Abstract
In adult mammals, growth of new vasculature from extant blood vessels (angiogenesis) is rare in the absence of pathology. However, nonpathogenic angiogenesis occurs in the cycling ovary when the avascular postovulatory follicle transforms into a highly vascularized corpus luteum (CL). To improve our understanding of molecular mechanisms that regulate nonpathogenic vascular growth, we characterized the expression of two secreted matricellular proteins associated with angiogenesis, SPARC and thrombospondin (TSP), in postovulatory preluteal follicles and CL of hormone-primed immature rats. By indirect immunofluorescence with specific antibodies, we found SPARC in the cytoplasm of granulosa cells and thecal cells of preluteal follicles, in connective tissue cells of the ovarian interstitium, and in the oocyte nucleus. Administration of a luteinizing stimulus (chorionic gonadotropin) increased the expression of SPARC in granulosa cells. TSP was prominent in the basement membranes of growing follicles. Many cells in the early vascularizing CL expressed both SPARC and TSP. Neovascularization of CL was accompanied by expression of SPARC in nascent vessels and concentration of TSP in central avascular areas. In mature CL, steroidogenic luteal cells expressed both SPARC and TSP. Luteal cells of regressing CL retained SPARC to a variable degree but did not express TSP. The observed changes in expression of SPARC and TSP during development of the CL support distinct roles for these matricellular proteins in nonpathological morphogenesis and angiogenesis.
I
The rapid rate of neovascularization of the developing CL and the subsequent regression of vasculature that accompanies degeneration of the CL in the absence of pregnancy make the CL an ideal model for studies of molecular processes that mediate and modulate vascular growth (Bagavandoss et al. 1983; Augustin et al. 1995; Modlich et al. 1996; Tsukada et al. 1996). Despite this fact, molecules that regulate angiogenesis have been studied to a limited extent in the cycling ovary, although vascular endothelial cell growth factor (Kamat et al. 1995; Gordon et al. 1996), matrix metalloproteinases 2 and 9 (Tsang et al. 1995; Goldberg et al. 1996), tissue inhibitor of metalloproteinases 1 and 2 (Goldberg et al. 1996), urokinase plasminogen activator (uPA) (Bacharach et al. 1992), and plasminogen activator inhibitor-1 (PAI-1) (Bacharach et al. 1992) have been implicated in this process.
Among proteins that influence angiogenesis are the matricellular class of proteins (Sage and Bornstein 1991), two of which are Secreted Protein Acidic and Rich in Cysteine (SPARC) and thrombospondin-1 (TSP). Although SPARC and TSP are not related structurally, both proteins are associated with capillaries in vivo and both share certain functional characteristics. For example, SPARC and TSP inhibit cell attachment and spreading and cause partial detachment and rounding of spread cells in vitro (Sage 1997). SPARC has minimal effects on angiogenesis in vivo. However, peptide fragments of SPARC have angiogenic properties (Jendraschak and Sage 1996). TSP is believed to be anti-angiogenic in vivo (Bagavandoss and Wilks 1990; Tolsma et al. 1993). However, it is proposed that TSP can stimulate angiogenesis by indirect effects on nonendothelial cell types (Tuszynski and Nicosia 1996). In this article we report on the expression of SPARC and TSP during development of the CL of the rat.
Materials and Methods
Animals
Long-Evans rats, maintained on a 12-hr:12-hr light:dark cycle at 24C, were fed standard rodent chow (Prolabs; St Louis, MO) and water ad libitum. Immature female rats were obtained as needed by breeding of the adults. Use of animals in this study conformed to the guidelines published by the National Research Council.
Induction of Follicle and CL Development
Immature female rats 25 days of age were made pseudopregnant by injection of 15 IU of pregnant mare's serum gonadotropin (PMSG; Sigma Chemical, St Louis, MO) followed 48 hr later by 5 IU of human chorionic gonadotropin (hCG) (Sigma). Pseudopregnant rats were sacrificed at specific intervals and the ovaries were collected for immunohistochemical analyses. Specific stages of development of follicles or CL were represented by ovaries from a minimum of three animals.
Immunohistochemistry (IHC)
Isolated ovaries were fixed in Bouin's fluid (24 hr at room temperature), dehydrated, embedded in paraffin, and sectioned at 6 μm. Before IHC, deparaffinized sections were incubated for 10 min in 50 mM NH4Cl to quench aldehydeinduced fluorescence. Selected ovaries were frozen in OCT embedding medium (Lab-Tek Products; Naperville, IL) and were sectioned at 8 μm with a cryotome.
