Abstract
Biotinylation of antibodies is an established method for producing systems for detection of antigens. We currently aim to develop liposomal targeting vectors for gene transfer into transgenic gonadal tumor cells expressing the luteinizing hormone (LH) receptor (R). We have biotinylated (
Keywords
G
The detection of LH receptors in tissue sections has thus far been achieved using radioactively labeled LH/ CG (Rajaniemi and Vanha-Perttula 1972; Midgley 1972, 1973) and immunocytochemistry (Loosfelt et al. 1989; Visintin and Luborsky 1989; Vu-Hai et al. 1990). The avidin-biotin-peroxidase system has widely replaced radioactivity in biochemical detection methods. Biotinylated hormone derivatives have been used in receptor detection (Michel and Parsons 1988; Childs and Unabia 1989; Childs et al. 1989; Newman et al. 1989). Immunocytochemistry reveals the existence of specific structures of the gonadotropin receptors in selected tissues but it cannot provide direct evidence for the biological activity of receptor protein detected.
To create a targeting molecule for certain gonadal tumors and to achieve the detection of functional LH receptors using nonradioactive labeling, we set out to study the feasibility of using biotinylated hCG for these purposes.
Materials and Methods
Biotinylation of hCG and Bovine Serum Albumin (BSA)
Biotin isothiocyanate (BITC) (Wallac; Turku, Finland) (1.33 mmol/liter) was incubated with 13.3 m mol/liter hCG (CR-121, 11,500 IU/mg; National Institutes of Health, Bethesda, MD) or BSA for 4 hr in 50 mmol/liter sodium carbonate buffer (pH 9.8) at 23C. The endproducts were separated from unbound BITC by gel filtration. The biotinylated product was eluted with TBS buffer (trisaminomethane 50 mmol/liter, sodium chloride 154 mmol/liter, pH 7.75) through PD-10 (Pharmacia; Uppsala, Sweden) and NAP-10 (Pharmacia) (because of increased sample volume after the first elution) Sephadex G25 columns, according to the manufacturer's instructions. The degree of biotinylation of hCG was determined with 2-(4′-hydroxyazobenzene) benzoic acid (HABA) (Pierce; Rockford, IL) -reaction according to the manufacturer's instructions, as earlier described (Green 1965).
[125I]-Iodo-hCG Binding to Rat Testicular Membranes
Both B-hCG and intact hCG, at dilutions of 0.1–400 ng in 50 μl Dulbecco's PBS with 0.1% BSA (DPBS-BSA), were mixed with 10,000 cpm (100 μml) of [125I]-iodo-hCG in DPBS-BSA and 100 μl of rat testicular membrane (one testis per 5 ml DPBS-BSA) in a total volume of 250 μl/tube, and incubated in triplicate for 24 hr at 23C as described earlier (Catt et al. 1976). After the incubation, 4 ml of DPBS-BSA was added to each tube and the tubes were centrifuged for 30 min at 2000 × g (4C). The supernatants were removed and the specific activities of the pellets were measured using a γ-spectrometer. All the procedures using rats were approved by the University of Turku Ethical Committee on Use and Care of Animals.
Binding of B-hCG to Coated Microtitration Plates
A time-resolved immunofluorometric Delfia hCG kit (Wallac) was used to determine the binding of 0.2–200 ng B-hCG/ microtitration plate to monoclonal hCG antibodies according to the instructions of the manufacturer. B-hCG (0–200 ng) was applied to streptavidin-coated microtitration plates from the same manufacturer to detect the binding of the hormone derivative to both streptavidin and the same europiumlabeled second monoclonal antibody as used in the Delfia hCG kit.
Stimulation of cAMP Production by B-hCG
One day before stimulation, the BLT-1 Leydig tumor cells (Kananen et al. 1996) were plated on 24-well culture dishes (Greiner Labortechnik; Frickenhausen, Germany) at a density of 10 = cells/well in 500 μl of culture medium. The cells were incubated in the presence of B-hCG, native hCG, or forskolin (Sigma; St Louis, MO) and a final concentration of 0.2 mmol/liter of 3-isobutyl-1-methylxanthine (MIX; Aldrich-Chemie, Steinheim, Germany) was added to the incubation media for 4 hr. At the end of the incubations the media were collected and assayed for extracellular cAMP concentrations using an RIA as described before (Harper and Brooker 1975).
