Abstract
Carboxypeptidase E (CPE) is highly concentrated in neuroendocrine tissues and is the only carboxypeptidase detected in mature secretory vesicles. Carboxypeptidase D (CPD), a carboxypeptidase with CPE-like activity, is widely distributed in tissues and is present in the trans-Golgi network. Previous work had shown that both CPE and CPD are expressed in the human placenta and that CPD is expressed at much higher levels than CPE. The present work provides evidence for the co-localization of CPE and CPD to basal plate extravillous trophoblasts and maternal uteroplacental vascular endothelial cells, chorionic villous endothelial cells, amnionic epithelial cells, and umbilical venous and arterial smooth muscle cells. Whereas the intensity of CPD immunostaining is similar in the placenta and umbilical cord, CPE staining in the placenta is much weaker than in the umbilical cord, suggesting that CPD plays a more important role in the processing of placental peptides. Immunoelectron microscopy of umbilical venous smooth muscle cells shows subcellular localization of both enzymes to the rough endoplasmic reticulum. In addition, CPE is present just subjacent to the cell membrane. The difference in cellular and subcellular localization between the two enzymes indicates that they perform distinct functions in the processing of placental peptides and proteins.
Keywords
C
CPD has been localized to the trans-Golgi network (Varlamov et al. in press) within pituitary corticotropes and pancreatic β-cells. The subcellular localization, broad tissue distribution, and CPE-like properties of CPD suggest that this enzyme plays some role in peptide processing. However, the absence of CPD from mature secretory vesicles indicates that this is not the primary role of CPD. Instead, it is likely that CPD participates in the processing of proteins that are secreted via the constitutive pathway, such as growth factors and growth factor receptors (insulin receptor, insulin-like growth factor receptor, and others). Interestingly, whereas CPE is expressed at only low levels in the human placenta (Fricker 1988), CPD is highly expressed in this tissue (Song and Fricker 1996).
Although a variety of both growth factors and neuroendocrine peptides have been identified and localized in the human placenta and umbilical cord (Rao et al. 1995; Petraglia et al. 1996), including pro-opiomelanocortin-derived peptides (Laatikainen et al. 1987; Margioris et al. 1988; Cooper et al. 1996) and corticotropin- releasing hormone (Grino et al. 1987; Challis et al. 1995), the enzymes that process bioactive peptides in these tissues have generally not been studied. We speculated, on the basis of the previous finding that CPD is expressed at higher levels than CPE in the human placenta, that CPD may substitute for CPE in placental peptide processing, as in the CPE deficient fat/fat mouse. We also took specific interest in the expression of CPE and CPD in the umbilical cord, and in umbilical vascular smooth muscle in particular, because of their potentially critical contribution to modulation of fetal blood supply by control of umbilical vascular tone. It was therefore of great interest to identify the placental and umbilical cord cell types that contain immunoreactivity for both enzymes and to consider these in relation to putative substrates.
Materials and Methods
Antibodies
Two rabbit polyclonal antisera were used for immunostaining of each enzyme, one directed against the C-terminal portion of the enzyme and a second antiserum directed against the N-terminal portion of the enzyme. The specificities of the anti-CPE antisera and of the anti-CPD antisera were previously characterized by Western blotting (Varlamov et al. 1996; Song and Fricker 1996; Xin et al. 1997).
Human Tissue
Frozen tissue sections taken from both the fetal and maternal sides of the central portion of placentas were retrieved from the human placental frozen tissue bank, which stores placental tissues at -80C from over 2000 patients who delivered at Georgetown University Hospital. Tissue from five patients with normal spontaneous vaginal deliveries without induction at 40 weeks of gestation, with no history of gestational diabetes mellitus, pregnancy-induced hypertension, or pre-eclampsia, were examined for chorionic plate, chorionic villous, and basal plate staining. Segments of fresh umbilical cords were collected in the delivery room from patients at Jack D. Weiler Hospital, sectioned, snap-frozen with optimal cutting compound and liquid nitrogen, and stored at -80C. All studies involving human tissue were conducted with IRB approval (IRB Protocol Number 1199707276.)
