Abstract
Microscopic evaluation of whole-mount colons stained with methylene blue and/or hexosaminidase has identified putative preneoplastic lesions in the colons of rodents treated with carcinogen and in the grossly normal colons of humans. Enzyme histochemistry with glycol methacrylate sections has permitted the identification of putative premalignant lesions in rodent livers, human and rodent colons, and human prostates. Immunohistochemistry with paraffin-embedded tissues has been used to identify and characterize putative premalignant lesions in human colons and prostates.
Keywords
T
Materials and Methods
Specimens and Animal Protocols
Grossly normal human colonic mucosa and sections of human prostate were obtained for us by the Western Division of the Cooperative Human Tissue Network of the National Cancer Institute located at Case Western Reserve University. For experimental animal protocols, the reader is referred to the original articles cited for the specific studies.
Identification of Aberrant Crypt Foci (ACF) in Whole-mount Colons
Bird (1987) first identified ACF in whole-mount preparations of colons from carcinogen-treated rodents. The colons are rapidly removed, cleaned in an isotonic solution, such as 0.9% NaCl at 4C, and fixed flat. The type of fixative and the conditions of fixation are dictated by the subsequent techniques that are to be used. For routine microscopic evaluation of ACF in unembedded tissue after staining for 3–5 min with 0.2% methylene blue (Chroma-Gesellschaft Schmid, distributed by Roboz Surgical Instruments, Washington, DC), an extended fixation in formalin (neutral-buffered, 10% w/v, phosphate buffer, pH 7.0) (Fisher Scientific; Pittsburgh, PA) at room temperature (RT) works well. If subsequent enzymehistochemical evaluation of the ACF is planned, fixation in 2% paraformaldehyde in 0.1 M sodium phosphate buffer (pH 7.4) for 2 hr at 4C (Pretlow et al. 1990,1993b) is required to preserve the activity of some enzymes. For immunohistochemistry, initial fixation for 1 hr in formalin or 4% paraformaldehyde at RT works well for most antigens (Pret-low et al. 1994c). More recently, we have obtained better morphology with formalin fixation for 1 hr at 4C. A more even distribution of stain is obtained if the stained tissue is held for 30 min or more in 1% paraformaldehyde at 4C before it is viewed. The wet, fixed tissue is placed on a glass microscope slide with the mucosal side up and is viewed with a regular microscope or a dissecting microscope at X 30–100 magnification [rodent colon has smaller crypts (Figure 1A) and requires higher magnification than is required for the larger crypts found in human mucosa (Figures 1B and 1C)]. ACF are not as easily identified in sections of embedded colons as they are in unembedded colons. To assist in the location of ACF or other specific microscopic lesions after they are embedded, the lesions are painted with permanent ink (Davidson Marking System; Bradley Products, Bloomington, MN) with a fine needle while viewed under the microscope.
Fixed, unembedded tissue can be stained for the demonstration of enzyme activity, such as hexosaminidase, by using the same substrates as used for enzyme histochemistry with histological sections (Pretlow et al. 1993b). Alternatively, we found that colon tissue can be stained with methylene blue, evaluated, and then stained for demonstration of hexosaminidase to enable direct comparison of the two methods. Others (Caderni et al. 1995) have demonstrated sialomucins and sulfomucins in whole-mount colon mucosa stained with high-iron diamine-alcian blue (HID-AB).
Enzyme Histochemistry with Glycol Methacrylate-embedded Tissue
Tissues embedded in glycol methacrylate (JB-4 embedding kit, Polysciences, Warrington, PA; molds and block holders, Energy Beam Sciences, Agawam, MA) can be stained with water-soluble reagents and can be cut at 2 μm (Beckstead et al. 1981). These relatively thin sections, compared to the usual 4–6-μm paraffin sections and 10-μm frozen sections, provide excellent preservation of morphology and a larger number of serial sections of very small lesions. For preservation of enzymes, all of the fixation and embedding steps are carried out at 4C, and acetone dehydration is preferred to dehydration with ethyl alcohol (Beckstead et al. 1981; Pretlow et al. 1987, 1990; Barrow et al. 1990). Multiple enzyme histochemical techniques have identified putative premalignant lesions in glycol methacrylate-embedded specimens of liver (Pretlow et al. 1987). Similar approaches have been useful in the study of human colon tissue (Pretlow et al. 1991) and the colons of carcinogen-treated animals (Barrow et al. 1990; Pretlow et al. 1990).
