Abstract
The aim of this study was to investigate the distribution pattern of Ca2+ - and Mg2+ -dependent ecto-ATPases on the surface of rat brain capillary endothelial cells (ECs) in control and lipopolysaccharide (LPS)-treated animals. Ecto-ATPases in the membrane of vascular endothelial cells are suggested to play a crucial role in thromboregulation. Loss of this enzyme activity after oxidative stress and upregulation of the enzyme chain hydrolyzing extracellular ATP after transient forebrain ischemia have also been reported. We used histochemistry to localize the activities of this enzyme on ECs and found pH- and cation-dependent changes in the localization of enzyme activity both in control and in LPS-treated animals. These findings suggest the presence of more than one ecto-ATPase enzyme on the surface of rat capillary ECs. The different behavior of ECs after LPS treatment is the target of further investigations. The increased ecto-nucleotidase activity might play a role in nucleotide-mediated cellular responses after bacterial infections.
E
The source of nucleotides and their release mechanisms have been studied mostly in the vascular and nervous systems. Nucleotides are released into the bloodstream by damaged cells in the vessel wall (Born and Kratzer 1984) or by platelets during thrombus formation (Mills et al. 1968). The dense granules in blood platelets contain approximately 1 M of ATP and ADP (Gordon 1986), which is released by exocytosis during platelet activation and aggregation (Mills et al. 1968). Vascular endothelial cells release ATP via an undetermined mechanism (Pearson and Gordon 1979) after stimulation with trypsin, thrombin, extracellular calcium, or norepinephrine (Pearson and Gordon 1979, 1985; Milner et al. 1990a,b; Yang et al. 1994; Hashimoto et al. 1995). Mechanical forces, such as osmotic shock or shear stress, applied to cells also lead to the release of intracellular ATP (Petruzzi et al. 1994; Grierson and Meldolesi 1995). The enzyme activity is stimulated by a number of divalent cations, of which Ca2+ and Mg2+ are the most relevant ones (Plesner 1995; Nagy et al. 1997).
Ecto-ATPases are related to several different proteins (Dzhandzhugazyan and Bock 1993,1997; Handa and Guidotti 1996; Vasconcelos et al. 1996). The most studied ecto-ATPase is a membrane-bound 95-kD glycoprotein, named CD39 and originally identified on activated human B lymphocytes (Kansas et al. 1991; Maliszewski et al. 1994; Handa and Guidotti 1996; Wang and Guidotti 1996). The cDNA of ecto-ATPase from rat brain shows high similarity to the mouse and human CD39 cDNA, and is identical to the sequence of rat CD39 (Kegel et al. 1997). Ecto-ATPase from chicken gizzard smooth muscle and from rat brain shows a high homology to CD39 (Kirley 1997; Kegel et al. 1997). Marcus and colleagues (1997) found identical regions in the sequence of endothelial cell ADPase and CD39. This enzyme, which possesses both ATPase and ADPase activities, may have a pivotal role in thromboregulation (Marcus et al. 1991,1997; Marcus and Safier 1993; Kaczmarek et al. 1996; Wang and Guidotti 1996). Its soluble form is a potential therapeutic agent for inhibition of platelet-mediated thrombotic diseases (Gayle et al. 1998).
In this study, we used an enzyme histochemical method at the electron microscopic level, which is appropriate for localizing putative ecto-ATPase enzyme activity. We investigated the effect of lipopolysaccharide (LPS) on capillary endothelial cells (ECs). This bacterial endotoxin has potent proinflammatory properties toward many cell types, including ECs (de Vries et al. 1996; Lu et al. 1996; Spolarics et al. 1996; Yang et al. 1996; Defazio et al. 1997; Bourke et al. 1998). We wanted to know whether there is a difference in the cation-dependent enzyme activity pattern after LPS treatment and using fixatives with different pHs. This would indicate the presence of different ATPase enzymes. In the presence of Ca2+ or Mg2+, pH- and cation-dependent changes occurred in the localization of enzyme activities after LPS treatment. These results suggest the presence of several ecto-ATPases on the surface of capillary ECs, presumably with different physiological roles.
Materials and Methods
All of the general reagents were purchased from Sigma Chemical, (St Louis, MO) unless otherwise specified. This study was carried out in accordance with the guidelines of the Institutional Ethical Committee for Experimental Animals.
LPS Treatment
Male Wistar rats (about 200-250 g) were injected with LPS (2 mg/kg body weight) IP 2 hr before sacrifice (Kovacs and Sawchenko 1996; Spolarics et al. 1996).
