Abstract
Thymidine phosphorylase (dThdPase) is an essential enzyme for activation of the oral cytostatic drug capecitabine and its intermediate metabolite, doxifluridine, to 5-fluorouracil in tumors. Methods to estimate dThdPase expression in tumor tissue might be useful to predict the efficacy of capecitabine and doxifluridine in cancer patients. We established a new monoclonal antibody (MAb), 1C6–203, applicable for dThdPase immunohistochemistry and compared its staining characteristics with those of a previously established MAb, 654–1. In 4% paraformaldehyde-fixed colorectal carcinoma, 1C6–203 and 654–1 stained cancer cells in 19/30 and 9/30 patients, respectively. In 10% formalin-fixed colorectal carcinoma, 1C6–203 and 654–1 stained cancer cells in 18/30 and 6/30 patients, respectively. In negative 10% formalin-fixed tissues, microwave treatment improved the positivity of 654–1-stained cancer cells. These results suggest that an epitope recognized by 1C6–203 is resistant to epitope masking by formaldehyde fixation, whereas that for MAb 654–1 is sensitive. Therefore, MAb 1C6–203 might be more suitable than MAb 654–1 for evaluating dThdPase expression in colorectal carcinoma.
Keywords
T
On the other hand, dThdPase is identical to platelet-derived endothelial cell growth factor (PD-ECGF) and is reported to promote angiogenesis (Ishikawa et al. 1989; Haraguchi et al. 1994). dThdPase/PD-ECGF is also believed to be fundamental to tumor growth (Hanahan and Folkman 1996; Saeki et al. 1997) and metastasis in gastric cancer (Maeda et al. 1996; Tanigawa et al. 1996).
However, in colorectal carcinoma tissues, the frequency of positive dThdPase expression in tumor cells and tumor stroma remains to be investigated. A monoclonal antibody (MAb) against dThdPase, 654–1 (Nishida et al. 1996), used for staining of clinical specimens (Takahashi et al. 1996; Amaya et al. 1997), gives a rather low frequency of positive staining in colorectal cancer tumor cells unless antigen is retrieved by microwave treatment. This MAb may not be suitable for evaluating tumor cell positivity in formalin-fixed colorectal carcinoma.
Recently, we developed a new anti-human dThdPase MAb by using a recombinant dThdPase as an antigen for staining formalin-fixed sections. In this study, we compared our new antibody with those currently in use in clinical research.
Materials and Methods
Immunization and Hybridoma Preparation
BALB/c mice were immunized with 50 μg of recombinant dThdPase, prepared as described elsewhere (Moghaddam and Bicknell 1992), in complete Freund's adjuvant. Boosters containing 50 μg of recombinant dThdPase in complete Freund's adjuvant were given twice at 4-week intervals. Six days after the final booster, the mice were sacrificed and their dissociated spleen cells were fused with P3×63-Ag8.653 myeloma cells (Kearney et al. 1979) using standard fusion technique. Hybridoma clones that produced anti-dThdPase antibody were screened by antibody capture assay. Clones that produced an antibody useful for immunohistochemistry were further screened by immunohistochemistry of paraffin-embedded human colorectal, breast, and gastric cancer tissues fixed with 10% neutral buffered formalin. Tissue specimens used for hybridoma screening were from the tissue archives of our laboratory. Isotypes of clones were determined with an Immuno Pure Monoclonal Antibody Isotyping kit I (Pierce; Rockford, IL).
Antibody Capture Assay
A 96-well microtiter plate (Nunc-immunoplate Maxisorp; Nunc, Roskilde, Denmark) was incubated with 0.25 μg/ml of the recombinant dThdPase in 10 mM PBS (pH 7.6) overnight at 4C. The coated plate was then incubated with 3% (w/v) skim milk in PBS (blocking buffer) for 1 hr at room temperature (RT). The plate was washed with PBS containing 0.05% Tween-20 and 0.05% sodium azide and kept at 4C until used. Hybridoma culture supernatants were dispensed onto a plate coated with the recombinant dThdPase. The plate was (a) incubated at 37C for 1 hr and washed with PBS containing 0.05% Tween-20 (washing buffer), (b) incubated with 1000-fold diluted anti-mouse IgGγ conjugated with horseradish peroxidase (KPL; Gaithersburg, MD) for 1 hr at 37C and washed, and (c) incubated with a substrate solution containing 3,3′,5,5′-tetramethylbenzidine (TMB) and H2O2 (TMB microwell peroxidase substrate system; KPL) for 10–20 min at RT. The peroxidase reaction was stopped by addition of 1 M phosphate solution, and absorbance was read at 450 nm with a plate reader (BIO-RAD Model 3550; Bio-Rad Laboratories, Hercules, CA).
