Abstract
In situ hybridization (ISH) has proved to be an invaluable molecular tool in research and diagnosis to visualize nucleic acids in their cellular environment. However, its applicability can be limited by its restricted detection sensitivity. During the past 10 years, several strategies have been developed to improve the threshold levels of nucleic acid detection in situ by amplification of either target nucleic acid sequences before ISH (e.g., in situ PCR) or the detection signals after the hybridization procedures. Here we outline the principles of tyramide signal amplification using the catalyzed reporter deposition (CARD) technique, present practical suggestions to efficiently enhance the sensitivity of ISH with CARD, and discuss some applications and possible future directions of in situ nucleic acid detection using such an amplification strategy.
Keywords
I
In recent years, therefore, several strategies have been developed to amplify the nucleic acid targets (target amplification) or (immuno)cytochemical detection signals (signal amplification) in situ (Table 1). In addition, increased absolute amounts of hybridized probes were used successfully to improve the ISH detection sensitivity, e.g., cocktails of oligonucleotides or multiple cRNA probes (Trembleau and Bloom 1995; Komminoth 1996).
Approaches to amplify nucleic acid target sequence and (immuno) cytochemical detection signals in situ (for review see Komminoth and Werner 1997).

Target Amplification
In general, target amplification techniques combine PCR and ISH to visualize specific amplified DNA and RNA sequences in cell and tissue preparations. In theory, these in situ PCR techniques are straightforward and include sample fixation and pretreatment that allows the primers, nucleotides, and polymerase enzymes to enter the cell but avoids the loss of the generated amplificants, PCR amplification in the cell, and direct or indirect (by ISH) detection of the amplificants. The practical procedure, however, is associated with several obstacles, such as low amplification efficiency (restricted sensitivity), poor reproducibility (restricted specificity), and difficulties in quantification of the results (Höfler 1993; Komminoth and Long 1993). These findings are caused by a number of events as a consequence of PCR amplification in situ, such as the inevitable diffusion of PCR products, inextricably bound up with the denaturation steps, from the site of synthesis inside and/or outside the cells, followed by the possible extracellular generation of amplificants. In addition, the final results of direct in situ PCR can be influenced by incorporation of labeled nucleotides into nonspecific PCR products and the generation of nonspecific PCR products resulting from mispriming, from fragmented DNA undergoing “repair” by DNA polymerase (“repair” artifacts), or from priming of nonspecific DNA or cDNA fragments (“endogenous priming” artifacts). These kinds of artifacts can also be observed in apoptotic cells or samples that have been pretreated with DNase before in situ RT-PCR for mRNA detection (Sällström et al. 1993; Long and Komminoth 1997). Therefore, it has been recommended to use a multitude of different controls to allow adequate interpretation of in situ PCR results and, in addition, to use ISH preferably after PCR (= indirect in situ PCR) to check or increase the specificity of the amplified nucleic acids (Long and Komminoth 1997).
In situ PCR must therefore still be considered a rather cumbersome ISH method, in which sample pretreatment consists of fixation and protease digestion in combination with heating (thermal cycling) during nucleic acid amplification (by PCR). Moreover, the increase in detection sensitivity compared with conventional ISH is rather limited, even after optimization of the procedures (Mee et al. 1997), and cell morphology and nucleic acid localization are often poor. Furthermore, the question arises of whether the increase in the final ISH signal intensity is really caused by the in situ amplification of target sequences alone or is rather the result of heating the specimen by thermal cycling, because it has been shown that microwaving or thermal cycling without in situ PCR can also result in increased sensitivity of ISH (Sperry et al. 1996; Pilling et al. 1997). In this respect, we would like to report a similar effect localizing human centromere 9-specific DNA in routinely fixed, paraffin-embedded tissue sections, in which it was possible to perform a primed in situ labeling reaction with a chromosome 9-specific primer with positive outcome only after pepsin digestion in combination with thermal cycling in buffer without PCR reagents (unpublished results).
