Abstract
A one-step procedure for the synthesis of different tyramide conjugates, which can be utilized in the catalyzed reporter deposition (CARD) amplification system, is described. Succinimidyl esters of biotin, digoxigenin, and of the fluorochromes fluorescein, rhodamine, aminomethylcoumarine acetic acid, and Cy3 were coupled to tyramine in dimethylformamide (DMF) adjusted to a pH of 7.0–8.0 with triethylamine (TEA). The coupling reaction can be performed within 2 hr and the reaction mixture can be applied without further purification steps. Furthermore, trinitrophenyl (TNP)-tyramide was prepared by adding 2,4,6,-trinitrobenzenesulfonic acid to tyramine dissolved in either MilliQ/DMF basified with TEA or in an NaHCO3 (pH 9.5) buffer. A subsequent precipitation of the TNP-tyramide resulted in a high-yield isolation of this conjugate. The synthesized tyramide conjugates were applied successfully in single- and multiple-target in situ hybridization (ISH) procedures to detect both repetitive and single-copy DNA target sequences in cell preparations with high efficiency. The described approach provides an easy and fast method to prepare a variety of tyramide conjugates in bulk amounts at relatively low cost.
Keywords
A
Free primary amino groups can be quantitatively acylated by NHS esters in organic media within a few minutes and with minimal risk of hydrolysis of the active esters, leading to inefficient coupling or impure products (Lomant and Fairbanks 1976; Hopman et al. 1986b). Therefore, we coupled the NHS esters of biotin (BIO), digoxigenin (DIG), fluorescein (FLU), rhodamine (RHO), aminomethylcoumarin (AMCA), and Cy3 to tyramine in water-free dimethylformamide to which the base triethylamine (TEA) was added in a 1.25-fold equimolar amount to deprotonate the amino group of the tyramines. These reaction conditions have already proved to be successful in the synthesis of mercury binding sulfhydryl-hapten ligands, used to detect mercurated nucleic acid probes in situ (Hopman et al. 1986a,b). Alternatively, trinitrophenyl-tyramides (TNP-tyr) were synthesized by mixing equal molar amounts of 2,4,6,-trinitrobenzenesulfonic acid (TNBS) and tyramine in MilliQ basified with TEA or in a carbonate buffer.
The applicability of the newly synthesized tyramides was tested in single- and multiple-target ISH procedures visualizing both repetitive and single-copy target sequences in cell preparations with CARD amplification fluorescence or brightfield detection systems.
Materials and Methods
Reagents for the Synthesis of Tyramide Conjugates
The N-hydroxysuccinimide esters of biotin [sulfosuccinimidyl-6-(biotinimide)hexanoate], abbreviated as BIO-NHS (mw 557), fluorescein [5-(and 6)-carboxyfluorescein] succinimidyl ester (FITC-NHS), abbreviated as FLU-NHS (mw 473), rhodamine [5-(and 6)-carboxytetramethylrhodamine] succinimidyl ester (TRITC-NHS), abbreviated as RHO-NHS (mw 528), and coumarin (succinimidyl-7-amino-4-methylcoumarin-3-acetic acid (AMCA-NHS; mw 330) were purchased from Pierce (Rockford, IL). The N-hydroxysuccinimide esters of digoxigenin (digoxigenin-3-0-succinyl-∊-aminocaproic acid-N-hydroxysuccinimide ester; DIG-NHS, mw 659) and Cy3 (succinimidyl ester; Cy3-NHS, mw 766; one cap contains 200 mg) were purchased from Boehringer-Mannheim (Mannheim, Germany) and Amersham Life Science, (Arlington Heights, IL), respectively. Tyramine-HCl (mw 173) was purchased from Sigma Chemical (St Louis, MO), and dimethylformamide (DMF; water-free) and triethylamine (TEA; 7.2 M) were purchased from Pierce.