For demonstration of hexosaminidase activity and immunohistochemical demonstration of the incorporation of 5-bromo-2′-deoxyuridine (BUdR) (Sigma; St Louis, MO) in serial sections, the initial fixation of colon tissue was reduced to 15 min in 2% paraformaldehyde at 4C, followed by a 1-hr wash in 3% sucrose in 0.1 M phosphate buffer (pH 7.4), and fixation in 100% acetone for 2 hr under vacuum (Pretlow et al. 1994b). More recently, the glycol methacrylate-embedding procedures were adapted for use with sections of whole human prostates for demonstration of altered enzyme activity with different pathological conditions (Monger et al. 1994).
Immunohistochemistry with Paraffin-embedded Tissue
Many antigens can now be demonstrated in formalin-fixed, paraffin-embedded tissues with antigen retrieval methods (Gown et al. 1993; Shi et al. 1991,1997). Mouse monoclonal antibodies to demonstrate the following antigens were obtained from the following suppliers: carcinoembryonic antigen (clone COL-1; Zymed, San Francisco, CA), nm23 (NCL-nm23; Vector Laboratories, Burlingame, CA), and BUdR (Chemicon; Temecula, CA). Biotinylated horse anti-mouse IgG was obtained from Vector; streptavidin-biotinylated horseradish peroxidase complex from Amersham (Arlington Heights, IL); Superfrost-Plus-treated slides from Fisher Scientific.
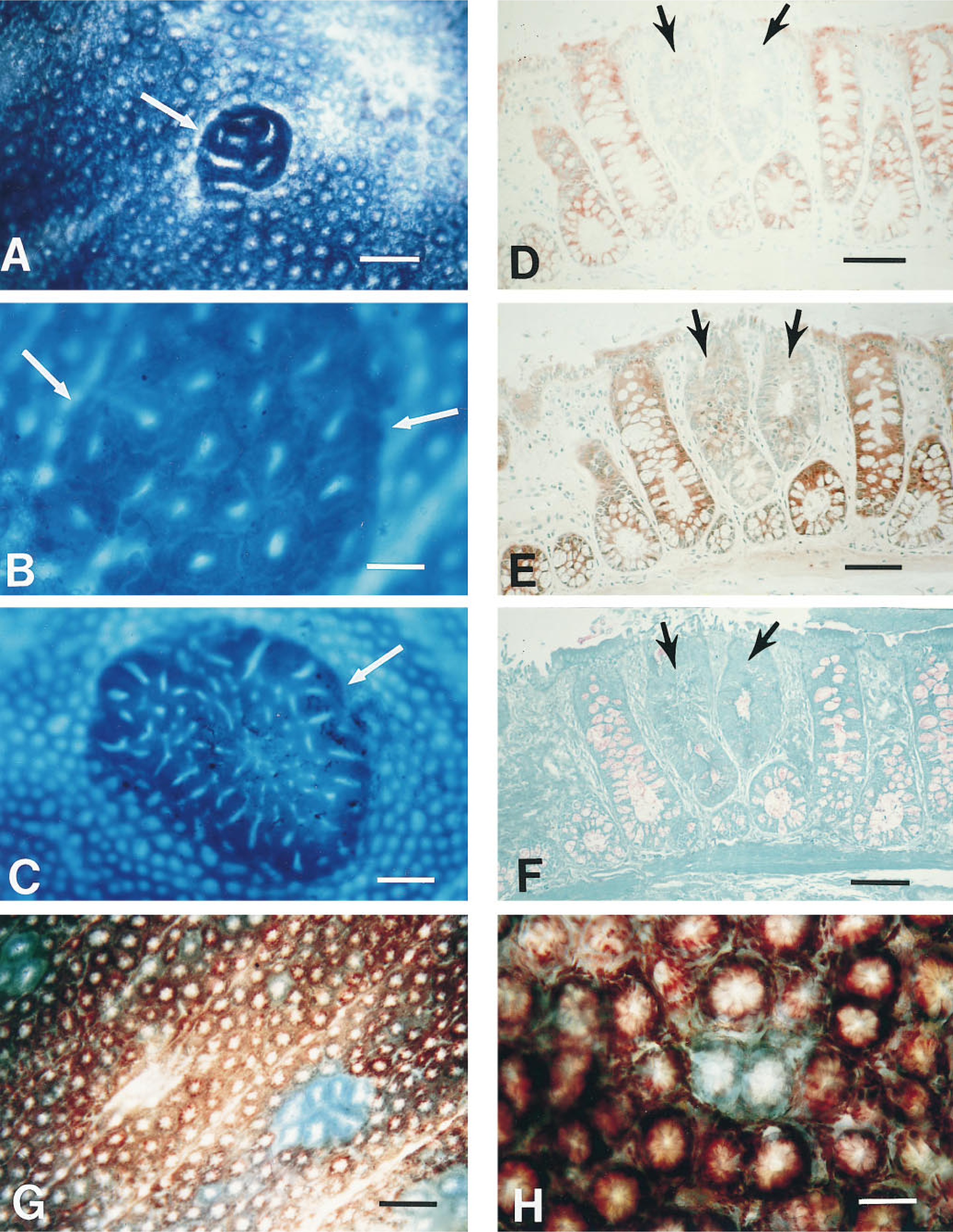
Aberrant crypt foci (arrows) in methylene blue-stained whole-mount colon from a carcinogen-treated rat (
Results and Discussion
Enzyme-altered Foci in Liver Carcinogenesis