In summary, initial enthusiasm has now changed into a more realistic and sometimes skeptical attitude about the practical potential of in situ PCR in research and clinical studies. As a consequence, in situ signal amplification approaches have been more and more explored during the past couple of years (Table 1), of which the CARD signal amplification method appears to be the most promising.
CARD Signal Amplification
CARD signal amplification was introduced by Bobrow et al. (1989,1991,1992) for use in immunoblotting and ELISA assays and is based on the deposition of a large number of haptenized tyramine molecules by peroxidase activity (Figure 1). Tyramine is a phenolic compound and horseradish peroxidase (HRP) can catalyze the dimerization of such compounds when they are present at high concentrations, probably by the generation of free radicals (Zaitsu and Ohkura 1980). If applied in lower concentrations, such as those used in the signal amplification reaction, the probability of dimerization is reduced, whereas the binding of the highly reactive intermediates to electron-rich moieties of proteins, such as tyrosine, at or near the site of the peroxidase binding site is favored. In this way, a lot of extra hapten molecules can be introduced at the hybridization site in situ. Visualization of deposited tyramides can be performed either directly after the CARD reaction with fluorescence microscopy, if fluorochromelabeled tyramides are used, or indirectly with either fluorescence or brightfield microscopy, if biotin, digoxigenin, or di- or trinitrophenyl are used as haptens, which can act as further binding sites for anti-hapten antibodies or (strept)avidin conjugates (in the case of biotinylated tyramides) (Speel et al. 1995b). In addition, fluorescein and rhodamine can be used as haptens, because specific antibodies against these fluorochromes are commercially available from several companies.
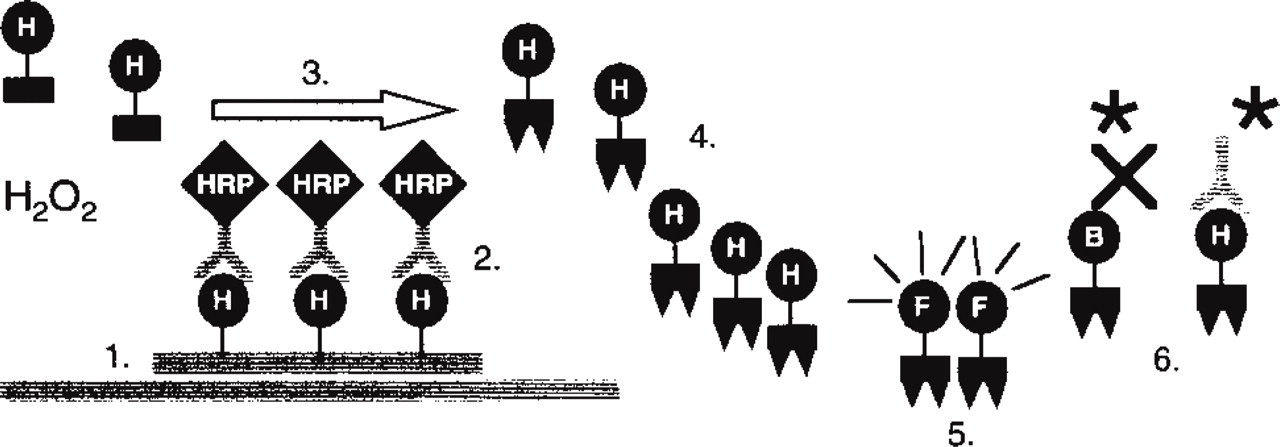
CARD signal amplification for ISH. 1. Hybridization in situ with a hapten (H = biotin, digoxigenin, dinitrophenyl, fluorochrome)-labeled probe. 2. Application of an anti-hapten antibody conjugated to horseradish peroxidase (HRP). 3. CARD signal amplification with hapten-labeled tyramine and H2O2. 4. Deposition of tyramide radicals to tyrosine moieties of proteins in situ in the vicinity of the site of synthesis. 5. Direct visualization of fluorochrome-labeled tyramines (Hapten H = fluorochrome F). 6. Indirect visualization of biotin-labeled or hapten-labeled tyramides with, respectively, (strept)avidin or anti-hapten antibody conjugates labeled with fluorochromes or enzymes (∗). Enzyme activity can be visualized utilizing enzyme precipitation reactions (Speel et al. 1995).
