Abstract
17β-hydroxysteroid dehydrogenase (17β-HSD) controls the last step in the formation of all androgens and all estrogens. At least six 17β-HSD isoenzymes have been identified. The recently cloned Type 5 17β-HSD transforms 4-dione into testosterone. To gain a better understanding of the role of this enzyme in reproductive tissues, we immunocytochemically localized the enzyme in human male and female reproductive organs. In the ovary of adult premenopausal women (25-40 years of age), immunostaining was found in corpus luteum cells. In the uterus, staining was detected only in the epithelial cells of the endometrium. Immunolabeling was also detected in the mammary gland, a positive reaction being detected in epithelial cells of acini and intralobular ducts as well as in the surrounding stromal cells. In the testis, strong staining was seen in the Leydig cells, and a weak but specific reaction was occasionally detected in Sertoli and germ cells. In the prostate, specific labeling was observed in alveoli and stromal fibroblasts. In alveoli, all the basal cells were generally labeled, whereas the luminal cells exhibited variations in immunoreactivity. In all the reproductive organs examined, specific staining was routinely detected in the walls of blood vessels, including the endothelial cells. These results indicate a cell-specific localization of Type 5 17β-HSD in the different human reproductive organs, thus suggesting new mechanisms of local androgen and estrogen formation that may play an important physiological role.
I
To obtain information about the precise cellular localization of Type 5 17β-HSD and then a better knowledge of the role of this enzyme in reproductive tissues, we developed specific antibodies against human Type 5 17β-HSD and then immunocytochemically localized the enzyme in male and female human reproductive organs.
Materials and Methods
The samples of reproductive tissues, i.e., testis, prostate, ovary, uterus, and breast, were all obtained at surgery. The specimens used in the present immunocytochemical studies had a volume of about 2.5 cm3, with the exception of prostate chippings, which were of smaller sizes (approximatively 0.4 cm3). They were fixed in 4% paraformaldehyde in 0.2 M phosphate buffer (pH 7.4) within 15 min after they had been dissected out. The average fixation time was 12 hr. The tissues were then dehydrated through increasing concentrations of ethanol, cleared in toluene, and embedded in paraffin. Ovaries, uterus, and breast tissues were obtained from premenopausal patients (25-40 years of age). Prostate tissue was obtained from patients with symptomatic benign prostatic hyperplasia undergoing transurethral prostatectomy. The age of male patients ranged from 55 to 70 years. In each case, a minimum of three separate tissues was studied and the results were shown to be consistent.
Preparation of a Human Type 5 17β-HSD Antibody
The peptide sequence N-GLDRNLHYFNSDSFASHPNYPYS located at amino acid position 297-320 of the human Type 5 17β-HSD (manuscript in preparation) was synthesized by le Service de Séquence de Peptides de l'Est du Québec (SSPEQ) (Québec, Canada) and was purified on HPLC. New Zealand rabbits (2.5 kg) received an
Immunoblot Analysis
Human embryonal kidney cells (293) were transfected with CMV-neo vectors expressing human Type 5 17β-HSD, 3α-HSD Types 1 and 3, and 5α-reductase Types 1 and 2, respectively. Stable transfectants were selected by their resistance to 10-7 M G-418. Positive clones were further confirmed by their ability to efficiently transform the corresponding substrate (unpublished data). Forty μg of the homogenate of each cell line was electrophoresed on a 5-15% SDS-PAGE as described (Luu-The et al. 1994) before being transferred to nitrocellulose filters using a Bio-Rad (Hercules, CA) apparatus for 4 hr at 60 V. Blots were treated three times with 5% fat-free milk in PBS containing 0.1% Nonidet P-40 for 30 min. The antiserum developed against the Type 5 17β-HSD peptide was diluted to 1:1000 and the blot was then incu bated at 4C for 18 hr with the diluted antiserum. The blot was then washed three times with PBS containing 5% fat-free milk and 0.1% Nonidet P-40. After incubation with horseradish peroxidase-conjugated anti-rabbit IgG (diluted 1:200) for 2 hr, membranes were washed and bound antibodies were detected with ECL detection reagents (Amersham; Arlington Heights, IL) and exposure to Hyperfilm.
Immunocytochemistry
The tissues were serially cut at 7 μm and sections mounted on glass slides. The sections were deparaffinized, hydrated, and incubated overnight at 4C with the human Type 5 17β-HSD antiserum diluted 1:1000 in Tris-saline, pH 7.6. The sections were then washed and incubated at room temperature for 4 hr with peroxidase-labeled goat anti-rabbit γ-globulin (Hyclone; Logan, UT) diluted 1:500 as previously described (Pelletier et al. 1995). Endogenous peroxidase activity was eliminated by preincubation with 3% H2O2 for 30 min and peroxidase was then revealed during incubation with 10 mg of 3,3'-diaminobenzidine in 100 ml of Tris-saline buffer containing 0.03% H2O2. The intensity of the staining was controlled under the microscope. The sections were then counterstained with hematoxylin. Control experiments were performed on adjacent sections by substituting nonimmune rabbit serum (1:1000) or the Type 5 17β-HSD antiserum (diluted 1:1000) absorbed with an excess (10-6 M) of the synthetic peptide used to raise the antibodies.
Results
The immunoblot analysis demonstrated that the antiserum reacted only with Type 5 17β-HSD (Figure 1). No crossreactivity was detected either with 3α-HSD Types 1 and 3, which share approximately 80% identity with 17β-HSD and are present in the testis, mammary gland, and ovary (Khanna et al. 1995; Dufort et al. 1996; Penning 1997), or with 5α-reductase Types 1 and 2, two enzymes that are abundant in prostate tissue (Anderson and Russell 1990; Pelletier et al. 1998).
