR was found in pulmonary endocrine cells (Seldeslagh and Lauweryns 1993), and in young mice (3 weeks onwards) most airway epithelial cells have been reported to be immunostained (Rozengurt et al. 1990).
The present study was carried out in the mouse, a species in which only postnatal studies concerning the presence of ET in lung have been made, rendering contradictory results. Immunocytochemical techniques were applied in a systematic manner to developing (fetal and early postnatal) and adult lungs to determine the time of appearance of ET-IR, not reported to date, as well as the further time course pattern along pulmonary development.
Materials and Methods
Pathogen-free Swiss mice bred at the Centro de Investigación en Farmacología Aplicada (CIFA; University of Navarra, Spain) were used in this study. Lungs of 36 mice were studied: eight adult lungs (six of them pregnant females: P-ad) and 28 subjects at various stages of development [gestational Days (E) 17, 18, and 19 and postnatal days (P) 0, 1, 2 and 6], the age at which mouse lung is considered to be mature (Four subjects of each age).
The adults were anesthetized with 12.5% urethane (1 ml/100 g body wt). The abdomen of pregnant females was opened and the fetuses were rapidly removed from the uterus and chilled in ice. The newborn mice were anesthetized by hypothermia (Danneman and Mandrell 1997). The chest of fetuses, newborns, and adults was opened and lungs were removed, fixed by immersion in Bouin's fluid for 20–24 hr, and embedded in paraffin.
Immunocytochemistry
Paraffin sections (4 μm thick) were mounted on glass slides and immunocytochemical staining was performed using the avidin–biotin complex (ABC) method (Hsu et al. 1981). After removal of paraffin with xylol followed by 5 min in absolute ethanol, endogenous peroxidase was blocked by treatment with 3% hydrogen peroxide in absolute methanol for 30 min. Sections were hydrated through a graded series of ethanol and then placed in Tris-HCl-buffered saline (TBS: Tris buffer 0.05 M, pH 7.36, and NaCl 0.5 M). Nonspecific binding sites were blocked with 5% normal swine serum in TBS for 30 min at room temperature (RT). Sections were then incubated overnight at 4C with the primary antiserum. Four polyclonal antisera raised against ET-1 (1914, 1915, 2020, 2092) and one against big-ET-1 (2049) previously characterized (Inagaki et al. 1991) were used. An antiserum against calcitonin gene-related peptide (CGRP; 1204) was used because CGRP has been proved to be the most reliable and sensitive immunohistochemical marker for pulmonary neuroendocrine cells in the mouse (Wuenschell et al. 1996). These antisera were a kind gift from Dr. Polak (Hammersmith Hospital; London, UK). A polyclonal antiserum to Type II pneumocytes [surfactant protein C (SP-C), a kind from Dr. Whitsett (Children's Hospital Medical Center, Cincinnati, OH)] was used. Sections were rinsed in TBS (5 min) and then incubated for 30 min at RT with biotinylated swine anti-rabbit immunoglobulins (1:200 in TBS; E353; Dakopatts, Glostrup, Denmark). After a second rinse in TBS, sections were treated for 30 min at RT with avidin–biotin–peroxidase complex (1:100 in TBS; K355; Dakopatts). The peroxidase activity was demonstrated by 0.03% 3,3′-diaminobenzidine tetrahydrochloride (D-5637; Sigma, St Louis, MO) in sodium acetate/acetic acid 0.1 M, pH 5.6, containing 2.5% ammonium nickel sulfate, 0.2% β-
Controls
Several controls were performed: (a) omission of the primary antibody or substitution by a non-immune serum; (b) positive control with mammalian tissues (rat stomach); and (c) absorption tests with the corresponding antigens. Antisera were preincubated for 12 hr at 4C with their respective synthetic antigens at a concentration of 0.1–10 nmol per ml of optimally diluted primary antiserum before application to tissue sections.
Results
In the present study, using various antisera raised against ET-1, IR was found in airway epithelial cells in both developing and adult mouse lung.