Detection of LH Receptors in Rat Tissue Sections by B-hCG
Ten-μm frozen tissue sections of rat testis, ovary, and skeletal muscle were washed and incubated with 10 mg/liter B-hCG in DPBS for 2 hr at 37C. Then the slides were washed and incubated for 30 min at 23C with the ABC reagent of the Vectastain ABC kit according to the instructions of the manufacturer (Vector Labs; Burlingame, CA). After washing, the bound peroxidase enzyme was visualized by a nickel-diaminobenzidine solution (nickel chloride 70 mg/liter, DAB 200 mg/liter, hydrogen peroxide 0.002% in TBS, pH 7.5, 2 min at 23C), followed by a silver enhancement method (silver nitrate 1 g/liter, ascorbic acid 200 mg/liter, sodium acetate 80 g/liter, cetylpyridinium chloride 100 mg/liter, sodium tungstate 5 g/liter, acetic acid 0.56%, and Triton X-100 0.06%, mixed immediately before use, 1–7 min at 23C) as described previously (Merchentaler et al. 1989; Mullink et al. 1992). As controls, the sections were incubated with biotinylated BSA (B-BSA) or preincubated for 2 hr with a 100-fold excess of intact hCG at 37C, or the rats were pretreated sc with a 1000 IU/kg injection of hCG (Pregnyl; Organon, Oss, The Netherlands) 48 hr before sacrifice.
Immunohistochemistry of LH Receptor
Frozen sections of rat ovary semiparallel to those labeled with B-hCG (see above) were Bouin-fixed and preincubated with 4% normal goat serum in TBS for 30 min at 23C. The antiserum (kindly provided by Dr. P. Roche, Mayo Clinic, Rochester, MN), containing polyclonal peptide antibodies raised in a rabbit against the N-terminal peptide sequence 15–38 of the rat LH/CG receptor, was applied to the rat ovarian sections at a dilution of 1:500 in 1% BSA-TBS and incubated for 16 hr at 4C. After washing, biotinylated antirabbit IgG antibodies (1:250 dilution of Vectastain ABC kit secondary antibody solution in 1% BSA-TBS) were placed on sections for 30 min at 23C. After addition of the secondary antibody the procedure was continued as described for labeling with B-hCG (see above).
Results
HABA chemical reaction showed an average biotinylation degree of 3.5 ± 0.2 (SD) biotin molecules per hCG molecule for the biotinylated product. B-hCG was able to compete in parallel fashion with [125I]-iodo-hCG for specific binding to rat testicular membranes (Figure 1). The amount needed for 50% displacement was approximately sixfold higher than that of native hCG. After the biotinylation of hCG, the Delfia hCG kit test showed qualitatively the existence of a compound recognizable by the monoclonal anti-hCG antibodies of the kit (data not shown). Similarly, application of B-hCG to the streptavidin-coated microtitration wells indicated binding to streptavidin and thereafter to a monoclonal antibody raised against hCG (data not shown).
