Abstract
The reasons for the failure of clinical islet transplantation remain obscure. Islet isolation, however, exposes the islet to variety of cellular stresses, including disruption of the cell-matrix relationship, an event associated with apoptosis. The cell-matrix relationship is characterized by an interaction between cell surface integrin receptors and matrix molecules of the surrounding basement membrane (BM). The purpose of this study was to characterize integrin expression and the distribution of the peri-insular BM in human, porcine, canine, and hamster pancreas, and after routine islet isolation. Whereas islets in the porcine pancreas do not have a demonstrable BM, islets in the human, canine, and hamster pancreas have an almost continuous BM with very little direct exocrine to endocrine cell-cell contact. After islet isolation, the BM was destroyed, only to be reestablished during the period of culture. In the pancreas of all four species, integrin α3 was expressed only on islet cells, and integrin α5 was present on islet cells as well as on acinar, centroacinar, and duct cells. Integrin αV was detected only in human and canine pancreas. Integrin β1 was demonstrated only in the human pancreas. In isolated islets, integrin α3, α5, and αV expression decreased during the culture period and the intensity of the staining was observed to be coincident with the distribution of the BM. In summary, this is the first report of integrin expression in hamster, canine, porcine, and human islets. After islet isolation, the altered islet cell-matrix relationship is reflected both in the decrease in integrin expression and in the destruction of the peri-insular BM. These profound changes will need to be considered as the process of islet isolation for transplantation is refined.
I
Factors that mediate cell survival include those found in the local microenvironment, such as extracellular matrix (ECM), and those associated with the cell itself, the integrin family of cell surface proteins (Stoker et al. 1990). An important role of cell-matrix interactions in the formation of islet structure and the maintenance of islet integrity has been suggested by previous studies (Montesano et al. 1983; Lucas-Clerc et al. 1993). Therefore, the consequences of a complete cell-matrix disruption on islet cell survival would be profound.
ECM is present in two forms, interstitial matrix and basement membrane (BM). In the pancreas, BM contains laminin, fibronectin, and collagen Types IV and V (Ingber 1993). The specific role of these matrix molecules in the pancreas has yet to be determined, but ECM, and BM in particular, is crucial for cell survival and maintenance of the normal epithelial phenotype (Emerman and Pitelka 1977; Hay 1993).
Integrins, expressed on virtually every cell type, are a diverse class of αβ heterodimeric receptors through which cells interact with matrix proteins, providing the physical basis for cell adhesion (Juliano and Haskill 1993; Schwartz and Ingber 1994; Kantengwa et al. 1997; Longhurst and Jennings 1998). More importantly, integrins are authentic receptors that transduce an extracellular stimulus into a variety of intracellular signals (Damsky and Werb 1992; Richardson and Parsons 1995; Longhurst and Jennings 1998) which, in turn, regulate intracellular events, including differentiation, cell growth (Damsky and Werb 1992; Yurochko et al. 1992), and cell survival (Schwartz and Ingber 1994; Clark and Brugge 1995; King et al. 1997). In the pancreas, integrins have been studied in cancer and pancreatitis, but little is known of their expression and function in islets (Hall et al. 1991; Weinel et al. 1992; Levine et al. 1994; Shimoyama et al. 1995).
In this study we characterized the presence and distribution of the islet BM and the expression of integrins in normal human, porcine, canine, and hamster pancreas. We then compared the findings to those obtained from purified islets after isolation.
Materials and Methods
Sources of Pancreatic Tissue and Isolated Islets
Pancreata from female Syrian Golden hamsters (body weight 85–100 g; n = 3) and mongrel dogs of both sexes (2–4 years old, body weight 10–20 kg; n = 4) were excised under general anesthesia. All procedures were in compliance with Canadian Council for Animal Care standards and were approved by the institutional animal care committee. Pancreatic islets from porcine pancreas were obtained from Boehringer Mannheim Company (Indianapolis, IN) (n = 4) and porcine pancreata (n = 3) were a gift from Dr. Ray C.J. Chiu (Montreal General Hospital, Research institute, Canada). Human pancreata were procured from cadaveric donors (ages 25–60 years; n = 3). Prior consent for research use of these tissues was in accordance with the policies of Quebec-Transplant, the local organ procurement agency.
Chemicals and Antibodies
All chemicals for routine use were obtained from GIBCO (Gibco; Burlington, Ont, Canada). For islet isolation, Lib-erase and Liberase CI (Boehringer Mannheim) were used for both human and canine pancreata, and collagenase P was used for hamster pancreata (Boehringer Mannheim; Montreal, QC, Canada). For islet cell culture, CMRL1066 supplemented with 10% fetal bovine serum (FBS) was purchased from GIBCO. The following primary antibodies were used for immunocytochemical staining. Monoclonal anti-human integrin α1, α2, α3, β1, and β2 antibodies, and polyclonal anti-human integrin α5, α6, and αV antibodies were purchased from Chemicon International (Temecula, CA), monoclonal anti-human insulin antibody, polyclonal anti-human glucagon and somatostatin antibodies were obtained from Biogenex (San Ramon, CA), and monoclonal anti-human collagen IV and polyclonal anti-human laminin were purchased from Sigma (St Louis, MO). The optimal concentrations of all primary antibodies were preliminarily determined.