Tyramide Synthesis in Organic Media
Table 1 summarizes the different amounts of active esters (X-NHS), tyramine, and TEA to be combined in a one-step synthesis of tyramide conjugates. The NHS esters are prone to hydrolysis and are light-sensitive. For this reason, the active esters were freshly dissolved only shortly before tyramide synthesis. All esters were dissolved in DMF at concentrations as indicated in Table 1 to obtain an active ester stock solution “A” (10 mg per ml DMF). The tyramine-HCl stock solution “B” was prepared by dissolving 10 mg/ml DMF (equivalent to 58 μ mol) to which a 1.25-fold equimolar amount of TEA (10 μl of 7.2 M stock solution; 72.5 μmol) was added. For efficient acylation, the active esters (stock solution A) were added in 1.1 times equimolar amounts to the tyramine stock solution B and left at room temperature (RT) in the dark for 2 hr. The synthesized tyramide conjugates were diluted with ethanol to obtain stock solution “C” of 1 mg/ml. The tyramide stock solutions could be safely stored for at least 8 months at 4C without any reduction in reactivity.
Reaction conditions for the one-step synthesis of several tyramide conjugates
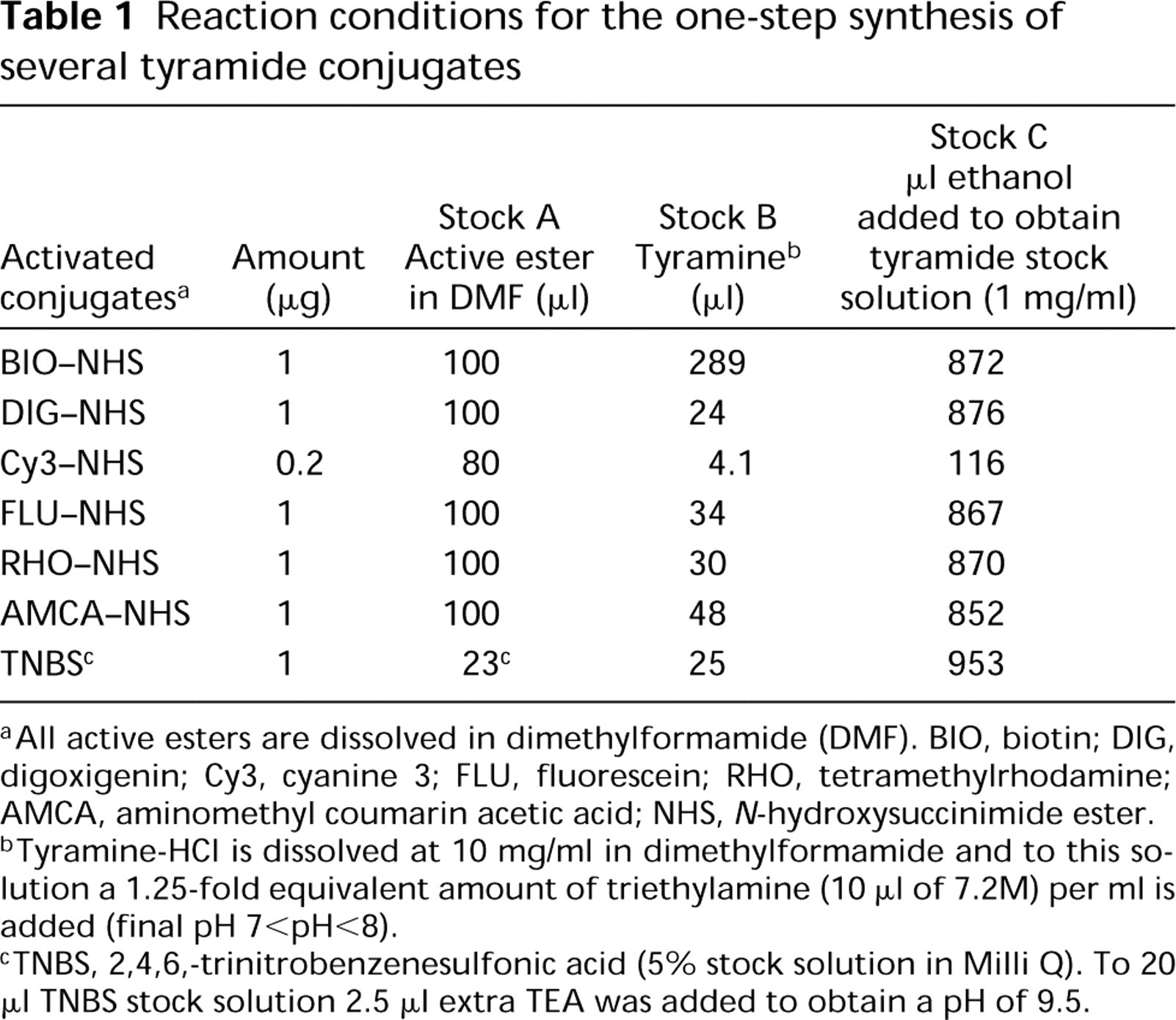
All active esters are dissolved in dimethylformamide (DMF). BIO, biotin; DIG, digoxigenin; Cy3, cyanine 3; FLU, fluorescein; RHO, tetramethylrhodamine; AMCA, aminomethyl coumarin acetic acid; NHS, N-hydroxysuccinimide ester.