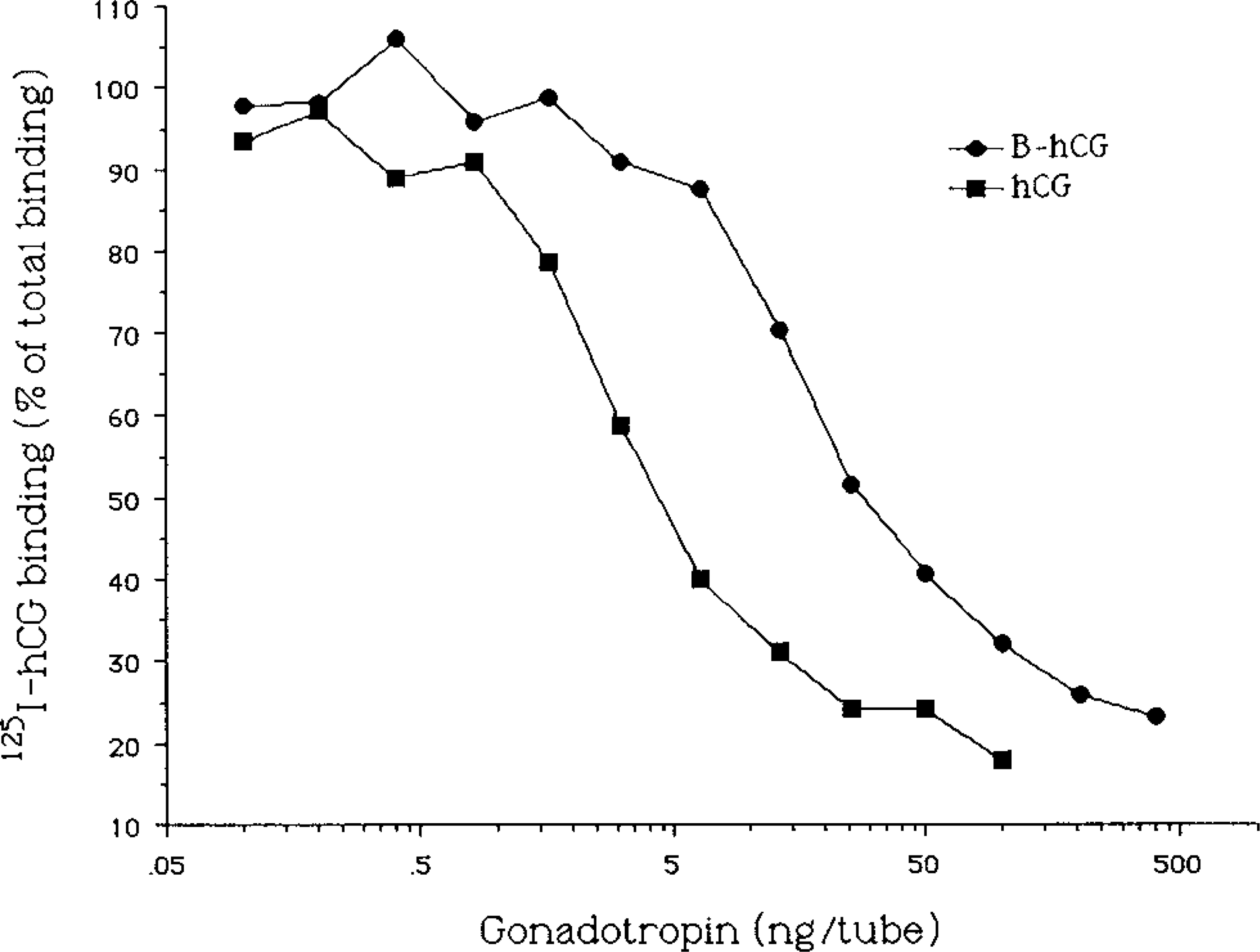
Inhibition of binding of [125I]-iodo-hCG to rat testicular embranes by biotinylated (B-hCG) and native hCG. A total of 0–400 ng B-hCG or 0–100 ng native hCG per tube was incubated for 24 hr with rat testicular membranes and [125I]-iodo-hCG (10,000 cpm/tube). Binding of [125I]-iodo-hCG at each dose is presented as percentage of total binding in the tubes with no unlabeled hormone. Nonspecific binding was determined in the presence of a 100-fold excess of nonlabeled hCG and was subtracted from the binding used for calculation of the results.
cAMP production by BLT-1 cells was induced with B-hCG. The concentration needed for stimulation of a similar level was approximately 10-fold compared to native hCG (Figure 2), which is in good agreement with the binding inhibition data shown in Figure 1.