Islet Isolation and Culture
Human pancreata were removed from cadaveric donors after an in situ flush with UW solution (Dupont Pharma; Montreal, QC, Canada). Canine pancreata were removed from anesthetized dogs without the use of an in situ preservation solution. Islets were separated from the surrounding exocrine tissue by enzymatic digestion with Liberase [2.5 mg/ml Liberase in Hanks' balanced salt solution (HBSS)] supplemented with 0.1 mg/ml DNase I (Boehringer Mannheim) at 37C using a semiautomated technique described by Ricordi et al. (1988). Purification was achieved by density gradient separation in a three-step discontinuous EuroFicoll gradient using a COBE 2991 Cell Processor (COBE BCT; Denver, CO). The final preparation usually consists of 70–90% dithizon-positive tissue fragments with diameters of 50–500 μm.
Collagenase P was used for the digestion of hamster pancreata. Purification was carried out by a two-step discontinuous bovine serum albumin (BSA) gradient (Sigma), as previously described (Metrakos et al. 1992). Islets were collected and washed with minimal essential medium (MEM), and the final step included the hand-picking of islets.
After overnight incubation, islets were cultured in suspension in CMRL1066 medium supplemented with 10% FBS at 37C in a CO2 incubator (95% air/5% CO2). Porcine islets isolated at the Indianapolis facility were shipped the same day, by overnight courier, in serum-supplemented CMRL1066. Islets selected at random during the digestion period, before and after purification, and then at 2-day intervals for at least 10 days were fixed in 4% paraformaldehyde (PFA) overnight at 4C.
Reticulin Staining
The reticular fibers of the basement membrane were stained by silver impregnation techniques (Culling 1974). Deparaffinized sections were oxidized in acidified potassium permanganate, bleached in 1% oxalic acid, and mordanted in iron alum. Each step was rinsed in water. The sections were treated with silver solution until the section became transparent. After counterstaining, the reticulin fibers appeared black.
Immunocytochemistry
The fixed islet samples from each time point were embedded in 2% agarose according to a standardized protocol of dehydration and paraffin embedding. Sets of 10 serial sections (thickness 4 μm) were cut from the paraffin blocks. Consecutive sections of pancreata or isolated islets were immunostained for integrins, pancreatic hormones, and matrix proteins, using the avidin-biotin (AB) complex method (streptavidin-biotin-horseradish peroxidase complex; Dako, Glostrup, Denmark), as described previously (Wang et al. 1994). For integrin α3 and collagen IV, a pretreatment with either 0.1% trypsin or 0.1% protease was used. The sections were incubated overnight at 4C with the appropriate primary antibodies. Bound antibodies were visualized with the ABC immunoperoxidase system using biotinylated goat anti-mouse and goat anti-rabbit immunoglobulin G (IgG) (Amersham; Poole, UK), and developed with DAB (3,3′-diaminobenzidinetetrahydrochloride; Sigma) as chromogen. For negative controls, the primary antibodies were omitted.
A quantitative evaluation of the number of cells stained was performed. Three randomly selected sections of the first 10 islets from each species were used for counting, and at least 200 acinar cells, duct cells, or isolated islet cells were counted. Data are expressed as mean percentage ± SD.
Results
Expression of Basement Membrane Components
The human (Figure 1A), canine (Figure 1D), and hamster (Figure 1G) pancreas each showed the presence of a continuous peri-insular BM. In the porcine pancreas, the BM around islets was either discontinuous or absent (Figure 2A). Within the islet, BM was observed only around capillaries.
The BM of isolated islets was lost immediately after enzymatic digestion (Figures 1B, 1E, and 1H). After 5 days of culture, the BM was gradually recovered (Figures 1C, 1F, and 1I). Unlike the in situ findings, isolated porcine islets developed a continuous BM within 5 days of culture (Figure 2C).
Immunocytochemical localization of laminin and collagen IV demonstrated that they were components of the peri-insular BM membrane in the pancreas, although staining was of very low or only moderate intensity. Laminin and collagen IV were destroyed during the islet isolation process and were absent immediately after isolation. Immunocytochemical staining, however, returned by Day 5 in culture.
