Abstract
Membranous (M)-cells are specialized epithelial cells of the Peyer's patch domes that transport antigens from the intestinal lumen to the lymphoid tissue. Vimentin is a reliable marker for M-cells in rabbits. Using immunohistochemistry (IHC), a subpopulation of epithelial cells has recently been identified in ordinary rabbit ileal villi, which are vi-mentin-positive and share morphological characteristics with the M-cells of the domes. To test the hypothesis that these cells represent M-cells outside the organized lymphoid tissue, lectin labeling and tracer uptake experiments were performed. Lectins specific for N-acetyl-glucosamine oligomers selectively bound to the vimentin-positive villous cells but not to M-cells in the domes. Microbeads instilled into the ileal lumen were taken up by M-cells within 45 min but not by the vimentin-positive cells in the villi. Lectin-gold labeling on ultrathin sections revealed that the lectin binding sites were located in the brush border and in vesicles in the apical cytoplasm. The vimentin/lectin-positive cells shared ultrastructural characteristics with the so-called “cup cells.” We conclude (a) that the vimentin-positive cells in ordinary villi represent cup cells but not M-cells, (b) that they are readily detectable by (GlucNAc)N-specific lectins, and (c) that they do not transcytose experimental tracers. Although the specific function of cup cells is still obscure, they most probably represent a cell type distinct from M-cells of the domes with respect to both function and expression of the two new markers.
T
Immunohistochemistry (IHC) for the intermediate filament protein vimentin can be used to detect M-cells in the rabbit (Gebert et al. 1992; Jepson et al. 1992). Many functional studies have confirmed that vimentin expression and tracer transport are closely related (Jepson et al. 1993b,d; Meynell et al. 1999; Mantis et al. 2000). Recently, individual vimentin-positive cells in the villi of the rabbit ileum, i.e., outside the domes of the Peyer's patches, were described (Fujimura and Iida 2001; Iwatsuki et al. 2002). Because these cells possessed morphological characteristics of (immature) M-cells, e.g., short microvilli and an electron-lucent cytoplasm (Bye et al. 1984), they were considered to represent villous M-cells (Iwatsuki et al. 2002). The presence of M-cells in ordinary villi would imply that antigenic matter can be transported from the lumen into the epithelium and lamina propria and that the potential pathogens come into contact with immunocompetent cells outside the organized gut-associated lymphoid tissue (GALT). Because the composition of lymphocyte subtypes and antigen-presenting cells largely differs between the organized lymphoid tissue of the domes and the diffusely distributed lymphoid cells of ordinary villi (Ermak and Owen 1986; Bjerke et al. 1988; Farstad et al. 1994; Brandtzaeg et al. 1999), it is unclear what kind of immune response could be initiated by the assumed M-cell-mediated antigen transport in the villi.
The aim of the present study was to test the hypothesis that the vimentin-positive cells in the villi of the rabbit ileum possess structural, histochemical, and functional characteristics of M-cells. Therefore, we tested potential histochemical markers to identify similarities and differences between the two vimentin-positive cell populations. Because lectins are useful tools to differentiate epithelial cell types, we used a panel of 26 lectins, including all typical saccharide specificities, that are typically expressed on intestinal epithelial cells. Using confocal fluorescence microscopy and lectin-gold-labeled ultrathin sections, we identified specific markers for the suspicious vimentin-positive cells in the villi and characterized their ultrastructure. In addition, uptake studies were performed using fluorescent latex microbeads as particulate tracers to investigate whether transepithelial transport similar to that of dome epithelial M-cells is performed by the vimentin-positive cells of the villi.
Materials and Methods
Animal Experiments and Tissue Preparation
Adult New Zealand rabbits (n = 11), fed standard laboratory diets, were sacrificed by cervical dislocation or intracardial injection of T61 (Hoechst, Frankfurt/M., Germany). The peritoneal cavity was opened and small pieces of the ileum, some including Peyer's patches, were removed and briefly rinsed in saline. Samples for fluorescence microscopy were frozen and stored in liquid nitrogen. Those for the electron microscopic methods were stored in the fixatives described below. The animal experiments had been approved by the local government of Lower Saxony/Germany (TS-no. 604-42502-94/719 and 509-42502-98/131).
Lectin Labeling
Cryosections (5-μm, 12-μm, 30-μm, and 60-μm thick) were fixed in a mixture of methanol and acetone for 10 min at −20C and transferred to PBS. The sections were incubated with a solution containing an anti-vimentin monoclonal antibody (MAb) (dilution 1:800 or 1:1600; clone V9; Sigma, Deisenhofen, Germany) conjugated to the red fluorescent dye Cy3 and one of the lectins (5–20 μg/ml) for 2 hr.
The following lectins conjugated with fluorescein (FITC) or biotin were tested in preliminary experiments to identify specific binding to the vimentin-positive cells: Abrus precatorius (APA), Arachis hypogaea (PNA), Bandeiraea simplici-folia I, isolectin B4 (BS-I-B4), Bandeiraea simplicifolia II (BS-II), Bauhinia purpurea (BPA), Canavalia ensiformis (ConA and succinylated ConA), Datura stramonium (DSA), Glycine max (SBA), Helix aspera (HAA), Helix pomatia (HPA), Jacalin (JAC), Lotus tetragonolobus (LTA), Lycopersicum esculentum (LEA), Maackia amurensis (MAA), Phaseolus vulgaris (PHA-L and PHA-E), Phytolacca americana (PWA), Psophocarpus tetragonolobus (WBA), Ricinus communis I (RCA-I), Sambucus nigra (SNA), Solanum tuberosum (STA), Triticum vulgaris (WGA), Ulex europaeus I (UEA-I), Vicia villosa (VVA), and Wisteria floribunda (WFA).
Biotinylated lectins (BPA, DSA, LTA, STA, WBA) were detected using streptavidin conjugated to the green fluorescent dye AlexaFluor 488 (Molecular Probes; Leiden, The Netherlands). The lectins MAA and SNA were purchased from EY Labs (San Mateo, CA), the others from Sigma. Nuclei were stained using the blue fluorescent dye bisbenzimide Hoechst 33258 (0.1 μg/ml in PBS for 1 hr; Sigma). Controls were performed by replacing the primary vimentin antibody with buffer and by preincubation of the lectins with their corresponding mono-, di-, or oligosaccharides overnight.