Incubation of B-hCG on rat testicular sections showed almost exclusive binding into the interstitial compartment of the tissue (Figure 3A). In ovarian sections, the luteal, thecal, and interstitial cells were labeled, whereas the immature granulosa cells showed no staining (Figure 3B). The skeletal muscle was not labeled by B-hCG (Figure 3C). The control gonadal sections incubated with native hCG or B-BSA instead of B-hCG, or preincubated with excess native hCG (Figure 3E), or sections of the gonads of rats pretreated with high doses of hCG, showed no B-hCG binding (data not shown). In the testis, the low-level punctate intratubular binding was not totally blocked by preincubation with native hCG before B-hCG. The muscular wall of the gonadal blood vessels showed clear labeling with B-hCG (Figure 4).
Semiserial sections of the rat ovaries showed a similar pattern of labeling when incubated with either the rat LHR antibody or B-hCG. The internal parts of the corpora lutea showed more intense labeling by immunohistochemistry (Figure 5).
Discussion
These results demonstrate that the biotinylated hCG binds to LHR in the binding assay and in rat gonadal tissue sections and that it induces cAMP production in vitro. On the sections, the visualization of LHR was performed by using the avidin-biotin-peroxidase system for labeling. The monoclonal antibodies used in Delfia microtitration well kits showed affinity with the biotinylated derivative. At the receptor site, the B-hCG had affinity that was about 10-fold lower than that of native hCG, which is still acceptable for most of the applications, being on the same order as that of native human LH (Huhtaniemi and Catt 1981; Cameron and Stouffer 1982).
The cAMP production and binding affinity of B-hCG were similarly approximately 10-fold lower compared to native hCG. Therefore, it appears that biotinylation has little effect on the regions of hCG important for receptor binding and signal transduction in the cAMPdependent pathway.
Amplification of the detected signal by using multiple biotin binding sites of avidin gives the assays surpassing sensitivity. The systems employing biotinylated derivatives are relatively fast and easy to perform (Jeltsch et al. 1993; Macielag et al. 1994; Zecchini et al. 1995). LH/CG-R detection in tissue sections using B-hCG requires about 3 hr from start to completion.
LHR expression has been reported in interstitial cells in the rat testis (Catt et al. 1976) and in preovulatory granulosa cells and thecal, luteal, and interstitial cells of the rat ovary (for review see Richards 1980). The slight difference in the binding pattern of B-hCG with respect to immunocytochemisty in the rat ovarian tissue sections may reflect the existence of translated alternatively spliced LH/CG-R mRNA variants (Segaloff et al. 1990; Wang et al. 1990; LaPolt et al. 1991; Sokka et al. 1992), or different stages in processing of the receptor protein (Visintin and Luborsky 1989). Using a ligand probe of a receptor enables the detection of functional receptor proteins. Antibodies detect merely a specific structural feature of the receptor molecule, e.g., a peptide sequence, with no direct indication of the physiological function.