From the earliest studies of the induction of tumors in experimental animals, it was known that there is a long latency period between the first application of carcinogen and the final appearance of tumor(s) (reviewed in Pitot 1981). By the mid 1960s it was recognized that histological sections of grossly normal appearing liver from an animal treated with a carcinogen just a few weeks earlier contained discrete areas or “islands” of aberrant enzyme activity that are generally referred to as enzyme-altered foci (reviewed in Pitot 1981; Peraino et al. 1983). Serial sections of liver demonstrated that many foci displayed multiple phenotypic alterations, such as an increase in γ-glutamyl transpeptidase activity and a decrease in bile canalicular ATPase activity (Pitot 1981; Peraino et al. 1983; Pretlow et al. 1987). Some of these phenotypic changes, such as increased glucose-6-phosphate dehydrogenase, the ratelimiting step in the pentose phosphate pathway (Weber 1977), reflect alterations that directly affect the increased rates of growth that occur during progression of the neoplastic process. Other alterations are not as well understood. These enzyme-altered foci in liver have been demonstrated to be monoclonal (Scherer and Hoffmann 1971; Rabes 1983) and are believed to be the earliest identifiable progeny of initiated cells. Hundreds of studies over the past 30 years have supported the hypothesis that these focal populations of enzyme-altered cells are putative premalignant lesions, i.e., at least some of these foci have the potential to progress to hepatocellular carcinomas (Peraino et al. 1983). Therefore, enzyme histochemistry has provided a tool with which to identify and follow the biochemical and molecular events that take place between initiation or the first application of carcinogen and the appearance of tumors in liver.