Western Blotting Analysis
Fifty μg of human tumor homogenates was electrophoresed on 5–20% SDS-polyacrylamide gradient gels (PAGEL; ATTO, Tokyo, Japan) according to the method of Laemmli (1970), and proteins in the gels were electrophoretically transferred to a polyvinylidine difluoride membrane (Millipore; Bedford, MA). The polyvinylidine difluoride membrane was treated with a 20 mM Tris-buffered saline (pH 7.5) containing 3% (w/v) skim milk for 1 hr. Then the membrane was incubated overnight at 4C with 5 μg/ml of established MAb 1C6–203. The membrane was washed with Tris-buffered saline containing 0.05% Tween-20 and then incubated with biotinylated anti-mouse IgG after incubation with avidin-biotin-peroxidase complex (Vectastain ABC elite kit; Vector Laboratories, Burlingame, CA). After washing, the membrane was developed using the Konica Immunostain HRP-1000 (Konica; Tokyo, Japan).
Patients and Methods
All tissues used in this study were obtained from surgically resected specimens. Carcinomas were analyzed from colorectal adenocarcinoma (n = 30), gastric adenocarcinoma (n = 5), and breast cancer (n = 5). The histopathological diagnosis was confirmed in all patients by the Department of Surgical Pathology (Asahikawa Medical College Hospital).
Specimens were washed in ice-cold 0.1 M PBS (pH 7.4) and immersed in either 4% paraformaldehyde or 10% formaldehyde in 0.1 M phosphate buffer (PB, pH 7.4). After incubation for 12 hr at 4C, the tissues were kept at 4C in 25% sucrose in PB (pH 7.4) until they sank. Then tissue specimens were embedded in paraffin and consecutively cut into 4–6-μm-thick sections from each tumor block, and mounted on gelatin-coated glass slides. The sections were deparaffinized with xylene and rehydrated with 98% ethanol. In dThdPase-negative cases, consecutive deparaffinized sections were placed in 0.1 M citrate buffer (pH 6.0) and antigen retrieval was performed by heating in a microwave oven for 5 min twice (500 W). After soaking in 0.1 M PB containing 3% blocking serum and 0.3% Triton X-100, the slides were incubated with mouse MAb 654–1 against human dThdPase (10 μg/ml) or MAb 1C6–203 (10 μg/ml), overnight at 4C. The slides were washed with PBS, incubated with secondary antibodies, stained with the ABC staining technique (Vector Laboratories), and developed with diaminobenzidine tetrahydrochloride in PBS containing 0.03% H2O2. The sections were counterstained with hematoxylin and viewed and photographed using a Nikon microscope. As a control for nonspecific staining, mouse IgG1κ (ICN Pharmaceuticals; Aurora, OH) was used as primary antibody.
We examined 200 cells, including cancer cells or various stromal cells in tumor tissue, and normal mucosal cells or various stromal cells in normal tissue, to determine whether the cells were dThdPase-positive. Because more than 90% of normal colorectal tissues showed less than 5% epithelial cell staining for dThdPase, specimens were regarded as dThdPase-negative when less than 5% of 200 cells were stained and as positive when more than 5% of them were stained. Specimens were evaluated independently by two experienced pathologists, who were blinded to the dThdPase antibody type and the fixation.
Statistics
The two-sided Fisher exact test was used to assess the difference in the frequency of dThdPase immunostaining among the various groups shown in Table 1. p < 0.05 was regarded as statistically significant.
Results
Specificity of the Anti-dThdPase MAb
Anti-dThdPase MAb 1C6-203 is a mouse IgG1κ. Figure 1 shows Western blotting analysis of various human cancer tissue homogenates with the MAb 1C6-203. 1C6-203 reacted only with a protein band having a molecular mass of 55 kD, which corresponds to dThdPase protein in breast, gastric, and colorectal cancer tissue homogenates. The specificity of 1C6-203 was consistent with that of the previously reported anti-dThdPase antibody 654-1 (Nishida et al. 1996), which is also mouse IgG1κ.