Integrin Expression
The data on integrin expression are summarized in Table 1. The presence of β2, α1, and α6 was not detected in this study (not shown). Integrin β1 was strongly positive in human acinar cells, much less so in human islet cells, and negative in all other species of pancreas, except for duct cells of the porcine pancreas. Some isolated canine and porcine islets demonstrated positive staining, but those cells appeared to be associated with ducts. Islet cells from all four species were positive for integrin α3 (Figure 3). Integrin α2 was positive only in porcine islets. Interestingly, the intensity of integrin α2 immunoreactivity in isolated porcine islets appeared to increase from Day 1 to Day 5, but the staining was then lost at Day 9. Integrin α5 was strongly expressed in islet, epithelial, and endothelial cells (Figure 4). In the human and hamster pancreas, staining for integrin α5 was positive in acinar cells (Figure 4A), whereas in the canine and porcine pancreas, positive staining was obtained only for the centroacinar cells. Human and canine pancreas also showed strong staining for integrin αV (Figure 5), but negative results were found in porcine and hamster pancreas. There was positive integrin α5 and αV staining in isolated islet cells of all four species, which was coincident with the distribution of the BM at Day 5 of culture (Figures 4C and 5C).
The correlation of integrin expression and pancreatic hormones was determined by immunocytochemical staining in consecutive sections (Figure 3). Cells in isolated islets were positive for integrin α3, α5, and αV. The staining was strong the day after islet isolation, but then gradually decreased in intensity during the period of culture (Figures 3C, 4C, and 5C). A similar decrease in staining was also observed for insulin (Figure 3D), glucagon, and somatostatin (data not shown).
Discussion
This is the first report of integrin expression on islet cells in the hamster, canine, porcine, and human pancreas. This comparative study, in which islets from four commonly used species were examined before and after islet isolation, is unique and contributes new and important information to the field of islet cell biology. We also show that the peri-insular basement membrane is destroyed during the process of islet isolation.
The only previous report of integrin expression in the human pancreas is that by Hall et al. (1991). Our characterization of integrin expression, however, is not entirely in agreement with their findings, primarily because they failed to demonstrate any integrin expression on islet cells. The absence of integrin expression on islets appears highly unlikely (Kantengwa et al. 1997), and any differences can be attributed to several factors, including tissue handling and the specific antibodies employed in the respective studies.
The pattern of integrin expression on acinar, duct, and islet cells in the normal pancreas of the four different species was similar but not identical. Moreover, integrin expression also varied among cell types in each species. This observation is interesting from an ontogenetic viewpoint, given that the duct cell is presumed to give rise to both islet and acinar cells (Pictet et al. 1972). This observation is important because these differences could be used to identify specific cell types in studies of cell differentiation, particularly in vitro.
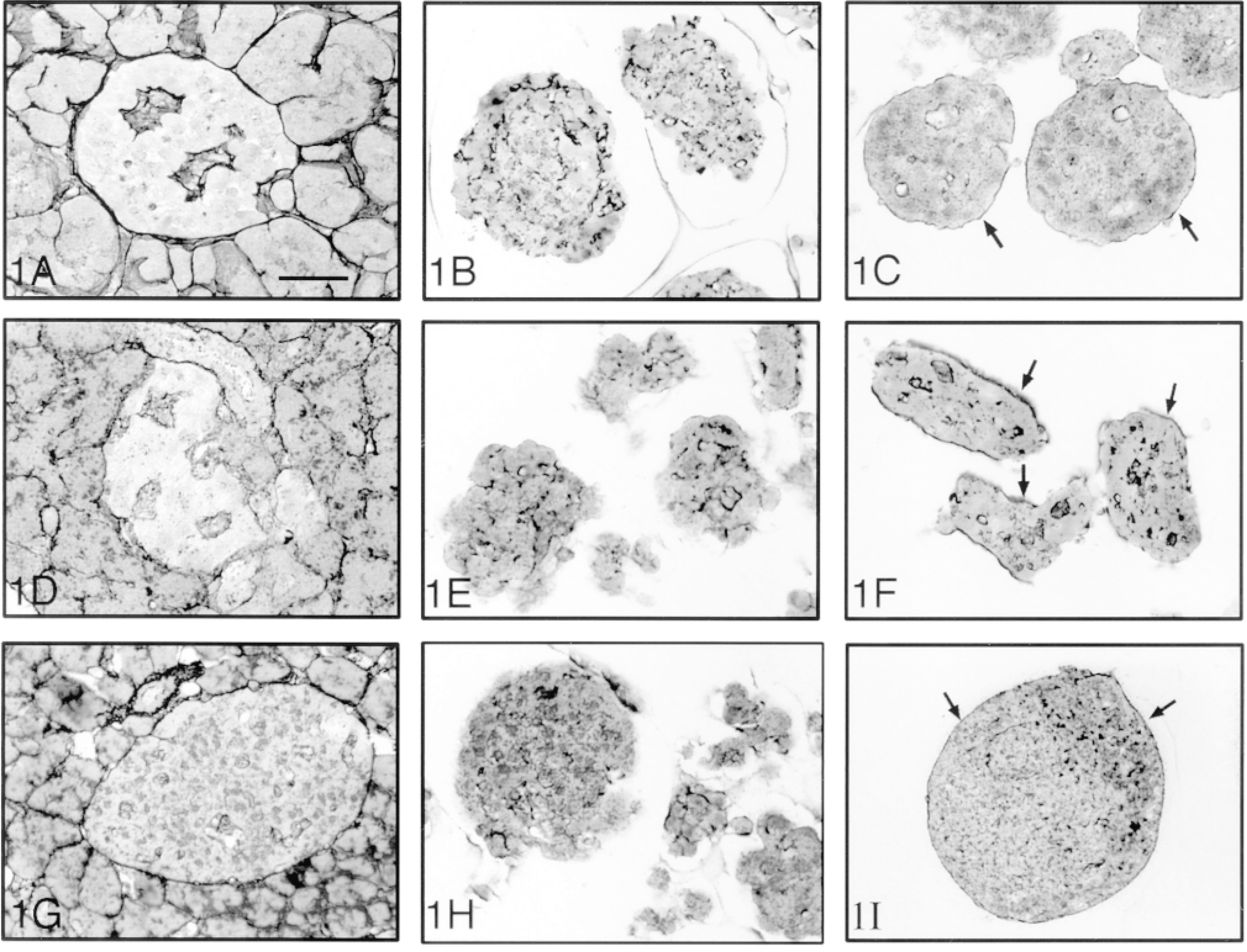
Reticulin staining in human (A-C), canine (D-F) and hamster (G-I) pancreas, and of isolated islets on Day 1 (B,E,H) and Day 5 (C,F,I) of culture. An intact basement membrane, demonstrated around the islet while still in the pancreas, was lost immediately after isolation and then recovered again at Day 5 (arrows). Bar = 28 μm.