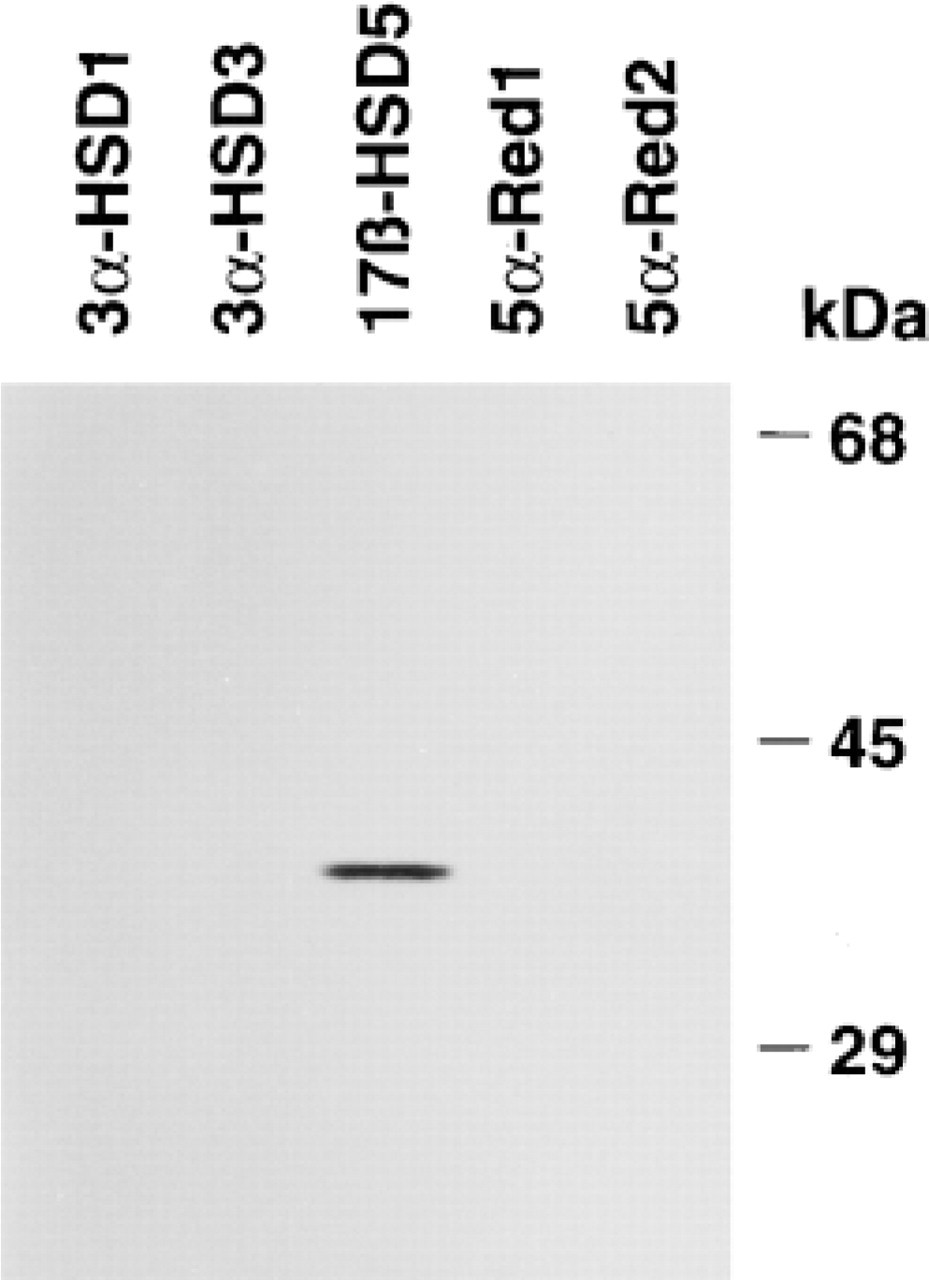
Specificity of the antiserum used in the immunostaining of Type 5 17β-HSD, as established by immunoblotting. The immunoblot analysis indicates that the antiserum specifically reacts with Type 5 17β-HSD. Types 1 and 3 3α-HSD, which share 84% and 86% amino acid identity with Type 5 17β-HSD, respectively, and Types 1 and 2 5α-reductase, two other androgen-synthesizing enzymes, were used as negative controls.