For lectin double labeling, cryosections were incubated with a mixture of LEA-FITC (20 μg/ml), STA-biotin (10 μg/ml), and unconjugated anti-vimentin antibody V9 (1:800). After rinsing in PBS the sections were incubated in a mixture of streptavidin conjugated to the red fluorescent dye AlexaFluor 555 (2.5 μg/ml; Molecular Probes) and a goat anti-mouse antibody conjugated with the infrared-emitting fluorescence dye AlexaFluor 647 (1:100; Molecular Probes). Nuclei were counterstained with bisbenzimide as described above.
Fluorescence Microscopy
Cryosections of lectin labelings and microbead uptake experiments were examined using a conventional fluorescence microscope equipped with separate filter sets for blue, green, and red fluorescence, and a digital CCD camera (Zeiss; Jena, Germany). To resolve cellular details in thick sections (30-μm thick or more), individual sections were examined using a Zeiss LSM 510UV Meta confocal laser scanning microscope, equipped with lasers for 364-nm, 488-nm, 543-nm, and 633-nm excitation. In addition to fluorescence channels, a differential interference contrast (DIC) image was recorded simultaneously from each focal plane that allowed morphological details to be identified. Digital 3D multichannel data sets were processed using LSM 5 software (Zeiss), Photoshop software (version 6.0; Adobe, Edinburgh, UK), and the software package Erdas Imagine for geographical imaging (version 8.4; Erdas, Atlanta, GA).
Ultrathin Sections and Lectin-Gold Labeling
Small tissue samples were fixed in a solution of 2% formaldehyde and 0.05% glutaraldehyde (grade I; Sigma) in 0.1 M phosphate buffer, pH 7.3, for 4 hr at 4C. They were then rinsed in PBS containing 1%
The lectin-gold labeling was performed in PBS containing 1.5% bovine serum albumin-c (BSA-c; Biotrend, Cologne, Germany). Free aldehyde groups were blocked in a drop of this buffer (PBS-BSA) containing 0.7%
Freeze-fracture Replicas
Small pieces of ileal mucosa, some of them containing a dome of a Peyer's patch, were briefly rinsed in Ringer's solution and immediately fixed in a solution of 2% formaldehyde, 2.5% glutaraldehyde, and 2 mg/ml CaCl2 in 0.1 M sodium cacodylate-HCl buffer, pH 7.3, at 4C. The freeze-fracture and replication of the fracture face were performed in a BA 360 M freeze-fracture device equipped with a QSG 201 quartz crystal thin film monitor and an EVM 052 electron beam gun (all from Balzers; Liechtenstein) at 2.7 × 10−4 Pa and −100C. The replicas were cleaned in commercial bleach, chromic acid, and distilled water, collected on formvar-coated copper grids, and examined in a Philips EM400T electron microscope.
Microbead Uptake Experiments
Adult New Zealand rabbits (n = 10) were anesthetized with 100 mg ketamine (Alprecht; Aulendorf, Germany) IM, 5 mg midazolam (Curamed; Karlsruhe, Germany) IV, 10 mg Propofol-Lipuro 1% (Braun; Melsungen, Germany), and 150 mg Temgesic (Essex; Munich, Germany) IV. After intubation, the rabbits inhaled a mixture of oxygen and isoflurane (Abbot; Wiesbaden, Germany) for 1 hr or longer.
The abdominal cavity was opened and isolated loops of 20–30-mm length, some containing a Peyer's patch, were prepared in situ from the ileum without disturbing the blood supply. They were then filled with a suspension of green fluorescent latex microbeads (diameter 0.5 μm; Polysciences, Eppelheim, Germany) in a dilution of 1:20 in PBS. The loops were removed after 45 min, briefly rinsed in saline, and frozen in liquid nitrogen. The animals were sacrificed by IV injection of T61. Vimentin labeling and microscopy were performed as described above.
Results
Vimentin-positive Epithelial Cells
Cryosections of rabbit ileal Peyer's patches labeled with the monoclonal anti-vimentin antibody (clone V9) revealed that M-cells, comprising almost 50% of all epithelial cells in the domes, were intensely labeled, whereas enterocytes and lymphocytes of the dome epithelium remained unstained. In villi distant from the Peyer's patch domes, individual columnar cells were vimentin-positive (V9+; Figure 1a). These epithelial cells were columnar in shape, reached the gut lumen with a brush border, and contained a network of vimentin filaments that were most dense in the perinuclear cytoplasm (Figure 1b). The V9+ epithelial cells comprised 1.23 ± 1.05% of all epithelial cells in the villi (58 of 5724 cells counted in 24 randomly selected villi from five individuals). In many cases the nuclei of the V9+ villous epithelial cells lay above the level of the nuclei of the surrounding enterocytes (Figure 1b and 2a). The density of V9+ cells varied slightly from villus to villus so that one to five V9+ cells were found per section plane of a single villus. The V9+ cells were only rarely sectioned along their long axis in conventional cryosections (5-μm), so that either the apical or the basal cell pole was present in the section plane. Therefore, thick sections (e.g., 30-μm) examined by confocal microscopy were used for the following lectin labeling.
Lectin Labeling
Preliminary tests with the panel of 26 lectins listed above revealed that the lectins LEA and STA selectively labeled the V9+ cells in the villi (Figures 1–3). Enterocytes and goblet cells, which could readily be identified in DIC, remained unstained. Weak staining of mucus in goblet cell secretory granules and in the gut lumen was seen in the majority of the samples. This labeling intensity with strong binding of LEA and STA to V9+ cells was found in 8/11 rabbits examined in total. The remaining individuals showed less intense lectin labeling and stronger staining of mucus in the goblet cells. The two lectins bound to the apical cell pole of the V9+ cells, probably to the brush border and apical cytoplasm (Figure 1 and 2a). Confocal microscopy of thick sections revealed that, when the apical cell pole of a V9+ cell was in the volume of the section, it was strongly positive for LEA (or STA). High-resolution optical scans demonstrated that the binding pattern in the apical cytoplasm was granular and that the basolateral membrane was lectin-positive in some of the V9+ cells.