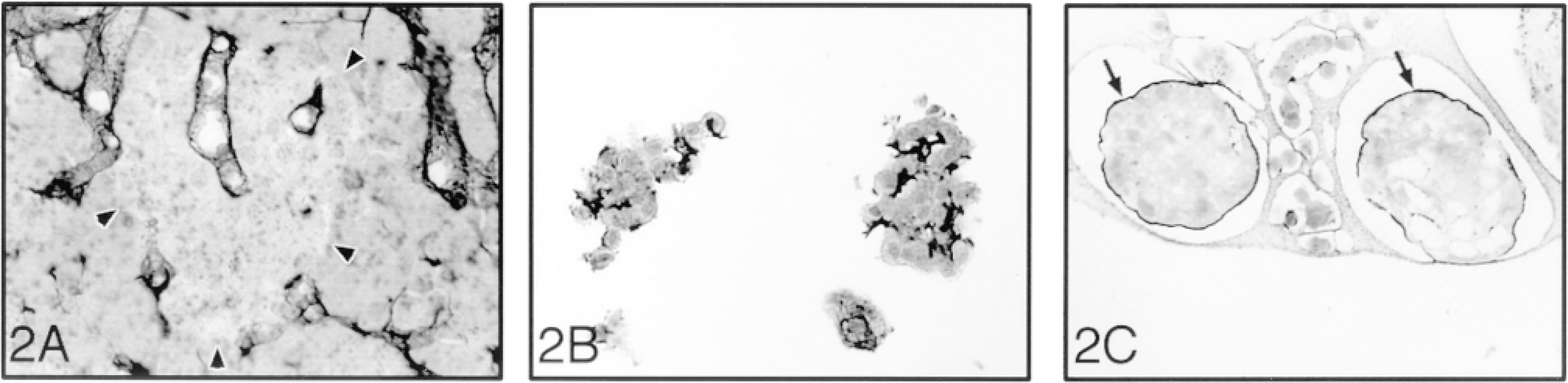
Reticulin staining in the porcine pancreas (A) and of isolated islets on Day 1 (B) and Day 5 (C) of culture. The islet basement membrane is absent in the porcine pancreas (arrowheads). After 5 days of culture, an intact BM was observed around the islets (arrows). Bar = 28 μm.
The role of integrin family members in the regulation of apoptosis has recently been recognized, based, in part, on their capacity to trigger discrete intracellular phosphorylation events and to initiate gene transcription (Bates et al. 1995). After islet isolation, the expression of specific integrin subtypes was found to be either unchanged or diminished. In particular, the expression of integrin α5 appeared to be reduced in all four species. This finding may be of considerable importance in view of a recent report that linked integrin α5β1 to support of cell survival through the upregulation of the expression of bcl-2, an important regulatory protein that inhibits apoptosis (Zhang et al. 1995). In addition, the absence of integrin-mediated signals may contribute to the activation of the stress-activated protein kinase (SAPK) JNK, whose downstream effectors are also associated with the induction of apoptosis (Meredith and Schwartz 1997). In this context, it is relevant that we have recently confirmed that islet cell apoptosis rapidly ensues after islet isolation (Paraskevas et al. 1997a) and that nuclear and DNA fragmentation is preceded by the activation of JNK and another SAPK, p38 (Paraskevas et al. 1997b,c). Therefore, the structural alterations that occur in integrin expression as a result of islet isolation appear to have important functional consequences for cell survival.