In developing lungs (Figures 1 and 2) ET-1-IR was detected for the first time at E-18 (Figure 1A and 1B) in upper airways (bronchi and large bronchioles) and gradually extended towards the rest of the bronchiolar tree (Figure 2). Early ET-1-IR was scarce and was mainly located in the apical cytoplasm of epithelial cells (Figure 1B). While development continued (end of gestation and first days of postnatal life), the immunostained area gradually increased, extending towards the base of the cells (Figure 1C and 1D). From approximately P-6 onwards, the entire cytoplasm of epithelial cells was immunoreactive (Figure 1E and 1F). In bronchiolar epithelium (Figure 2), ET-1-IR, although appearing later, showed a similar cytological pattern, sparse and mainly apical at the beginning (Figure 2B) and then extending throughout the cell (Figure 2C and 2D). The application of antiserum against CGRP and ET-1 to serial reversed-face sections showed that fetal CGRP-positive endocrine cells (Figure 2E) did not stain with the antiserum against ET (Figure 2F).
The time of appearance of ET-1-IR depended not only on the size of airways but also on the antiserum used. In bronchi, the earliest IR (E-18) was obtained with the 2020 antiserum (Figure 1A and 1B). Conversely, 2092 antiserum gave IR for the first time in postnatal lungs (P-1; data not shown). Nevertheless, the evolution pattern of ET-1-IR (apical to the entire cytoplasm) was similar with all the antisera.
Big-ET-1-IR showed in the epithelium a similar developmental pattern to that of ET-1: first in upper airways, first in the apical cytoplasm, and extending afterwards towards the rest of the cell. However, big-ET-1 was detected later in development (P-2 lungs) than ET-1 (data not shown).