In cryosections that contained both ileal villi and Peyer's patch domes, the M-cells were identified by their vimentin content, their cell shape, and their typical association with clusters of lymphocytes. Neither immature M-cells at the base of the domes nor mature M-cells on the upper flanks bound the lectins STA and LEA (Figure 2b). LEA and STA binding epithelial cells were even completely absent from the more than 50 domes screened in total. The remaining 24 lectins tested displayed different labeling patterns as described previously (Gebert and Hach 1993), but none of them specifically bound to both populations of V9+ cells (i.e., dome epithelial M-cells and V9+ villous epithelial cells).
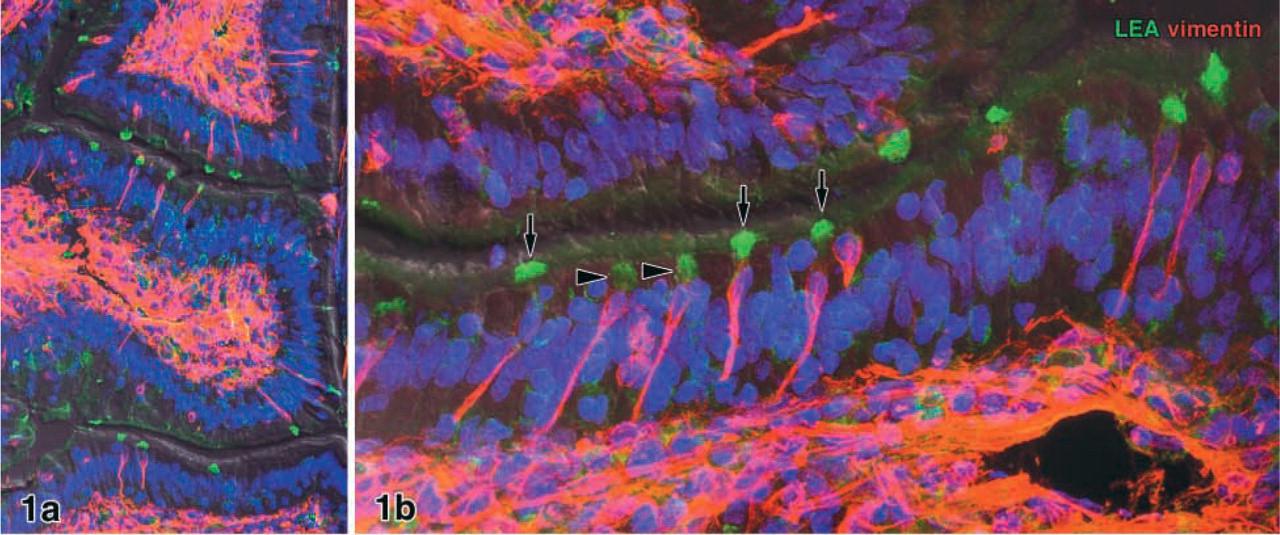
Cryosections 20 μm thick of rabbit ileum. Individual vimentin-positive (V9+) epithelial cells (red) are found in the villous epithelium. Confocal microscopy reveals that the V9+ epithelial cells selectively bind the lectin LEA at their apical cell pole (green). When the brush border of a V9+ cell is in the section volume, it is strongly labeled by the lectin (arrows in