Electron microscopic demonstration of Ca2+ - or Mg2+ -dependent ecto-ATPases at pH 7.5 was as described by Kittel et al. (1996). Control and LPS-stressed rats were anesthetized with Nembutal (Sanofi Philaxia; Budapest, Hungary), then perfused briefly through the ascending aorta with a 0.05 M Na-cacodylate buffer containing 0.25 M sucrose, 2 mM CaCl2, followed by a mixture of ice-cold 3% paraformaldehyde (Merck; Darmstadt, Germany) and 0.5% glutaraldehyde (Taab; Aldermaston, Berks, UK), 0.25 M sucrose in 0.05 M cacodylate buffer (pH 7.4) for 30 min. The brains were quickly removed and 2-3-mm-thick tissue blocks from the cerebrum were postfixed in the same solution for 15 min. Vibratome sections 50 μm thick were cut and washed in cacodylate buffer with 0.25 M sucrose (pH 7.4) for 45 min. Then the sections were incubated in a medium containing Tris-maleate buffer (70 mM, pH 7.4), ATP (1 mM), CeCl3 (2 mM), MnCl2 (5 mM), 1 mM levamisole (inhibitor of alkaline phosphatases) (Amersham; Poole, UK), 1 mM ouabain (Na+,K+ -ATPase inhibitor; Merck), and 50 μM α,β-methylene-ADP (5′-nucleotidase inhibitor), as well as CaCl2 (1.5 mM) and EDTA (0.1 mM) or MgCl2 (1.5 mM) and EGTA (0.1 mM) for 45 min at 37C. Incubation was followed by three rinses in Tris-maleate buffer. The samples were postfixed in 1% OsO4 (Taab) dissolved in cacodylate buffer for 30 min. After rinses in distilled water, tissue slices were dehydrated in graded ethanol (en bloc-stained with 2% uranyl acetate in 70% ethanol for 30 min), processed through propylene oxide, and embedded in Epon (Fluka; Buchs, Switzerland). Ultrathin sections were cut and examined in a Hitachi 2001 transmission electron microscope (Hitachi; Tokyo, Japan). To demonstrate the specificity of the reaction product, in some experiments either the ATP or the ions were omitted from the incubation medium. Control experiments were carried out in the presence of both cations at pH 7.5 and pH 9.5.
Demonstration of Ca2+ - or Mg2+ -dependent Ecto-ATPases After Fixation at pH 9.5
The procedure was the same as above except for the washing and the fixative solutions. We used a 0.9% NaCl solution for rinsing the vessels. The fixative solution contained 4% para-formaldehyde, 0.1 M NaOH, and 0.1 M Na2B4O7-10 H2O at pH 9.5. After fixation and washing, the sectioning was carried out in Tris-maleate buffer.
Results were obtained from five different experiments with parallel sampling for both fixation procedures.
Results
To demonstrate the Ca2+ - and Mg2+ -dependence of ecto-ATPDase activity in the capillary ECs in rat brain cortex, two fixative solutions were used at pH 7.5 and 9.5. The ecto-enzyme activity was represented as a Ce-phosphate deposit at the site of liberation of phosphate during the hydrolysis of extracellular ATP or, as the next step of the enzyme activity, from ADP. As the control experiments demonstrate, when Ca2+ and Mg2+ were used together, the ecto-ATPase activity was very strong on both (luminal and basal) surfaces of the ECs. This activity was independent of the pH of the fixative solution. There was also some activity in the extracellular matrix (ECM) (Figures 1a and 1b).