Applications of In Situ CARD Signal Amplification
CARD signal amplification with biotinylated tyramides has been easily adapted for immunohistochemistry by Adams (1992), allowing an increase in sensitivity of up to 1000-fold compared with conventional avidin-biotinylated enzyme complex (ABC) procedures (Adams 1992; Berghorn et al. 1994; Merz et al. 1995; Sanno et al. 1996; Werner et al. 1996; Von Wasielewski et al. 1997). In these studies, the amplification factor was assessed by performing serial dilutions of the primary antibody. In most cases, however, the increase in sensitivity seems to be in the range of five- to 50-fold, and sometimes even less. As a consequence of this variability, the optimal dilution for every antibody needs to be determined. CARD signal amplification has also been applied to visualize antigens or incorporated BrdU in fluorescence microscopy (Chao et al. 1996; Van Gijlswijk et al. 1997; Van Heusden et al. 1997) or electron microscopy (Mayer and Bendayan 1997), and has further been used in double stainings with two unconjugated primary antisera raised in the same species (Shindler and Roth 1996), where one antibody could be applied at a concentration below the detection limit of fluorescently labeled secondary antibodies (used to detect the second antibody), but still sufficient for detection with CARD.
Since 1995, CARD has further been implemented in detection procedures for both DNA and RNA ISH on cell preparations and tissue sections. With signal amplification, the ISH sensitivity could be improved in the range of two- to 100-fold, enabling the detection of (a) repetitive and single-copy (up to the level of 1-5 KB) DNA sequences in cell preparations, (b) up to three different DNA sequences (repetitive as well as single-copy) simultaneously in cell preparations (Speel et al. 1997; Van Gijlswijk et al. 1997; Hopman et al. 1998a), (c) low- and single-copy human papillomaviruses and other microorganisms in cell and tissue preparations, and (d) rRNA and mRNA ranging from high to low abundance in cell and tissue preparations (Table 2).