In adult lung (Figure 3), IR for ET-1 and big-ET-1 was obtained in most epithelial cells of both bronchi and bronchioles (Figures 3A, 3C, and 3D). Among the cell types present in airway mouse epithelium, Clara cells were clearly identified as immunoreactive for ET-1 (Figure 3C). Conversely, ciliated cells, when their identification was certain, were negative (Figure 3C). CGRP-positive endocrine cells (Figure 3E) were also negative for ET-1 (Figure 3F). The specificity of IR was tested by absorption controls (Figure 3G and 3H).
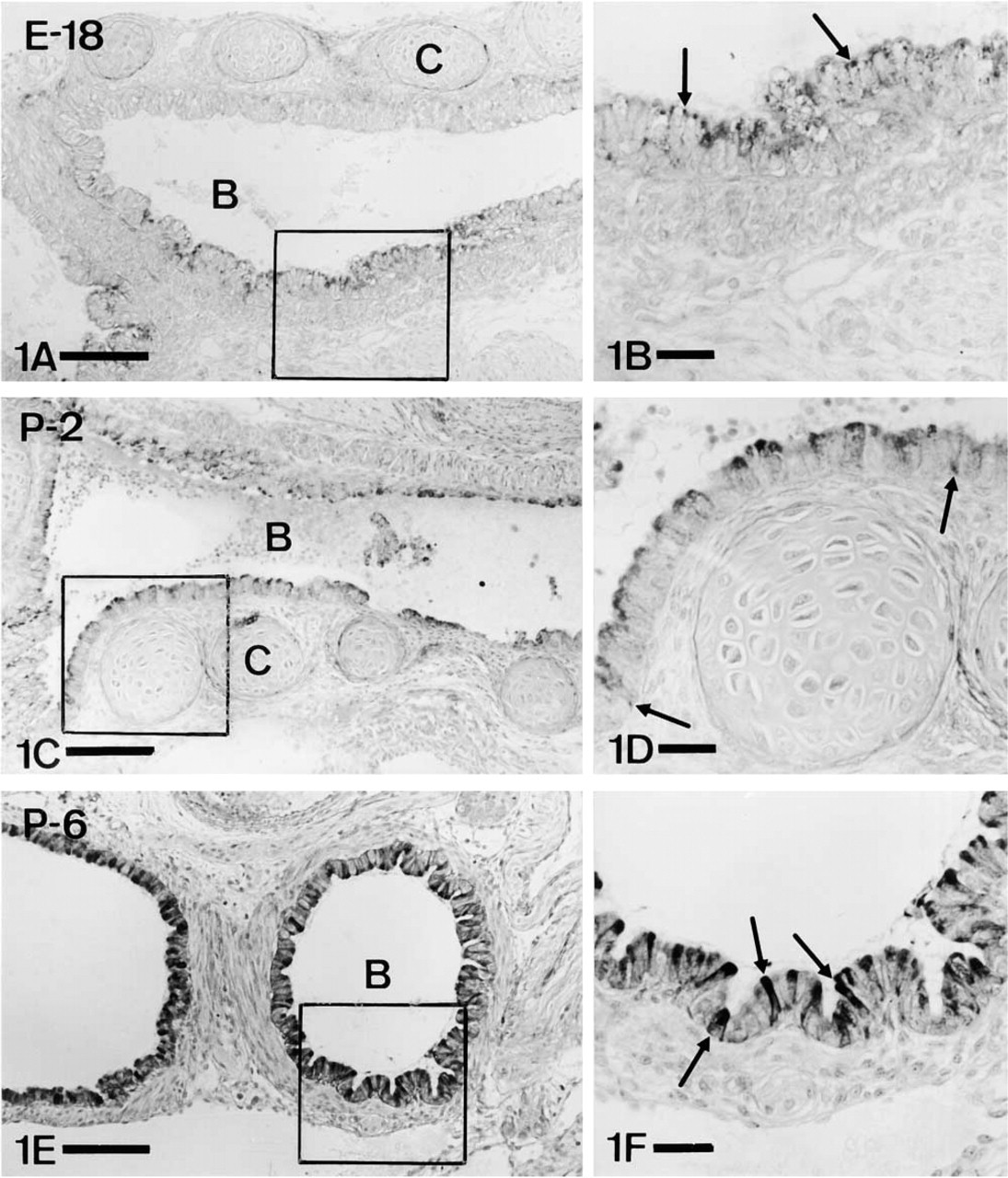
Appearance and evolution of ET-1 immunoreactivity in bronchial epithelium. B, bronchus; C, cartilage. (
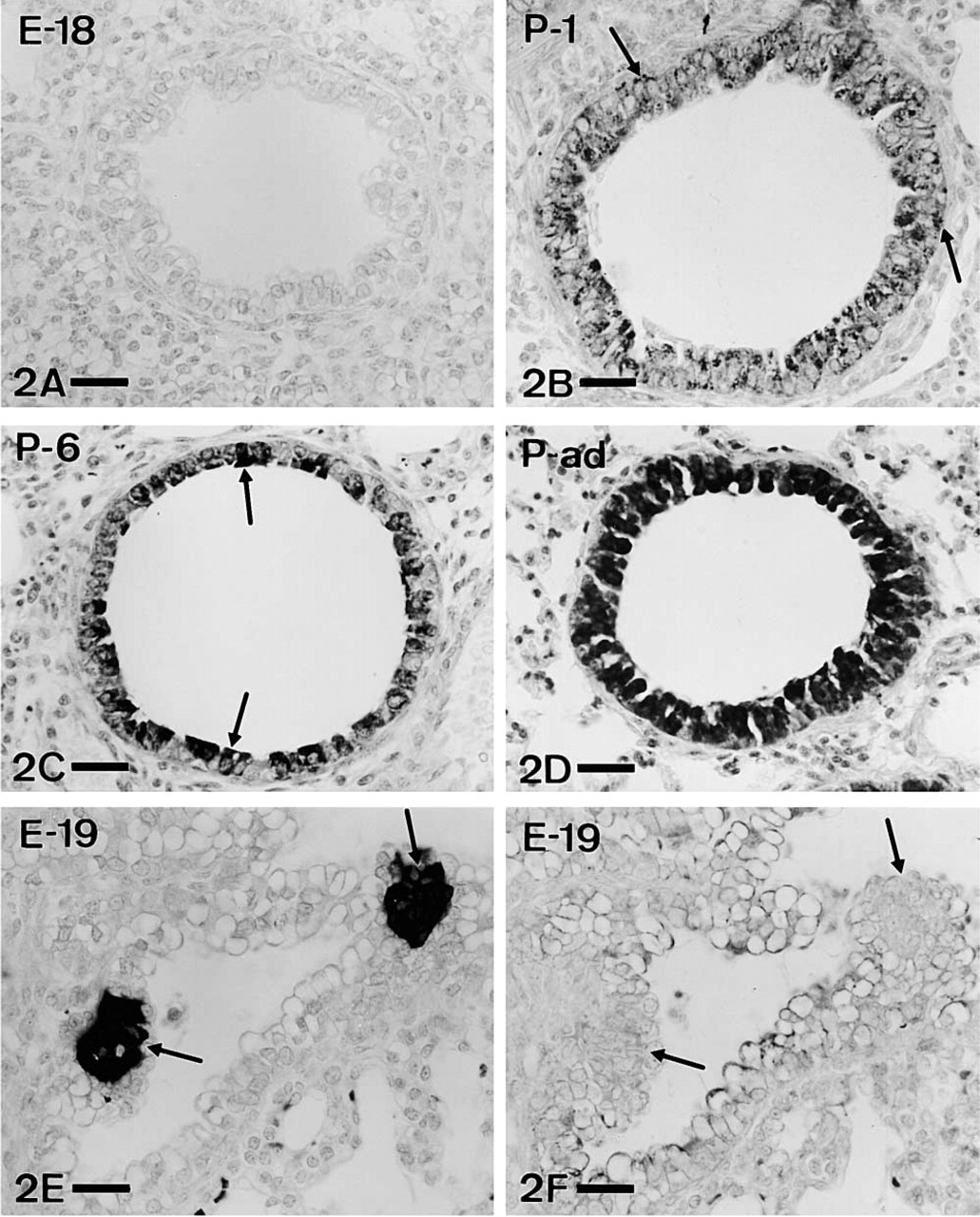
Appearance and evolution of ET-1 immunostaining in bronchiolar epithelium. (
In adult lungs, in addition to epithelial cells, ET-1-IR and big-ET-1-IR were obtained in part of the smooth muscle population of airway walls. Positive myocytes were especially numerous in bronchi (Figure 4A–4C). In blood vessels, some smooth muscle cells also exhibited immunostaining (Figure 4D and 4E), although usually with weaker intensity than in airways. Parenchymal cells of adult lungs also showed IR (Figures 4F). Comparing IR for ET and the marker of Type II pneumocytes (SP-C), a similar general distribution pattern was observed (Figure 4F and 4G), although no clear cell–cell correspondence was found. No IR in endothelial cells was found in any age with any of the antisera used.
Discussion
Little is known concerning the appearance and evolution of ET in mammalian lung development. In the present work, IR for ET-1 and big-ET-1 was detected in the airway epithelium of both developing and adult mouse lungs. IR was found in most epithelial cells (Clara cells). Conversely, IR in airway and vascular muscle and parenchymal cells was observed only in adults.
The perinatal role of ET is now being studied. ET could have both a constrictor or a dilator effect on pulmonary smooth muscle and appears to play a role in the maintenance of the bronchial and vasomotor tone both in gestation (Ivy et al. 1994) and after birth (Pinheiro and Malik 1993). Studies with knockout mice deficient in ET-1 showed that this peptide is essential for physiological control of