Immunohistochemistry
Tissue blocks were warmed to -20C, mounted on a microtome chuck with optimal cutting compound, and sectioned at 4 μm. After air-drying for at least 1 hr and desiccating for at least 24 hr, sections were fixed in PBS-4% paraformaldehyde for 20 min. After extensive washing in PBS, sections were permeabilized with 0.2% Triton X-100 for 15 min. After subsequent washing in PBS, sections were incubated in a humidified chamber with PBS-5% bovine serum albumin (BSA) for 30 min to reduce nonspecific binding. Excess buffer was blotted off and the primary antiserum, or preimmune serum for negative controls, was added, diluted 1:1000 for placental staining and 1:200 for umbilical cord staining, in PBS containing 1% BSA and 0.1% Tween 20. After a 1-hr incubation, the sections were washed in 0.2% Tween 20 in PBS and immunostaining was achieved using an avidin-biotin complex developer kit (Dako; Carpinteria, CA) and 3,3′-diaminobenzidine as substrate. Sections were counterstained with Mayer's hematoxylin.
Double Labeling
Sections of chorionic villous parenchyma were incubated sequentially with anti-CPD antiserum and anti-CD34 mouse monoclonal antibodies (Dako). Staining was developed with fluorescein-labeled anti-rabbit Ig antibodies and Texas redlabeled anti-mouse Ig antibodies (Vector; Burlingame, CA) according to the manufacturer's instructions. Tissue sections were examined with a Bio-Rad (Richmond, CA) confocal microscope.
Immunoelectron Microscopy
Immunohistochemistry was performed as described above, but tissue sections were cut by hand, incubated with 10% sucrose in PBS for 30 min, and kept floating in buffer throughout the staining protocol. After immunoperoxidase-DAB staining the sections were washed in PBS and postfixed in 1% osmium tetroxide with 0.01% potassium ferrocyanide for 1 hr. The tissue was dehydrated in a series of ascending ethanols from 50 to 100%, cleared in acetonitrile, infiltrated, and then embedded with a mixture of araldite- epon resin. Thin sections were examined unstained with a JEOL 100S electron microscope.
Results
For each enzyme, the same pattern of immunostaining was obtained with both the antiserum directed against the C-terminal end and the antiserum directed against the N-terminal portion of the protein. Furthermore, the staining patterns were essentially the same in tissues from five different patients. CPD and CPE appear to be co-localized in some placental cell types and in the umbilical cord. Whereas intense CPD immunostaining is observed in both the placenta and umbilical cord, CPE staining is intense in the umbilical cord but relatively weak in the placenta.
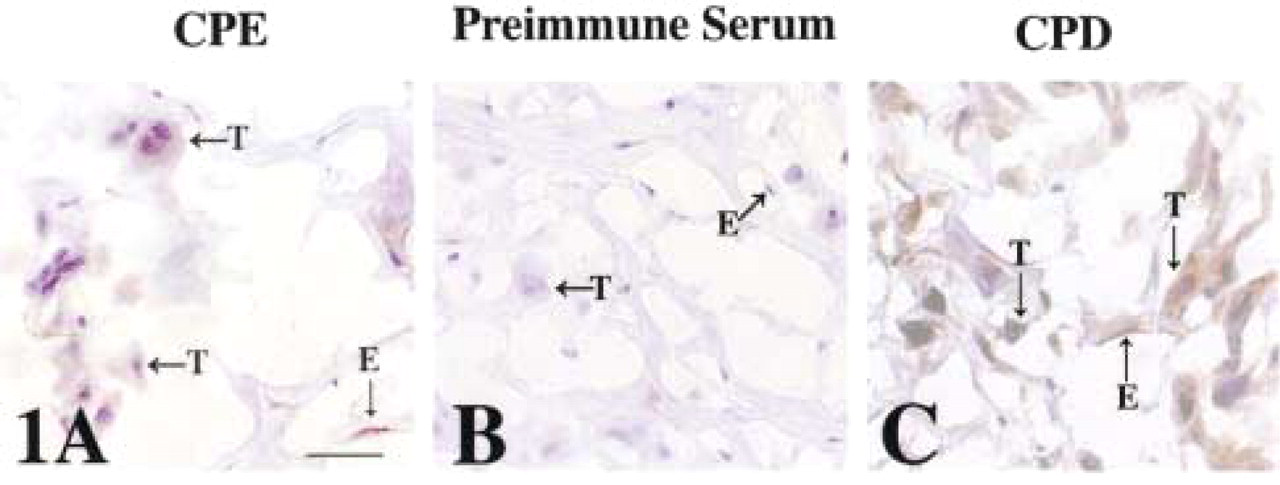
Specifically, in the basal plate, CPE immunostaining is present in extravillous invasive trophoblasts (Figure 1A, T) and in vascular endothelial cells (Figure 1A, E). The trophoblast staining is intracellular, with the brown DAB reaction product consistently localized to the perinuclear region of the cell (Figure 1A, T). The endothelial cells show a uniform cytoplasmic immunoreactivity for CPE (Figure 1A, E). No staining is seen in the basal plate when preimmune serum is substituted for immune serum (Figure 1B). CPD immunostaining is present in the same cell types of the basal plate that contain CPE immunoreactivity (Figure 1C). CPD staining is also perinuclear in the trophoblast (Figure 1C, T) and uniform in the cytoplasm of the maternal endothelial cell (Figure 1C, E).