Tyramine-HCl is dissolved at 10 mg/ml in dimethylformamide and to this solution a 1.25-fold equivalent amount of triethylamine (10 μl of 7.2M) per ml is added (final pH 7<pH<8).
TNBS, 2,4,6,-trinitrobenzenesulfonic acid (5% stock solution in Milli Q). To 20 μl TNBS stock solution 2.5 μl extra TEA was added to obtain a pH of 9.5.
Our synthesized BIO-tyr was compared to the BLAST kit obtained from Du Pont NEN (Boston, MA) and the Catalyzed Signal Amplification (CSA) System from DAKO (Glostrup, Denmark). Our fluorochrome tyramide conjugates were compared with the TSA-Direct system from NEN Life Science Products (Renaissance TSA amplification kit).
TNP-Tyramide Synthesis in Aqueous Media
TNP-tyr was prepared by two different protocols: (a) by mixing equimolar amounts of 2,4,6-trinitrobenzenesulfonic acid (TNBS) (mw 690, 5% solution in water; Pierce) and tyramine with an excess of TEA to obtain a pH of 9.5. The reaction was performed for 2 hr at RT; and (b) by mixing equimolar amounts of TNBS (20 mg/400 ml) and tyramine dissolved in 0.3 M NaHCO3 buffer, pH 9.5. This reaction was performed for 1 hr in the dark at RT in a 10-ml plastic tube, with mixing on a vortex every 15 min. Even after the first 15 min a precipitate of TNP-tyr was formed. To complete the reaction the mixture was left at RT for another 1 hr. The precipitate was pelleted, and the supernatant was removed and washed subsequently with the carbonate buffer and MilliQ. The pellet was dried in a vacuum excitatory and dissolved in 0.2 ml DMF to which 0.8 ml ethanol was added.
Cell Processing
A 70% ethanol suspension of the human transitional cell carcinoma line T24 (Bubeník et al. 1973) (DNA index 1.6; trisomic for 1q12, tetrasomic for 1p36, and trisomic for 4p16) and methanol:acetic acid (3:1)-fixed human lymphocyte metaphase spreads were pretreated by pepsin digestion to improve DNA probe and conjugate penetration, as previously described (Hopman et al. 1988; Speel et al. 1993).
DNA Probes and Labeling Procedures
The probes for chromosome 1q12 (pUC 1.77), 1p36 (p1-79), 1q42-43 (pH5SB), and 4p16 (c5.5) have been described by Cooke and Hindley (1979), Buroker et al. (1987), Little and Braaten (1989), and Landegent et al. (1986), respectively. The DNA probes were labeled with biotin-11-dUTP (Enzo Diagnostics; New York, NY), digoxigenin-11-dUTP, or fluorescein-12-dUTP (Boehringer-Mannheim) in a standard nick-translation reaction and used in single-, double-, or triple-target ISH procedures.
ISH Procedure
The DNA probes described above were used at a concentration of 0.4 ng/μg (pUC 1.77), 1 ng/μg (p1-79), or 5 ng/μg (pH5SB and c5.5) and hybridized in different combinations in a hybridization buffer containing 50% formamide, 2 X SSC (0.3 M NaCl, 30 mM Na-citrate) pH 5.0, 10% dextran sulfate, 0.2 mg/ml herring sperm DNA as carrier DNA, and 0.2 mg/ml yeast tRNA as carrier RNA. An excess of total human placenta DNA was added as competitor DNA when c5.5 (100 X excess competitor) or pH5SB (250 X excess competitor) was included. Ten μg hybridization buffer was added to each slide under a coverslip (20 X 20 mm). Denaturation was performed on the bottom of a metal box in a water bath at 70C for 3 min and hybridization was performed overnight at 37C. The slides were washed twice for 5 min at 42C with 2 X SSC containing 0.05% Tween 20, followed by two 5-min washes at 60C 0.1 X SSC and a 5-min wash with 4 X SSC, pH 7.0, containing 0.05% Tween 20 (Buffer A) at RT.