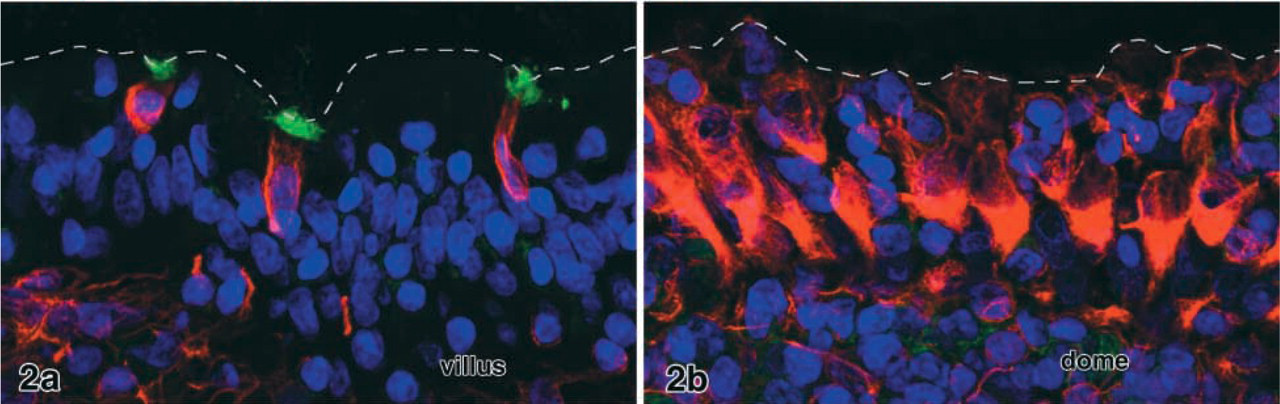
Villous epithelium (
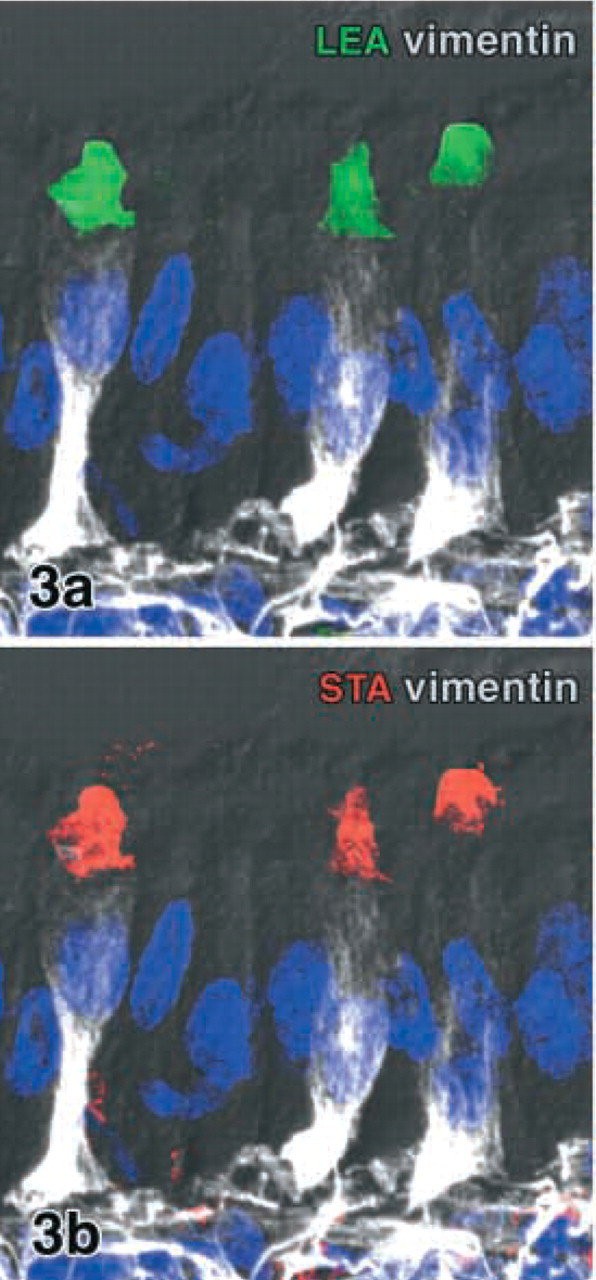
Vimentin-positive cells in the rabbit villous epithelium triple labeled for LEA (green), STA (red), and vimentin (infrared, white). Binding sites of LEA (
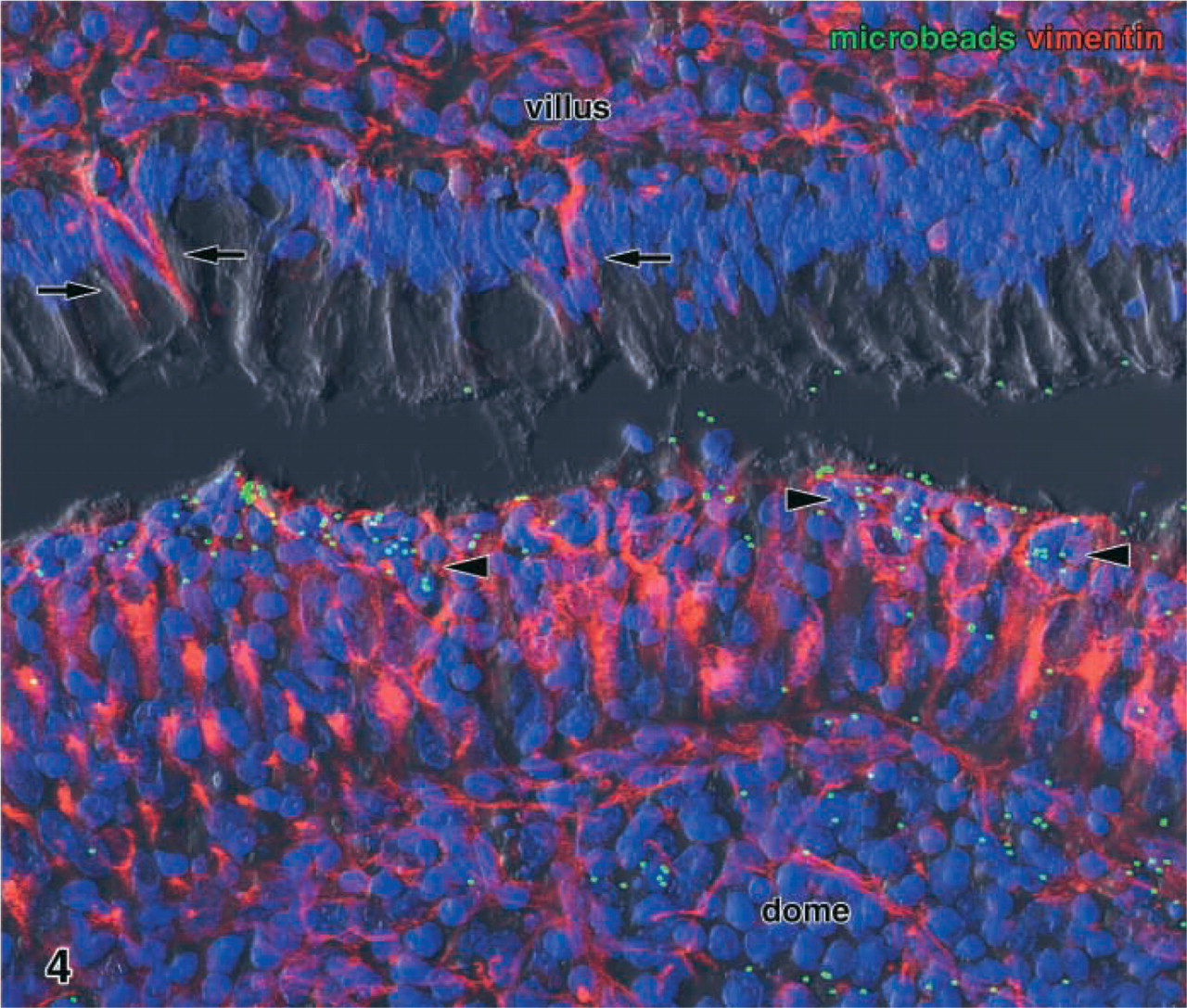
Fluorescent latex microbeads (green dots), instilled in vivo into the gut lumen for 45 min, were avidly taken up by the M-cells (red, vimentin immunolabeling) of the dome epithelium (lower half). The tracer was not only enriched in the pockets of the M-cells (arrowheads) but was also transported to the subepithelial lymphoid tissue (lower right corner). Although some of the microbeads adhered to the brush border of the villous epithelium (upper third), neither enterocytes nor vimentin-positive epithelial cells of the villi (arrows) endocytosed the tracer. Maximal projection of 19 confocal sections. Green, latex microbeads; red, vimentin; blue, nuclei; dark gray, DIC. Original magnification ×580.