ventilation (Kurihara et al. 1994; Kuwaki et al. 1996). Other effects of ET, such as proliferation of pulmonary epithelial cells, smooth muscle, and fibroblasts, have been described (Janakidevi et al. 1992; Peacock et al. 1992; Glassberg et al. 1994; Murlas et al. 1995). Therefore, ET could act in mouse lung as a growth factor during development, as suggested in other species (Giaid et al. 1991; Katsura et al. 1993).
In relation to the appearance and evolution of ET in mammalian lung there is no concordance in the data shown by different authors both in the same or in different species. In our study, ET-1-IR was demonstrated in most developing mouse airway epithelial cells, as reported in fetal rat (Katsura et al. 1993). Conversely, in developing human lungs, endocrine cells have been reported to be the cell type immunoreactive for ET (Giaid et al. 1991). Therefore, interspecific differences appear to exist in the epithelial cell type expressing ET in pulmonary development. Other differences found among the three species during development are time of ET detection, staging of maximal levels of ET, and distribution of ET in large and small airways. Comparing the relative gestational ages among mouse, rat, and human, it could be concluded that ET-IR appeared later in mouse lung (terminal sac stage) than in human and rat, both at the pseudoglandular stage of pulmonary development (Giaid et al. 1991; Katsura et al. 1993). In relation to changes of ET level expression, in mouse IR increased progressively, reaching a maximum in the mature lung, whereas in rat the strongest IR was detected at the pseudoglandular stage, disappearing after birth. Such differences, however, may not be so drastic because ET mRNA has been detected in rat at late gestation (MacCumber et al. 1989; Katsura et al. 1993). Finally, the early distribution of ET in airways differs between mouse (appearing first in bronchi and large bronchioles) and rat (detected first in small intrapulmonary airways; Katsura et al. 1993).