Sections processed for IHC were washed in PBS-0.2% BSA, blocked with PBS-10% BSA (30 min/25C), and exposed to PBS-0.2% BSA that contained: (a) a mouse monoclonal antibody (MAb) SSP2 against Domain I of murine SPARC (1:2000 dilution) (Porter et al. 1995), (b) a mouse MAb against human platelet TSP (MAb P12; Immunotech, Westbrook, ME) at 20 μg/ml (Clezardin et al. 1986), or (c) a rabbit polyclonal antiserum against human platelet TSP (1:10 dilution) (O'Shea and Dixit 1988). Control sections were incubated with either PBS-10% BSA or normal serum. Labeled sections were washed in PBS-0.2% BSA, exposed to secondary antibodies conjugated to fluorescein- or rhodamineisothiocyanate (Organon Teknika; Durham, NC), washed, and mounted under coverslips in Vectashield (Vector Laboratories; Burlingame, CA). Selected sections were double labeled with anti-SPARC and anti-TSP antibodies.
Specificity of TSP and SPARC Antibodies
The specificity of the anti-SPARC and anti-TSP antibodies has been characterized in previous studies. The P12 MAb against TSP immunoprecipitates the 160-kD polypeptide of TSP from human platelet releasates (Clezardin et al. 1986). The rabbit polyclonal antiserum against purified human platelet TSP immunoprecipitates both human and murine TSP (O'Shea and Dixit 1988), recognizes all chymotryptic fragments of TSP on Western blots (Majack et al. 1988), crossreacts against mouse tissues and rat cells by immunofluorescence (O'Shea and Dixit 1988; Majack et al. 1988), and specifically labels frozen sections of tissues from mouse, goldfish, newt, and Xenopus (Hoffman et al. 1994). Neither the MAb nor the polyclonal antiserum against TSP distinguishes TSP-1 from TSP-2. Anti-SPARC MAb SSP2 was raised against a synthetic peptide corresponding to region 1.1 within Domain 1 of murine SPARC and recognizes murine, bovine, and human SPARC in Western blot and immunoprecipitation assays (Porter et al. 1995). Reactivity with other proteins was not observed, and the reactivity towards murine SPARC was inhibited by prior incubation of the purified antibody with either bovine or murine SPARC peptide 1.1 (Porter et al. 1995). In IHC studies, SSP2 labeled human tissues with specificity and exhibited a high level of reactivity with murine parietal yolk sac (PYS-2) teratocarcinoma cells (Porter et al. 1995), a cell type that synthesizes high levels of SPARC in vitro (Sage et al. 1989). A specific affinity of SSP2 for rat SPARC is predicted on the basis of a close homology between region 1.1 of murine SPARC (GENBANK, accession M20684-92) and rat SPARC (DDBJ, accession D28875) that exceeds the homologies between murine region 1.1 and the corresponding sequences from bovine SPARC (SWISS-PROT, accession P13213) and human SPARC (GENBANK, accession M25738-46).
Results
Expression of SPARC and TSP During the Growth of Follicles
No fluorescent labeling of sections was observed when secondary antibodies were applied in the absence of primary antibodies (data not shown). In the PMSGprimed ovary, interstitial cells and follicular thecal cells were labeled extensively with anti-SPARC IgG (Figure 1A). Follicular granulosa cells expressed SPARC to a lesser degree than was observed in thecal cells or interstitial cells (Figure 1A). After injection of hCG, expression of SPARC by follicular granulosa cells increased relative to that of thecal cells (Figure 1B). Granulosa cells of the corona radiata that surrounded maturing oocytes within antral follicles (Figure 1C) exhibited significant levels of SPARC in the cytoplasm. SPARC was not found in the cytoplasm of oocytes. However, the nuclei of oocytes were strongly immunopositive for SPARC (Figure 1C). In contrast, granulosa cells did not express SPARC in their nuclei. TSP was expressed at high levels in the basement membranes of specific growing follicles (Figure 2). However, this expression was variable (not shown).