Lectin double labeling of LEA with STA revealed that the two lectins bound to cells with identical subcellular labeling patterns (Figure 3). After preincubation of the lectins LEA and STA with N,N',N”-triace-tylchitotriose, their binding intensity was reduced although not completely abolished.
Lectin-Gold Labeling on Ultrathin Sections
Ultrathin sections gold-labeled with LEA or STA showed weak overall binding of the lectins to most tissue components and moderate binding to the brush border of enterocytes. A small number of epithelial cells (up to two per cross-sectioned villus) intensely bound the two lectins on their brush border (Figure 5 and 6). These individual lectin-positive epithelial cells were found in all of the five rabbits examined by lectin-gold labeling. The cells were columnar in shape and possessed microvilli that were shorter and slightly thicker than those of the neighboring enterocytes (Figure 5a). The brush border of many of these cells was indented so that the luminal surface was concave or cup-like (Figure 5a and 5b). The cytoplasm was less electron-dense than that of adjacent enterocytes, and small electron-lucent vesicles were present in its apical parts; most of them were lectin-positive (Figures 5a and 6). Some of the LEA+ and STA+ cells bound the lectin along the basolateral membrane (Figure 6b). The binding patterns of the lectins LEA and STA were almost identical. Other structures moderately labeled by the two lectins in the villous epithelium were the cell surface of intraepithelial lymphocytes and mucous granules in the goblet cells. The apical cell pole of a few enteroendocrine cells, identified by their typical electron-dense granules, was labeled by the LEA lectin, but such lectin-positive enteroendocrine cells were extremely rare and were found in only 3/85 grids examined in total.
The epithelial cells of the domes, i.e., M-cells and dome epithelial enterocytes, did not bind the lectins LEA and STA on their apical membrane (Figure 5b). However, most of the lymphocytes present in M-cell pockets were intensely stained along their cell surface (arrows in Figure 5b). Preincubation of the two lectins with their corresponding oligosaccharides resulted in reduced but not completely abolished binding. Gold labeling performed with other lectins as controls (WGA, UEA-I, PWA, DSA) revealed completely different labeling patterns, with no lectin specifically binding to the LEA+/STA+ cells and the M-cells in the domes.
Freeze-fracture Electron Microscopy
Freeze-fracture replicas of the rabbit ileum contained the epithelial layer of the villi, consisting of enterocytes, goblet cells, and a few enteroendocrine cells, but brush cells, often seen in rat and mouse ileum, were not observed. Individual epithelial cells with the morphological characteristics of the V9+/LEA+/STA+ cells described for ultrathin sections could be identified in the freeze-fracture replicas as well. Compared to adjacent enterocytes, these cells possessed an often concave, cup-like brush border (Figure 7a) as well as shorter and thicker microvilli (length about 1.2 μm; diameter 123 ± 7 nm, n=41; enterocytes 107 ± 7 nm, n=23). Whereas intramembranous particles (IMPs) present on P-faces were randomly distributed over the microvilli of enterocytes, the V9+/LEA+/STA+ cells contained linear aggregates of IMP on their P-faces, and corresponding grooves on the E-face of the microvillous membrane (Figure 7b). These arrays consisted of globular and rod-shaped particles and short fibrils that were orientated along an axis tilted 45° or less to the longitudinal axis of the microvillus. The present investigation and a previous detailed freeze-fracture study on the rabbit Peyer's patch dome epithelium (Gebert and Bartels 1991) revealed that epithelial cells with such typical arrays of IMPs were absent from the dome epithelium and that both mature and immature M-cells did not possess these unique membrane structures.
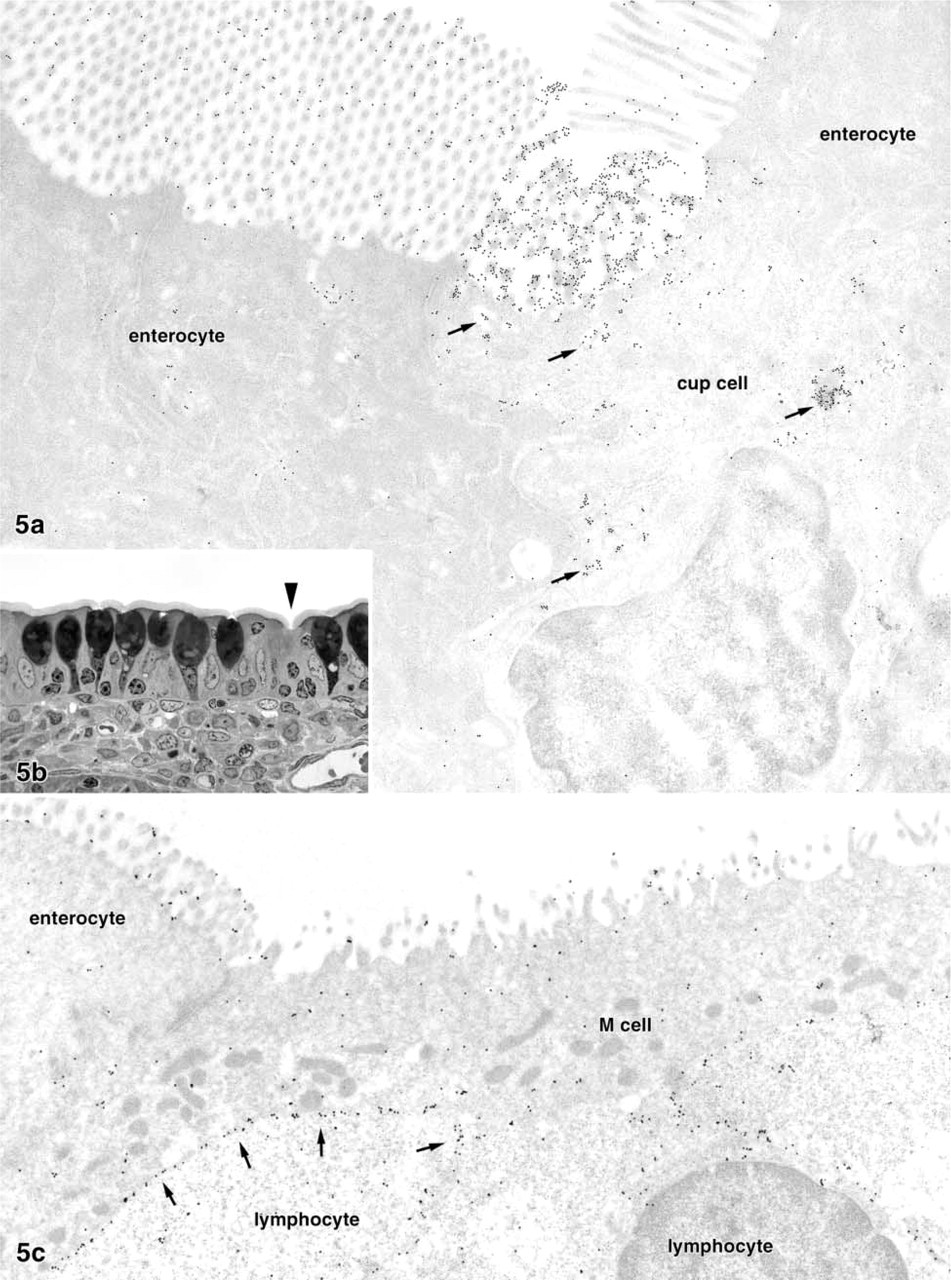
LEA-positive epithelial cell in a lectin-gold-labeled ultrathin section (LEA-gold 10 nm,
Microbead Uptake Experiments
Cryosections of ileal Peyer's patches instilled with latex microbeads in vivo displayed large numbers of green fluorescent particles in the lumen above the domes and around the villi (Figure 4). Most domes contained many microbeads and could easily be identified in the sections without further M-cell labeling, even at low magnification. The particles were most numerous in the epithelium at the flanks of the domes and in the underlying lymphoid tissue. When micro-beads were present in the cytoplasm of dome epithelial cells, these cells were V9+ and therefore represented M-cells (Figure 4).