CPE (
CPE (
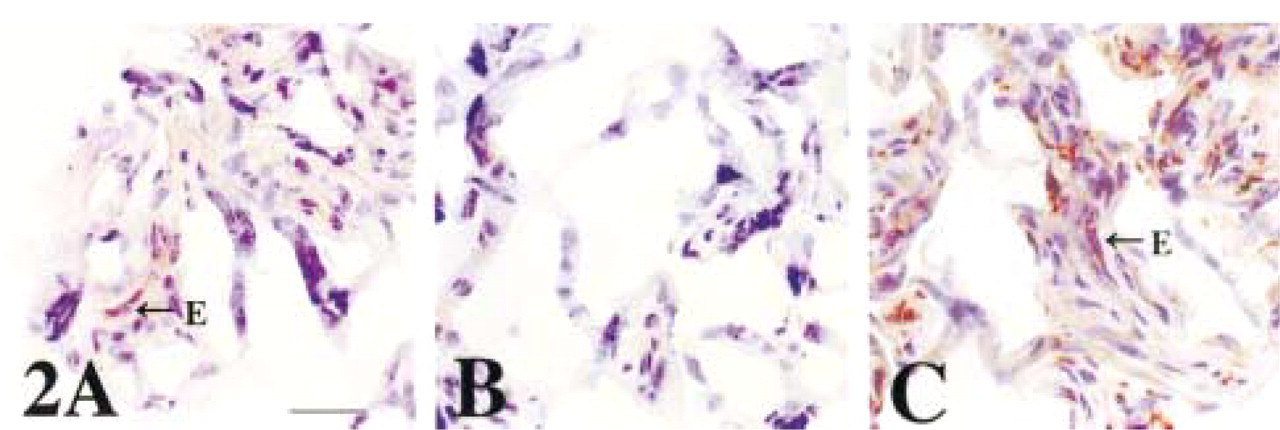
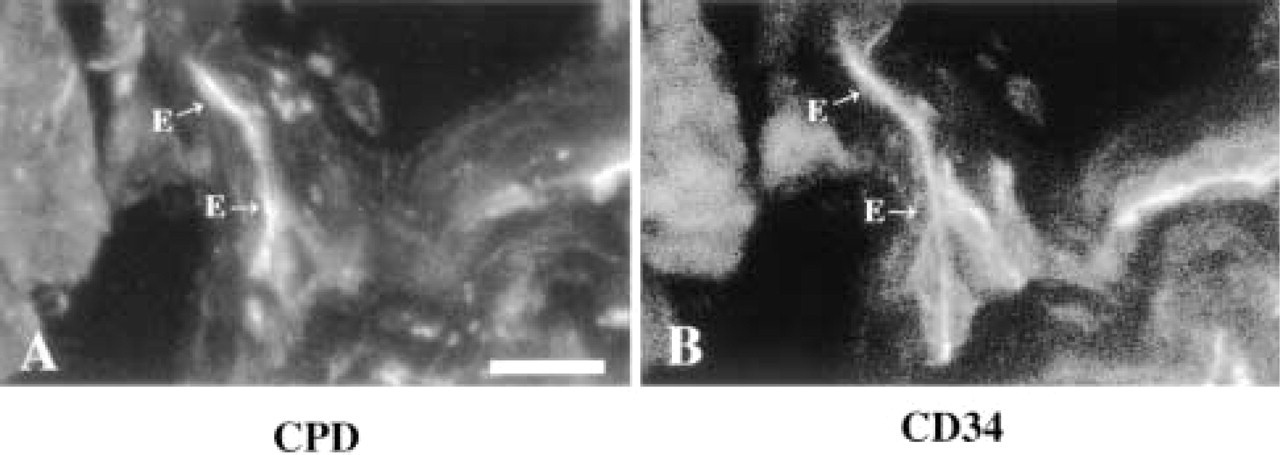
In the chorionic villi, weak CPE staining is consistently observed in the fetal endothelial cells (Figure 2A, E; negative control Figure 2B), but not in the trophoblast. Intense chorionic villous immunostaining with CPD antisera is also consistently observed in the fetal endothelial cells (Figure 2C, E). To confirm that the chorionic villous cells containing this intense CPD immunoreactivity are endothelial cells, the same tissue sections incubated with anti-CPD antiserum were double stained with monoclonal CD34 antibodies. Figure 3A shows labeling with fluorescein-tagged anti-rabbit antiserum, which represents CPD immunoreactivity. Figure 3B contains the same microscopic field as Figure 3A, but the filter is changed to reveal the Texas red-labeled anti-mouse antiserum, which represents CD34 immunoreactivity. By double labeling, CPD colocalizes with the endothelial cell marker CD34.