SPARC and TSP in the Developing CL
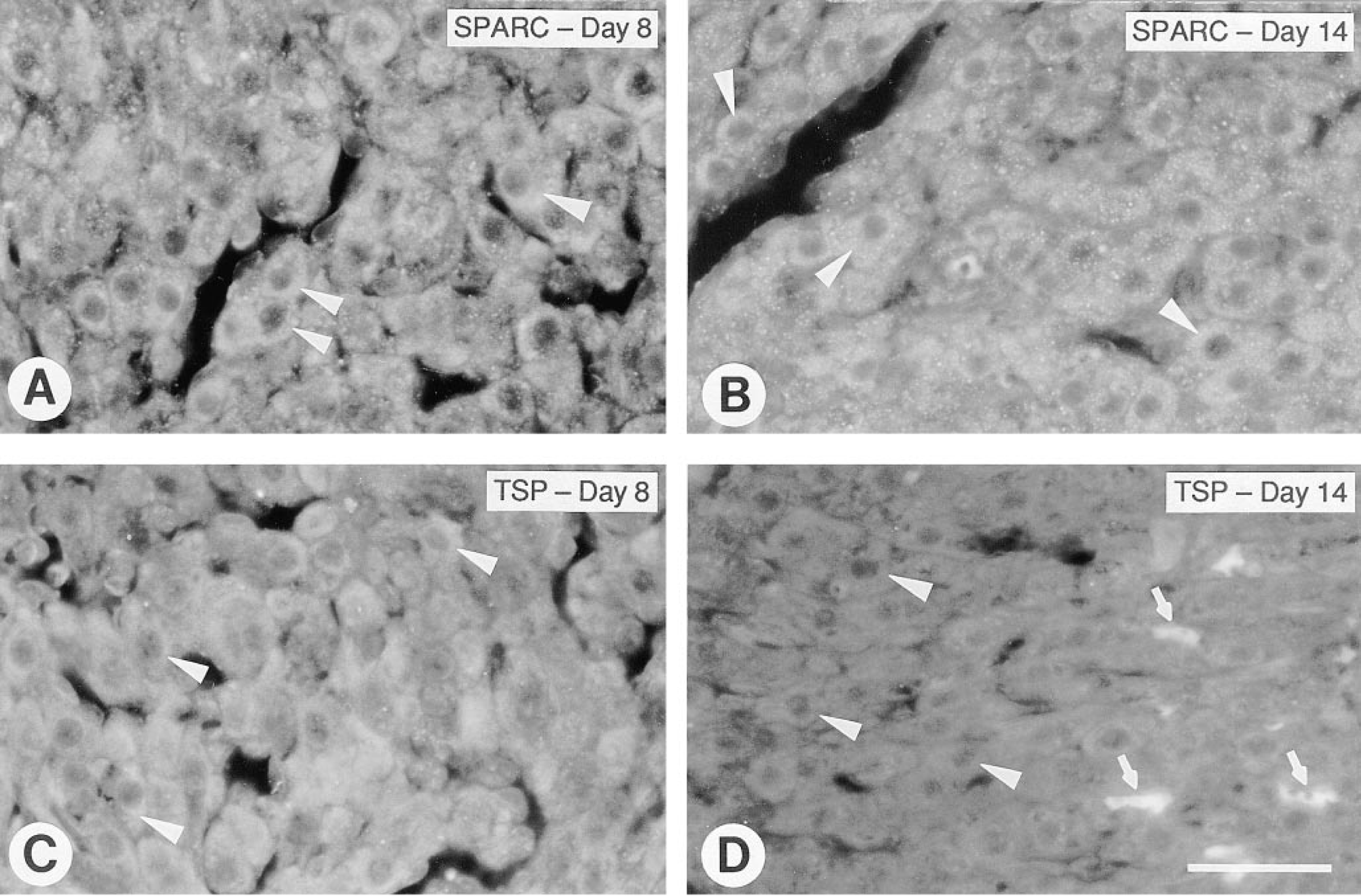
During the earliest stages of development of the CL, granulosa cells did not express detectable levels of SPARC (not shown). Granulosa cells in CL undergoing neovascularization produced both SPARC and TSP, and many cells expressed both proteins concomitantly (Figure 3). With the appearance of well-defined vasculature in CL, SPARC was expressed by microvessels throughout the CL as well as in the avascular center of the CL (Figure 4A). TSP was concentrated only in the avascular center of the CL (Figure 4B).