The presence in mouse lung of IR for big-ET-1, the precursor of ET, was an expected result. Nevertheless, big-ET-1-IR was detected later (newborns) than ET-1-IR (fetuses). Apart from the possible lower sensitivity of the antiserum against big-ET-1 used, this unexpected result could be due to differences in the activity of the ET-1 converting enzyme compared to the rate of big-ET-1 translation in different stages of development. In fact, in pig lung a greater activity for the converting enzyme in the early postnatal period has been suggested (Liben et al. 1993).
In the present work we found ET-1-IR in most airway epithelial cells of developing and adult mouse, confirming the data obtained by part of the authors in several mammalian species. Immunoreactive cells found in the present study have been clearly identified as Clara cells, as reported in mice and rats (Rozengurt et al. 1990; Nakanishi et al. 1999). This is also in agreement with the synthesis and secretion of ET-1 by guinea pig Clara cells in vitro (Laporte et al. 1995). Conversely, in the present study we never obtained ET-1-IR in endocrine CGRP-positive cells, although co-localization of both peptides was found in neonatal mouse (Seldeslagh and Lauweryns 1993). According to the present and previous results, intra- and interspecific differences in relation to the distribution of ET in airway epithelium appear to exist, as indicated for other peptides (Polak et al. 1993).
The finding that in mouse ET-IR was located in the apical region of airway epithelial cells suggests the secretion of ET towards the airway lumen in both developing and adult lungs. In fact, apical ET-IR is the only one present in the epithelium at the beginning, before extending towards the base. Apical secretion of ET would explain the presence of ET in the bronchoalveolar lavage (Aarnio et al. 1994; Shennib et al. 1995). On the other hand, studies with cultured guinea pig tracheal epithelial cells suggest that release of ET occurs predominantly in a basal direction (Noguchi et al. 1995). In this manner, ET could act through a paracrine mechanism on the airway smooth