Frequency of dThdPase expression in colorectal carcinoma using immunohistochemistry with different fixations and different antibodies
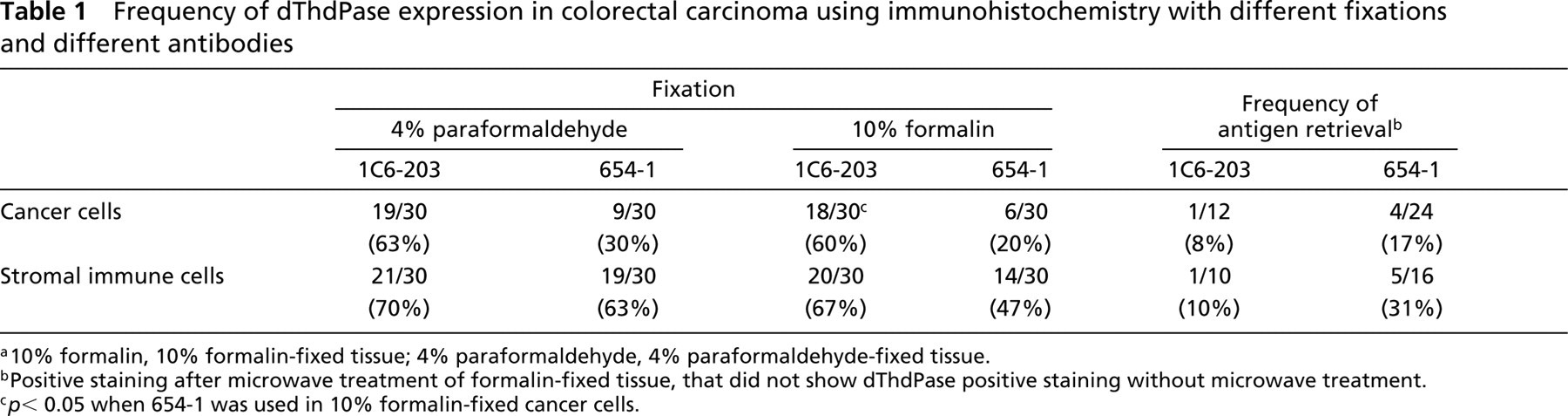
a10% formalin, 10% formalin-fixed tissue; 4% paraformaldehyde, 4% paraformaldehyde-fixed tissue.
bPositive staining after microwave treatment of formalin-fixed tissue, that did not show dThdPase positive staining without microwave treatment.
c p < 0.05 when 654–1 was used in 10% formalin-fixed cancer cells.
dThdPase Expression in Colorectal, Breast, and Gastric Carcinoma Tissues
In 10% formalin-fixed colorectal tumor tissues, 1C6–203 stained both tumor cells and stromal cells, whereas 654–1 stained mainly stromal cells and not tumor cells in many cases (Figure 2). However, in breast and gastric tumor tissues, both 1C6–203 and 654–1 stained tumor cells and stromal cells (Figure 3).
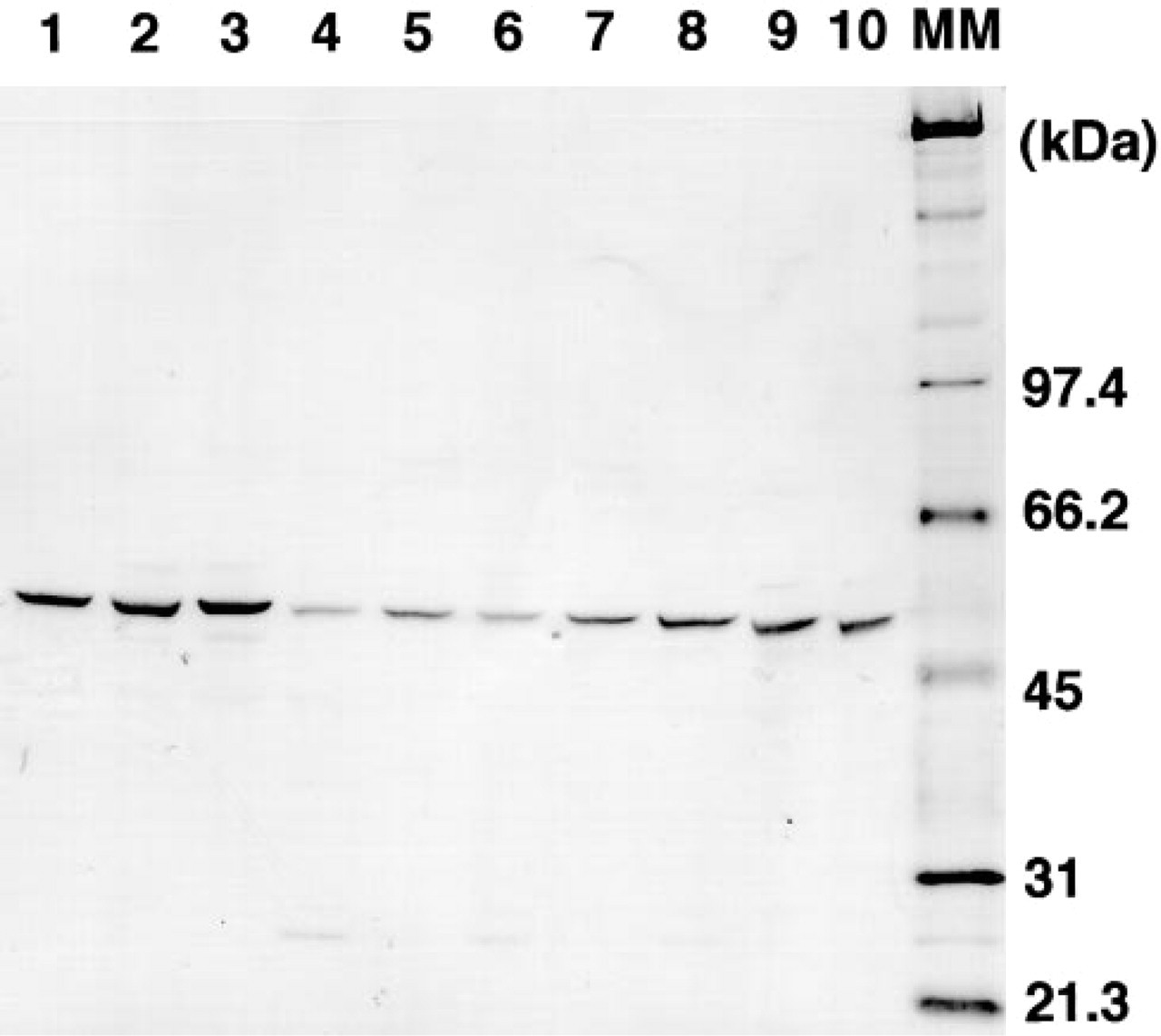
Western blotting analysis of human cancer tissue homogenates with anti-dThdPase MAb 1C6–203. Fifty-μg homogenates of breast tumor (Lanes 1–3), gastric tumors (Lanes 4–6), and colorectal tumor (Lanes 7–10) were subjected to SDS-PAGE and blotted onto Immobilon-P membrane. MM, molecular mass marker.