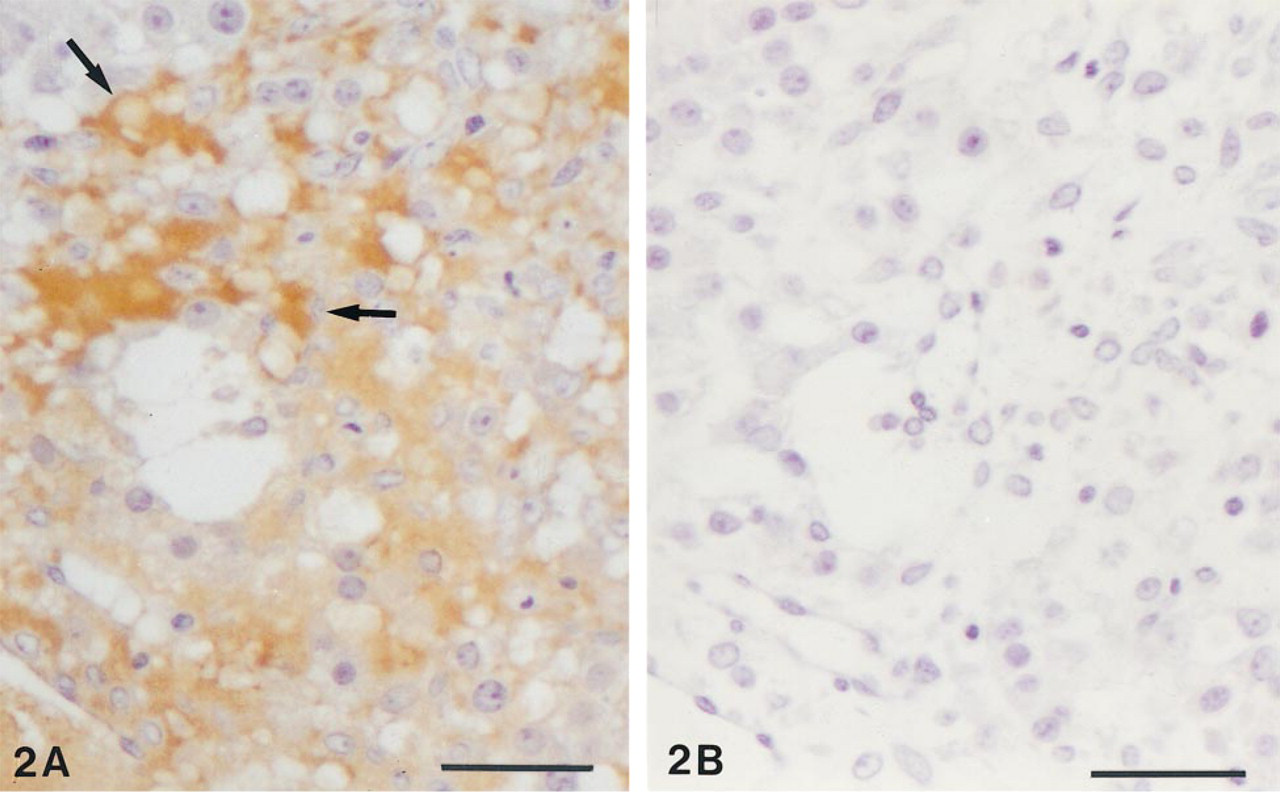
In the ovary, immunolabeling was detected only in corpora lutea. In these structures, most of the luteal cells were moderately labeled (Figure 2A). Both large cells originating from the granulosa layer and small cells originating from the theca interna were immunolabeled. As shown in Figure 2B, immunoabsorption with the antigen completely prevented the staining. No labeling could be detected in the different types of follicles, including the secondary and preovulatory follicles, whereas specific staining was routinely detected in the walls of blood vessels (Figure 3). In the uterus, specific immunolabeling was detected in the endometrium, the myometrium being completely unlabeled. Both epithelial cells lining the glands and those covering the surface were strongly reactive (Figure 4). In the glandular epithelium, both the secretory (functional) cells and the basal cells (when they could be identified) showed a positive reaction. The endometrial stroma was devoid of labeling.
In mammary glands from premenopausal women, strong specific staining could be detected in the epithelial cells of acini and intralobular ducts and in stromal fibroblasts (Figure 5). In both cell types, the labeling appeared mainly cytoplasmic. Staining was also detected in the walls of blood vessels, including endothelial cells. In interlobular (Figure 5) and lactiferous ducts, staining also was detected in the epithelial cells. When the antiserum was absorbed with the antigen, no labeling could be detected (not shown).
In the testis, strong staining was found in the Ley-dig cells (Figure 6). In approximatively 10% of tubules, weak labeling could be observed in the seminiferous epithelium (not shown). Sertoli as well as germ cells, especially spermatogonia and spermatocytes, showed immunolabeling. In control sections, no labeling could be detected in Leydig cells as well as seminiferous epithelium. In the prostate, immunoreactivity was detected in alveoli and stroma (Figures 7 and 8). In alveoli, staining was consistently found in the basal cells of the epithelium, whereas the luminal secretory cells exhibited variation in immunoreactivity. Whereas in some alveoli no luminal cells were labeled (Figure 7), other alveoli contained a large number of positive luminal cells (Figure 8). In general, the staining of luminal cells was weaker than that observed in basal cells. In the stroma, labeling was observed in the fibroblasts, the unstained cells probably being smooth muscle cells. Specific immunolabeling was found in the endothelial cells of capillaries, veins, and arteries. In veins, all the fibroblasts in the walls were stained, whereas in arteries, the tunica adventitia, but not the tunica media, was immunolabeled. When the antiserum was immunoabsorbed with the antigen, no staining could be detected.
Discussion
To precisely determine the structures and the cell types producing Type 5 17β-HSD in human reproductive tissues, we have developed antibodies against a synthetic peptide designed on the sequence of the Type 5 17β-HSD protein (aa 297-320). The antibodies appear to be specific, as verified by immunoblot analysis as well as immunoabsorption studies with the antigen in immunocytochemical studies.
The present data clearly indicate that, in each of the reproductive tissues investigated, the enzyme expression occurs in specific cell types. It is now quite clear that Type 5 17β-HSD and Type 2 3α-HSD are the same enzyme (Lin et al. 1997; Dufort et al. 1999). It remains possible that some Type 2 3α-HSD activity might be present in the cells immunolabeled with antibodies to Type 5 17β-HSD. On the other hand, because previous results from this laboratory have indicated that all the reproductive tissues from human and rhesus monkey have an androgenic 17β-HSD activity (Labrie et al. 1997), it can be suggested that the conversion of 4-dione to testosterone is related to activity of Type 5 17β-HSD. Because Type 3 17β-HSD has been identified in the testis (Andersson et al. 1995), androgenic activity observed in this organ might also be related, at least in part, to Type 3 17β-HSD activity. Thus far, Type 3 17β-HSD has not been found in other reproductive tissues.
In the testis, the presence of Type 5 17β-HSD in the Leydig cells suggests that these cells are involved in the conversion of 4-dione to testosterone needed for the internal male reproductive structures (epididymis, seminal vesicles, and vas deferens) as well as all secondary sex organs. Because 3β-HSD is also localized in Leydig cells (Levy et al. 1959; Pelletier et al. 1992), it is likely that DHEA from the general circulation is converted to 4-dione in these endocrine cells. The respective contributions of Type 5 17β-HSD and Type 3 17β-HSD remain to be established. Recently, it has been shown that impairment of Type 3 17β-HSD (Geissler et al. 1994) leads to the well-characterized male pseudohermaphroditism.