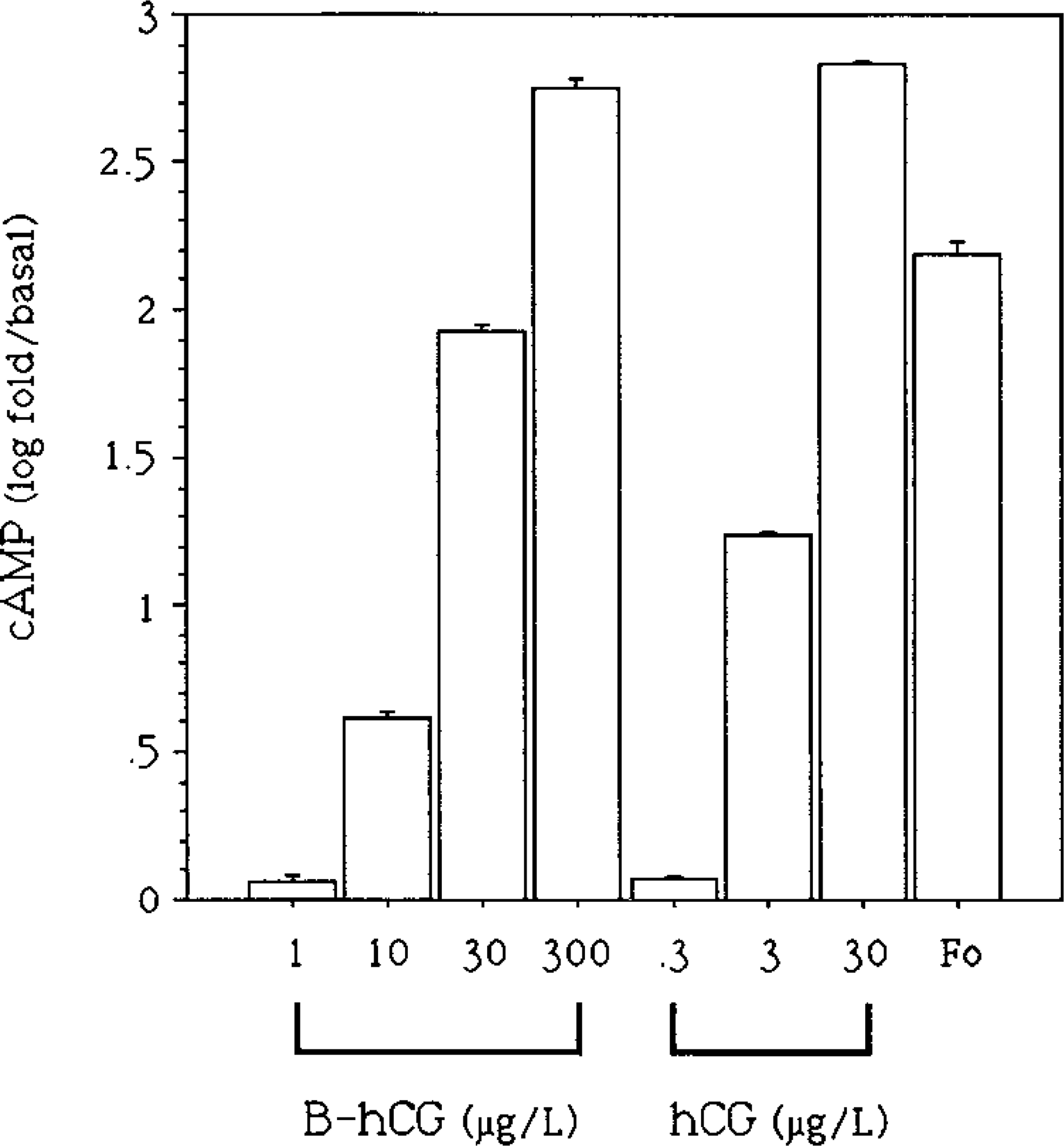
cAMP response of BLT-1 cells to increasing doses of B-hCG and native hCG and a single dose of forskolin (0.01 mmol/liter) (Fo). Each bar is the mean 1 SEM of four replicate incubations. One of three similar experiments is presented.
