Abstract
Corneas of diabetic patients have abnormal healing and epithelial adhesion, which may be due to alterations of the corneal extracellular matrix (ECM) and basement membrane (BM). To identify such alterations, various ECM and BM components and integrin receptors were studied by immunofluorescence on sections of normal and diabetic human corneas. Age-matched corneas from 15 normal subjects, 10 diabetics without diabetic retinopathy (DR), and 12 diabetics with DR were used. In DR corneas, the composition of the central epithelial BM was markedly altered, compared to normal or non-DR diabetic corneas. In most cases the staining for entactin/nidogen and for chains of laminin-1 (α1β1γ1) and laminin-10 (α5β1γ1) was very weak, discontinuous, or absent over large areas. Other BM components displayed less frequent changes. The staining for α3β1 (VLA-3) laminin binding integrin was also weak and discontinuous in DR corneal epithelium. Components of stromal ECM remained unchanged even in DR corneas. Therefore, distinct changes were identified in the composition of the epithelial BM in DR corneas. They may be due to increased degradation or decreased synthesis of BM components and related integrins. These alterations may directly contribute to the epithelial adhesion and wound healing abnormalities found in diabetic corneas.
Keywords
D
These and other defects are likely to result from alterations in epithelial adhesion, migration, differentiation, and renewal. Extracellular matrix (ECM) and BM components acting through cell surface adhesive receptors, integrins (Giancotti 1997), play an important role in the adhesion, migration, and differentiation of various cells (Azar et al. 1992; Kurpakus et al. 1992; Zieske et al. 1994). We therefore hypothesized that corneal epithelial abnormalities in diabetes may be due to altered BM structure and/or epithelial integrin expression. However, despite abundant information on normal corneal integrin patterns and BM structure (Lauweryns et al. 1991; Tervo et al. 1991; Virtanen et al. 1992; Masur et al. 1993; Stepp et al. 1993; Trinkaus-Randall et al. 1993; Ljubimov et al. 1995, 1996a; Tuori et al. 1996), their changes in diabetes remained unknown.
Consequently, the purpose of this investigation was to identify the specific changes in individual BM and ECM components and in integrins that occur in the diabetic human cornea, with special reference to DR. Such a study was a necessary first step in understanding of the molecular mechanisms of corneal epithelial abnormalities in diabetes. Because of lack of pertinent data, we needed to study many BM components and integrins to single out those that were specifically altered in diabetes and DR. We conclude that corneas from patients with DR have specific alterations in the distribution of major epithelial BM components, entactin/nidogen, laminin-1 and laminin-10, and of an integrin receptor, α3β1 (VLA-3), reported to bind to these components. In contrast, corneas from diabetic patients without DR showed such alterations much less frequently. This suggests that, as retinal diabetic disease worsens, concomitant alterations of the corneal epithelial BM occur in parallel. Our study therefore provides the necessary background information for future investigations of the role of changes in specific BM components and integrins in DR-associated corneal dysfunction.
Materials and Methods
Tissue
Human autopsy corneas were obtained within 30 hr after death from the National Disease Research Interchange (Philadelphia, PA). They included age-matched corneas from 15 non-diabetic individuals (normal group), 10 diabetics without DR, referred to as non-DR diabetics (four IDDM and six NIDDM), and 12 diabetics with DR (nine IDDM and three NIDDM). All corneas were bisected, embedded in OCT (Miles; Elkhart, IN), and cryostat sections were studied.
Immunohistological Analysis
Indirect immunofluorescent staining of cryostat sections, their treatment with urea to reveal type IV collagen epitopes, and photography were performed as described (Ljubimov et al. 1995). Routine specificity controls (without primary or secondary antibodies) were negative. Monoclonal antibodies were used as straight hybridoma supernatants or at 10–20 μg/ml when purified, and polyclonal antibodies were used at 20–30 μg/ml. At least two independent experiments were performed for each marker, with identical results. Abnormalities in the immunofluorescent distribution of a specific protein, such as appearance of staining (Figure 5, α1(IV) chain, tenascin-C, fibrillin-1), markedly decreased staining (Figure 6, α3β1, β1 integrins), discontinuity (Figure 2, laminin γ1, entactin/nidogen; Figure 4, α3(IV) chain) and absence of staining (Figure 2, laminin α5,β1) were taken into account only if they were reproducible on serial sections and/or with different antibodies (e.g., to different chains of laminin).
Antibodies
Well-characterized antibodies were used to α1-α6 chains of Type IV collagen, to α2, α2, α4, β1, β2 and γ1 chains of laminin, to laminin-5 (α3β3γ2), to entactin/nidogen, to fibronectin eighth Type III repeat, to Types VI, VII, XII, and XIV collagen, and to perlecan and bamacan core proteins (Ljubimov et al. 1995; 1996a,b; Miner et al. 1997; Tiger et al. 1997). Antibodies to Types I, III, and V collagen were from Southern Biotechnology (Birmingham, AL), antibodies to fibrillin-1 (clone 11C1), tenascin-C (clone TN-2), and decorin core protein were from Chemicon International (Temecula, CA). An antibody to cellular fibronectin (clone IST-9) was from Sera-Lab (Crawley Down, UK), and an antibody to Type VIII collagen (clone 9H3) was from Seikagaku America (Rockville, MD). The monoclonal antibody 4C7 to α5 chain of laminin was from Chemicon International. This antibody was previously believed to react with laminin α1 chain and was only recently shown to recognize the α5 chain instead (Tiger et al. 1997). Antibodies to α5β1 integrin heterodimer, α3β1 integrin heterodimer (clone M-KID 2), to α6 (clone NKI-GoH3), α3 (clone P1B5), α2,α1 (clone FB12 to I domain), β1 (clone HB1.1), and β4 (clone 3E1) integrin subunits, and cross-species-absorbed fluorescein- and rhodamineconjugated secondary antibodies were from Chemicon International.