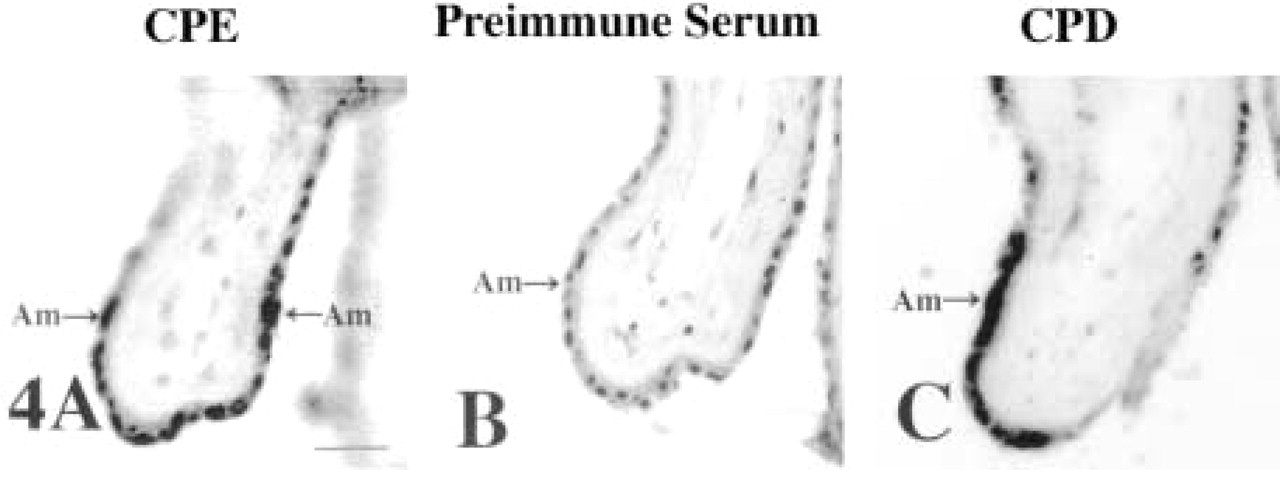
In the chorionic plate, focal CPE immunostaining was obtained in the amnion (Figure 4A; negative control, Figure 4B). CPD immunoreactivity, on the other hand, is present in many amniotic epithelial cells (Figure 4C). Both CPD and CPE amnion staining appear perinuclear.