Most histologically normal colorectal mucosal cells were not stained with MAb 1C6-203, similar to the results with MAb 654–1, in both 4% paraformaldehyde-fixed and 10% formalin-fixed tissues. The percentage of cells expressing dThdPase was less than 5% of 200 cells in 28 (93.3%) of 30 4% paraformaldehyde-fixed normal tissues, and in 29 (96.7%) of 30 10% formalin-fixed tissues when stained with MAb 1C6-203. Various stromal dThdPase-expressing cells were detected in normal interstitum. Of 30 normal tissues, 14 (47%) and 12 (40%) showed more than 5% of examined stromal immune cells in 4% paraformaldehyde-fixed and 10% formalin-fixed tissues, respectively.
dThdPase-expressing cancer cells in colorectal carcinoma were detected by MAb 1C6-203 in 19 (63.3%) of 30 paraformaldehyde-fixed tissues and 18 (60%) of 30 formalin-fixed tissues, respectively (Table 1). In dThdPase-expressing cancer cells, dThdPase staining was observed in nucleus and/or cytoplasm (Figure 2A). Although various degrees of stromal dThdPase expression were detected in most colorectal cancer specimens, including immune cells (macrophages and lymphocytes), fibroblasts, and endothelial cells, they occurred predominantly in immune cells (macrophages and lymphocytes) in the interstitum adjacent to cancer cells (Figure 2B). Of 30 colorectal tumors, 21 (70%) and 19 (63%) showed more than 5% of positive stromal immune cells in 4% paraformaldehyde-fixed and 10% formalin-fixed tissues, respectively (Table 1).