Statistical Analysis
This was performed using a double-sided Fisher's exact test (In- Stat software program; GraphPad Software, San Diego, CA).
Results
Laminin Chain Distribution in Normal Human Corneas
Human cornea can be topographically divided into the central part, which has the collagenous Bowman's layer under the epithelial BM, and the peripheral corneoscleral limbus, which harbors epithelial stem cells but does not have Bowman's layer (Ljubimov et al. 1995). Previously, the corneal distribution of laminin and Type IV collagen isoforms has been described in detail (Ljubimov et al. 1995,1996a; Tuori et al. 1996). However, the antibody 4C7 commonly used to recognize laminin α1 chain was recently shown to react with a ubiquitous α5 chain instead (Tiger et al. 1997). To reveal corneal expression of laminin α1 chain, we have previously used an 11D5 antibody (see Ljubimov et al. 1995) that gave identical staining patterns to 4C7 and also may have recognized the α5 chain. Therefore, the distribution of laminin chains in the normal human cornea first had to be revisited using other antibodies with a properly defined α-chain specificity. Such antibodies became available only very recently (Miner et al. 1997; Tiger et al. 1997).
In normal adult corneas, the laminin α5 chain-specific antibody 4C7 yielded the same staining pattern as 11D5: the epithelial central and limbal BM, limbal blood vessels, and the endothelial face of Descemet's membrane (DM) were strongly positive (Figure 1). Laminin α1 chain was present in the epithelial BM throughout the cornea, and on the endothelial face of DM (Figure 1). Limbal blood vessels exhibited weak to no staining (not shown). Some keratocyte staining was observed for both the α1 and α5 chains (Figure 1). The α4 chain could not be detected with the antibody used (Figure 1). These and previous data (Ljubimov et al. 1995; Tuori et al. 1996) suggest that multiple laminins may be present in the corneal epithelial BM, including laminin-1 (α1 β1γ1), laminin-5 (α3 β3γ2), laminin-6 (β1γ1), and laminin-10 (α5β1γ1), with more isoforms in the limbus (see Miner 1998 for nomenclature). In DM, chains of laminin-1 and laminin-10 have been detected (Figure 1; Ljubimov et al. 1995).
Basement Membrane Abnormalities in Diabetic Retinopathy Corneas
In the corneoscleral limbus, all ECM and BM components studied had a normal distribution in all non-DR diabetic or DR corneas. In addition, the corneal stromal components decorin, bamacan, and Types I, III, V, VI, and XII collagen were unchanged compared to normal corneas (not shown). However, profound alterations were revealed at the level of the epithelial BM in the central part of DR corneas. In most DR cases, staining for chains of laminin-1 and laminin-10, α1 (nine of 12 cases), α5 (10 of 12 cases), β1 (10 of 12 cases) and γ1 (eight of 10 cases), and for entactin/nidogen (seven of 11 cases) was very weak, discontinuous, or absent from parts or whole central epithelial BM (Figure 2; Table 1). In contrast, these components displayed strong and continuous staining in normal corneas (Figures 1 and 2). The incidence of the abnormal distribution of laminin-1, laminin-10, or entactin/nidogen in the DR group was significantly higher (p>0.03) than in either non-DR diabetic or normal group. Laminin chains α2 and β2 did not appear in the central epithelial BM or DM of non-DR diabetic or DR corneas and were seen only in the limbal BM, similar to normal corneas (not shown). In about half of the DR cases, alterations similar to laminin-1 and entactin/nidogen were observed in the central epithelial BM for α3 and α4 Type IV collagen chains (Figure 3; Table 1), although differences from the non-DR diabetic group were not significant. Laminin-5, Type VII collagen, α5 and α6 chains of Type IV collagen (Figure 4; Table 1), and perlecan (not shown) were rarely altered even in DR corneas. In the epithelial BM, fibronectin was also normal in most cases.
In DM, the only consistent change in DR group was the absence of fibronectin (both total and cellular, six of nine cases, Figure 5), which was found at the stromal aspect of DM in normal corneas (Ljubimov et al. 1995). This change was also detected in some non-DR diabetic corneas. In addition, in some non-DR diabetic corneas and especially in DR corneas, several ECM components appeared that were not present in normal central corneas. These were α1- α2 Type IV collagen abnormally deposited in the epithelial BM, fibrillin-1 deposited in BM and stroma, and tenascin-C mainly deposited in stroma (Figure 5). The deposition of these components may be related to diabetesassociated corneal edema, because they also appeared in human corneas with bullous keratopathy, an edematous blistering disease (Ljubimov et al. 1996a).