Some fluorescent microbeads were attached to the brush border of the villous enterocytes, but no particles were seen in the epithelium and the lamina propria of the villi (Figure 4), except for a few regions where the epithelial layer was damaged and disrupted (probably because of mechanical alterations during ligation of the gut or dissection of the tissue). More than 300 V9+/LEA+ cells of the ileal villi were examined for microbeads lying in their cytoplasm or in close vicinity to them in the epithelium. None of these V9+/LEA+ cells contained fluorescent particles in the cytoplasm, and no particles were found close to them in the epithelium or lamina propria. In addition, adherence of microbeads to the brush borders of these cells was extremely rare and no more frequent than that of villous enterocytes.
Discussion
Previous studies on GALT demonstrated that the M-cells in the dome epithelia of rabbits contain large amounts of vimentin, whereas this intermediate filament protein is absent from other dome epithelial cells (Gebert et al. 1992; Jepson et al. 1992). Since that time, vimentin has been employed for the detection of M-cells at other mucosal sites of the rabbit, such as the large intestine, tonsils, and airways (Gebert and Hach 1993; Jepson et al. 1993a; Gebert et al. 1996). Functional studies confirmed that the vimentin-containing cells, but not the other cells in the dome epithelium, perform transcytosis of experimentally applied tracers (Jepson et al. 1993b,d) and therefore represent the antigen-sampling (M)-cells.
The presence of individual vimentin-containing epithelial cells in ordinary villi of the rabbit ileum outside the Peyer's patches has recently been reported, and it was hypothesized that these columnar cells might represent cup cells (Fujimura and Iida 2001) or M-cells similar to those in the domes of GALT (Iwatsuki et al. 2002). In the present study we reproduced these experiments using different antibodies, detection techniques, and microscopic methods, and found that 1–2% of the villous epithelial cells in normal rabbit ileum contain vimentin filaments. However, our results show that these vimentin-containing cells represent a population distinct from M-cells, because they differ considerably from mature and immature M-cells in the glycosylation pattern of their apical and cytoplasmic membranes and do not transcytose an experimentally applied tracer. In addition, the vimentin-containing cells in the villi bear linear arrays of particles in their apical membrane, which are known to be characteristic for the so-called “cup cells” described for various species (Neutra 1979; Madara 1982; Madara and Carlson 1985).
The vimentin-positive epithelial cells in the villi are columnar in shape, contain electron-lucent vesicles in their apical cytoplasm and possess relatively short microvilli. They therefore resemble the immature M-cells found near the base of the Peyer's patch domes (Bye et al. 1984; see also Iwatsuki et al. 2002). Immature M-cells are typically associated with only a few or even no lymphocytes and do not possess an elaborate pocket-like invagination of the basolateral membrane (Bye et al. 1984). Although such morphological characteristics were likewise observed for the vimentin-containing cells in the villous epithelium (present study; and Iwatsuki et al. 2002), lectin histochemistry revealed that the two cell types differ in the expression of N-acetyl-glucosamine oligomers. The lectins LEA and STA, which are both specific for di-, tri-, and tetramers of this saccharide (Allen and Neuberger 1973), intensely bound to the vimentin-containing cells in the villi but not to mature and immature M-cells of the domes. Interestingly, the lectin DSA, which has a similar although not identical saccharide specificity (Crowley et al. 1984), did not bind preferentially to these cells, indicating that cup cells express a constant and highly defined saccharide sequence in their brush borders. This interpretation is further supported by our lectin double labeling, demonstrating a constant binding of LEA and STA to all vimentin-containing epithelial cells in the villi.