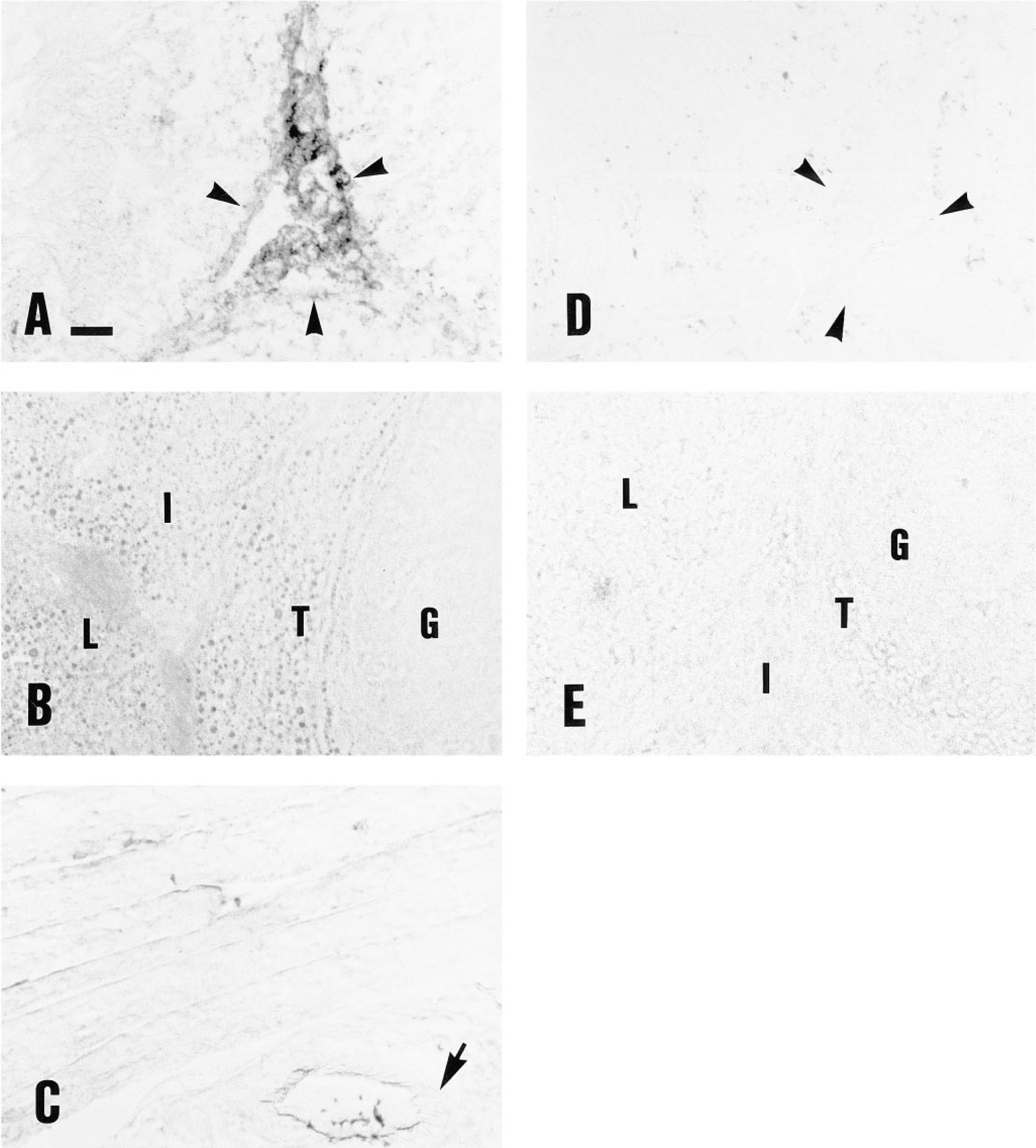
Labeling of rat testicular (
In an attempt to improve the current method, biotinylation of the common α-subunit of the glycoprotein hormone family (TSH, LH, FSH, and CG) would leave the selectivity-determining β-subunit intact. After coupling the α- and β-subunits, this method could yield an easily detectable hormone with highly restored binding affinity.