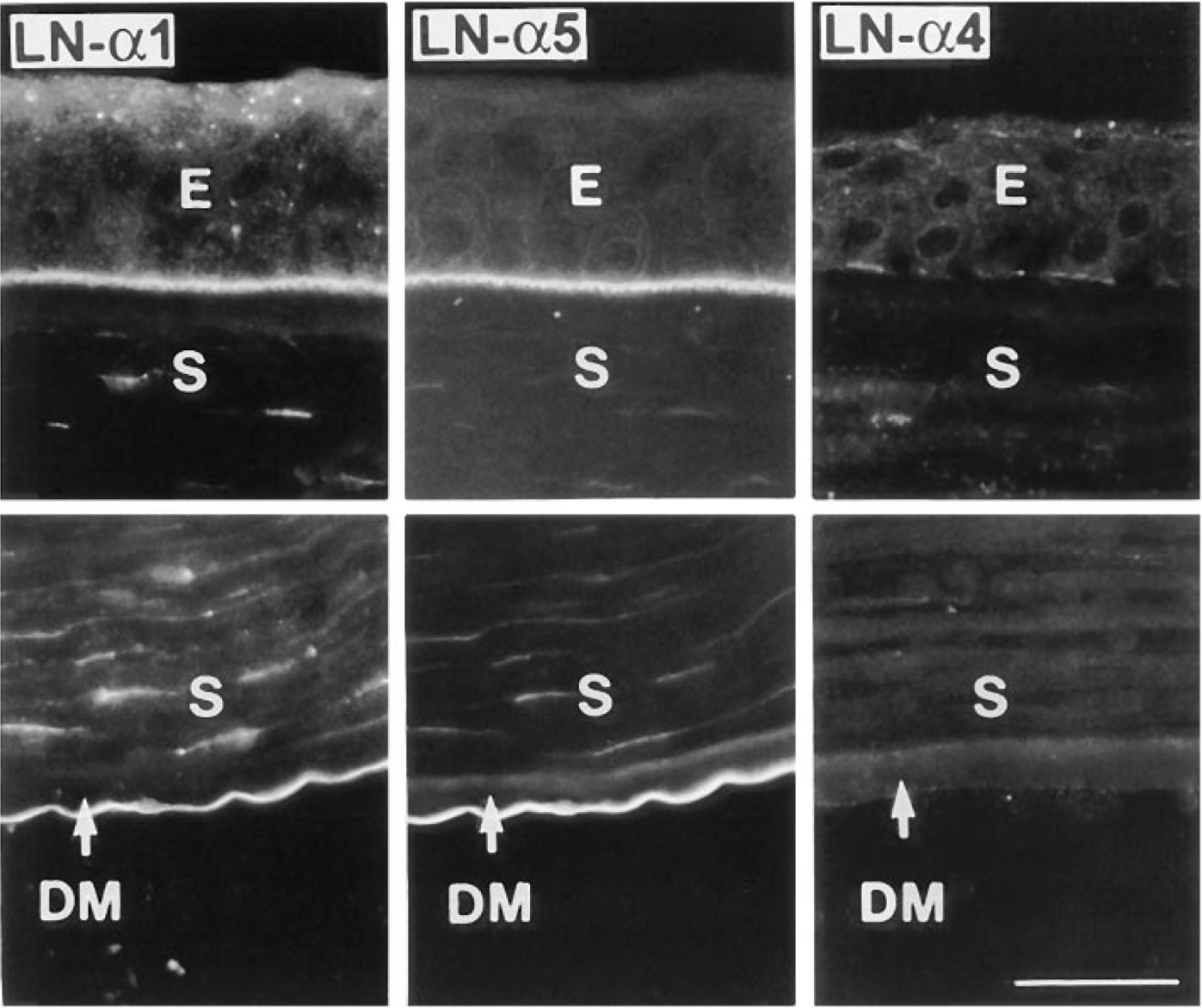
Distribution of α1, α4 and α5 laminin chains in the normal adult human corneal epithelial BM (upper row) and DM (lower row). α1 and α5 chains (double labeling) co-distribute in both BMs, whereas α4 chain is absent. Note keratocyte staining for α1 and α5 chains. Only central corneas are shown. Bar = 40 μm.
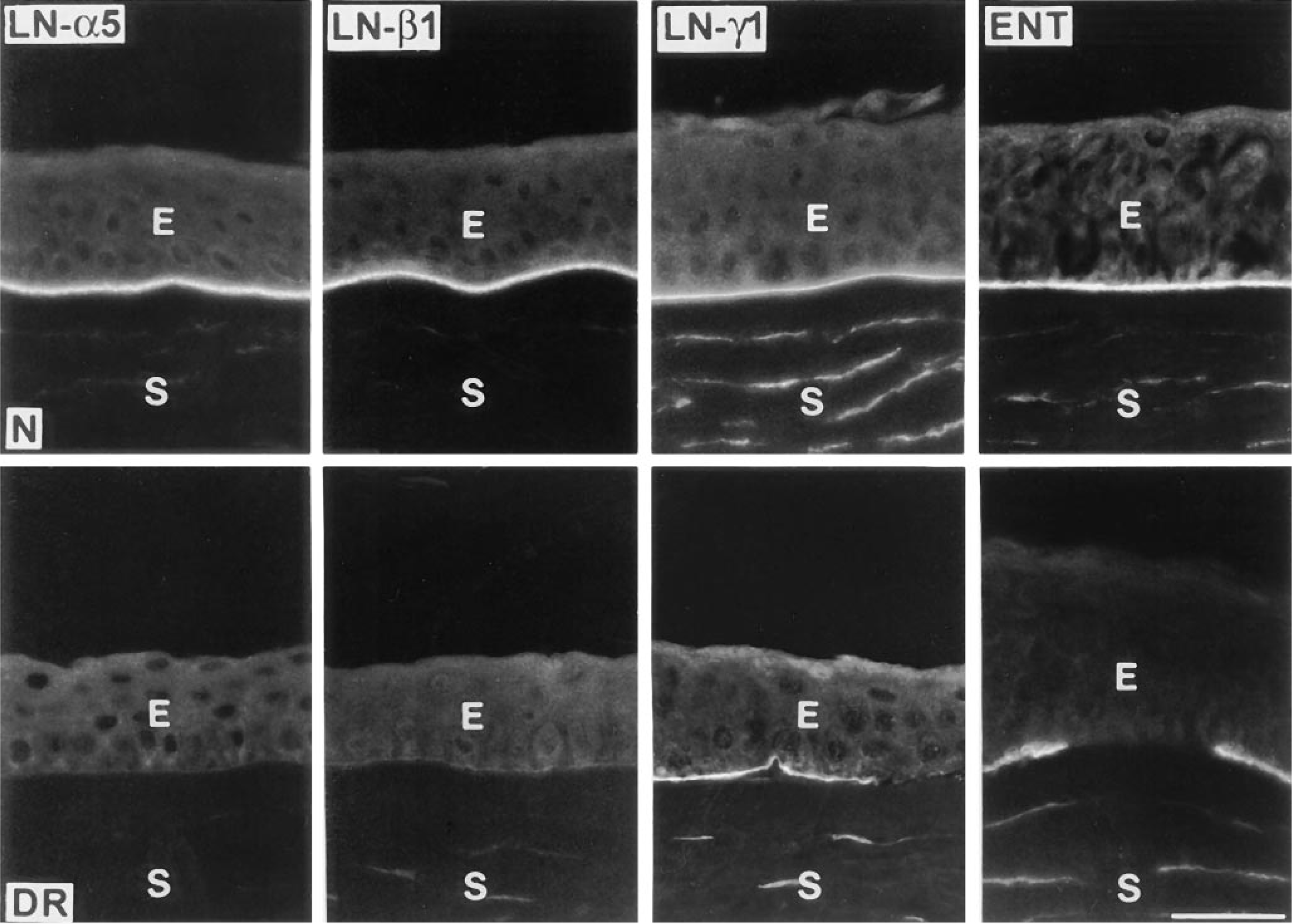
Representative distribution of laminin-10 chains (α5, β1, γ1) and entactin/nidogen in normal (N, upper row) and DR (lower row) human corneas. Note disappearance of staining in DR corneas for laminin α5 and β1 chains and discontinuity of staining for laminin γ1 chain and entactin/nidogen. Laminin-1 (α1β1γ1) was altered in a very similar way to laminin-10 (not shown here). E, epithelium; S, stroma; LN, laminin; ENT, entactin/nidogen. Bar = 40 μm.