Cytochemical Detection Procedures
To reduce background staining in the cytochemical detection procedures, the slides were preincubated for 10 min at 37C with 4 X SSC, pH 7.0, containing 5% nonfat dry milk (Buffer B), followed by dipping in Buffer A (see above). For all the detection procedures, the avidin conjugates were diluted in Buffer B, and all the antibody conjugates were diluted in PBS containing 0.05% Tween 20 (Buffer C) and 2% normal goat serum (NGS). After each incubation step of 20-30 min at 37C, the slides were rinsed twice in Buffer A in the case of avidin conjugates (ABC system) or Buffer C in the case of antibody conjugates.
Single-target ISH
The biotinylated pUC 1.77 probe was detected by peroxidase (PO)-conjugated avidin (Av-PO, 1:50 dilution; DAKO), followed by CARD amplification as previously described (Speel et al. 1997). After cytochemical detection of the probes, the different tyramide conjugates RHO-tyr 1:1000, BIO-tyr, DIG-tyr 1:500, TNP-tyr, AMCA-tyr, FLU-tyr, Cy3-tyr 1:250 (diluted from the 1 mg/ml stock solution C with concentration 1.8, 1.7, 1.5, 1.4, 3.0, 2.0, and 1.3 mM, respectively) were applied in PBS containing 0.1 M imidazole, pH 7.6, and 0.001% H2O2 (50 μl under a coverslip) for 5-10 min at 37C. Thereafter the slides were washed twice with Buffer C.
The tyramide conjugates were detected as follows: TNP-tyr was detected with rabbit anti-DNP (RADNP 1:100; DAKO) and FITC-labeled swine anti-rabbit IgG (SWAR-FITC 1:80; DAKO); DIG-tyr with FITC-labeled rabbit anti-DIG (RADIG-FITC 1:100; Boehringer); BIO-tyr with FITC-labeled avidin (Av-FITC 1:500; Vector, Brunschwig Chemie, Amsterdam, The Netherlands). The digoxigenin-labeled c5.5 probe was visualized by peroxidase (PO)-conjugated sheep anti-DIG Fab fragments (SHADIG-PO 1:100; Boehringer), followed by CARD amplification with DIG-tyr (see above), SHADIG-PO, and the PO-DAB reaction (see above).
Multiple-target ISH
For multiple-target ISH with fluorochrome-labeled tyramides, CARD detection of different probes was performed sequentially, with a 0.01 M HCl treatment for 10 min at RT between the individual detection steps to inactivate PO activity (Speel et al. 1994,1995). Labeled probes were detected as follows: BIO with AV-PO and optionally followed by biotinylated goat anti-avidin (GAA-BIO) and AV-PO; DIG with SHADIG-PO or mouse anti-DIG (MADIG) and PO-conjugated rabbit anti-mouse (RAM-PO); FITC with PO-conjugated anti-FITC (AFITC-PO 1:200; NEN Life Science Products, Boston, MA). In all cases, cytochemical detection was followed by CARD amplification with fluorochromelabeled tyramides. For multiple-target brightfield ISH, the DIG-labeled pUC1.77 probe was detected by alkaline phosphatase (APase)-conjugated sheep anti-DIG Fab fragments (SHADIG-APase 1:100; Boehringer) and the APase-Fast Red reaction (see below), followed by visualization of the biotinylated 1p36 probe with AV-PO, CARD amplification with BIO-tyr (see below), AV-PO, and the PO-DAB reaction (see below).
PO-DAB and APase-Fast Red reactions
PO-DAB Reaction (Brown Precipitate; Graham and Karnovsky 1966). Before use, 1 ml 3,3-diaminobenzidine tetrachloride (DAB; Sigma) in PBS (5 mg/ml stock) was mixed with 9 ml 0.1 M imidazole in PBS, pH 7.6, and 10 μg 30% H2O2. The slides were incubated with 0.1-1.0 ml for 5-15 min at 37C and washed three times for 5 min with PBS.