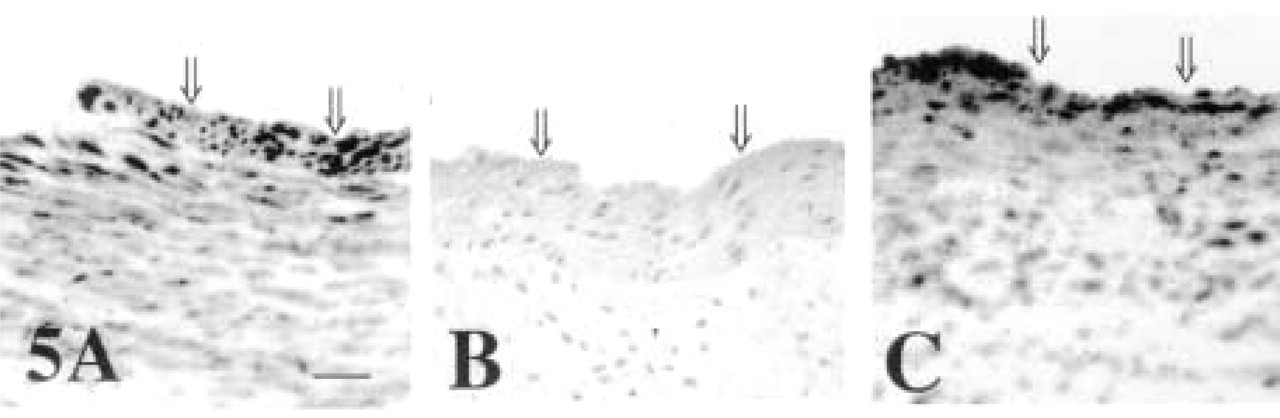
CPD and CPE appear to be co-localized in the umbilical cord to vascular smooth muscle cells of both the umbilical vein (Figure 5) and the umbilical arteries (Figure 6). Both CPD and CPE antisera produce intense perinuclear staining in cells of the umbilical venous wall (Figures 5A and 5C). All staining in the umbilical vein is eliminated with the substitution of preimmune serum for immune serum (Figure 5B). For both enzymes, the immunostaining is consistently more intense in the cells closest to the lumen, the smooth muscle cells just subjacent to the endothelial cells (Figures 5A and 5C, double arrows). The identity of the stained cells as smooth muscle cells is confirmed by electron microscopy (see below).
Co-localization of CPD (
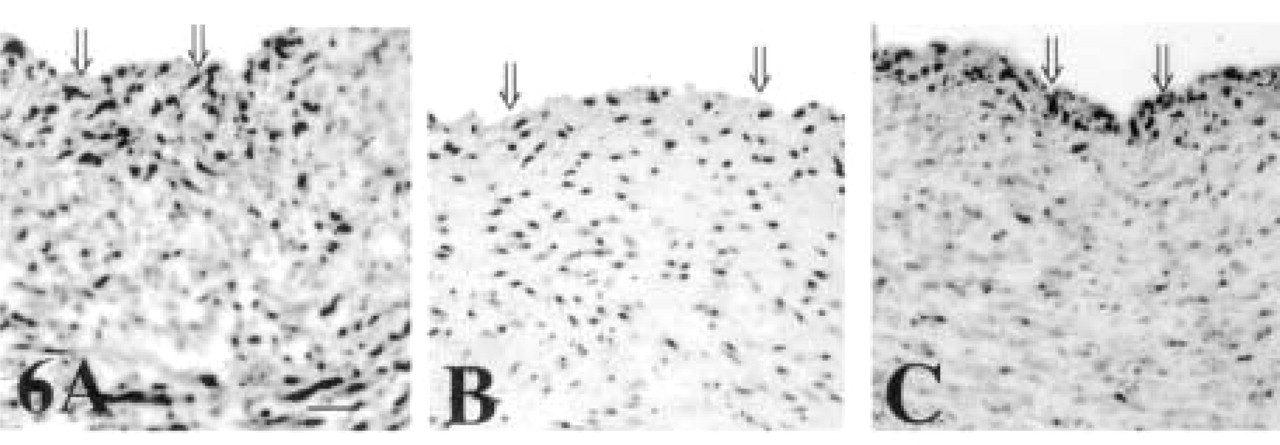
CPD and CPE umbilical artery staining is similar to immunostaining patterns of these enzymes seen in the umbilical vein. Intense perinuclear staining of CPE is present throughout the artery wall (Figure 6A). A very similar staining pattern is obtained with CPD antisera (Figure 6C; negative control Figure 6B), whereas no staining is obtained with preimmune serum (Figure 6B). For both enzymes, staining is particularly intense in the cells closest to the artery lumen, just subjacent to the endothelial cells (Figures 6A and 6C, double arrows).
In addition, intense immunoreactivity for both enzymes is focally present in Wharton's jelly cells (Figure 7). Umbilical cord sections stained with CPE antisera show dark perinuclear staining in focal Wharton's jelly myofibroblasts (Figure 7A, M). No staining is present in these cells after incubation with preimmune serum (Figure 7B). Reaction with CPD antisera, however, also produces intense perinuclear staining in focal umbilical myofibroblasts (Figure 7C, M).