SPARC and TSP in Mature and Regressing CL
In mature CL, expression of SPARC by vasculature was significantly diminished. SPARC and TSP were both expressed in the cytoplasm of luteal cells (Figures 5A and 5C). In rats, regression of the CL is maximal by Day 13 of PMSG-hCG-induced pseudopregnancy because serum progesterone returns to basal levels at this time (Horikoshi and Wiest 1971; Pang and Behrman 1979). We observed that some CL in regression at Day 14 of pseudopregnancy exhibited no obvious decline in luteal cell SPARC (Figure 5B), whereas other CL showed decreased expression of this protein (not shown). In contrast, expression TSP was diminished significantly in luteal cells of all regressing CL (Figure 5D).
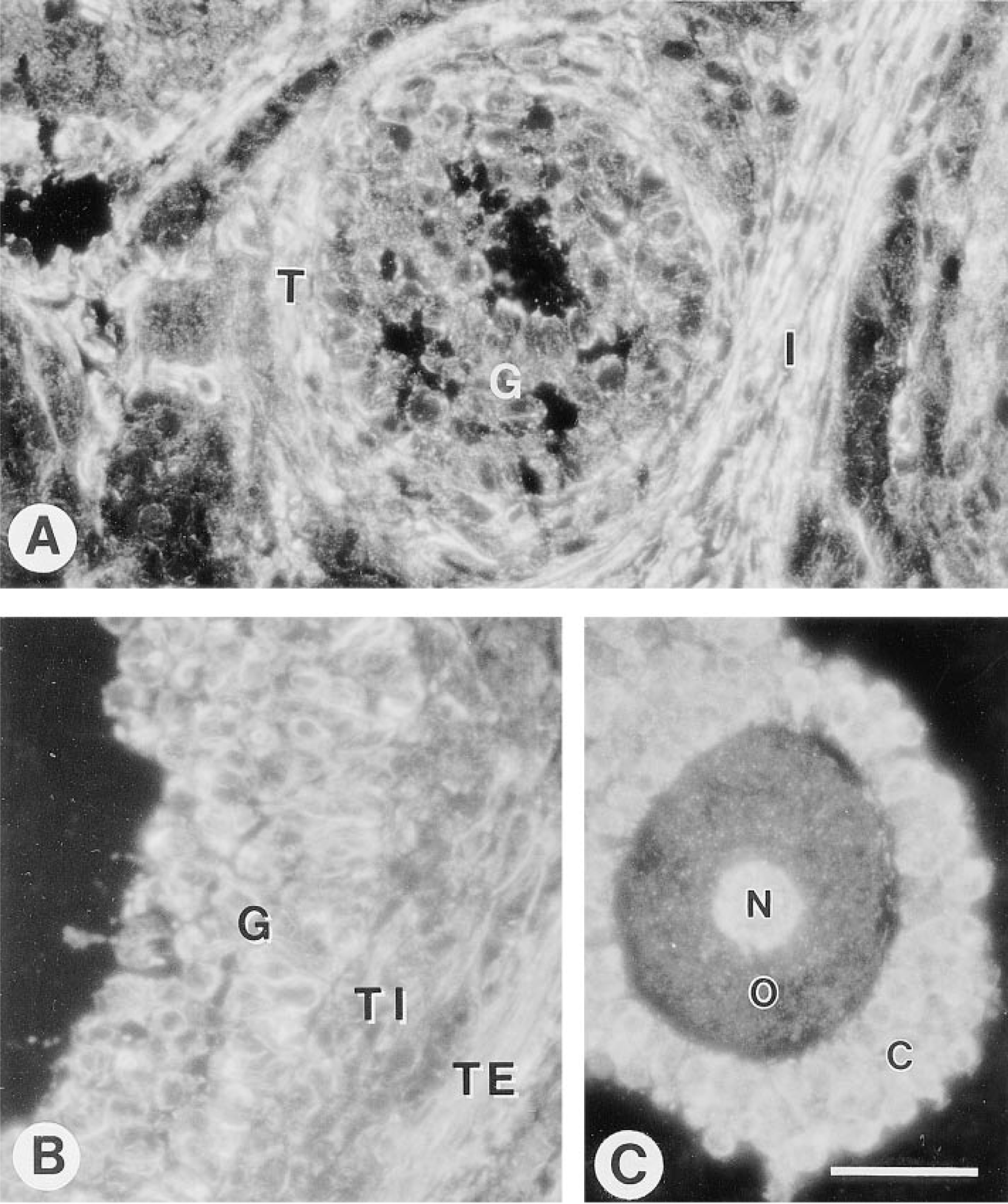
SPARC in the developing ovarian follicle.