APase-Fast Red Reaction (Red Precipitate;
Speel et al. 1993). Before use, 4 ml 0.2 M Tris-HCl, pH 8.5, containing 10 mM MgCl2 and 5% polyvinyl alcohol (PVA,
Microscopy
Microphotographs were made on a Leica DM RBE microscope equipped with the Metasystem Image Pro System (black-and-white CCD camera; Sandhausen, Germany). Images were captured using the ISIS program for FISH and for brightfield ISH additional green, red, and blue filters were applied. Images were processed in Adobe Photoshop 3.0 at a resolution of 120 pixels/inch.
Results
Tyramide Conjugate Synthesis
Table 1 summarizes the synthesis of seven different tyramide conjugates. For this purpose, a 1.1-fold molar excess of the active hapten- (BIO, DIG) or fluorochrome-(RHO, FLU, Cy3, AMCA) succinimidyl esters, freshly dissolved in DMF, was added to the tyramine. The tyramine was also dissolved in DMF adjusted with TEA to obtain a pH of about 7.0-8.0. After a 2-hr reaction, the synthesized tyramide conjugates were diluted to a final concentration of 1 mg/ml by adding ethanol without purification. These stock solutions were further diluted to working concentrations for ISH probe detection (see below), as indicated in Table 2.
The TNP-tyramide was synthesized by mixing the aqueous TNBS solution with tyramine-TEA (see above) or in 0.3 M NaHCO3 buffer, pH 9.5. Because TNBS is not prone to hydrolysis in water, the coupling reaction can be performed with an excess of base in both organic and aqueous reaction media. During the reaction of TNBS with tyramine in 0.3 M NaHCO3 buffer, the produced TNP-tyramide precipitated spontaneously, because these molecules are lipophilic and only slightly water-soluble. This precipitation reaction therefore allows the easy isolation of a pure reaction product while the unreacted tyramine and TNBS remain in solution. In this way, large amounts of this conjugate can be synthesized and isolated. The TNP-tyramide is dissolved in DMF and diluted with ethanol.
Application and Sensitivity of Tyramide Conjugates in Fluorescence ISH
All synthesized tyramides were tested in single-target ISH experiments on T24 cells, using the biotinylated probe for 1q12 and one detection layer of Av-PO. The fluorochrome-labeled tyramides (Cy3, FLU, RHO, and AMCA) were deposited in CARD amplification reactions for 5 min at 37C and could be visualized directly in the fluorescence microscope. T24 cells displayed three ISH signals per nucleus in all cases, as shown in Figure 1a for Cy3-tyramide. Depositions of the hapten-labeled tyramides (BIO, DIG, and TNP) required additional incubations with fluorescent detection conjugates to visualize them in the fluorescence microscope. For example, Figures 1b and 1c show the detection of chromosome 1q12 with TNP-tyramide/RADNP/SWAR-FITC and DIG-tyramide/SHADIG-FITC, respectively. Table 2 summarizes the working concentrations of the tyramide conjugates in the CARD amplification reactions for optimally localized fluorescent signals. All the tyramides could be applied at concentrations of 2-10μM in a 5-10-min amplification reaction at 37C in PBS-imidazole. The RHO-tyramide appeared to be the most sensitive conjugate, as was to be expected on the basis of previously described observations (Raap et al. 1995; Speel et al. 1997).