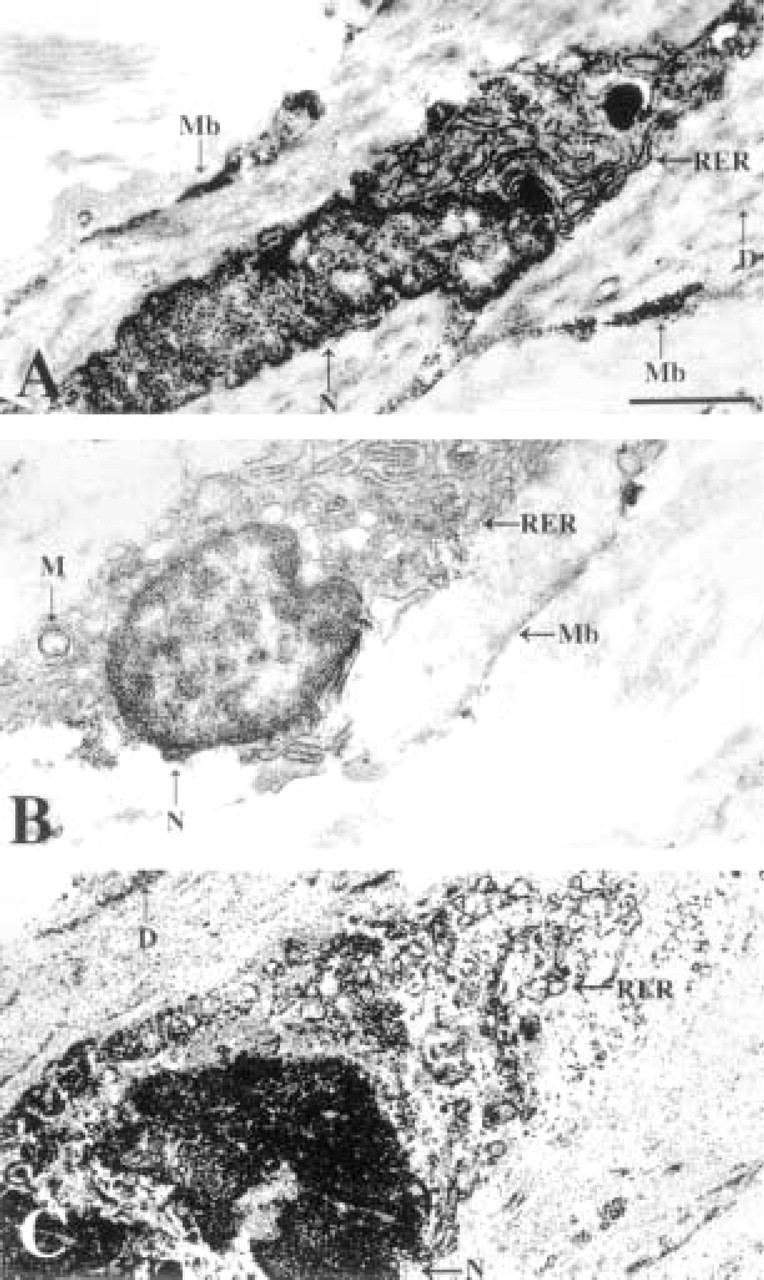
To confirm the identity of the stained umbilical venous cells as smooth muscle cells and to examine the subcellular localization of CPD and CPE in these cells, immunoelectron microscopy was performed. The location of the stained cells subjacent to the internal elastic lamina and the presence of dense bodies in the stained cells (Figure 8, D) indicate that they are smooth muscle cells. In tissue reacted with CPE antiserum, DAB reaction product is consistently associated with the rough endoplasmic reticulum that is in close proximity to the cell nucleus (Figure 8A, RER). The same staining pattern is obtained in tissue allowed to react with CPD antiserum (Figure 8C, RER), whereas the substitution of preimmune serum for immune serum eliminates staining (Figure 8B). Although CPD and CPE co-localize to the same cells in the umbilical vein at the light microscopic level, analysis by electron microscopy reveals distinct subcellular distributions. In addition to staining perinuclear rough endoplasmic reticulum, CPE antisera produce clusters of DAB reaction product subjacent to the cell membrane, following the entire perimeter of the cell (Figure 8A, Mb; Figure 9A, double arrows). This staining is not seen with either CPD antiserum (Figure 8C) or preimmune serum (Figures 8B and 9B).