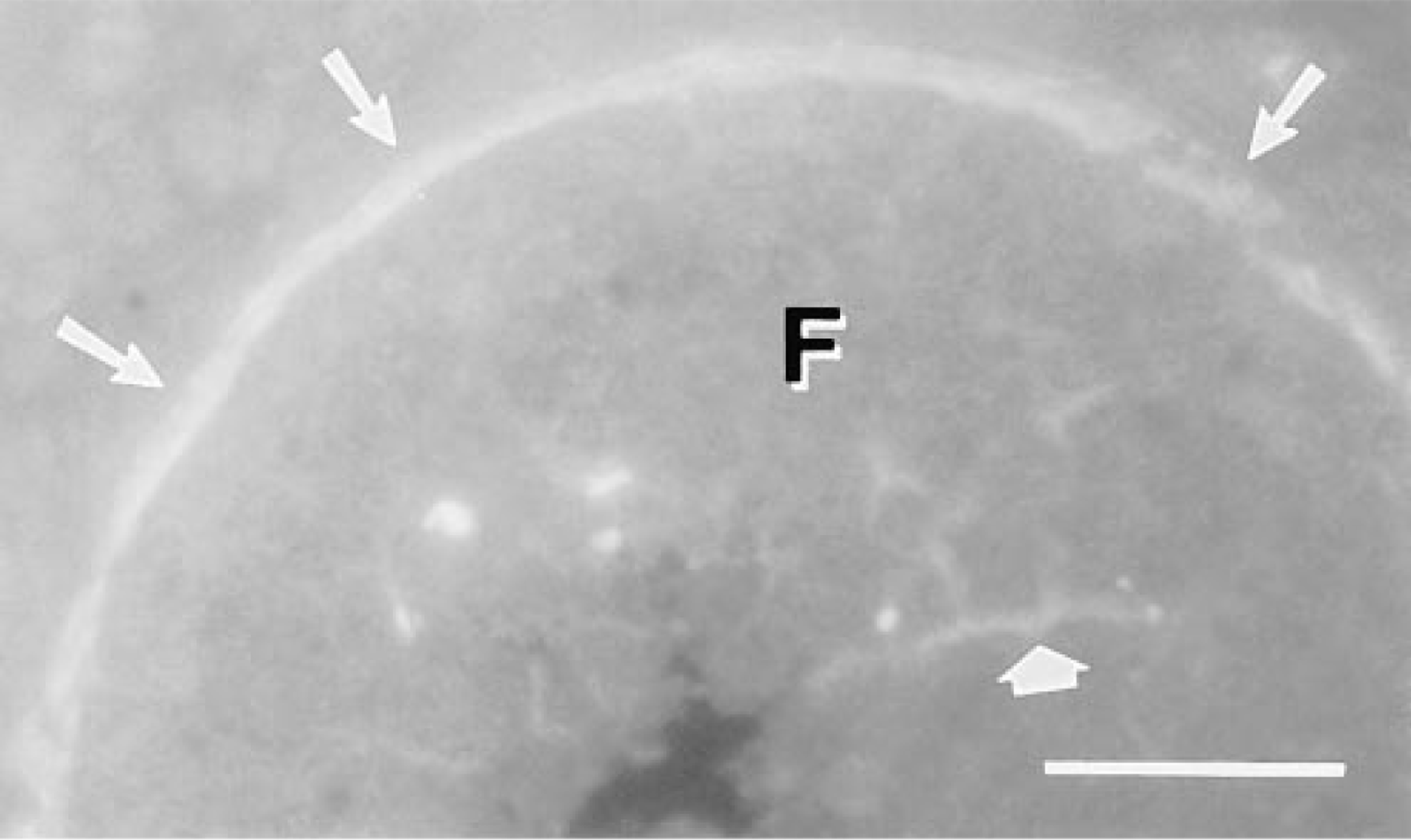
Localization of TSP in a growing follicle (F). TSP was distributed uniformly in the follicular basement membrane (narrow arrows) and appeared in scattered threadlike masses within the granulosa cell layer (broad arrow). Frozen section labeled with rabbit polyclonal anti-TSP antiserum. Bar = 50 μm.