Examples of studies applying CARD signal amplification to in situ hybridization
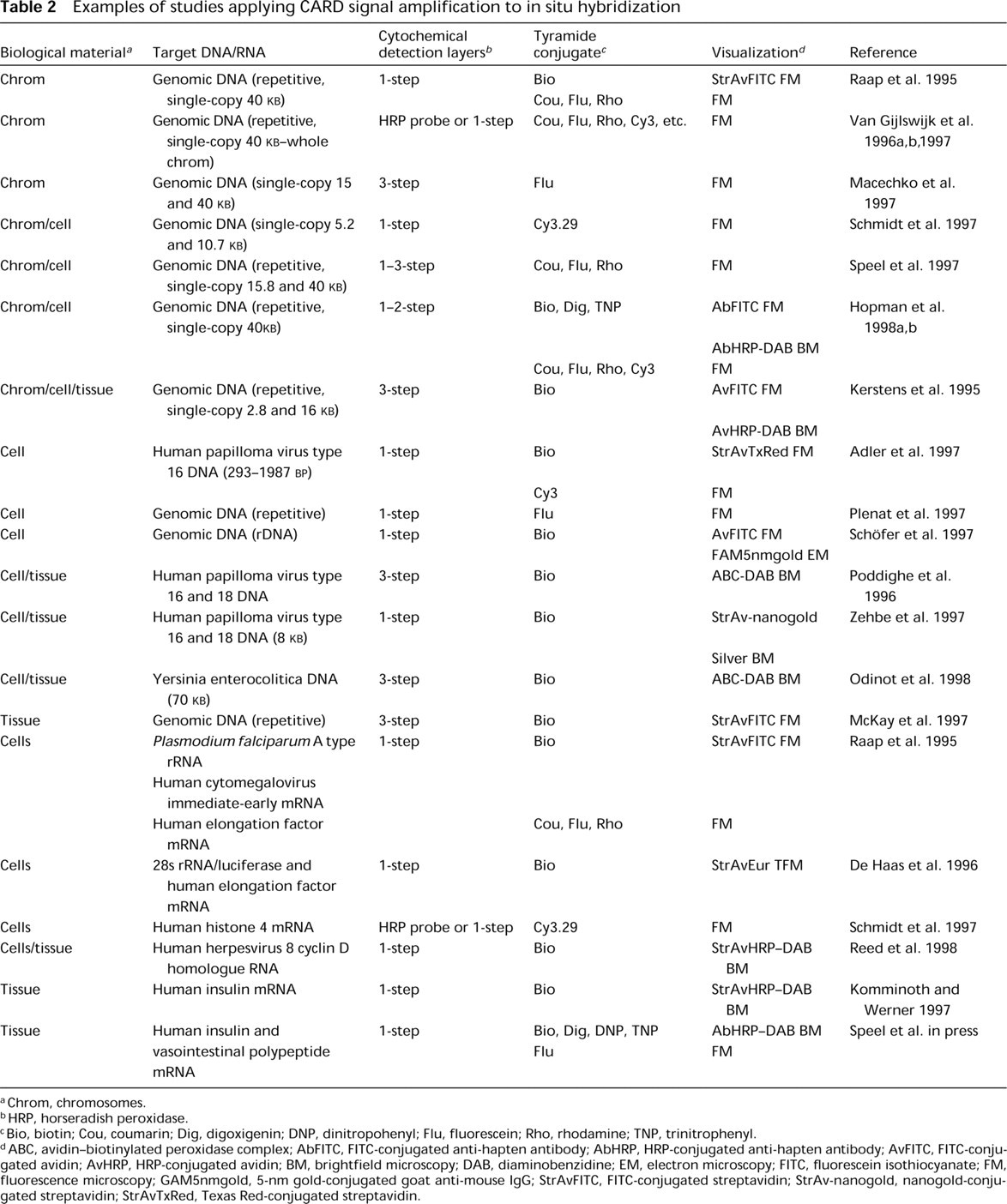
Chrom, chromosomes.
HRP, horseradish peroxidase.
Bio, biotin; Cou, coumarin; Dig, digoxigenin; DNP, dinitropohenyl; Flu, fluorescein; Rho, rhodamine; TNP, trinitrophenyl.
ABC, avidin-biotinylated peroxidase complex; AbFITC, FITC-conjugated anti-hapten antibody; AbHRP, HRP-conjugated anti-hapten antibody; AvFITC, FITC-conjugated avidin; AvHRP, HRP-conjugated avidin; BM, brightfield microscopy; DAB, diaminobenzidine; EM, electron microscopy; FITC, fluorescein isothiocyanate; FM, fluorescence microscopy; GAM5nmgold, 5-nm gold-conjugated goat anti-mouse IgG; StrAvFITC, FITC-conjugated streptavidin; StrAv-nanogold, nanogold-conjugated streptavidin; StrAvTxRed, Texas Red-conjugated streptavidin.
Practical Suggestions for ISH Using CARD Signal Amplification
The great advantage of using CARD signal amplification in ISH procedures is that it is applied after probe hybridization and stringent washings, so that the specificity of the probe hybridization is warranted. Therefore, in principle, proper conditions for sample fixation and pretreatment (including endogenous peroxidase inactivation) as well as hybridization in situ do not need to be modified compared with conventional ISH procedures. Only if nonspecific probe binding to the samples is suspected on the basis of control experiments should the probe concentration be further modified (decreased) for optimal results, e.g., in cases where complex DNA probes are used (e.g., chromosome painting, YAC, P1 or cosmid probes containing repetitive elements that need to be blocked by competitor DNA, such as Cot-I DNA) and an optimal balance is needed between probe and competitor DNA (Speel et al. 1997; Van Gijlswijk et al. 1997).