Epithelial Integrin Alterations in Diabetic Retinopathy Corneas
The next question was to determine whether the observed alterations of laminin-1, laminin-10, and entactin/ nidogen in DR corneal epithelial BM were accompanied by changes in the expression of specific integrin receptors on corneal cells that bind to these components. To this end, we studied the distribution of chains of most of the known laminin-binding integrins, including α6, α3 (also reported to bind entactin/nidogen), α2, α1, β4, and β1. One of these integrin chains, α1, was not found in any of the central corneas studied (not shown). In addition, corneas were stained for the fibronectin receptor α5β1, which served as a negative control because its ligand did not change in the epithelial BM of diabetic corneas. Because diabetic changes mainly concerned the epithelial BM, we will discuss below primarily the epithelial patterns of studied integrins. The endothelium and keratocytes of non-DR diabetic and DR corneas were positive for α5β1, α6, α3, α2, and β1 integrins, similar to normal corneas (not shown).
In normal corneas, the epithelial patterns of studied integrins and their subunits were identical to previously described patterns (Lauweryns et al. 1991; Tervo et al. 1991; Virtanen et al. 1992; Stepp et al. 1993; Trinkaus-Randall et al. 1993). In the epithelium of DR corneas (8/10 cases), the staining for α3β1 integrin was markedly weaker or discontinuous compared to non-DR diabetic or normal corneas (Figure 6; Table 1). Changes were more pronounced in suprabasal layers and on the basal surface of basal epithelial cells. In some DR corneas these alterations were local, whereas in other corneas they were seen in the majority of the epithelial cells. Identical results were obtained with antibodies recognizing the whole integrin heterodimer (Figure 6) or its α3 chain (not shown). The staining for β1 integrin was also weaker than normal or discontinuous in more than half of DR corneas (Figure 6; Table 1), unlike non-DR diabetic corneas. In contrast, the distribution of α6, β4 (Figure 6) and α5β1 (not shown) integrins did not change in either non-DR diabetic or DR corneas compared to normal corneas. Because the β1 integrin subunit, unlike α6 and β4, appeared to be reduced in DR corneas, the epithelial α6 subunit may mostly be part of α6β4 rather than of α6β1 integrin.
Alterations of BM, ECM, and integrins in diabetic corneas a

aFor each component, the number of cases with altered staining/total number of examined cases is shown.
bAlterations include markedly weaker staining than in normal corneas, discontinuities or absence of staining over small or large areas.
cSignificant differences between groups (p,0.03).
dLack of fibronectin staining was seen only in Descemet's membrane (DM) but not in the epithelial BM. For all other components, alterations were found at the epithelial BM or cell (integrins) level. Stromal ECM and DM components (I, III, V, VI, VIII, XII and XIV collagen, bamacan and decorin core proteins) were not changed in diabetes and are not shown in Table 1.