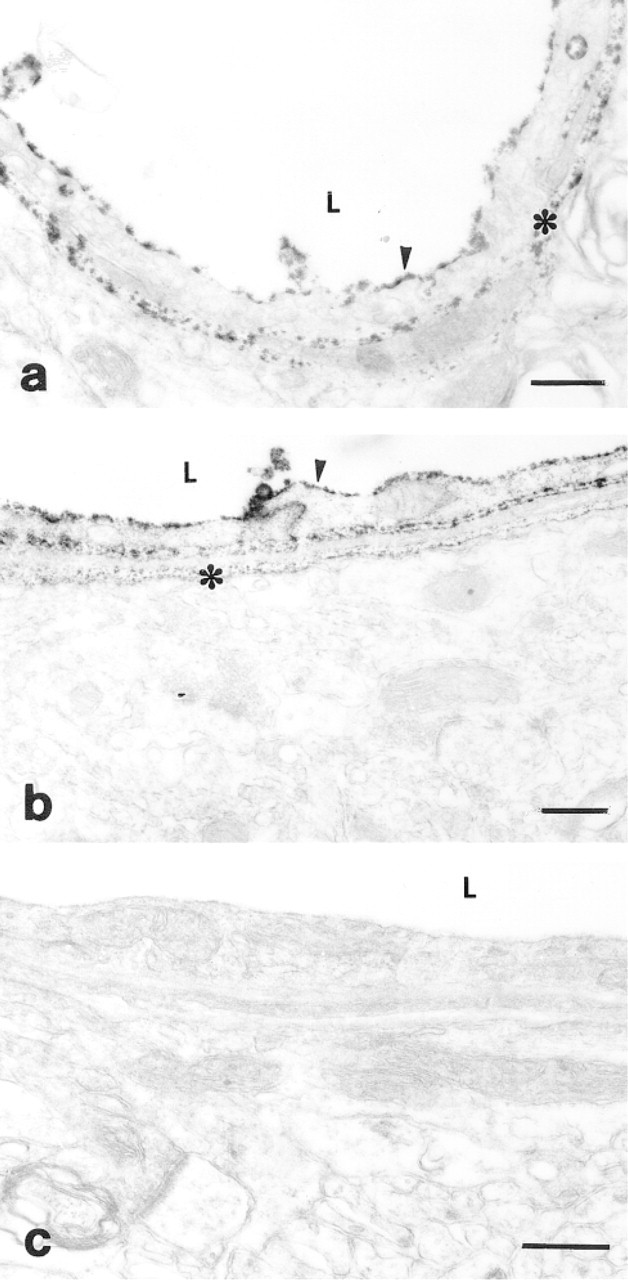
Electron micrographs of the ecto-ATPase activity. The enzyme is visualized as a Ce-phosphate deposit on the surface of capillary endothelial cells of the rat brain cortex auditory area.
Control experiments without substrate ATP or Ca2+ /Mg2+ did not show any deposit around the cells or on the cell membrane (Figure 1c).
After fixation at pH 7.5 and when the incubation medium contained only Ca2+ as cation, a fine cerium deposit was found both on the luminal and basal sides of the cell and in the ECM, as in the case when both cations were used (Figure 2a). However, if Mg2+ replaced Ca2+ in the incubation medium at pH 7.5 fixation, the activity on the luminal side was absent or reduced (Figure 2b). Enzyme activity did not change in the ECM.
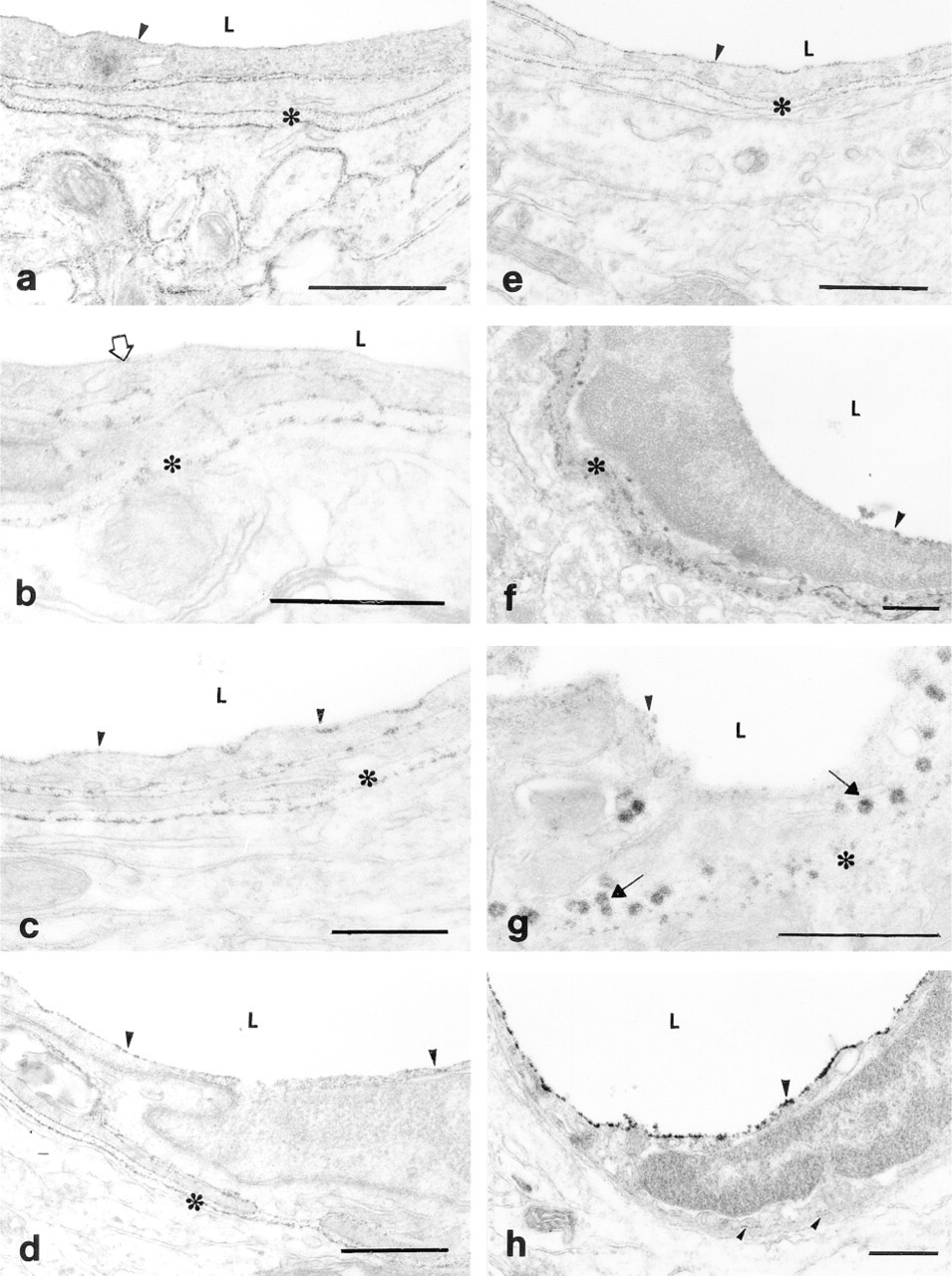
Ecto-ATPase activity in the rat brain cortex capillary endothelial cells at pH 7.5 (a-d). (a) Ca2+ -dependent ecto-ATPase activity. Fine Ce-phosphate deposit is seen on both apical (luminal) and basal sides of a capillary endothelial cell. There is also some deposit in the ECM. (b) Mg2+ -dependent ecto-ATPase activity. Fine Ce-phosphate deposit shows the ecto-ATPase activity on the basal side of an endothelial cell and in the ECM. The enzyme activity is absent or very weak on the luminal side. (c) After LPS treatment, Ca2+ -dependent ecto-ATPDase activity is similar to control conditions. (d) After LPS administration, Mg2+ -dependent ecto-ATPDase activity also appeared on the luminal surface of a capillary endothelial cell. Ecto-ATPase activity on endothelial cells after fixation at pH 9.5 (e-h). (e) Ca2+ -dependent ecto-ATPase activity is located on both sides of the endothelial cells. (f) Mg2+ -dependent e-ATPase activity is comparable to Ca2+ -dependent activity. (g) After LPS administration, strongly stained caveola-like membrane