With respect to cytochemical probe detection, closer examination of the literature as well as Table 2 shows that a number of different combinations of probe detection (one to three detection conjugate layers) and CARD signal amplification systems (utilizing different tyramides, amplification buffers, reaction times, and temperatures) were applied, and that optimization of each detection system was necessary to obtain a high signal-to-noise ratio. Because with CARD signal amplification both specific and nonspecific (background) ISH signals are greatly amplified, it is essential for nonspecific probe binding (see above) and detection to be avoided or kept at a minimum to apply this procedure successfully. Therefore, we recommend routine optimization of probe hybridization (if not already done for setting up conventional ISH), cytochemical probe detection, and CARD signal amplification for one's own experiment. For this purpose, an appropriate one- to three-step detection system should be selected and combined with either a commercially available signal amplification kit (available from NEN Life Science Products (Boston, MA) as Tyramide Signal Amplification (TSA) kits or from Dako (Glostrup, Denmark) as Genpoint kit used according to the manufacturers' instructions, or CARD signal amplification using hapten- or fluorochromelabeled tyramides diluted in an appropriate buffer, such as PBS buffer at pH 7.6 containing 0.1 M imidazole and 0.001% H2O2 (Speel et al. 1997, and in press; Hopman et al. 1998b). In our hands, discretely localized ISH signals of high intensity can be obtained by adjusting the number of cytochemical detection layers [e.g., one layer is sufficient for repetitive (centromeric) DNA detection but a minimum of two layers are recommended for DNA targets of 40 KB or less), the dilution of detection conjugates (usually the first detection layer can be diluted two- to tenfold further than in conventional detection systems), the tyramide concentration in the CARD amplification buffer (usually concentrations in the range of 2-12 μM are used (Hopman et al. 1998b)], and the reaction time (usually 5-15 min at room temperature or 37C). Also the use of viscosity-increasing polymers such as dextran sulfate or polyvinyl alcohol has been suggested for improved localization of tyramides (Van Gijlswijk et