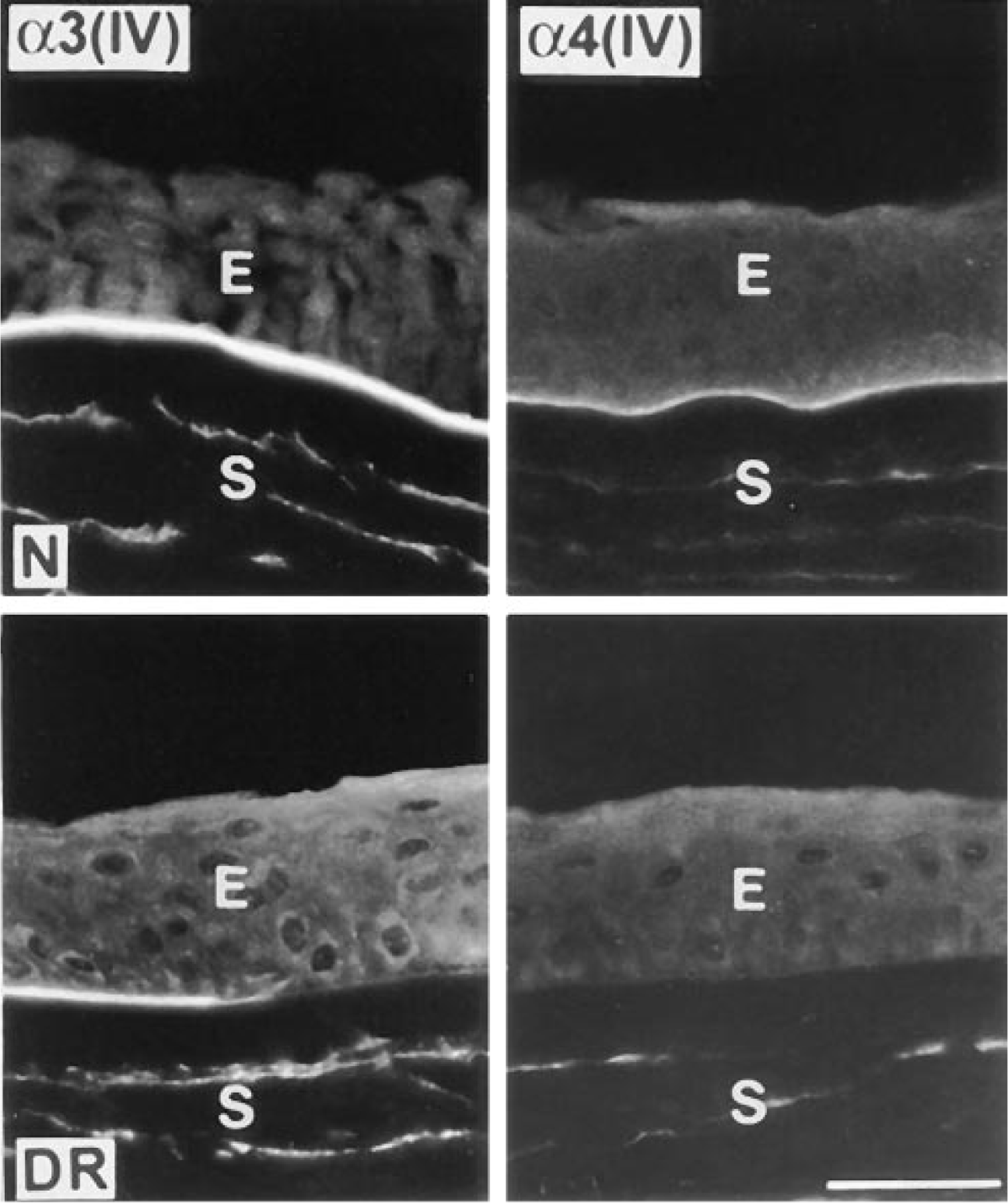
Alterations in Type IV collagen chains in DR corneas. Strong BM staining is seen in normal corneas (N, upper row). Note discontinuous staining for α3(IV) chain and absence of staining for α4(IV) chain in DR corneas (lower row). E, epithelium; S, stroma. Bar = 40 μm.
Discussion
Diabetic retinopathy is a severe ocular diabetic complication and a major cause of legal blindness. Although diabetes affects mostly the retina and iris (Lim and Murphy 1991), it also causes a corneal disorder, diabetic keratopathy, which involves epitheliopathy (altered epithelial barrier function, increased epithelial fragility, decreased basal cell adhesion and impaired healing), corneal edema resulting from endothelial alteration, and tear film dysfunction (Rao 1987; Meller et al. 1996; Ohashi 1997).
Diabetic nephropathy and retinopathy bring about profound changes in the ECM and BM in kidney and retina, respectively. Glomerular, retinal, and vascular BMs are thickened in diabetics and exhibit alterations in the expression of laminins, collagens, fibronectin, tenascin-C, proteoglycans, and some integrins (Østerby 1990; Nerlich and Schleicher 1991; Jin et al. 1996; Ljubimov et al. 1996b; Yokoyama and Deckert 1996; Regoli and Bendayan 1997). In diabetic corneas, only general BM changes have been described. They include BM thickening, decreased stromal penetration of anchoring fibrils, and greater susceptibility of epithelial BM to damage (Hatchell et al. 1983; Azar et al. 1989). The influence of diabetes on corneal BM/ECM components and integrins was not studied, which prompted us to analyze in detail their distribution patterns in non-DR diabetic and DR corneas.