In the prostate alveoli, immunoactive Type 5 17β-HSD has been detected predominantly in the basal cells. A similar finding has also been observed using in situ hybridization to localize Type 5 17β-HSD mRNA (El-Alfy et al. 1999). Because not only Type 5 17β-HSD but also 3β-HSD (El-Alfy et al. 1999) is highly expressed in basal cells, it can be suggested that testosterone synthesized by basal cells reaches the luminal cells in a paracrine way to be ultimately transformed into dihydrotesterone (DHT) in the luminal cells which contain 5α-reductase Types 1 and 2 (Levine et al. 1996; Negri-Cesi et al. 1998; Pelletier et al. 1998). The androgen would then exert its action in the luminal cells themselves, which contain androgen receptors (Sar et al. 1990; Orlowski and Clark 1991; Iwamura et al. 1994; El-Alfy et al. 1999), as an intracrine activity. Strong immunostaining for Type 5 17β-HSD has also been detected in the stromal fibroblasts, which have been shown by various techniques to contain 5α-reductase Types 1 and 2 (Eicheler et al. 1994; Levine et al. 1996; Pelletier et al. 1998) as well as androgen receptors (Kimura et al. 1993). Although the role of the steroidogenic enzymes in fibroblasts remains to be established, it is likely that DHT could act in the fibroblasts themselves (intracrine action) to modulate the activity of these cells.
(
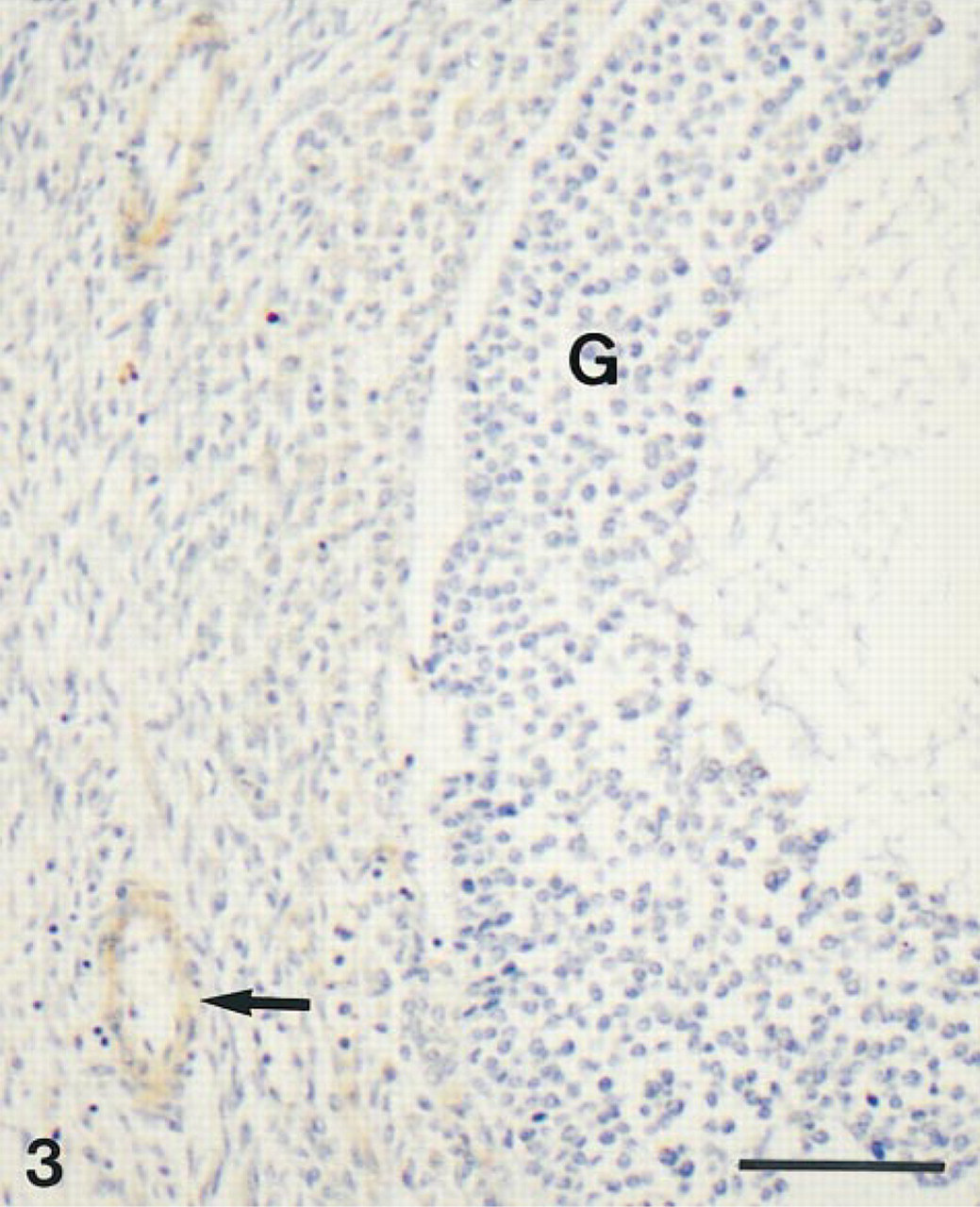
Low-power view of a portion of an antral follicle. No staining can be detected in granulosa cells (G). Note immunostained walls (→) of blood vessels. Bar = 100 μm.