Discussion
We have examined the expression of the matricellular proteins SPARC and TSP in the developing follicle and CL of the rat. The polyclonal anti-TSP antibodies used in this study do not distinguish between TSP-1 and −2. However, they are likely to recognize TSP-1 preferentially, based on confirmation by in situ hybridization studies (Iruela-Arispe et al. 1993; Reed et al. 1993). Moreover, the staining patterns obtained with the anti-TSP MAb and polyclonal anti-TSP antibodies were essentially the same. Because TSP-1 and −2 are homologues (Laherty et al. 1992) and both inhibit angiogenesis (Volpert et al. 1995; Panetti et al. 1997; Kyriakides et al. 1998), the functional consequence of expression of either form of TSP in the CL is likely to be similar. Future studies with type-specific antibodies will more clearly define the relationship between TSP-1 and −2 in the CL.
The variable expression of TSP by follicles at different stages of development is indicative of hormonal regulation. Indeed, expression of TSP by isolated rat granulosa cells in vitro is modulated by follicle-stimulating hormone (Dreyfus et al. 1992). Because TSP inhibits angiogenesis (Bagavandoss and Wilks 1990; Bagavandoss et al. 1993; Tolsma et al. 1993), the accumulation of TSP in basement membranes of immature follicles might prevent their premature vascularization.
SPARC is expressed in vivo where cells are proliferating, migrating, and undergoing structural reorganization, e.g., during embryonic development or during repair of wounds (Lane and Sage 1994). Ovarian granulosa cells, thecal cells, and interstitial cells undergo significant proliferative and reorganizational activity during folliculogenesis. Therefore, it is consistent that SPARC is expressed by all three cell types. Because SPARC inhibits the proliferation of cells in vitro (Sage 1997), it is possible that SPARC limits the rate of cell proliferation during growth of the follicle. An additional function of SPARC in vitro is counteradhesion (Sage and Bornstein 1991). By reducing adhesion of follicular cells to surrounding extracellular matrix, SPARC might facilitate the spatial redistribution of cells necessary for expansion of the follicle and formation of the antral chamber.