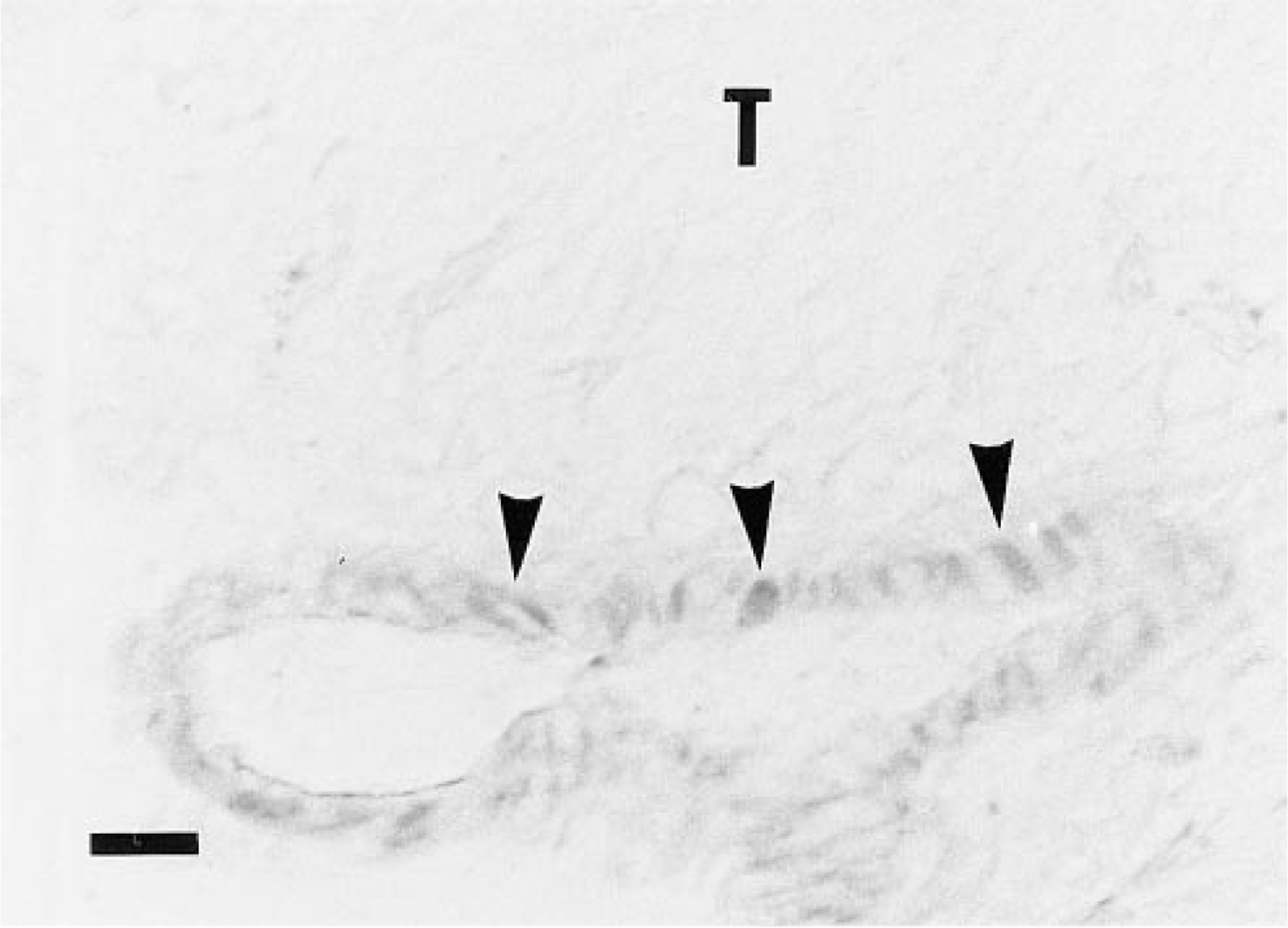
Binding of B-hCG to endothelial cells of testicular blood vessels in rat frozen tissue sections. Testicular blood vessel endothelia showed labeling (arrowheads), whereas skeletal muscle blood vessel endothelia (Figure 2C, arrow), used as negative controls, showed no binding of B-hCG. T, tubule lumen. Bar = 20 μm.
Selective gene transfer into cells expressing a specific receptor has been reported (Wu and Wu 1987; Wu et al. 1989; Cotten et al. 1990; Wagner et al. 1990,1991). We are now testing the applicability of B-hCG for targeted transfection into the LHR-expressing transgenic murine gonadal tumor cell lines previously established in our laboratory (Kananen et al. 1995,1996).