Characterization of integrin expression in the pancreas and isolated islets a
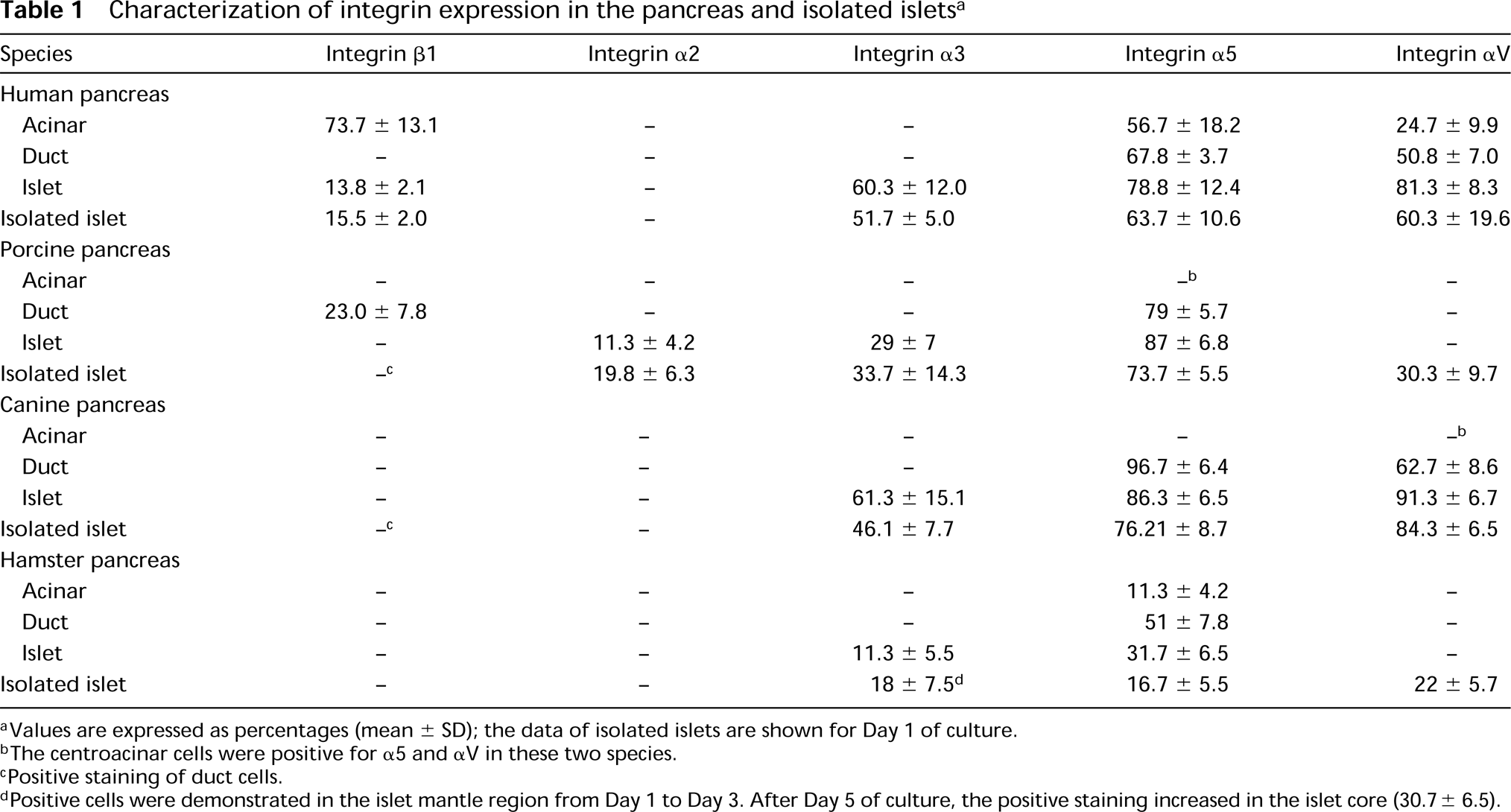
aValues are expressed as percentages (mean ± SD); the data of isolated islets are shown for Day 1 of culture.
bThe centroacinar cells were positive for α5 and αV in these two species.
cPositive staining of duct cells.
dPositive cells were demonstrated in the islet mantle region from Day 1 to Day 3. After Day 5 of culture, the positive staining increased in the islet core (30.7 ± 6.5).
The second morphological finding of the present study was the loss of the peri-insular BM as a result of the isolation process. BM is one of the two forms of extracellular matrix (ECM), the other being the interstitial matrix (Ingber 1993). ECM components have long been recognized as adhesive and support structures (Schnaper and Kleinman 1993). However, the ECM also plays a significant role in regulating the behavior of cells. Matrix proteins engender changes in cell shape and movement, bind growth factors, and facilitate cell-cell and cell-matrix interactions.