We compared frequencies of dThdPase staining in colorectal carcinoma using different fixations and different antibodies (Table 1). In 10% formalin-fixed colorectal cancer tissue, 18 (60%) of 30 cases displayed positive dThdPase immunoreactivity in carcinoma cells with MAb 1C6-203, in contrast to six (20%) of 30 with MAb 654–1. Twelve of the 24 negative specimens stained with MAb 654–1 were positive with MAb 1C6-203 (Figure 2). Therefore, the frequency of dThdPase expression was significantly reduced when MAb 654-1 was used in formalin-fixed tissues (p < 0.05). Despite this, there was no significant difference between 4% paraformaldehyde and 10% formalin with MAb 1C6-203.
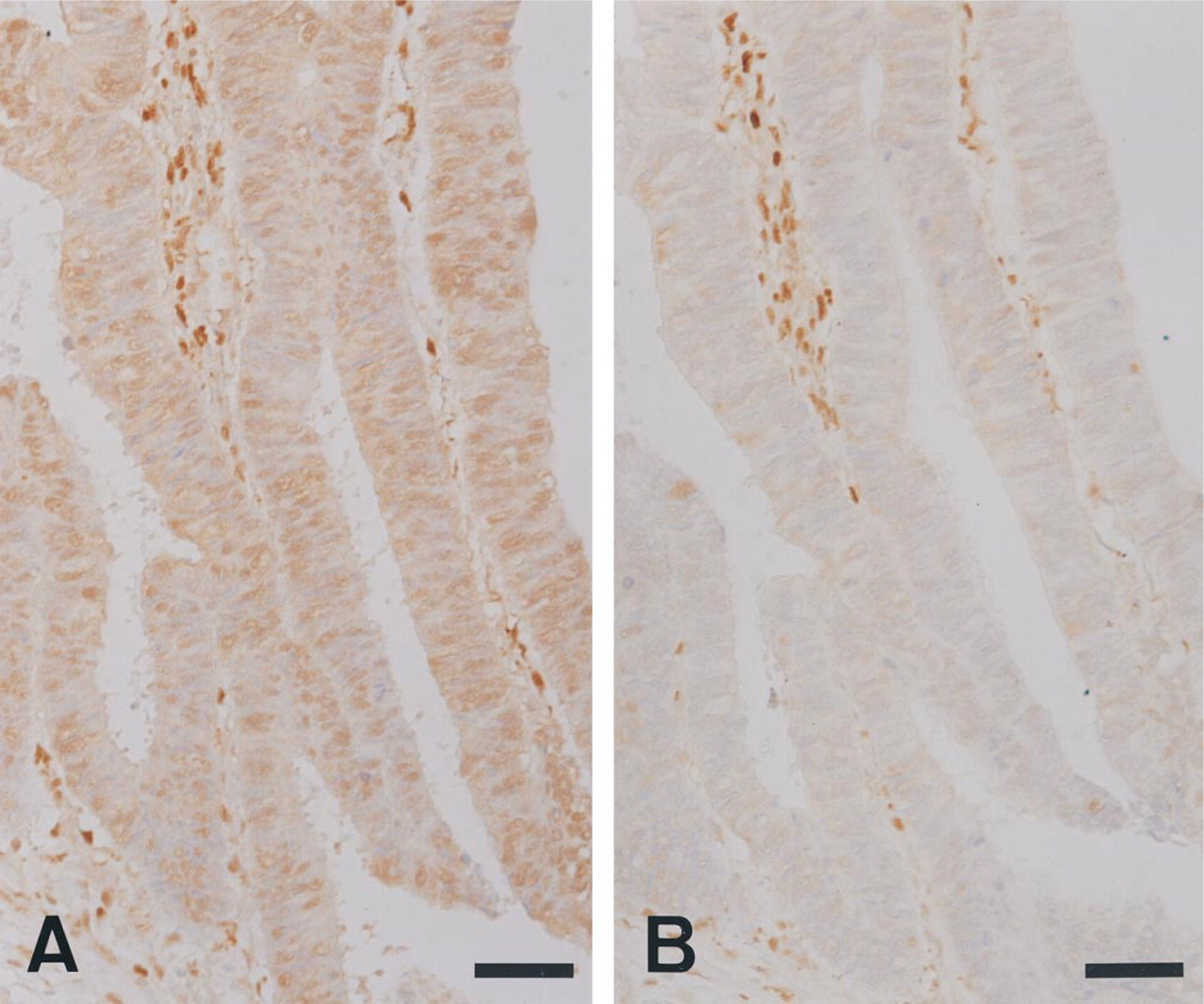
Immunohistochemical staining with a new MAb, 1C6–203, for dThdPase in 10% formalin-fixed colon cancer tissue (
In dThdPase-negative 10% formalin-fixed and 4% paraformaldehyde-fixed tissues, antigen retrieval was performed by microwave heating and slides were reexamined. MAb 654–1, but not MAb 1C6–203, gave a higher frequency of dThdPase expression in dThdPase-negative cases (Table 1).