structures appeared on the basal side of the endothelial cell, showing intensive ecto-ATPase activity. (h) After LPS treatment, significantly increased levels of Mg2+ -dependent ecto-ATPase activity are seen on the luminal surface of the endothelial cell and reduced activity on the basal side. Arrowheads, Ce deposits on the luminal membrane surface, indicating ecto-ATPase activity; open arrow, reduced amount or absence of deposit; arrow, caveola labeled with the Ce-phosphate reaction product of ecto-ATPase; asterisks, ecto-ATPase activity in the extracellular matrix; L, capillary lumen. Bars = 0.5 μm.
At pH 7.5 fixation, LPS treatment did not change the pattern in the presence of Ca2+ (Figure 2c). However, in the presence of Mg2+ some ecto-ATPase activity appeared on the luminal side, whereas the activity on the basal side remained at the same level (Figure 2d).
After fixation at pH 9.5, we observed only a slight difference in the appearance of ecto-ATPase activity, using either Ca2+ or Mg2+ in control animals. Some enzyme activity was present on the luminal and basal sides of the ECs (Figures 2e and 2f). This slight difference concerns the deposit on the luminal side of ECs using Mg2+ only (Figure 2f), which was not visible with fixation at pH 7.5.
LPS treatment after fixation at pH 9.5 changed the pattern of the enzyme activity with both cations used individually. With Ca2+ only, the enzyme activity of the basal side of the cell increased, especially in the pinocytotic vesicles. The presence of these caveola-like membrane structures in considerable numbers was observable after LPS treatment and in the presence of Ca2+ only (Figure 2g). If Mg2+ was used in the incubation solution, an abundant Ce deposit showed the increased ecto-enzyme activity on the luminal surface of the ECs. Staining on the basal side was observable only in trace amounts (Figure 2h).