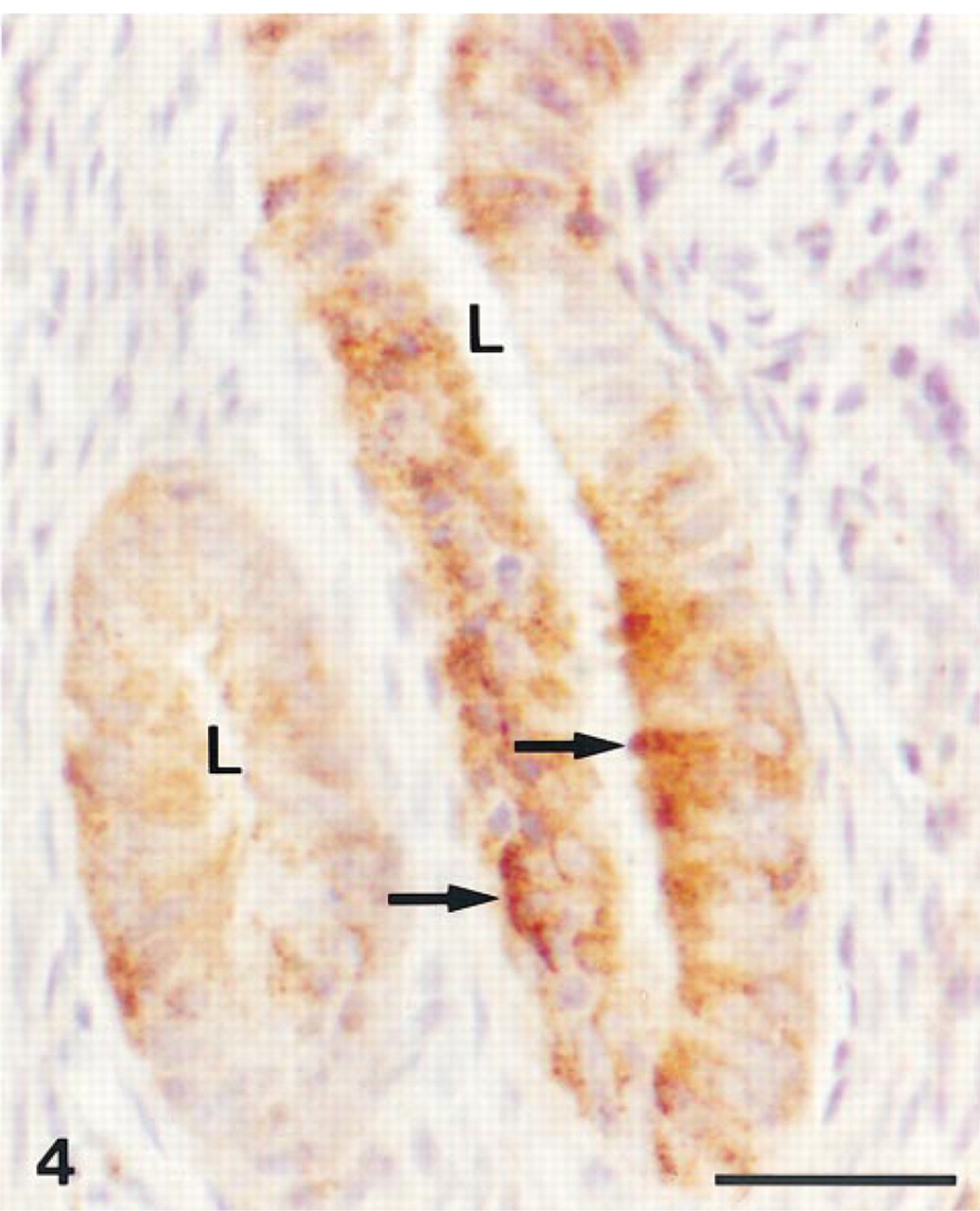
Uterus. Immunolabeling is observed in the epithelial secretory cells (→) of uterine glands. The stroma is unlabeled. L, lumen. Bar = 30 μm.
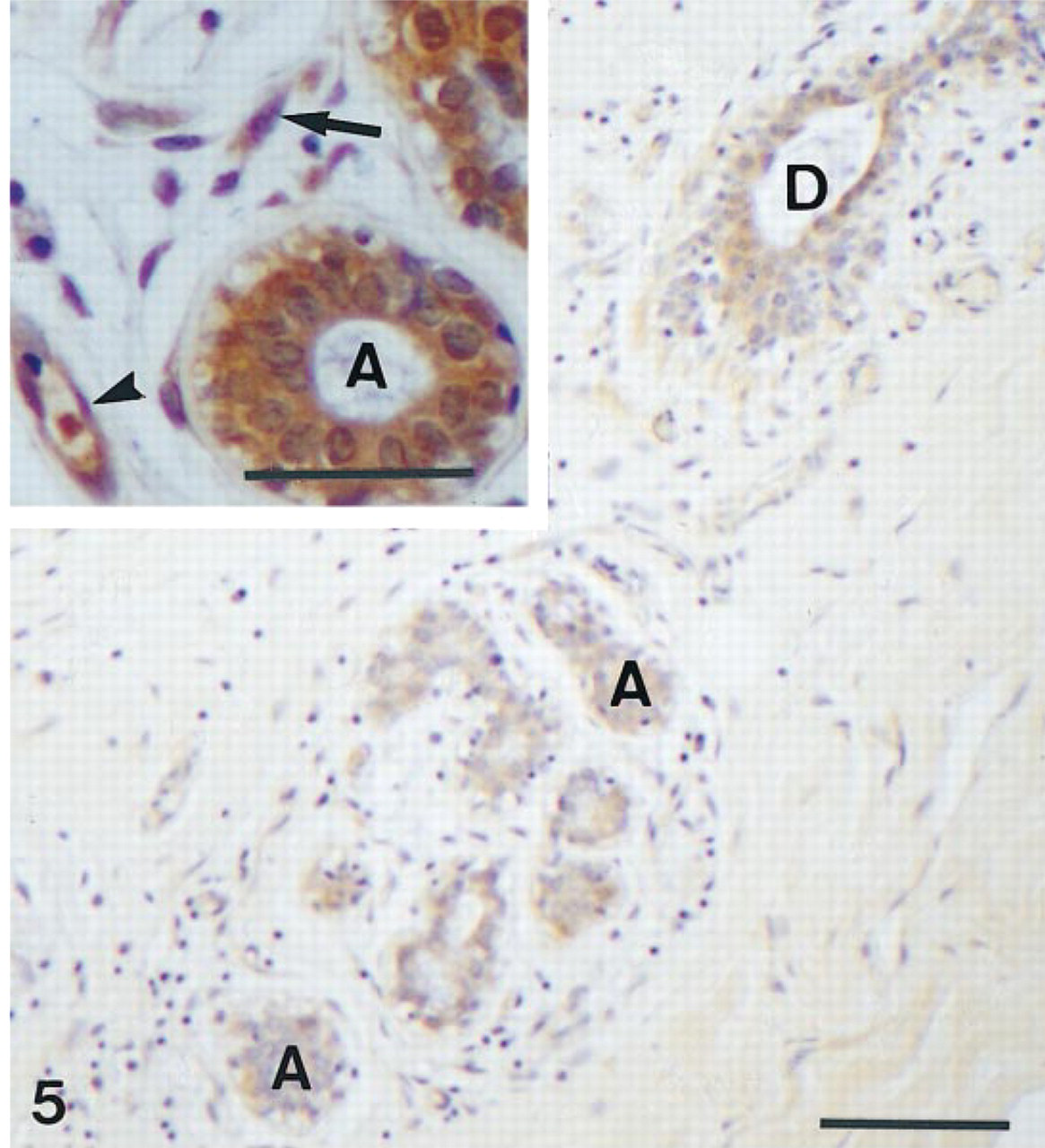