Working concentrations (μM) of different hapten-and fluorochrome-tyramides applied to ISH
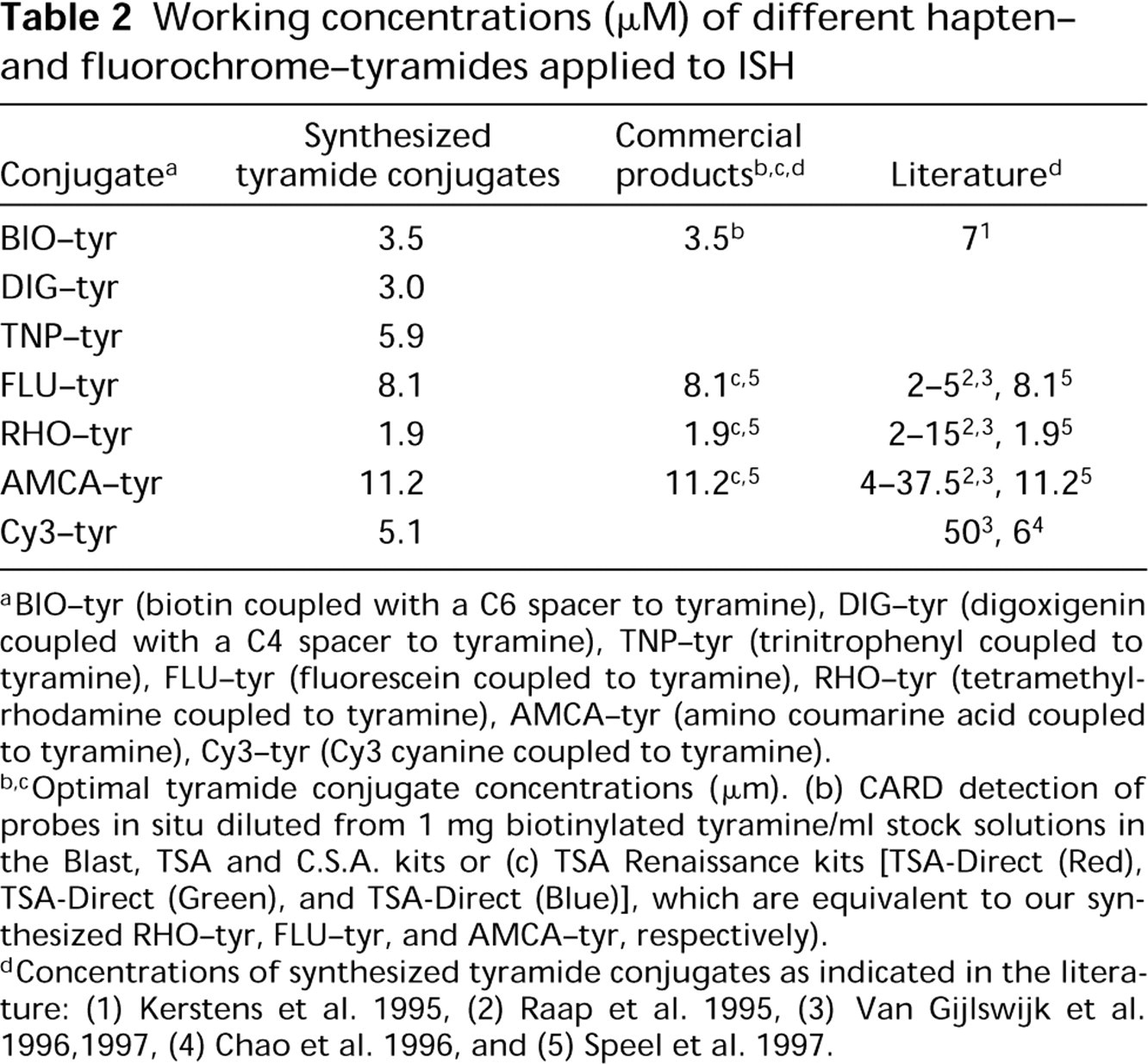
BIO-tyr (biotin coupled with a C6 spacer to tyramine), DIG-tyr (digoxigenin coupled with a C4 spacer to tyramine), TNP-tyr (trinitrophenyl coupled to tyramine), FLU-tyr (fluorescein coupled to tyramine), RHO-tyr (tetramethyl-rhodamine coupled to tyramine), AMCA-tyr (amino coumarine acid coupled to tyramine), Cy3-tyr (Cy3 cyanine coupled to tyramine).
Optimal tyramide conjugate concentrations (μm). (b) CARD detection of probes in situ diluted from 1 mg biotinylated tyramine/ml stock solutions in the Blast, TSA and C.S.A. kits or (c) TSA Renaissance kits [TSA-Direct (Red), TSA-Direct (Green), and TSA-Direct (Blue)], which are equivalent to our synthesized RHO-tyr, FLU-tyr, and AMCA-tyr, respectively).
Concentrations of synthesized tyramide conjugates as indicated in the literature: (1) Kerstens et al. 1995, (2) Raap et al. 1995, (3) Van Gijlswijk et al. 1996,1997, (4) Chao et al. 1996, and (5) Speel et al. 1997.