CPE (
CPE (
CPE (

CPE (
Subcellular localization of CPE (
Discussion
In the present work we provide immunohistochemical and immunoelectron microscopic evidence for the expression of two related carboxypeptidases in various cell types in the placenta and umbilical cord. Previous work had shown that CPD is highly expressed in human placenta (Song and Fricker 1996), whereas CPE is expressed at only low levels. Our immunohistochemical studies confirm this previous observation. Intense CPD immunostaining was present in basal plate extravillous trophoblasts and endothelial cells, chorionic villous endothelial and stromal cells, amniotic epithelial cells, and umbilical cord vascular smooth muscle cells, CPE immunoreactivity was relatively weak in the same cell types in the placenta and intense in the umbilical cord. Whereas the distribution of CPD between the placenta and umbilical cord is rather even, a much greater proportion of CPE immunoreactivity is observed in the umbilical cord than in the placenta. The same pattern in staining intensity was observed regardless of which combinations of CPD and CPE antisera were compared. A variety of hormones and neuroendocrine peptides have been previously found in the placenta by immunohistochemistry and support a role for the diffuse neuroendocrine system (DNES) in the modulation of placental function (Graf et al. 1996). The consistent staining pattern seen in the present work, coupled with the previous finding of low-level CPE expression in human placenta, suggests that CPD rather than CPE is the primary processing carboxypeptidase in these cell types.
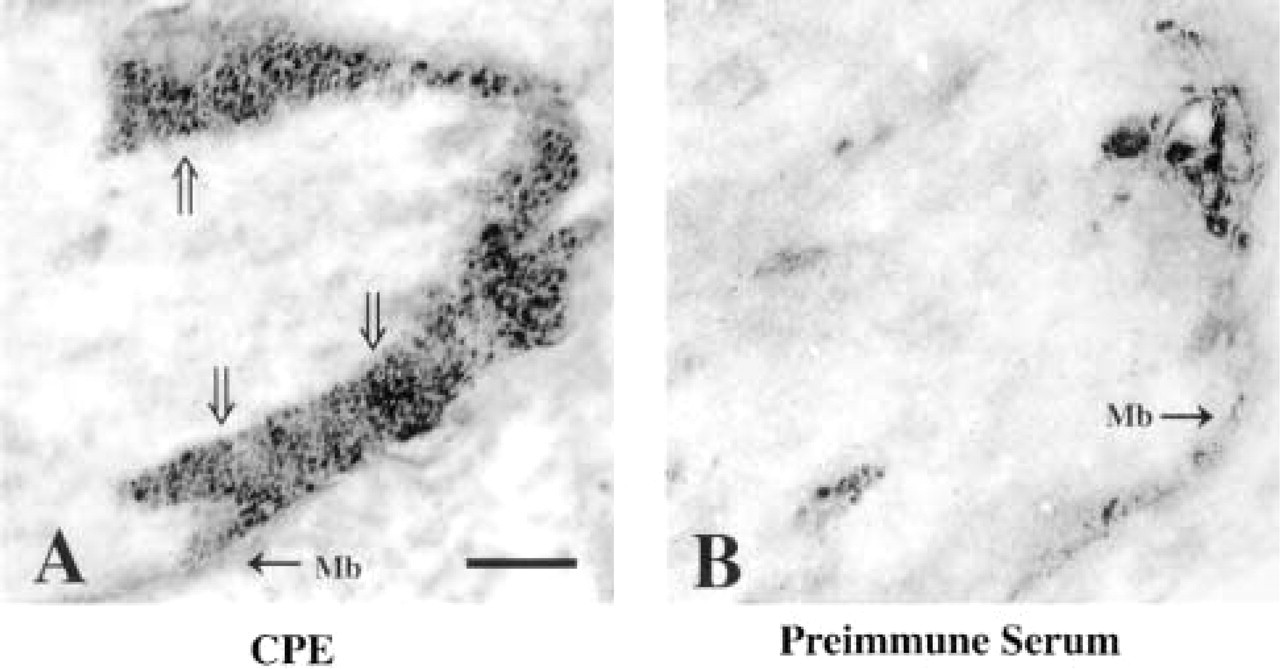
CPE immunostaining along umbilical venous smooth muscle cell membranes. (
Both the placenta and umbilical cord lack innervation and therefore must effect cell-to-cell communication under the influence of a variety of autocrine and paracrine factors. Many of these peptides are processed by the removal of C-terminal basic amino acid residues from intermediate peptide precursors. One example is insulin growth factor II (IGF-II), a singlechain polypeptide that shares 62% homology with proinsulin, a known CPE substrate (Naggert et al. 1995). IGF-2 has a broad distribution within the placenta (Fant et al. 1986; Shen et al. 1986; Zhou and Bondy 1992) and has been localized to syncytiotrophoblast and amnion by immunohistochemistry (Hill et al. 1993).