The composition and distribution of pancreatic ECM, and in particular the peri-insular BM, in rat, dog, pig, and human, have been reported by van Deijnen et al. (1993). In the pig, very little if any peri-insular capsule is present, and the structural integration of the porcine islet in the exocrine pancreas almost exclusively depends on cell-cell adhesion. In the canine and human pancreas the islets are almost completely encapsulated, with very little direct exocrineto-endocrine cell-cell contact. We examined the peri-insular BM in the pancreas and after islet isolation using both a reticulin stain and an immunocytochemical technique for specific identification of laminin and collagen IV. The two methods gave comparable results, and the findings in pancreatic tissue before islet isolation are in keeping with those reported by van Deijnen et al. (1993).
Although a variable loss of the peri-insular BM has also been reported by other investigators (Gray and Leow 1992), the effect of this on islet cell survival has not been previously considered. Degradation of ECM is known to be an early and critical event in organ regression in several systems (Meredith et al. 1993). Local disruption of ECM results in apoptosis in adjacent cells, whereas survival is promoted by allowing cells to adhere (Frisch and Francis 1994; Chen et al. 1997). It is not only the presence of ECM that is important, for it is well established that cell survival actually requires integrin-mediated adhesion to matrix proteins (Ruoslahti and Reed 1994). In fact, when islets are cultured either on or within solid matrices, improved islet survival and function are observed (Lucas-Clerc et al. 1993; Brendel et al. 1994; Metrakos et al. 1994).
ECM also plays an essential role in maintenance of cell differentiation (Lin and Bissell 1993). The disruption of the cell-matrix relationship may therefore be responsible, at least in part (Boukamp and Fusenig 1993; Hisaoka et al. 1993), for the loss of differentiated islet structure and function recently reported by us (Yuan et al. 1996) and others (Davalli et al. 1995; Kerr-Conte et al. 1996).
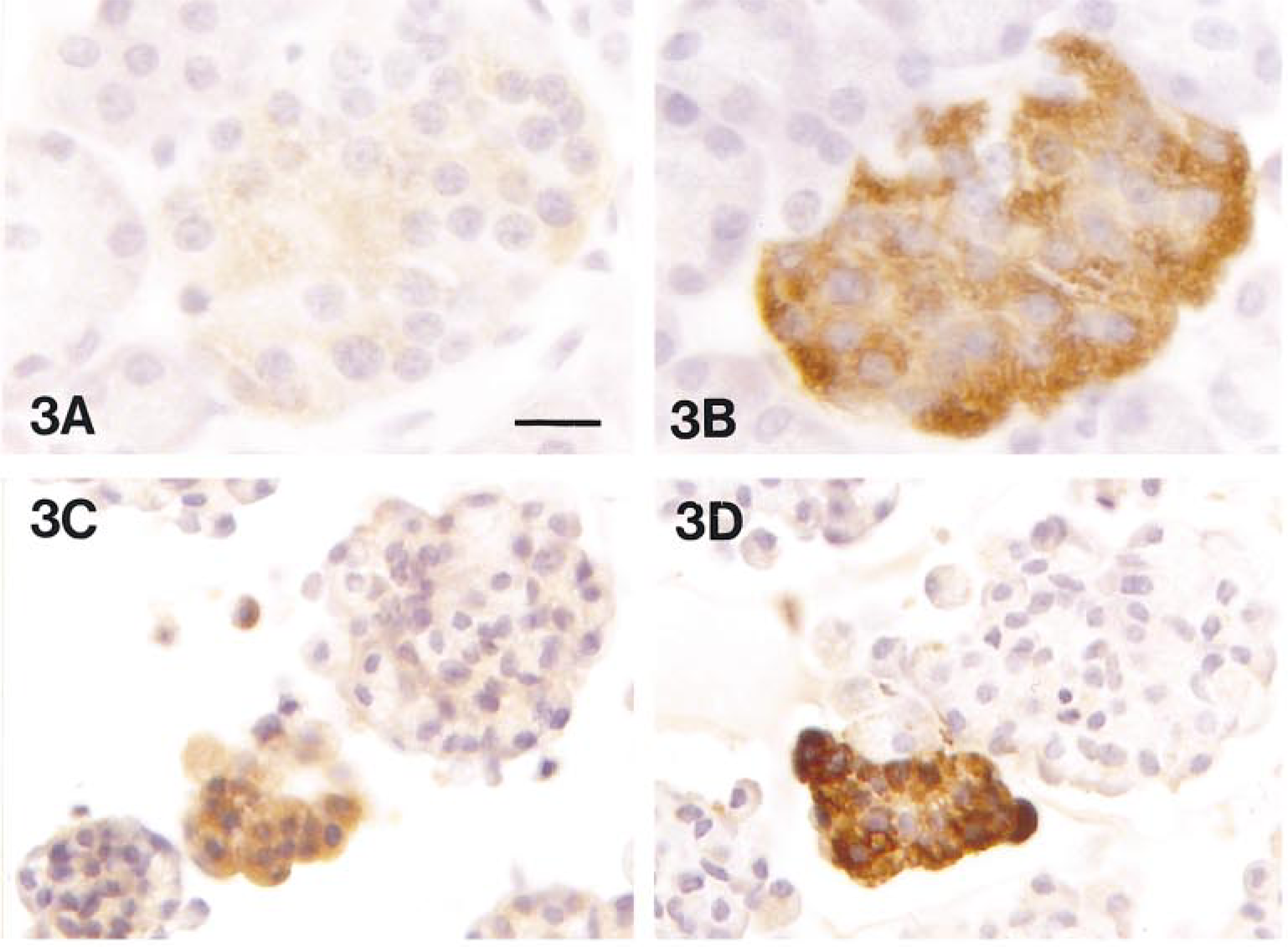
Consecutive sections stained by immunocytochemistry for α3 (A,C) and insulin (B,D) in normal human pancreas (A,B) and in isolated islets (C,D). The intensity of integrin α3 staining paralleled that of insulin staining both in situ and in isolated islets. Bars: A,B = 18 μm; C,D = 22 μm.