The applied tyramide conjugate concentrations were compared to the conditions recommended in the commercially available kits (BLAST, TSA Renaissance systems, or C.S.A. kits) and with the concentrations known from the literature (Raap et al. 1995; Chao et al. 1996; Speel et al. 1997; Van Gijlswijk et al. 1997). No differences were found when the commercially available tyramide conjugates were used for localizing the 1q12 targets in T24 nuclei. For the new DIG- and TNP- tyramides no reference values are available, but their optimal concentrations were within the same range as the other tyramide conjugates.
The synthesized fluorochrome-conjugated tyramides were also tested in multiple-target FISH procedures. Figure 1d shows a double-target FISH using AMCA- tyramide and Cy3-tyramide. The chromosomal targets 1q12 and 1p36 were subsequently detected, with SADIG-PO/Cy3-tyramide and Av-PO/AMCA-tyramide, resulting in discrete CARD signals. For this purpose, the PO activity remaining after the first CARD reaction step was inactivated by a mild acid incubation step before application of the next CARD detection step. In this way a triple-target fluorescence ISH experiment with CARD detection could also be performed. Figure 1e shows the localization of the 1q42-43, 1q12, and 1p36 targets on human chromosome 1 with AMCA-tyramide, RHO-tyramide, and FLU-tyramide, respectively. The chromosomes were not counterstained to avoid color overlap of the general DNA stains (DAPI or PI) with one of the CARD signals.
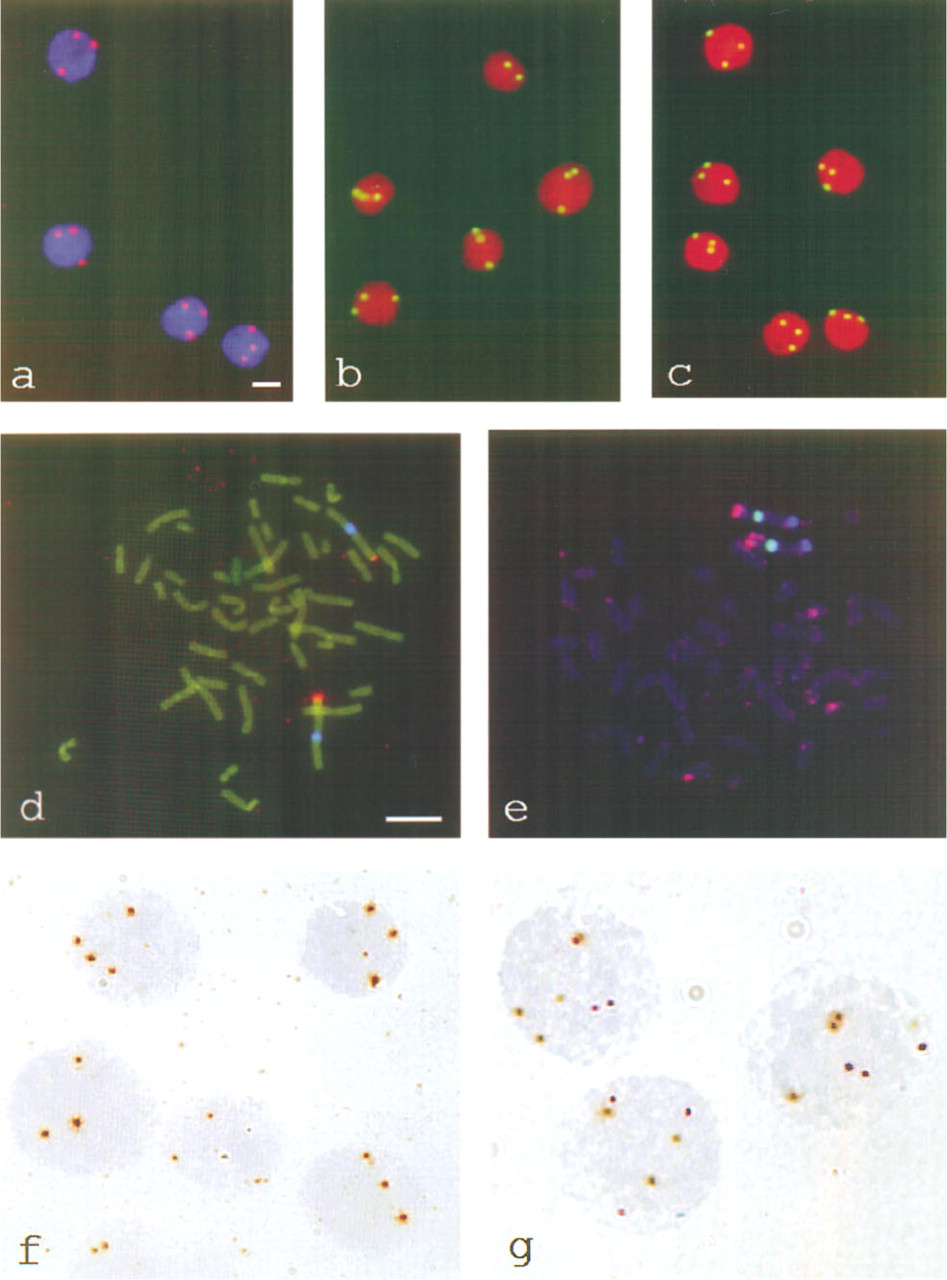
Fluorescence (
CARD Amplification in Brightfield ISH
The hapten-tyramides could also be applied to localize both repetitive and single-copy gene DNA sequences in brightfield ISH. As an example, Figure 1f shows the detection of the digoxigenin-labeled c5.5 cosmid probe with SHADIG-PO, DIG-tyramide, SHADIG-PO and the PO-DAB reaction. In the T24 interphase nuclei, both three and four ISH signals per nucleus could be visualized, as was expected from earlier unpublished results using fluorescence ISH (78% trisomy, 8% tetrasomy). Double-target brightfield ISH was carried out to detect 1q12 with APase-Fast Red, after which the 1p36 target could be detected using the BIO-tyramide. The latter target was detected in PO-DAB (Figure 1g).
Discussion
The one-step synthesis of tyramide conjugates in DMF (adjusted to pH 7.0-8.0 with TEA) resulted in products that could be successfully applied in the CARD amplification step for ISH. The hydrolysis of the active fluorochrome or hapten NHS esters was circumvented by performing the reaction in water-free medium and by adding TEA to deprotonate the amino group of tyramine-HCl. Raap et al. (1995) have synthesized fluorochrome-labeled tyramides