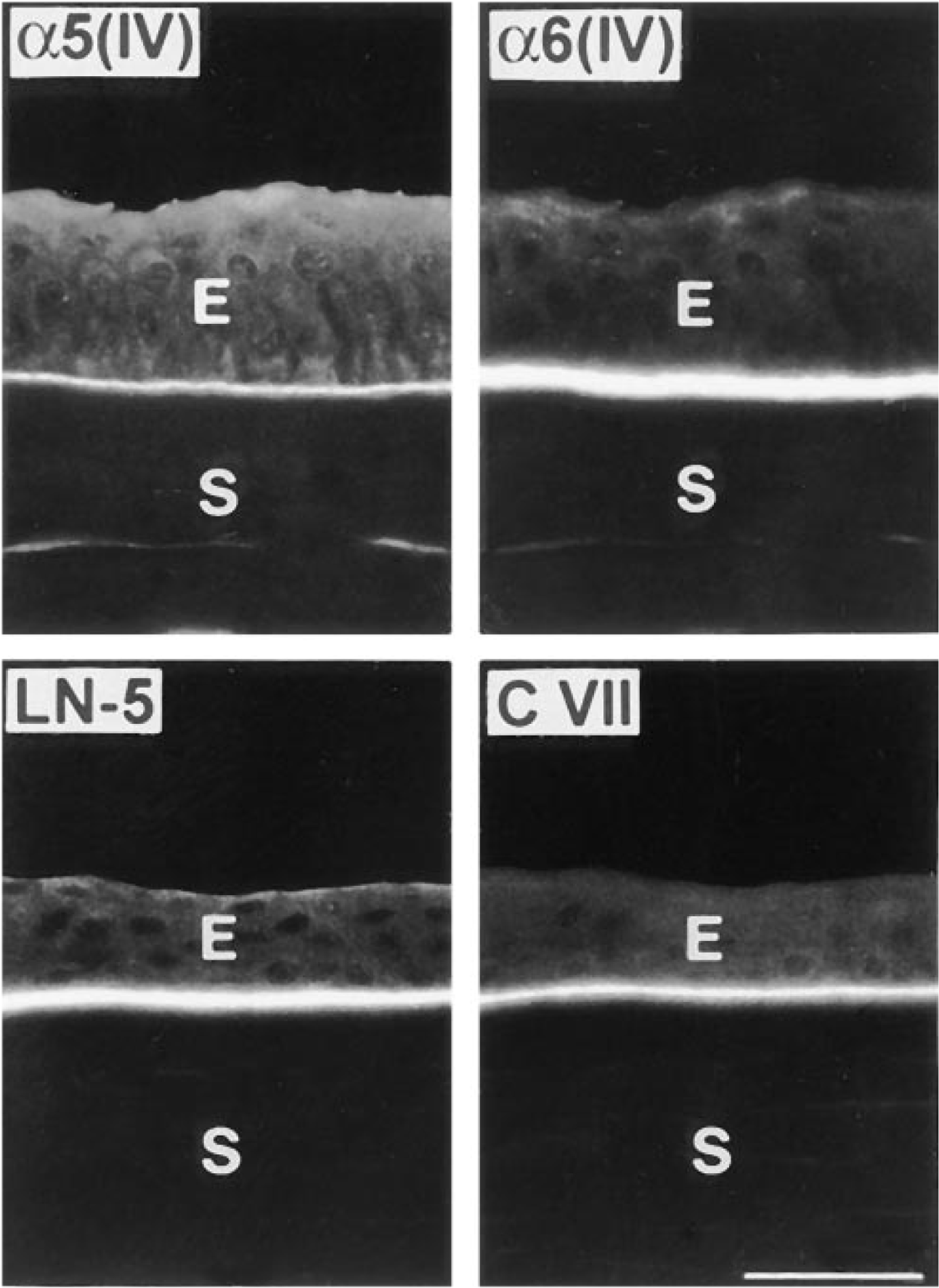
Normal distribution patterns of α5 and α6 chains of type IV collagen, laminin-5, and Type VII collagen in DR corneas. Sections of two DR corneas were double labeled for α5(IV) and α6(IV) chains or for laminin-5 and Type VII collagen. All proteins are continuous in the epithelial BM. E, epithelium; S, stroma; LN-5, laminin-5; C VII, Type VII collagen. Bar = 40 μm.
We show here that the epithelial BM composition in DR corneas is significantly altered. Major epithelial BM components, entactin/nidogen, laminin-1, and laminin-10, were markedly diminished in DR corneas as revealed by specific immunofluorescence. Alterations in other BM components, laminin-5, Type IV collagen isoforms, Type VII collagen, and fibronectin, were less pronounced and less common.
The next step was to analyze in DR corneas the fate of integrins that bind laminin and entactin/nidogen. Only one integrin studied, α3β1 (VLA-3), was significantly altered in the epithelium of DR corneas compared to both normal and non-DR diabetics (Table 1). This integrin has been reported to bind both isolated entactin/nidogen and laminins (Dedhar et al. 1992; Gresham et al. 1996; de Melker et al. 1997). Possibly its ubiquitous splice variant, α3Aβ1, is expressed in cornea, because the other variant, α3Bβ1, has a restricted tissue distribution (de Melker et al. 1997).
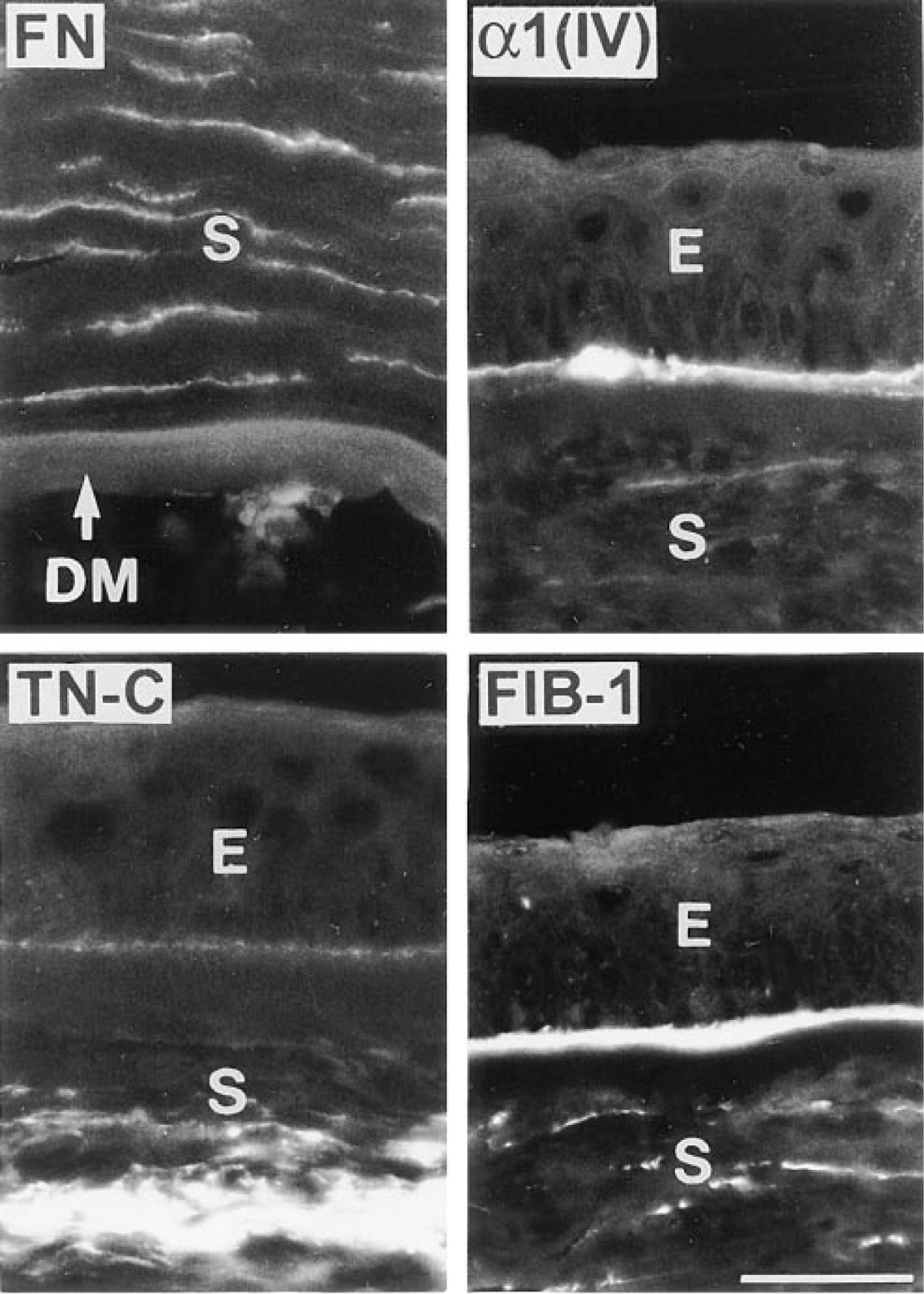
Frequent ECM abnormalities in DR corneas. Fibronectin (FN) is absent from Descemet's membrane (DM). α1(IV) chain appears in the central epithelial BM. Tenascin-C (TN-C) and fibrillin-1 (FIB-1), normally present only in the limbus, are expressed in the stroma (TN-C) and epithelial BM (FIB-1). E, epithelium; S, stroma. Bar = 40 μm.