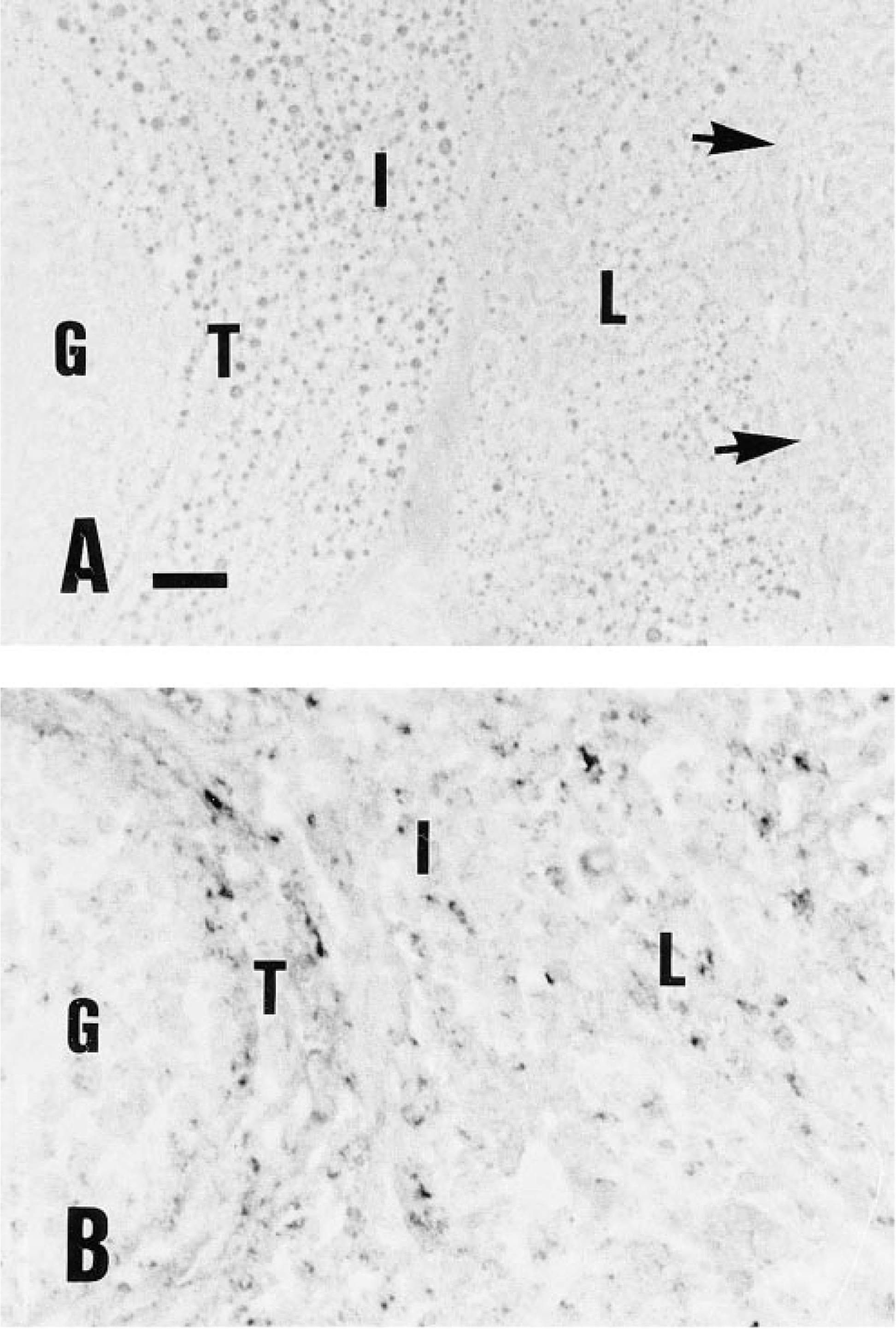
Comparison of rat ovarian frozen section labeling by B-hCG (A) and LH/CG-R immunocytochemistry (
Footnotes
Acknowledgements
Supported by grants from the Academy of Finland, the Sigrid Juselius Foundation, and the Finnish Cancer Fund. TP was supported by the Turku University Graduate School for Medical Sciences.
We thank Dr Kati Hakola, Dr Antti Rannikko, and Ms Aila Metsävuori for technical advice.