by mixing tyramine with different fluorochromes, both dissolved in dimethyl sulfoxide, which may result in irreproducible results, strongly depending on the pH of the solutions during the synthesis. Furthermore, Adams (1992) and Kerstens et al. (1995) synthesized biotinyl-tyramide by dissolving the active ester and tyramine-HCl in a borate buffer (pH 8.0), resulting in a slightly clouded solution, and agitated the solution overnight. The reaction started with reagents that were not properly dissolved; this also could lead to irreproducible results when these experiments are repeated. The approach as described here starts with reagents completely dissolved in DMF. Furthermore, hydrolysis was reduced to a minimum by adding a minimal amount of TEA. In addition, the reaction time was kept short compared to the other protocols (Bobrow et al. 1991; Adams 1992; Raap et al. 1995).
The synthesized tyramide conjugates were applied in model systems to test the sensitivity of immunocytochemical amplification systems. All synthesized products could be applied within the same concentration range, which indicates the efficiency of the synthesis, even though the purity of the products was not explicitly tested. Furthermore, for the known tyramides, these concentrations are comparable to the range of lowest concentrations reported in the literature (Table 2). The applicability and sensitivity of the haptenized tyramides in brightfield ISH are illustrated in Figures 1f and 1g. The intense signals for the cosmid probes illustrate the sensitivity and are comparable with the results obtained by Kerstens et al. (1994,1995). The large size of the ISH signal for the 1p36 target compared to the 1q12 target is the result of diffusion of the tyramides during the PO-tyramide reaction, followed by the PO-DAB reaction.
In conclusion, our approach for the synthesis of the tyramide conjugates is an easy method to prepare a variety of new substrates for peroxidase cytochemistry because many active esters of fluorochromes or other haptens are commerically available. The protocol minimizes problems arising from dissolving of these active esters and facilitates the coupling reaction.
Furthermore, the protocol is suitable for bulk production of tyramide conjugates. In particular, TNP-tyramide can be synthesized in gram quantities with high purity, opening the possibility of using this tyramide in a routine setting on a daily basis. In diagnostic pathology as well as other immunochemical studies, this could mean a significant reduction in the costs for primary antibodies, because these reagents can be diluted several more magnitudes.
Footnotes
Acknowledgements
Supported in part by the Dutch Cancer Foundation, grant no. IKL 92-07.