Laminin-binding integrin α1β1 (not shown) was not found in the corneal epithelium. Laminin-binding integrins α2β1 and α6β4 were generally not altered in DR corneas (Table 1). This might be due to preferential binding of α2β1 integrin to collagen and of α6β4, to laminin-5 (Niessen et al. 1994; Giancotti 1997), both of which were not significantly changed in DR corneas. α7β1 laminin binding integrin (Velling et al. 1996) was not analyzed here owing to lack of available antibodies.
The observed alterations in major human corneal epithelial BM components and in their binding α3β1 integrin appear to be DR-specific. In fact, in corneas from patients with bullous keratopathy, laminin and entactin/nidogen abnormalities were less severe and less common (similar to non-DR diabetics), and both α3β1 and β1 integrins retained a normal distribution (not shown). In addition, BM and integrin alterations were significantly more pronounced and more common in DR corneas than in non-DR diabetic corneas (Table 1). It should be noted that no similar alterations have been reported in retinas of DR patients or in kidneys of diabetic nephropathy patients. In contrast, there was an increased expression of BM proteins and integrins (Nerlich and Schleicher 1991; Jin et al. 1996; Ljubimov et al. 1996b).
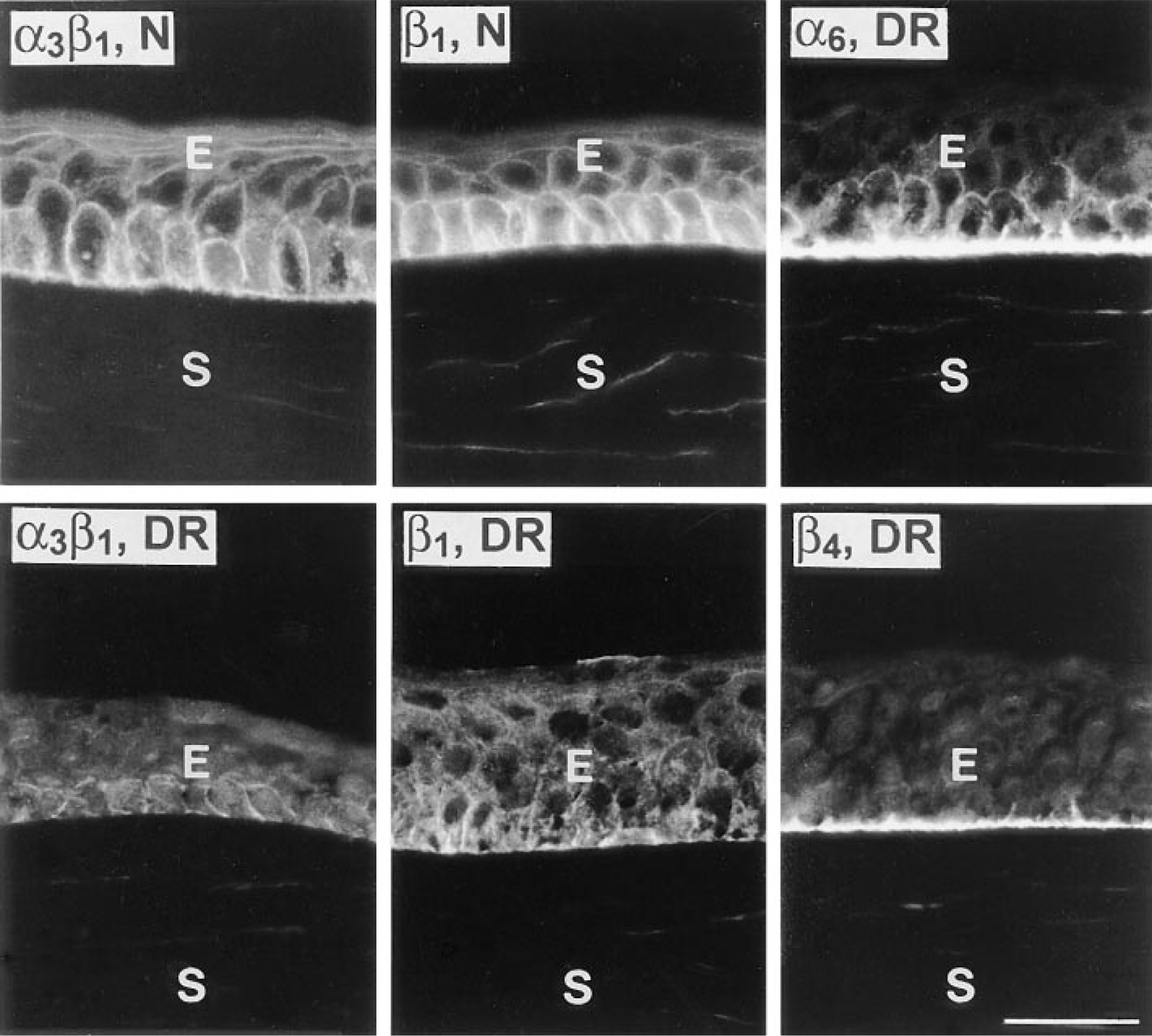
Integrins in normal and DR corneas. The staining for α3β1 integrin (left column) and β1 integrin subunit (middle column) is very weak and discontinuous in DR corneas compared to normal (N) corneas. At the same time, in DR corneas, patterns of α6 and β4 integrin subunits are typically normal (double labeling, right column). In view of reduced β1 subunit, but abundant β4 subunit, α6 may mostly complex with β4 to form α6β4 integrin. E, epithelium; S, stroma. Bar = 40 μm.