Discussion
Thymidine phosphorylase is an essential enzyme for activating the oral cytostatic drug capecitabine and its intermediate metabolite, doxifluridine, in tumors (Kono et al. 1983; Miwa et al. 1998). However, in clinical studies the correlation between capecitabine or doxifluridine efficacy and tumor level of dThdPase expression has not been fully investigated except a few cases (Toi et al. 1997; Ishii et al. 1999). In addition, dThdPase expression in tumor tissue has garnered attention because it is identical to PD-ECGF (Furukawa et al. 1992), an angiogenic factor, and correlates with tumor vascularity (Ishikawa et al. 1989; Haraguchi et al. 1994). Various carcinoma tissues are immu nostained with anti-dThdPase antibodies. Reportedly, dThdPase expression is associated not only with tumor hypervascularity but also with the prognosis for gastric (Maeda et al. 1996; Tanigawa et al. 1996) and colorectal cancers (Takahashi et al. 1996; Takebayashi et al. 1996a), hepatic metastasis of gastric cancer (Maeda et al. 1996; Tanigawa et al. 1996), and solid tumor proliferation (Takebayashi et al. 1996b). Recently, it was reported that dThdPase expression in tumor-associated stromal immune cells is also an important marker for prognosis (Toi et al. 1999).
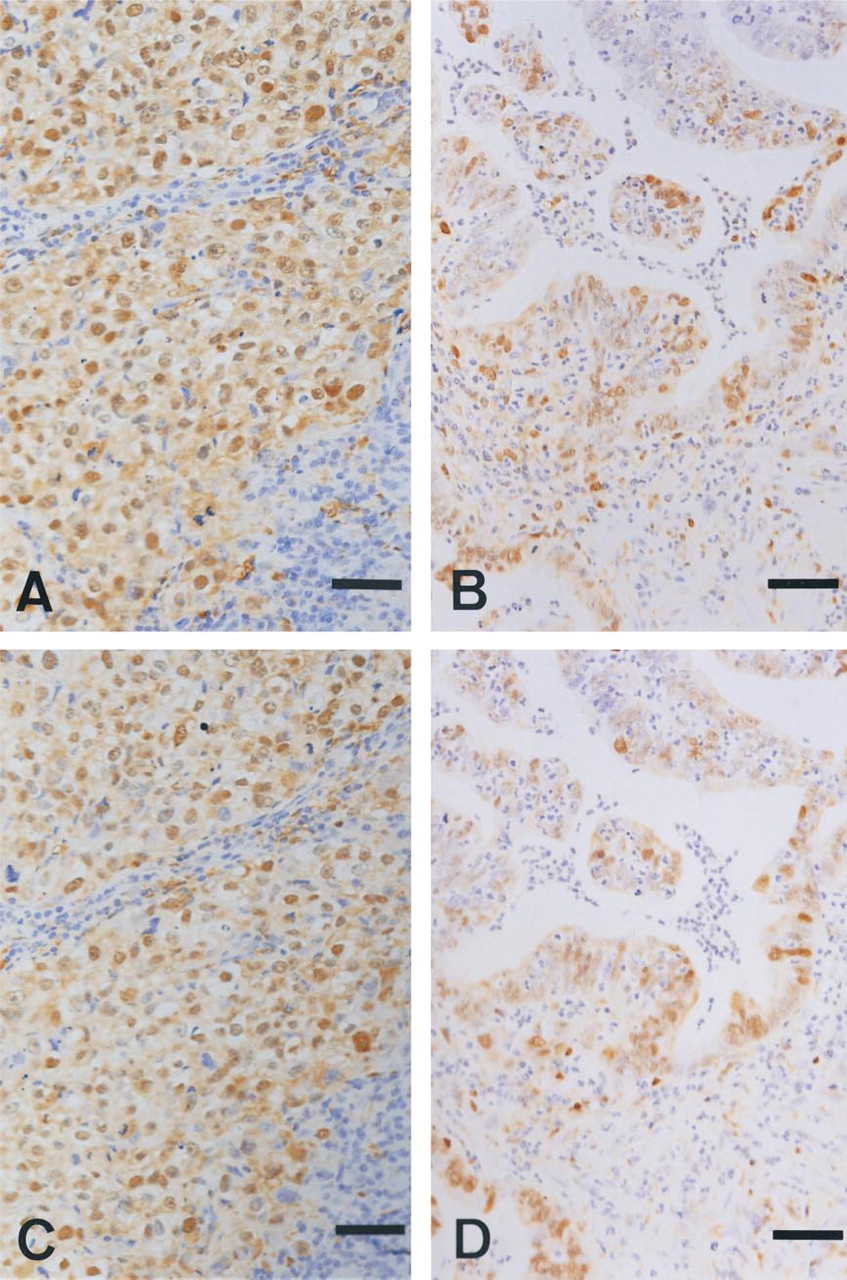
Immunohistochemical staining with a new MAb, 1C6-203, for dThdPase in 10% formalin-fixed breast cancer tissue (
There are indications that dThdPase immunoexpression in colorectal tumor cells of formalin-fixed tissue is weak when MAb 654–1 (Takahashi et al. 1996) is used, which is generally employed in clinical research (Nishida et al. 1996). The frequency of dThdPase-positive staining with MAb 654–1 in formalin-fixed colorectal cancer tissue was 20% (cancer cells) and 47% (stromal immune cells) in our experiment. This was almost equivalent to the dThdPase-positive frequency in tumor cells of formalin-fixed colorectal cancer tissue in previous reports (Takahashi et al. 1996; Amaya et al. 1997). However, anti-dThdPase MAb 1C6-203 showed a higher frequency of dThdPase expression than MAb 654-1 in formalin-fixed colorectal cancer, although both antibodies showed a similar high specificity of dThdPase in Western blotting analysis of tumor tissue homogenates. The higher frequency of dThdPase expression with MAb 1C6-203 that occurred in formalin-fixed colorectal