Mammary gland. Immunostaining is present in the secretory epithelial cells of acini (A) and an interlobular duct (D). Bar = 100 μm. (
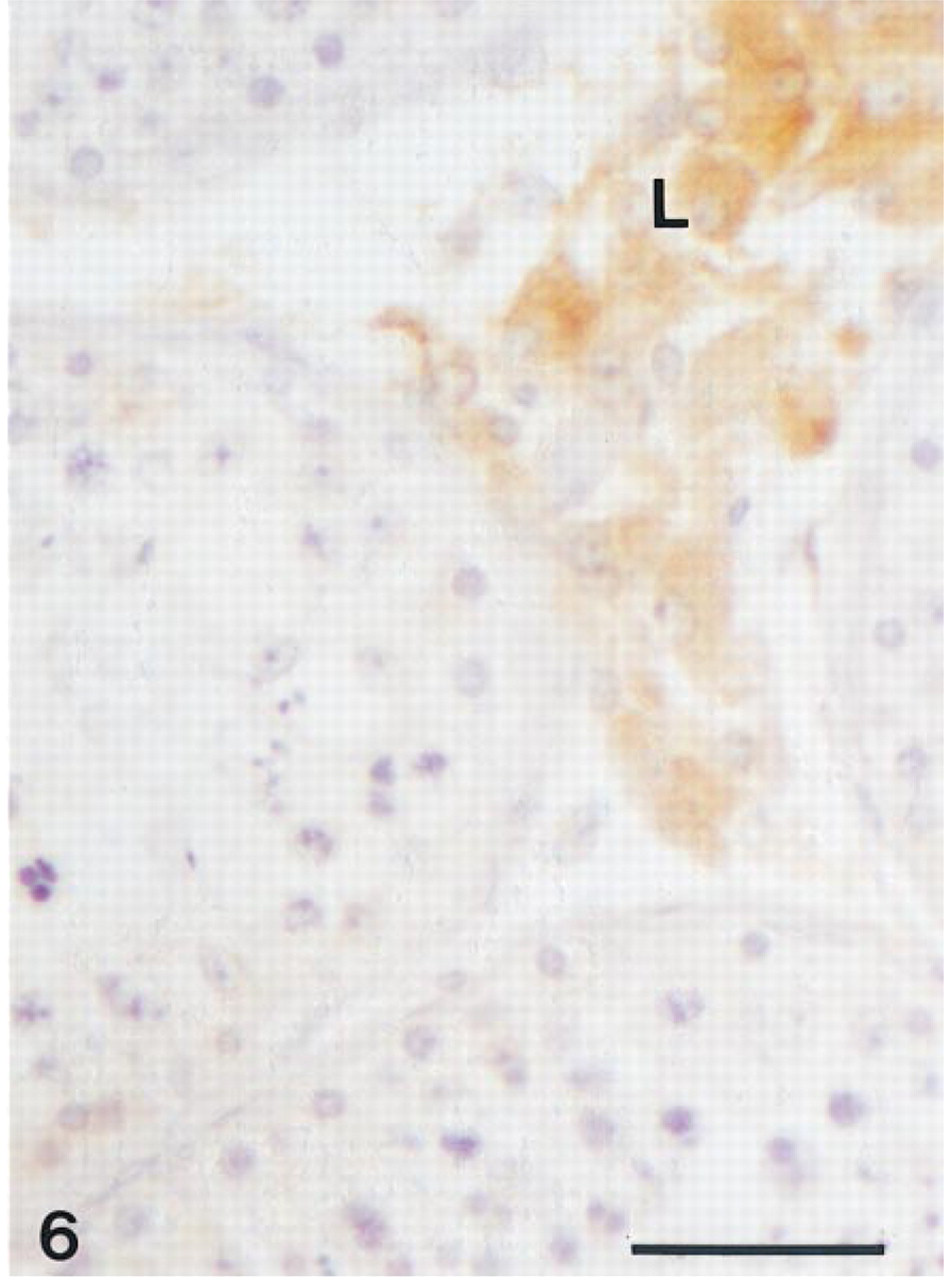
Testis. Leydig cells (L) are immunoreactive, whereas no labeling can be detected in the seminiferous tubules. Bar = 30 μm.