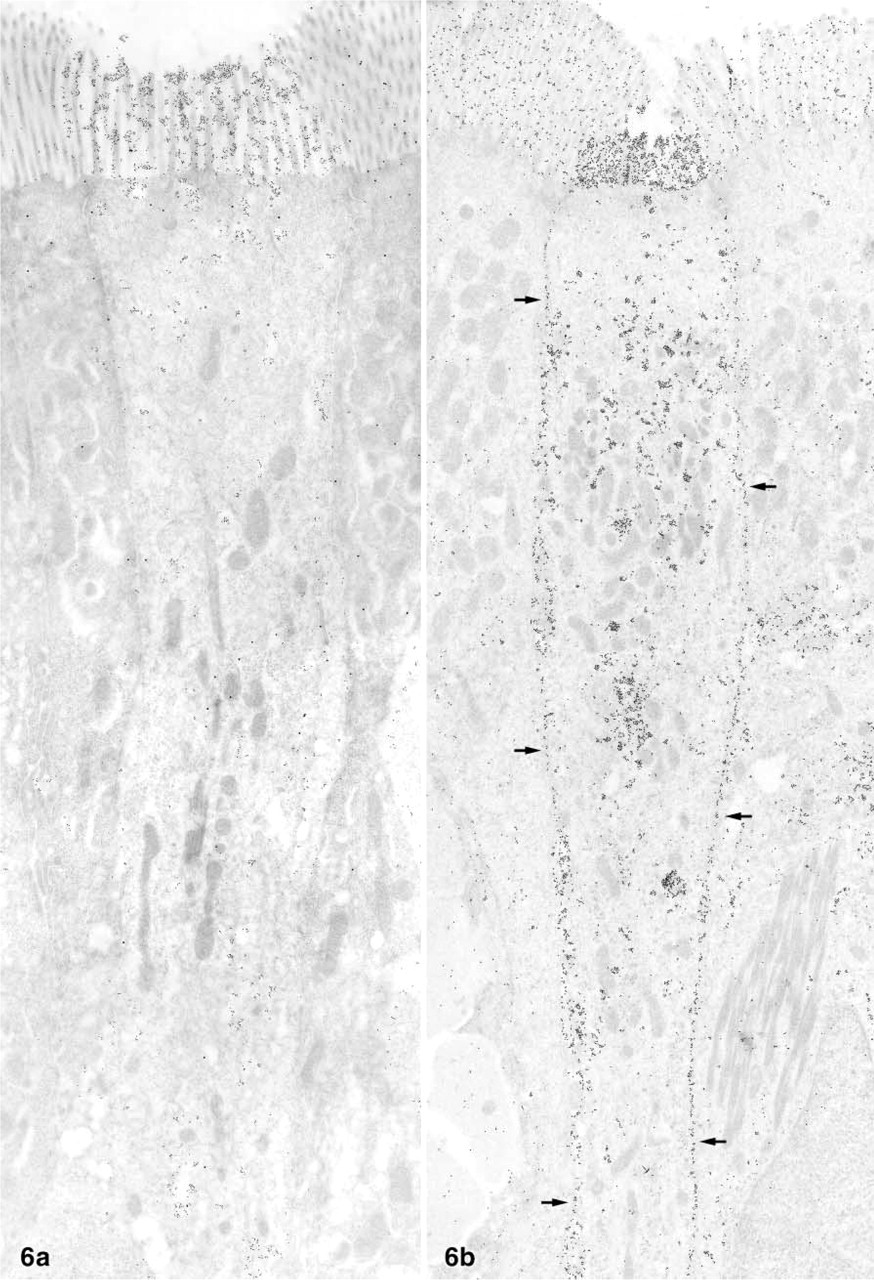
Survey micrographs of the ileal villous epithelium gold-labeled using the LEA lectin. The two cup cells depicted densely bind the lectin to their brush borders and to cytoplasmic vesicles. However, the cup cell in
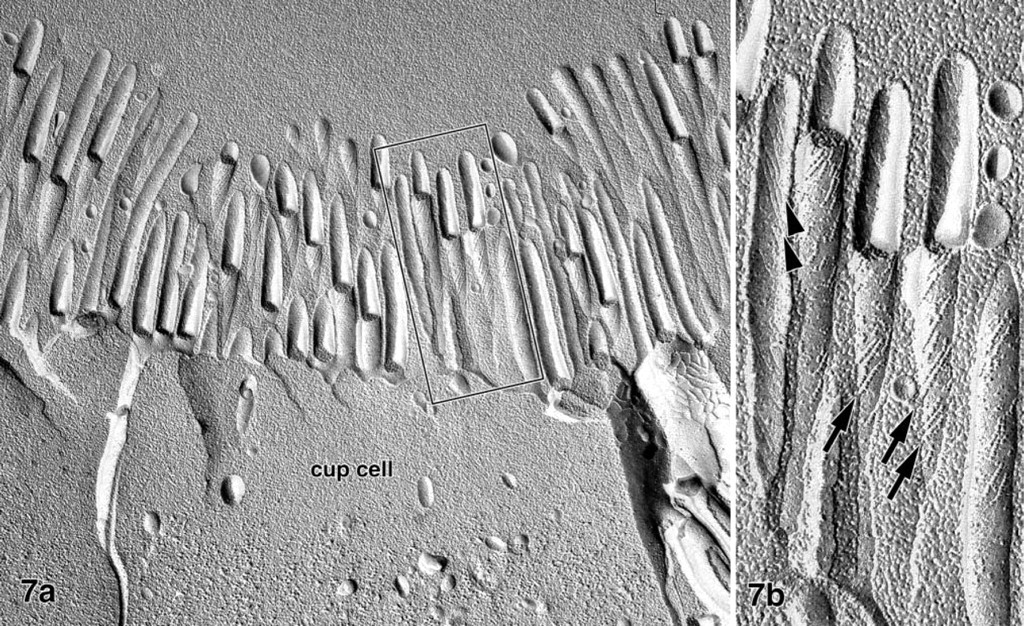
Freeze-fracture replica of the rabbit ileum. Whereas intramembranous particles are randomly distributed in the brush border membranes of most epithelial cells, a few cells (cup cells) contain linear arrays of globular and rod-shaped particles on the P-face of their microvillous membrane (arrows in
Lectins represent excellent markers for the distinction of epithelial cell types and differentiation pathways (Falk et al. 1994), and many studies have revealed that the M-cells of mice and rabbits express specific glycoconjugates on their apical membrane that were sucessfully used as M-cell markers (Clark et al. 1993; Gebert and Hach 1993; Jepson et al. 1993a; Gebert et al. 1996,1999). We therefore conclude that the differences in lectin binding between vimentin-containing cells in villi and domes reflect distinct glycosylation pathways in two different cell types. This view is further confirmed by studies in which other histochemical markers for rabbit M-cells were established, mostly unknown epitopes detected by monoclonal antibodies (Roy et al. 1987; Pappo 1989; Lelouard et al. 1999). Although these markers detected most if not all M-cells in the domes, marker-positive cells were described to be absent from the villi, indicating that these markers also might have discriminated M-cells and cup cells. Although M-cells of different GALT sites may display divergent glycocon-jugates in their apical membranes (Gebert and Hach 1993; Jepson et al. 1993a), no such variations have yet been described for different Peyer's patches in the same gut segment, e.g., the ileum, as investigated in the present study. Therefore, differences in the lectin binding properties among epithelial cells of the ileum most probably reflect distinct cell types.