Discussion
The distribution of the Ca- and Mg-dependent ecto-ATPases on rat brain capillary ECs was studied in control and LPS-treated animals. Cation preferences of ATPases showed region-dependence as previously suggested (Nagy et al. 1997; Wang and Guidotti 1998). We used different pHs for fixation because the biochemical studies reported ecto-ATPase activity at different pHs (Plesner 1995), but the exact regional distribution patterns of these data were not available.
Effects of LPS have been published on several enzymes such as Na+ -K+ -ATPase, Ca2+ -ATPase, GTPase and nitric oxide synthase (Kang 1990; Tanke et al. 1991; Lu et al. 1996; Liu and Sheu 1997). In some studies, ECs were used. In this work, the effect of LPS on ecto-ATPase activity on capillary ECs of the rat brain auditory area was investigated. During inflammation or vascular damage, ecto-nucleotidases can play an important role in neuroprotective processes by generating adenosine from ATP, released from damaged cells. This enzyme histochemical study demonstrates the presence of ion- and pH-dependent ecto-ATPases on the surface of capillary ECs. In control animals, when both cations were used in the incubation medium, the localization pattern of the ecto-ATPase did not show any difference dependent on the pH of the fixation. Ecto-ATPase activity was found on the luminal and basal surfaces of ECs and in the ECM. If only one cation was used, a different picture was observed in the presence of Mg2+ after fixation at pH 7.5. There was a reduced amount of deposits on the luminal surface, but the enzyme activity on the basal side and in the ECM did not change. The activity found in ECM showed the same characteristics under these experimental conditions as a membrane-bound ecto-ATPase. It may demonstrate the presence of a type of soluble ecto-ATPase. Similarly, a soluble ecto-ATPase (from Tetrahymena thermophila) related to the membrane-bound ecto-ATPase of smooth muscle has been described by Smith and colleagues (1997). However, we cannot exclude the possibility that this enzyme is a soluble apyrase.
Significant alterations occurred in the localization of the ecto-ATPase after LPS administration and fixation at pH 9.5. (Alternatively, when fixation was done at pH 7.5, the treatment with LPS did not induce visible changes.) With only Ca2+ in the incubation medium, many caveola-like membrane structures full of deposit indicating strong ecto-ATPase activity appeared on the basal side, whereas the activity on the luminal surface did not change. What might be the role of these membrane invaginations in the capillary ECs during inflammation? Caveolae, dynamic pieces of membrane, were first identified in ECs. They are suggested to take part in signal transduction and to play a major role in coordinating the interaction of the cell with its environment (Anderson 1993). Their appearance after/ during administration of an inflammatory agent and their high ecto-ATPase activity support this hypothesis. In contrast to the findings with Ca2+ -dependent ecto-enzyme, in the case of Mg2+ in the incubation medium, increased enzyme activity appeared on the smooth luminal surface of the ECs. This increased activity was not associated with caveolae. These findings indicate the presence of at least two different ecto-ATPases with different localization patterns. Although ecto-ATPases are active in the presence of both Ca2+ and Mg2+, they might have some ion-dependent “territorial dominance” on the EC surface in addition to their different sensitivity for pH and stress by injury or inflammation. Recently, several studies have appeared on the loss of ecto-ATPase activity on vascular ECs after injury or oxidative stress (Candinas et al. 1996; Koyamada et al. 1996; Robson et al. 1997; Gayle et al. 1998). The loss of ecto-ATPase activity may result in platelet aggregation, thrombosis, and xenograft rejection. However, there is also evidence of the upregulation of the enzyme chain hydrolyzing extracellular ATP after transient ischemia (Braun et al. 1998). The present findings support the possibility of induction of increased ecto-ATPase activity after inflammation or injury. The increased enzyme activity might play a significant role in the cellular responses after inflammation. Our findings suggest that the cellular responses to inflammation are twofold: (a) the increased Mg-dependent ecto-ATPase activity on the luminal surface increases the ATP-hydrolyzing capacity, and (b) the increased Ca-dependent ecto-ATPase activity in the caveolae may help in proper coordination of the endothelial cell with its environment.
Footnotes
Acknowledgements
Supported by a grant (OTKA) 019860 from the Hungarian Scientific Research Fund.
I wish to thank Drs E. Bacsy, S.C. Robson, and K.J. Kovacs for critical review of the manuscript and Ms A. Foldes, K. Lengyel, and O. Szalay for excellent assistance.