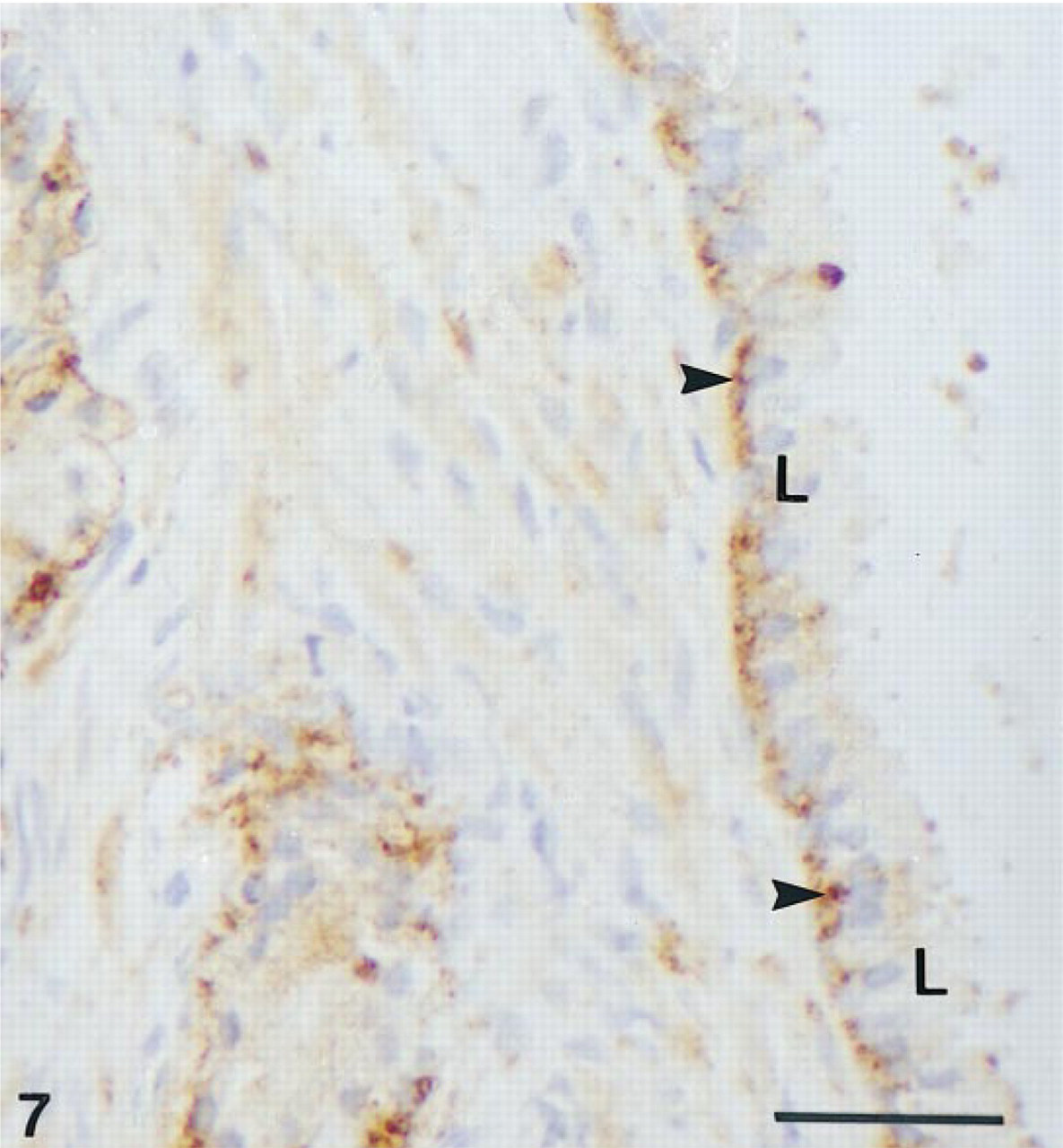
Prostate. Immunostaining can be detected in the basal cells (arrowheads) of an alveolus. The luminal cells (L) are unlabeled. Bar = 30 μm.