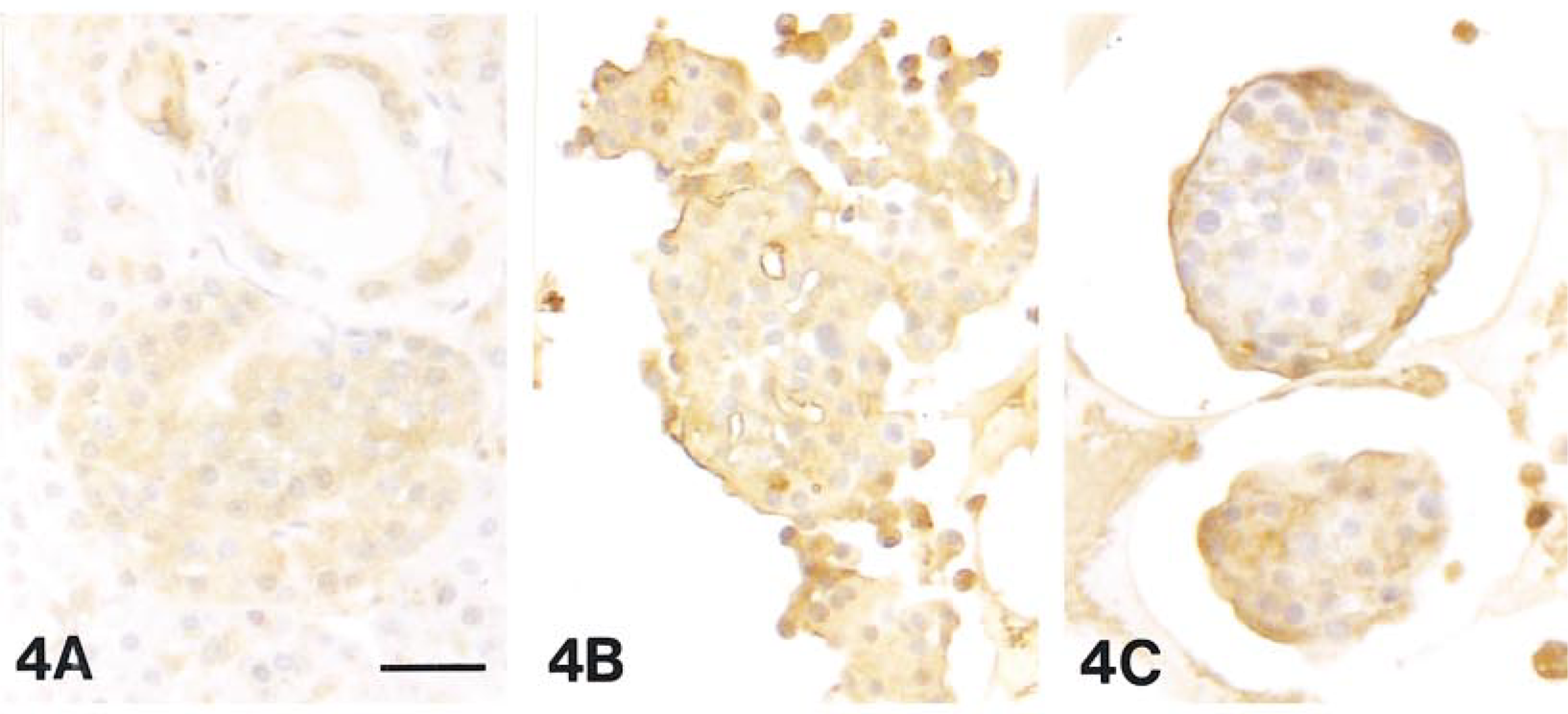
Immunocytochemical staining for α5 in the human pancreas (A) and in isolated islets on Day 1 (B) and Day 5 (C) of culture. The intensity of integrin α5 staining declined after the period of culture. Bars: A = 28 μm; B,C = 22 μm.