CPE and CPD may also be involved in the processing of the precursor to ET-1, a potent vasoconstrictor peptide produced by endothelial cells (Yanagisawa et al. 1988), which is implicated in the pathogenesis of pregnancy-induced hypertension and pre-eclampsia (Kamoi et al. 1990; Masaki and Yanagisawa 1992; Sudo et al. 1993), and pre-term labor (Romero et al. 1992; Marinoni et al. 1995) and is localized by immunohistochemistry to chorionic villous endothelial cells (Graf et al. 1996). ET-1 is unusual in that its final processing requires an endopeptidase, endothelin-converting enzyme-1 (ECE-1), which has the single known function of cleaving a Trp-Val bond in Big ET-1 to release the active peptide ET-1 (Matsumara et al. 1990; Takahashi et al. 1993). The synthesis of Big ET-1, in turn, requires first the processing of prepro ET-1 by an endopeptidase to produce Arg extended Big ET-1 (Turner and Murphy 1996), and then the removal of the C-terminal basic amino acid residue by a carboxypeptidase.
In the present work, CPD and CPE are co-localized to vascular smooth muscle cells and the myofibroblasts of Wharton's jelly in the umbilical cord. Both of these cell types contain immunoreactivity for epidermal growth factor and transforming growth factora (Rao et al. 1995), both of which require carboxypeptidase processing. Furthermore, vascular smooth muscle cells in other tissues produce insulin-like growth factor, insulin growth factor binding proteins 3, 4 and 6, platelet-derived growth factor, fibroblast growth factor (Cohick et al. 1993; Arnqvist et al. 1995; Delafontaine 1995), transforming growth factorb 1 and 2 and activin A (Kanzaki et al. 1995), and transforming growth factorb 3 (Stewart et al. 1996). Endothelin-1 is predominantly produced by endothelial cells and then binds to specific receptors on vascular smooth muscle cells, which, in fact, have been identified in the human umbilical artery and vein (Bogoni et al. 1996). Smooth muscle cells from human microvessels in culture were also shown to produce ET-1 (Anfossi et al. 1993).
At the ultrastructural level, the presence of immunoreactivity for both enzymes in the rough endoplasmic reticulum is consistent with their role in peptide and protein processing in umbilical venous smooth muscle cells. Although the two enzymes were co-localized at the light microscopic level, localization at the ultrastructural level also showed an important difference between CPE and CPD, i.e., the presence of CPE immunoreactivity in an additional site subjacent to the smooth muscle cell membrane. The difference in subcellular localization underlines the difference in function these two enzymes may have in peptide processing. CPE is probably involved in processing in the latter parts of the secretory pathway and CPD is more likely involved in processing that occurs in the Golgi/trans-Golgi network. The endogenous subcellular localization seen in umbilical venous smooth muscle cells in the present work is consistent with the immunocytochemical localization of both CPE and CPD previously seen in NIT cell lines, derived from mouse pancreatic β-cells (Varlamov et al. 1997) and with the subcellular localization of both enzymes recently seen in AtT-20 cells (Varlamov et al. in press; and unpublished work). The data presented suggest that both CPE and CPD are involved in the processing of at least some of the many placental and umbilical peptides that have autocrine and/or paracrine functions. The difference in immunoelectron microscopic localization between the two enzymes indicates that although there may be some overlap, they are involved in the processing of different neuroendocrine peptides and proteins.
Footnotes
Acknowledgments
Supported by National Institutes of Health Grants 1K08HD01209–01 (to SR) and DK-51271, DA-04494, and RSDA DA-00194 (to LDF).
We gratefully acknowledge Dr Yvonne Kress for her assistance with electron microscopy and Rachel Tucker for expert technical advice on immunohistochemistry.