muscle, where ET receptors have been detected (Koseki et al. 1989). The ET-IR pattern found in mouse fetal airway epithelium, extending from the apical cytoplasm towards the base, appears to indicate that the basal secretion of ET enhances along development. A similar cytological pattern of IR has been found in the same cells for the amidating enzyme PAM (Guembe et al. 1999), although no clear relationship between both substances can be stated.
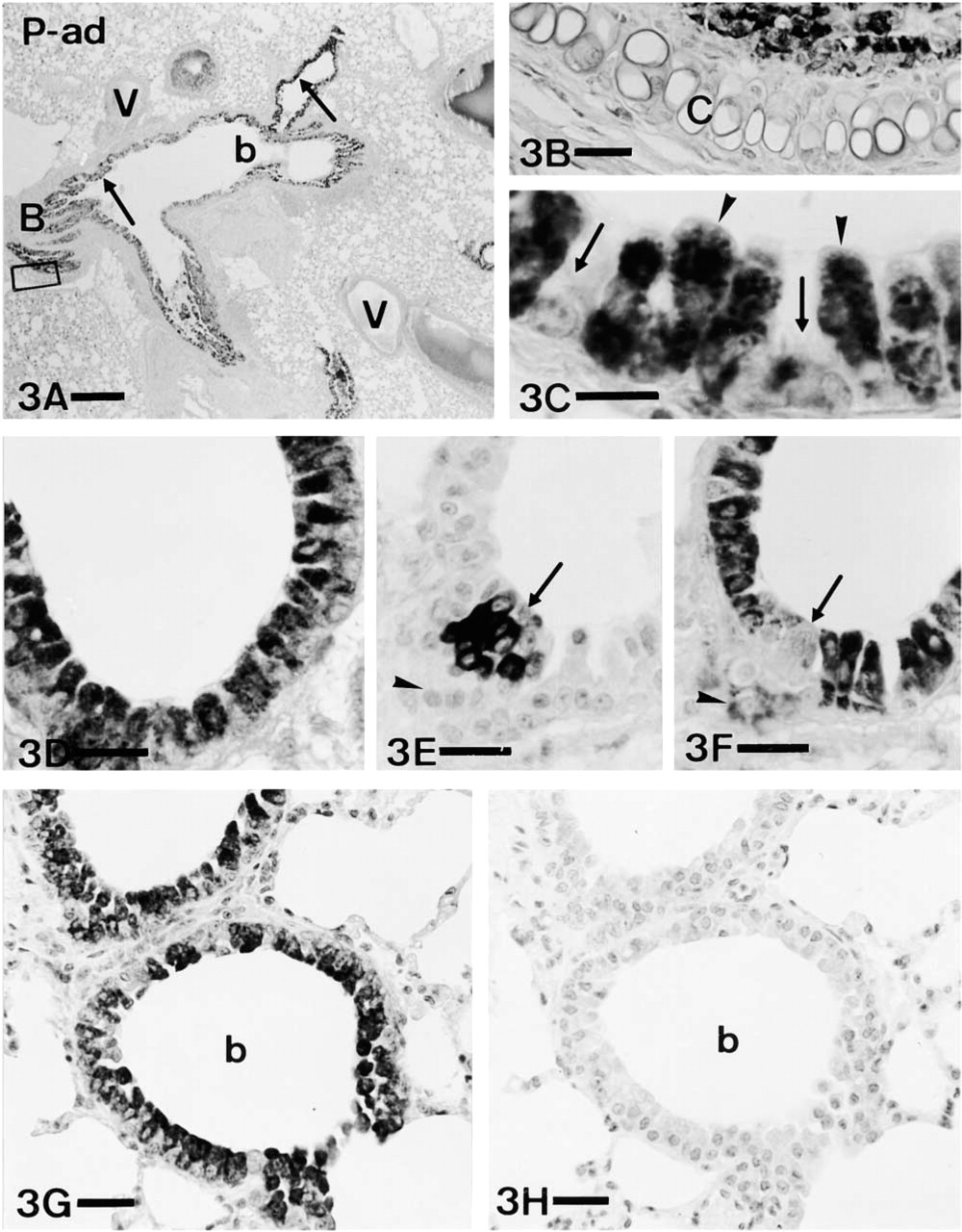
ET-1 in adult mouse airway epithelium. (
The presence of ET-1-IR in airway smooth muscle cells reported in the present work has been previously described in several mammalian species. Nevertheless, in mouse lung only some of the previous studies had reported ET-IR of smooth muscle (Seldeslagh and Lauweryns 1993). The production of ET-1 and the presence of ET-1 receptors in airway smooth muscle have been reported in vitro, suggesting an autocrine mechanism of action for ET-1 (Ergul et al. 1995).
As indicated, ET-IR was found in cells of pulmonary parenchyma and the distribution of these cells was similar to that of Type II pneumocytes. In agreement with this possibility, there are some reports on the expression of ET in this cell type (Rozengurt et al, 1990; Giaid et al. 1993; Crestani et al. 1998). The secretion of surfactant by Type II pneumocytes has been reported to be stimulated by ET (Sen et al. 1994). Therefore, ET could act through either an autocrine or a paracrine mechanism on Type II pneumocytes.
In summary, the appearance and gradual increase of IR for ET in mouse lung at late gestation and during the neonatal period in most airway epithelial cells supports the importance of ET in the perinatal period. According to our results, not only basal but also apical secretion of ET could take place both in development and in adulthood. Apical secretion could be present from the beginning, whereas basal secretion would appear later and appears to enhance along development. In addition to the reported control of ventilation in both development and adulthood, ET could also act as a growth factor in developing lung.