al. 1996a), which, however, proved not to be essential in our hands (see Figure 2). An even more rapid way to visualize nucleic acids in situ is the use of peroxidase-labeled probes, which can be detected directly with CARD signal amplification (Van Gijlswijk et al. 1996b; Schmidt et al. 1997). However, these procedures proved to be clearly less sensitive than the indirect procedures described above, although here the application of a second signal amplification reaction after detection of the first layer of deposited haptenized tyramides by a peroxidase-labeled anti-hapten antibody conjugate may be advantageous to further increase the detection sensitivity.
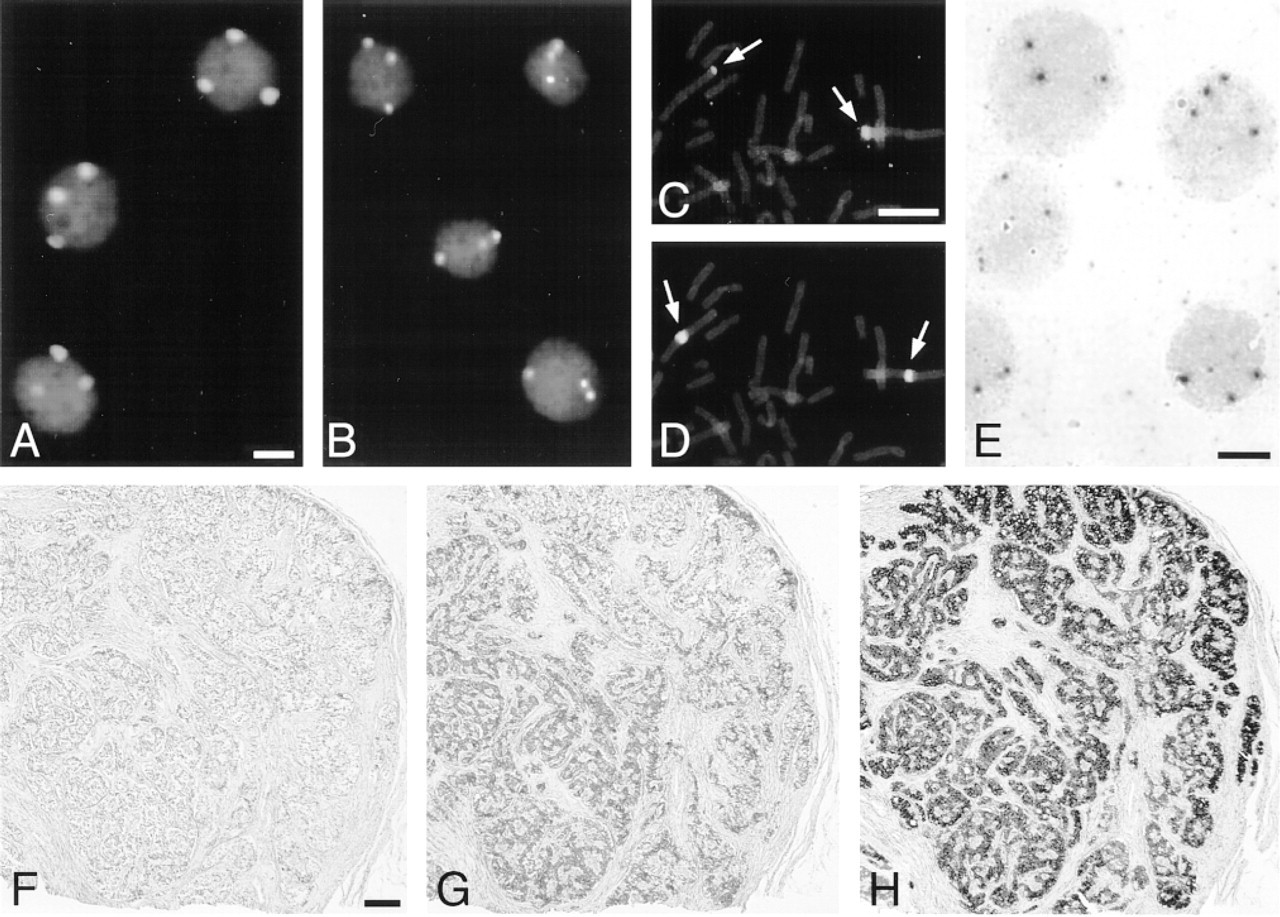
Results after ISH and CARD signal amplification on human cell preparations (A-E) and routinely fixed, paraffin-embedded tissue sections (F-H) [see also Table 2, Hopman et al. (1997), and Speel et al. (1997 and in press)]. (A,B) Fluorescence detection of three centromere 1 copies in bladder tumor cell line T24 using a chromosome 1-specific centromere probe (biotin-labeled), avidin-peroxidase, rhodaminetyramide (A) or TNP-tyramide/anti-TNP fluorescein (B) visualization, and propidium iodide counterstaining. (C,D) Fluorescence detection of chromosome 1p36 (C) and centromere 1 (D) on a lymphocyte metaphase spread using specific digoxigenin- and biotin-labeled probes, subsequent visualization with mouse anti-digoxigenin/goat anti-mouse peroxidase/rhodamine-tyramide and avidin peroxidase/coumarin-tyramide, respectively, and YOYO counterstaining. (E) Brightfield detection of chromosome 4p16 in T24 cells using a 40-KB digoxigenin-labeled cosmid probe, anti-digoxigenin-peroxidase/digoxigenin-tyramide/anti-digoxigenein-peroxidase and diaminobenzidine visualization, and hematoxylin counterstaining. Bars = 5 μm. (F-H) Brightfield detection of insulin mRNA in an endocrine pancreatic tumor (insulinoma) using an anti-sense digoxigenin-labeled oligonucleotide probe and conventional detection (F,G) or CARD signal amplification (H). Visualization with anti-digoxigenin-peroxidase (F), mouse anti-digoxigenin/horse anti-mouse biotin/avidin-biotinylated peroxidase (ABC) (G), or antidigoxigenin peroxidase/digoxigenin-tyramide/anti-digoxigenin-peroxidase (H), diaminobenzidine reaction and hematoxylin counterstaining. Bar = 200 μm.