cancers may be due to differences in epitope recognition between MAb 1C6-203 and MAb 654-1. MAb 1C6-203 was raised against recombinant human dThdPase and MAb 654-1 was directed against human dThdPase refined from a human colon cancer HCT116 xenograft. MAb 654-1 and MAb 1C6-203 did not interfere with each other in the antibody capture assay (data not shown). Moreover, the frequency of dThdPase expression revealed by MAb 1C6-203 was not improved by microwave processing, whereas the frequency of dThdPase expression with MAb 654-1 improved in formalin fixed tissue. The masking effect of formalin-fixation on the epitope is cancelled by microwave processing (Flossel et al. 1994; von Wasielewski et al. 1994). Therefore, MAb 654-1, but not MAb 1C6-203, may recognize the epitope close to the crosslink-age position induced by formalin fixation.
On the other hand, in contrast to colorectal cancers, dThdPase positive frequency and staining intensity of tumor cells in breast and gastric cancers estimated by two MAbs, 1C6-203 and 654-1, were similarly high in a preliminary analysis. In colorectal cancer tissue, dThdPase was expressed not only in tumor cells but also in tumor stromal cells in the present study. These findings are consistent with those previously reported (Maeda et al. 1996; Takebayashi et al. 1996b).
The above observations indicate that MAb 1C6-203 may be better than MAb 654-1 when formalin-fixed colorectal cancer tissue is used for dThdPase immunohistochemical analysis. However, further studies are needed to clarify whether MAb 1C6-203 can be used for clinical histological studies in a long-term formalin-fixed samples. At this time, we do not have enough information to correlate the site of dThdPase expression, such as tumor cell or stromal cell cytoplasmic staining or nuclear staining, with the antitumor activity of capecitabine/doxifluridine. This question requires further studies to investigate the correlation of expression levels and localization of dThdPase with the antitumor activity of these drugs.
In conclusion, MAb 1C6-203 for human dThdPase was newly developed and revealed a high frequency of dThdPase expression in formalin-fixed colorectal cancer tissues. Therefore, 1C6-203 should be useful for future clinical studies.
Footnotes
Acknowledgments
We thank Mr Yoshiyasu Satake and Ms Yoko Okada for expert technical assistance.