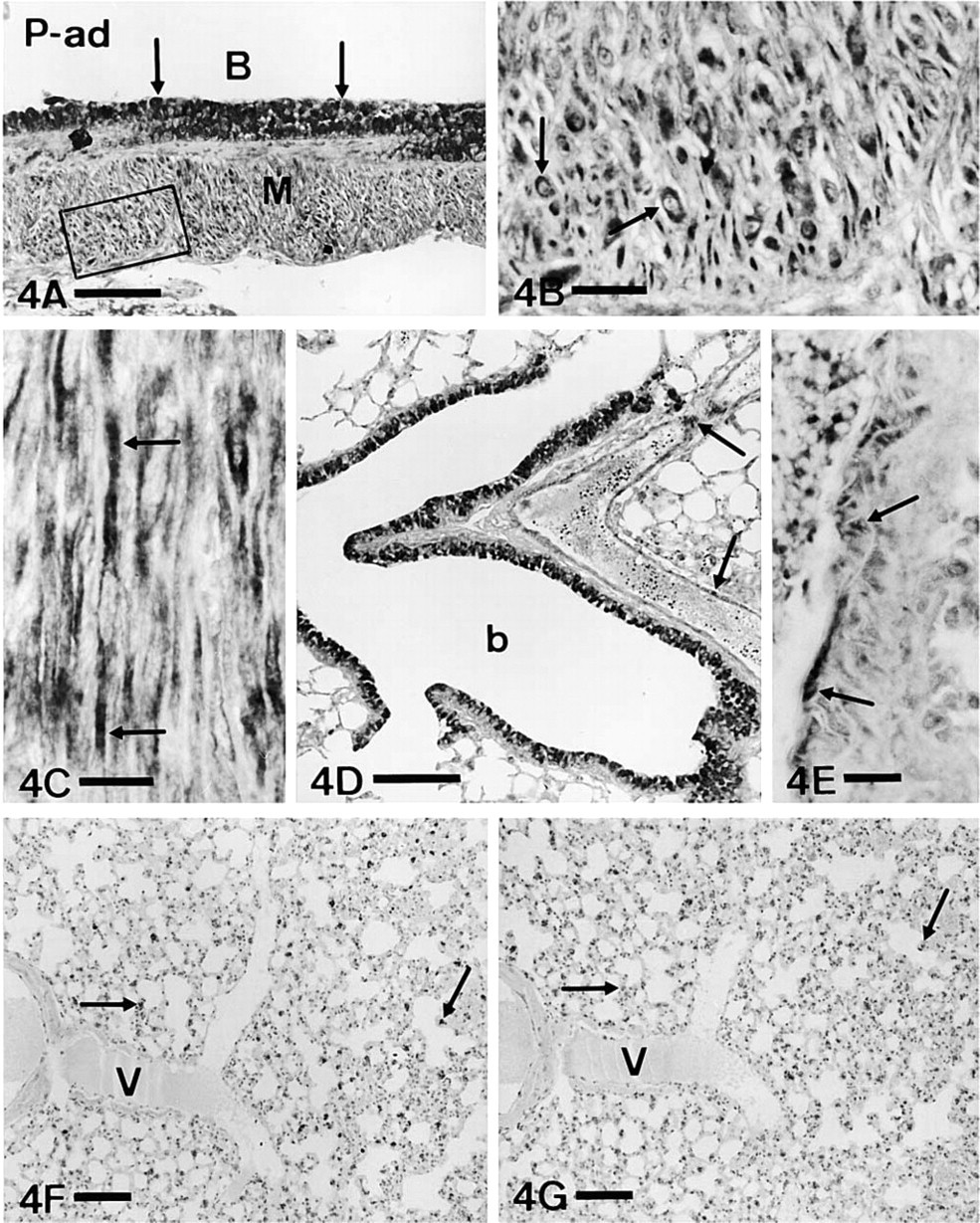
ET-1 immunoreactivity in smooth muscle and parenchyma. (
Footnotes
Acknowledgements
Supported by the Spanish Ministry of Education and Science (DGCYT project no. PB93-0711) and the University of Navarra (PIUNA), and by a grant from the Departamento de Educación y Cultura del Gobierno de Navarra, Spain (LG).
We would like to thank Prof J.M. Polak for the antisera against ET and CGRP and for critically reading the manuscript. We thank Dr Whitsett for the antiserum against SP-C. We thank I. Ordoqui and A. Urbiola for their technical assistance.