We find that the nuclei of maturing oocytes are markedly positive for SPARC. SPARC has also been found in nuclei of embryonic chick mesenchymal cells (M. Gooden, R. Vernon, and H. Sage, unpublished observations). Because extracellular SPARC is taken up by embryonic chick cells in vitro and is subsequently transported to the nucleus (M. Gooden, R. Vernon, and H. Sage, unpublished observations), SPARC within the oocyte nucleus might derive from extracellular sources, such as adjacent granulosa cells that provide trophic support to the oocyte. The function of SPARC in the oocyte nucleus is unknown. However, it is noteworthy that the protein calmodulin, which contains Ca+2 binding loops similar to those of SPARC, is implicated in the resumption of meiosis of the starfish oocyte (Santella and Kyozuka 1997).
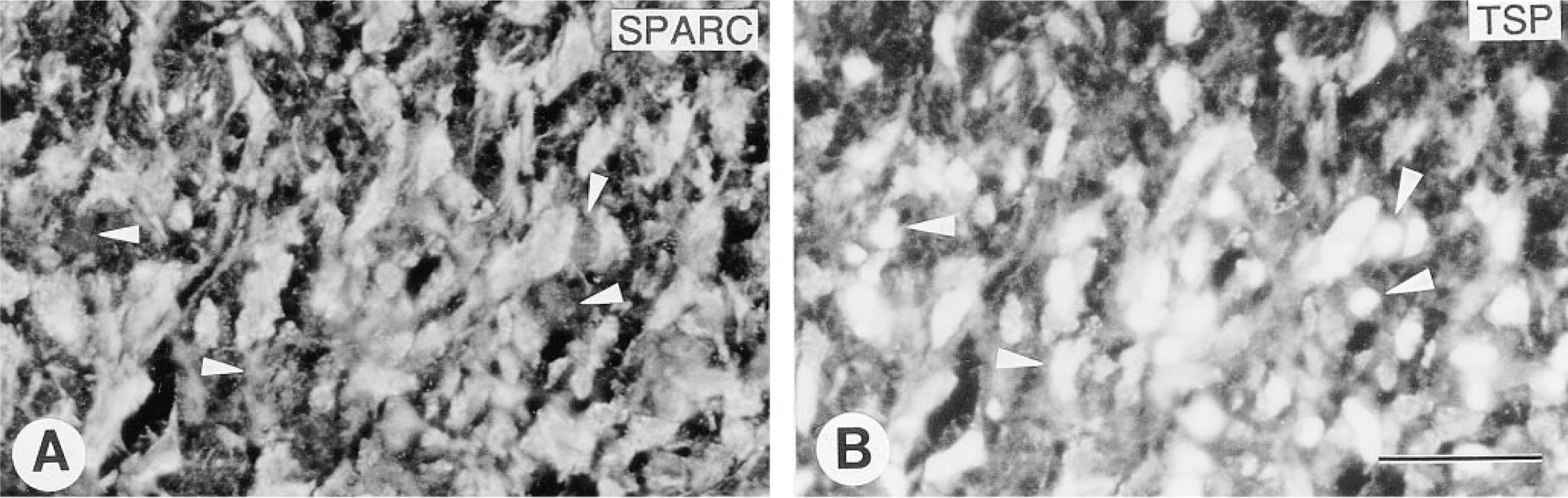
Expression of SPARC and TSP in an early CL. Sections were double labeled for SPARC
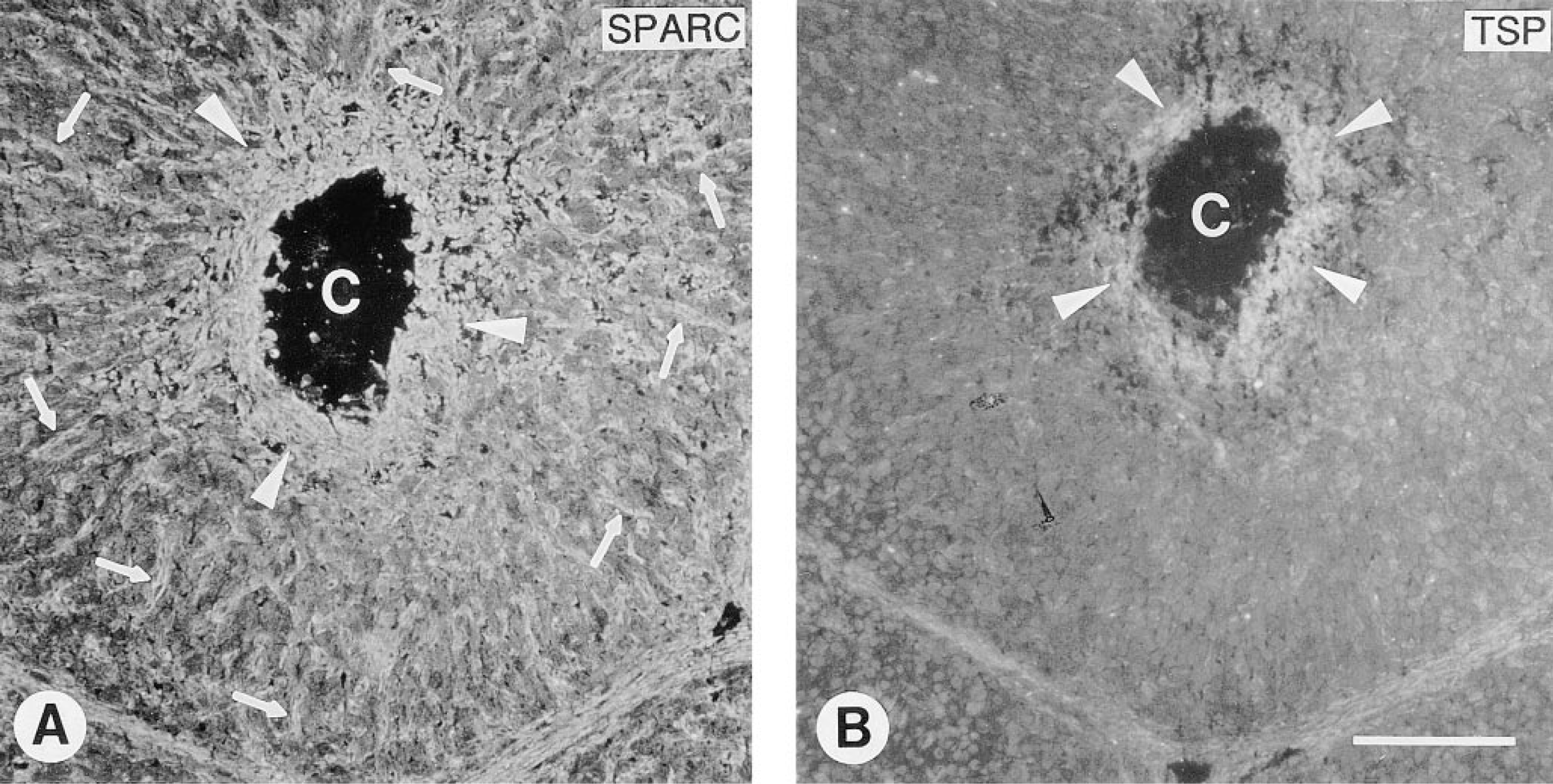
Expression of SPARC and TSP in a developing, vascularized CL. Sections were double labeled for SPARC
In developing CL, neovascularization of the granulosa layer progresses centripetally from the parent vasculature of the theca, which results in a steady reduction in the size of the central avascular area. In early stages of vascularization of the CL, we found TSP and SPARC in most preluteal granulosa cells. In contrast, in late CL where neovascularization was nearly complete, TSP was confined to the central avascular area, a result consistent with an anti-angiogenic function for TSP. TSP causes endothelial cells to decrease expression of uPA and to increase expression of PAI-1, responses that result in lower levels of matrix proteolysis and diminished angiogenesis (Bagavandoss et al. 1993; Sheibani and Frazier 1995). Ovarian granulosa cells produce both uPA and PAI-1, which are potentially susceptible to regulation by endogenous TSP. In this context, it is noteworthy that uPA and PAI-1, like TSP, are expressed in avascular areas of the CL (Bacharach et al. 1992).
Previous studies have reported that SPARC is expresssed by endothelial cells undergoing angiogenic morphogenesis in vitro and in vivo (Iruela-Arispe et al. 1991,1995; Reed et al. 1993). These results are consonant with our observation of elevated expression of SPARC in capillaries of the developing CL in comparison to mature vasculature in fully developed CL. Although intact SPARC has minimal effects on angiogenesis in vivo, specific peptide fragments of SPARC are angiogenic (and potentially anti-angiogenic). The co-expression of SPARC and TSP in the avascular center of the developing CL points to a potential antiangiogenic role for SPARC or SPARC peptides. Alternatively, the anti-angiogenic effects of TSP might counteract the angiogenic effects of SPARC peptides to yield a net inhibition of angiogenesis in this locale. Future studies will examine ways in which proteolytic activity within the CL might regulate the angiogenic properties of SPARC.