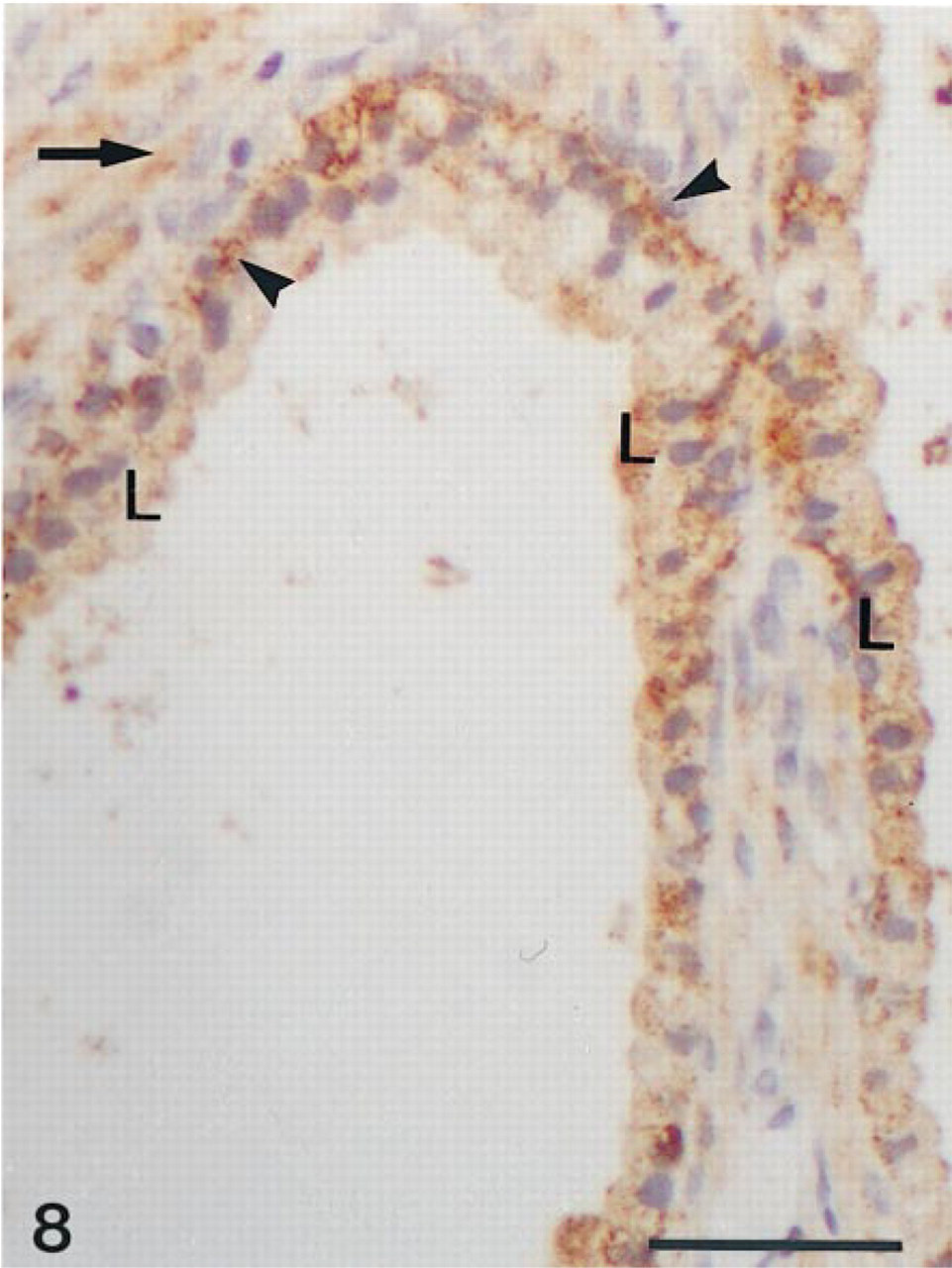
Prostate. Reaction product is observed in the epithelium of two alveoli. The basal cells (arrowheads) are more strongly stained than the luminal cells (L). Surrounding stromal cells are also labeled (→). Bar = 30 μm.
In the ovary, Type 5 17β-HSD immunoactivity has been found in the majority of luteal cells but not in the follicular cells. This is in contrast to the findings on 3β-HSD localization, which demonstrated the presence of this enzyme not only in luteal cells but also in theca interna cells (Pelletier et al. 1992). Because it is well known that in luteal cells testosterone is aromatized into estradiol (McNatly et al. 1979; Ohara et al. 1987), Type 5 17β-HSD might be involved in testosterone production, leading ultimately to estradiol synthesis. On the other hand, because androgen receptors have been detected in luteal cells during the midluteal phase and also in corpora lutea of the late luteal phase (Hild-Petito et al. 1991), it might be suggested that locally produced testosterone can exert an influence on the luteal cells themselves.
In the mammary glands, strong immunolabeling was detected in epithelial cells of both alveoli and ducts, as well as in stromal cells that probably represent fibroblasts. Because these cell types have been shown to contain androgen receptors (Ruizeveld de Winter et al. 1991; Kimura et al. 1993), the presence of Type 5 17β-HSD suggests that the intracellularly synthesized androgen may directly influence the activity of the same cells (intracrine activity). Estrogens produced after aromatization (Adams and Wong 1968; Adams et al. 1978) can also have an intracrine or autocrine activity. Thus far, the cellular localization of aromatase has not been reported.
In the uterus, Type 5 17β-HSD could be detected only in the epithelial cells of the endometrial glands. In the absence of androgen receptors in the uterus (Ruizeveld de Winter et al. 1991), locally produced testosterone might play a role as a precursor of estradiol, which could act on the epithelial secretory cells themselves, which contain estrogen receptors (Jensen and DeSombre 1972), in an intracrine fashion. Estradiol could also influence the stromal as well as the smooth muscle cells by a paracrine mechanism.
Another interesting finding was the localization of Type 5 17β-HSD in blood vessel walls, including the endothelial cells. This observation correlates very well with recent findings from this laboratory indicating the presence of 5α-reductase Types 1 and 2 mRNA in blood vessel walls in human prostate and skin (Pelletier et al. 1998). We have also recently shown that androgen receptors are detected in the endothelial cells and fibroblasts of blood vessels in the prostate (manuscript in preparation). It can be speculated that locally biosynthesized androgens are exerting a paracrine and/or intracrine action. It is also possible that these androgens are released into the blood circulation to reach some target tissues, although their global impact is likely to be minimal. Interestingly, Franck-Lissbrant et al. (1998) have shown that, in the rat prostate, testosterone could induce a rapid response of the vasculature which precedes growth of the glandular epithelium. It might well be that endothelial cells are stimulated by locally made androgens to produce paracrine growth factors that can promote the growth of the secretory epithelium. Further studies are required to elucidate the role of the steroids potentially biosynthesized by blood vessel walls.
In conclusion, the results obtained using specific antibodies to human Type 5 17β-HSD clearly indicate a cell-specific localization of the enzyme in human reproductive organs. In fact, endocrine steroid-secreting cells (Leydig and luteal), as well as epithelial secretory cells and stromal cells, express Type 5 17β-HSD. Therefore, the present data suggest new mechanisms of local androgen formation that may play an important physiological role in the different reproductive organs.