In the present study, no uptake of microbeads by vimentin-containing cells was observed in the villi, whereas the M-cells in the domes actively transcytosed the tracer and thus served as positive controls in this well-established antigen sampling model (Pappo and Ermak 1989; Jepson et al. 1993c; Meynell et al. 1999). It could be objected that the vimentin-containing cells in the villi are immature M-cells, as hypothesized by Iwatsuki et al. (2002). Immature M-cells in the domes, however, do transport tracers (Bye et al. 1984) and, within hours, develop into mature M-cells (Bhalla and Owen 1982; Bye et al. 1984) that acquire the typical morphological features of M-cells (e.g., a large pocket that contains clustered lymphocytes). The absence of such aggregates further supports our view that M-cells do not occur in ordinary villi and that the vimentin-containing cells in the villous epithelium represent a cell type distinct from M-cells.
Our lectin labeling at the electron microscopic level allowed the V9+/LEA+/STA+ cells of the villous epithelium to be identified ultrastructurally and revealed that they possess several characteristics of the so-called “cup cells” (Madara 1982). Because the unique feature of cup cells and of closely related cells described by Neutra (1979) in primate large intestine consists of linear arrays of particles in the microvillus membrane, freeze-fracture replicas were examined and they confirmed that the V9+/LEA+/STA+ cells possess such typical arrays and thus represent cup cells. Previous freeze-fracture studies revealed that the apical membrane of M-cells differs from that of enterocytes in the density of intramembranous particles (Madara et al. 1984; Gebert and Bartels 1991), but that arrays of particles characteristic for cup cells are absent from M-cell membranes. In cup cells, arrays of intramembranous particles are restricted to the microvilli (present study; and Madara 1982), while lectin binding was also found along the basolateral membrane and along that of cytoplasmic vesicles. It therefore appears not very likely that the intramembranous particles that form arrays in cup cell membranes are identical to the glycoconjugate that incorporates the binding sites for LEA and STA.
Cup cells comprise 1–6% of the epithelial cells in the ileum (present study; and Madara 1982) and therefore by far surpass the thoroughly studied enteroendocrine cells in frequency (the latter comprise 0.4% or less; Cheng and Leblond 1974). Nevertheless, the function of cup cells is still obscure, but it has been proposed that they are attachment sites for certain bacteria (Madara and Carlson 1985). In this context, the cup cell-specific glycoconjugates identified in the present study could serve as anchoring sites, because it is known that many pathogenic and commensal bacteria bind to oligosaccharide epitopes on epithelial surfaces via lectin-like substances on their outer membrane or pili (Wennerås et al. 1995). The presence of unique glyco-conjugates might reflect cup cell-specific glycoproteins that are involved in specific functions. The nature of such membrane-bound molecules in cup cells is unknown, but they might represent receptors associated with sensory or regulatory functions. Interestingly, it has been reported that individual villous cells in the rat and human intestine express the cystic fibrosis transmembrane conductance regulator (CFTR; Ameen et al. 1995; Hayden and Carey 1996). Because these cells resemble cup cells in both morphology and frequency (about 2% of the villous epithelial cells), it is tempting to speculate that the two cell types, and possibly a further cell type described by Heath et al. (1996), are identical. The present study provides for the first time histochemical markers for cup cells that might be employed in further attempts to elucidate the functions of these cell populations.
We conclude that the vimentin-containing epithelial cells in ordinary villi of the rabbit ileum represent cup cells but not M-cells. Although many ultrastructural, histochemical, and functional studies have been performed using various tracers and M-cell markers (Neutra 1998), no reports are available that can sustain the presence of M-cells outside the organised mucosal lymphoid tissues. The development of M-cells appears to be restricted to specialized crypts located at the periphery of the domes (Gebert et al. 1999; Lelouard et al. 2001), in which a unique microenvironment induces the formation of the M-cell lineage as a very early step of differentiation from stem cells (Neutra 1998). In addition, this milieu suppresses the development of goblet cells and that of cup cells, as shown in this study. It must be assumed that both induction and the immunological function of M-cells are closely related to a special GALT microenviroment that has recently been shown to be critically influenced by B lymphocytes (Golovkina et al. 1999; Debard et al. 2001).
Footnotes
Acknowledgements
Supported by the Deutsche Forschungsgemeinschaft (Ge647/3-1) and by the University of Lübeck (Schwerpunkt-programm Körpereigene Infektabwehr, C1).
The technical assistance of J. Endler, H. Manfeldt, C. Örün, G. Preiss, and D. Stöckmann is gratefully acknowledged.