We have recently introduced CARD signal amplification for our diagnostic nonradioactive oligonucleotide ISH procedure with digoxigenin-labeled probes to increase the sensitivity of the assay and to shorten the overall turnaround time of the assay (Speel et al. 1998). This approach allows, e.g., the detection of peptide hormone mRNA in tissue sections from routinely fixed, paraffin-embedded surgical samples within a single working day, and makes the assay suitable for routine diagnostic purposes. Furthermore, it allows the use of diaminobenzidine (DAB) as a chromogen and, as a consequence, the application of conventional counterstains and the mounting of slides in xylene-based mounting solutions, making the procedure more acceptable for the technician to perform and the pathologist to interpret. To date, most of the protocols use biotinylated tyramides for the amplification step, which are easy and inexpensive to synthesize (Bobrow et al. 1989,1991; Adams 1992; Kerstens et al. 1995; Raap et al. 1995; Plenat et al. 1997; Hopman et al. 1998b; Jacobs et al. 1998). However, similar to immunohistochemical procedures, the use of biotin is associated with significant disadvantages, especially in working with tissue sections. Therefore, in tissues with large numbers of endogenous streptavidin binding sites, such as liver or kidney, low signal-to-noise ratio and high background staining may be encountered. It is therefore desirable to be able to rely on differently labeled tyramides, e.g., with digoxigenin, dior trinitriphenyl (Hopman et al. 1998a; Speel et al. 1998), or fluorochromes (Schmidt et al. 1997; Van Gijlswijk et al. 1997; Hopman et al. 1998b; Speel et al. in press; also commercially available from NEN Life Science Products), which is now possible. These tyramide conjugates can also be used in multiple-target ISH approaches (Speel et al. 1997; Van Gijlswijk et al. 1997; Hopman et al. 1998a,b) or the combination of immunohistochemistry and ISH with signal amplification.
Although the increase in ISH sensitivity by using CARD signal amplification is obvious from the literature, speculation about the obtained amplification factor is difficult, because thus for only very few quantitative evaluations have been carried out. Moreover, because the tyramide deposition reaction runs very quickly, minor differences in amplification reaction time may lead to variations in the final signal intensities. Nevertheless, an amplification factor in the range of five- to tenfold, or possibly higher, together with preservation of distinct localization of ISH signals, seems to be a realistic indication for both DNA and mRNA ISH.
In conclusion, CARD signal amplification using labeled tyramides has significantly influenced the ISH methods performed on different biological specimens, because it is an easy, rapid, highly sensitive, and efficient procedure. As a consequence, sensitive ISH procedures incorporating CARD signal amplification may become the tool of choice for low-copy nucleic acid and antigen detection in situ and therefore may be suitable for diagnostic laboratories. In addition, these procedures will facilitate the analysis of DNA ISH signals, e.g., in the evaluation of chromosomal aberrations in cytological and histological materials (visualization with low-magnification objectives), and might help advance the development of automated ISH spot-counting by computer-assisted image generation and analysis. We expect that the now available spectrum of probe labels, detection systems, and tyramide conjugates for CARD signal amplification will simplify and further improve ISH and will promote multiple-target nucleic acid detection in situ and procedures combining ISH and immunophenotyping.
Footnotes
Acknowledgements
We thank P. Saremaslani for excellent technical assistence, N. Wey for computer-assisted reproductions, and Profs F.C.S Ramaekers (Department of Molecular Cell Biology and Genetics, University Maastricht, The Netherlands), Ph.U. Heitz, and J. Roth for continuous support.
Presented at the Histochemical Society Workshop, “Merging molecular biology with morphological techniques, in situ hybridization for the cellular localization of mRNA and DNA: State of the art 1998,” July 23, 1998, in San Diego, CA.