What could be the mechanisms of such alterations and why would they develop late in the course of the disease, with the advent of DR and proliferative DR? One possibility is that BM and/or integrin synthesis is decreased because of the action of growth factors abnormally expressed in the diabetic eye. Growth factor modulation of laminin-1, entactin/nidogen, and α3β1 integrin expression was shown in other systems (Schreiber et al. 1995; Narita et al. 1996; Nissinen et al. 1997). Elevated levels of fibroblast growth factor-2, insulinlike growth factor-I, and vascular endothelial growth factor have been found in the vitreous of DR patients compared to non-DR diabetics (Aiello 1997; Boulton et al. 1997). Some of these growth factors may also become elevated in DR corneas or may diffuse from the vitreous to the aqueous, this affecting corneal cell BM and integrin production.
Another possibility is that, in DR corneas, BM components and/or integrins may be altered because of their increased degradation by proteinases elevated in these corneas. Several lines of evidence support this hypothesis. Cultured diabetic human retinal endothelial cells have abnormal expression of matrix metalloproteinase-2 (MMP-2), which can cleave laminin (Grant et al. in press). In mouse mammary gland epithelium, entactin/nidogen is a specific target of MMP-3/stromelysin-1 (Alexander et al. 1996), and we have found increased expression of MMP-3 in DR corneas compared to normal and non-DR diabetic corneas (unpublished data). Additional experiments are needed to determine the actual mechanism(s) of laminin and entactin/nidogen decrease in the DR corneal epithelial BM.
The laminin γ1 chain is involved in the formation of stable complexes with entactin/nidogen (Mishima et al. 1996; Kadoya et al. 1997) that serves as a linker between laminin and Type IV collagen networks. This interaction is important for BM assembly, embryonic development, and tissue morphogenesis (Dziadek 1995; Kadoya et al. 1997). It is possible that reduced expression of γ1 chain-containing laminin-1 and laminin-10 in the epithelial BM of DR corneas could trigger coordinate alterations in entactin/nidogen. If proteolysis is involved in the DR corneal alterations, it may first affect entactin/nidogen, which is easily proteolysed (Dziadek 1995; Alexander et al. 1996; Kadoya et al. 1997). This, in turn, may lead to changes of laminins that complex with entactin/nidogen.
Interaction of corneal epithelial cells with BM components is known to modulate integrin expression patterns (Grushkin-Lerner et al. 1997). Therefore, BM changes in DR corneas may trigger respective changes in the integrin expression. Alternatively, the inhibition of α3β1 integrin expression by gene knockout has been recently shown to disrupt epidermal BM assembly (DiPersio et al. 1997). It can be suggested that downregulation of α3β1 integrin by some DR-activated factors may cause BM alterations observed in DR corneas. One such mechanism may be an increase in the expression of matrix metalloproteinases that degrade α3β1 BM ligands (Chintala et al. 1996).
The concerted reduction of expression of entactin/ nidogen, laminin-1, laminin-10, and of their binding α3β1 integrin in DR corneas may severely impair the adhesive and migratory properties of corneal epithelial cells. Such alterations in corneal cell-BM adhesion may be the mechanism underlying clinically observed diabetic abnormalities in epithelial barrier function, adhesion, epithelial integrity and wound healing. Finding ways to inhibit or retard corneal BM and integrin downregulation may prove useful in the development of novel therapeutics aimed at alleviating the symptoms of diabetic keratopathy.
Footnotes
Acknowledgements
Supported by the Iris and B. Gerald Cantor Foundation, the Discovery Fund for Eye Research, and the Skirball Program for Molecular Ophthalmology.
We thank Profs J.R. Couchman (University of Alabama, Birmingham, AL), E. Engvall (The Burnham Institute, La Jolla, CA), and A.F. Michael (University of Minnesota, Minneapolis, MN) for providing antibodies. Antibodies to laminin β2 chain produced by Dr J. Sanes and to Type IV collagen α1-α2 chains produced by Dr H. Furthmayr were obtained from the Developmental Studies Hybridoma Bank, Department of Biology, University of Iowa (Iowa City, IA), under contract N01-HD-2-3144 from the NICHD.