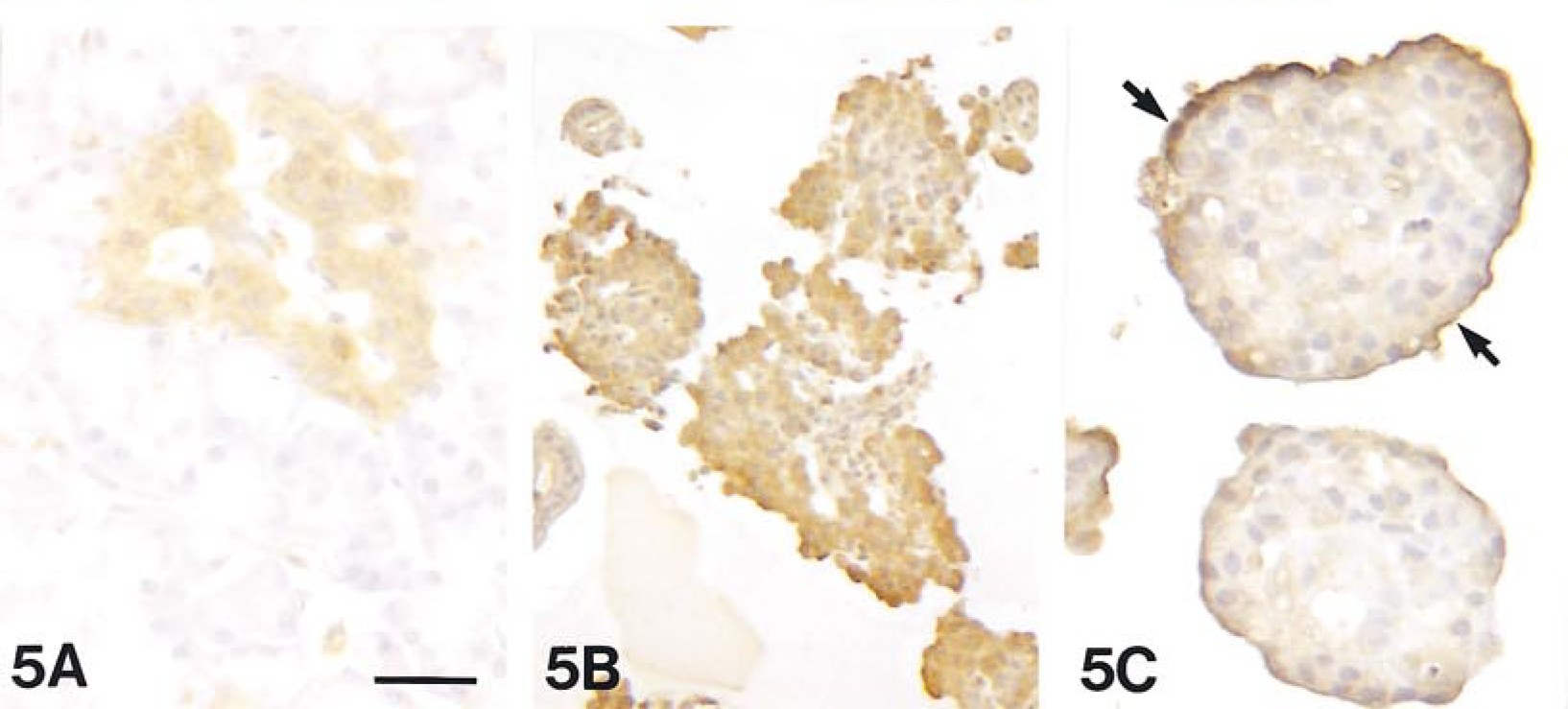
Immunocytochemical staining for αV in the canine pancreas (A) and in isolated islets on Day 1 (B) and Day 5 (C) of culture. Strong staining was observed primarily in the mantle region of islets at Day 5 (arrows). Bars: A,C = 28 μm; B = 45 μm.
In summary, we have characterized the integrin expression and distribution of the peri-insular BM in the human, porcine, canine, and hamster pancreas. We show that, after islet isolation, the BM is destroyed and integrin expression is altered. The resulting cell-matrix disruption could have profound implications for normal islet function and survival, both before and after islet transplantation. These alterations in normal islet structure and function offer a new explanation for the difference in the success of vascularized pancreas grafts compared to isolated islet transplants. If these preliminary findings are confirmed, then new strategies that take advantage of pharmacological manipulation of the cell-matrix relationship may offer a valuable new approach to improving the outcome of islet transplantation.
Footnotes
Acknowledgements
Supported by grants from the Sam Soloman Fund of the Canadian Diabetes Association (CDA) and the Medical Research Council (MRC) of Canada. Dr. Wang is supported by a fellowship from the Canadian Diabetes Association in honor of Herbert L. and Francis Nussbaum. Dr. Rosenberg is a senior clinician-scientist of the Fond de la Recherche Scientifique du Quebec (FRSQ).
We thank D. Agapitos, N. Malek, and A. Torrisi for expert technical assistance.