TSP was expressed by luteal cells of mature, functional CL but was lacking in luteal cells within regressive CL. Luteal cells in vivo and in vitro synthesize tissue- type plasminogen activator (tPA), which inhibits the production of progesterone by luteal cells (Liu et al. 1995). Because TSP indirectly stimulates the production of PAI-1, a potent inhibitor of tPA (Roberts et al. 1988), it is possible that endogenous TSP is necessary for maximal production of progesterone by luteal cells.
Expression of SPARC
A recent study has reported the expression of SPARC by small luteal cells of the ovine CL (Smith et al. 1996). The role of SPARC in the function of luteal cells is not clear. However, SPARC increases the permeability of endothelial cell monolayers in vitro (Goldblum et al. 1994). Thus, SPARC secreted by luteal cells might act in a paracrine manner to increase the permeability of the luteal vasculature, and thereby to facilitate the passage of steroid hormones and other bioactive molecules to and from the CL. In this context, it is noteworthy that SPARC is secreted by testicular Leydig cells which, like ovarian luteal cells, are steroidogenic and are in intimate contact with the microvasculature (Vernon and Sage 1989).
In summary, we have shown that the developing follicle and CL of the rat ovary exhibit distinctive staining patterns for two significant members of the matricellular class of extracellular proteins, i.e., SPARC and TSP. Although there was near-coincident expression of these proteins in granulosa and steriodogenic luteal cells, the distribution of SPARC and TSP in the invading vasculature of the CL was unique and moreover, was consonant with their known functions in angiogenesis. Therefore, preluteal follicles and developing CL provide a singular example of nonembryonic, nonpathologic angiogenesis in which two matricellular proteins might exert opposing effects by virtue of their distinctive localizations.
Footnotes
Acknowledgements
We are indebted to Dr B.C. Bruot for providing his laboratory facilities for conducting this research and to Dr Jennifer Marcinkiewicz for graciously sharing her histology facility. We also thank Drs V.M. Dixit and W.A. Frazier for providing the rabbit polyclonal antibody to TSP.