Enzyme-altered Foci in Colon Carcinogenesis
Knowing that enzyme-altered foci in liver had provided a tool that led to an increased understanding of liver carcinogenesis, we asked whether similar enzyme changes preceded morphological changes in the colon. For these studies we used serial sections of glycol methacrylate-embedded, grossly normal colons from F344 rats that had been treated with the colon carcinogen dimethylhydrazine (Barrow et al. 1988,1990). Multiple lesions with altered enzyme activities were identified in the distal colon and rectum of these carcinogen-treated rats (Barrow et al. 1990). Decreased expression of hexosaminidase activity (Figure 1D) identified the largest proportion (71–76%) of these lesions, which varied morphologically from normal to overtly dysplastic and in size from a single crypt to five or more crypts (Barrow et al. 1990). Like liver foci, these colon enzyme-altered foci frequently displayed multiple phenotypic abnormalities, including decreased α-naphthyl butyrate esterase activity (Figure 1E) and a reduction in goblet cells (Figure 1F). A decrease in hexosaminidase activity was also seen in colon tumors (Pretlow et al. 1992a; Pretlow and Pretlow 1997) that developed in these same animals. Hexosaminidase provides a marker with which to follow initiated colon epithelial cells throughout the carcinogenic process.
Aberrant Crypt Foci in Animals
While we were working on enzyme-altered foci in colons, Bird (1987) published the first description of ACF in whole-mounts of colons from carcinogen-treated rodents that had been stained with methylene blue and viewed at low power with a microscope. ACF are composed of one or more crypts that appear as a single focus and differ from other crypts in that they are larger, have a thickened layer of epithelial cells that stain darker, have an increased pericryptal area between them and the normal crypts, frequently have slit-shaped lumina, and are microscopically elevated above the adjacent normal crypts (Figures 1A−1C). The first question we asked is, are ACF enzyme-altered? To address this question, we marked ACF, identified in methylene blue-stained colons from azoxymethanetreated F344 rats, with permanent ink and embedded the tissue in glycol methacrylate (Pretlow et al. 1990). All of the first 30 ACF identified in this way had decreased hexosaminidase activity (Pretlow et al. 1990). Subsequent studies with hundreds of ACF in F344 rats have found greater than 99% of ACF with decreased hexosaminidase activity. Many of the ACF display additional phenotypic alterations, such as decreased α-naphthyl butyrate esterase activity or increased periodic acid-Schiff-reactive material (Pretlow et al. 1992a; Pretlow and Pretlow 1997), but the expression of these phenotypes is not altered uniformly in all ACF.
Having established that ACF are enzyme-altered, we wondered if all enzyme-altered foci in colons are ACF or if some enzyme-altered foci are morphologically normal. Our earlier studies with enzyme-altered foci in glycol methacrylate sections had suggested that some enzyme-altered foci were not larger than their adjacent normal crypts, but this was difficult to confirm because of the artifacts inherent in embedded tissue. By incubating fixed, intact, whole-mount colons from carcinogen-treated rats in the same substrate as was used for the histochemical demonstration of hexosaminidase activity in slides, we (Pretlow et al. 1993b) were able to demonstrate two populations of lesions with decreased hexosaminidase activity: one with aberrant morphology that resembles ACF (Figure 1G) and the other with normal morphology (Figure 1H). Although both lesions appear to be induced by carcinogen, the relationship between them is not known.
Multiple studies of ACF in animals (reviewed in Pretlow et al. 1992a; Pretlow 1994; Bird 1995; Pretlow and Pretlow 1997) support the hypothesis that ACF are putative premalignant lesions, some of which are capable of progressing to cancer. ACF are specifically induced by colon carcinogens (McLellan and Bird 1988), the number of crypts per focus increases with time after treatment with carcinogen (McLellan et al. 1991a), and the proliferative activity in ACF is increased (McLellan et al. 1991b; Pretlow 1994) and resembles that seen in colon tumors (Pretlow et al. 1994b). The finding of invasive cancer in a lesion identified in a methylene blue-stained preparation of colon as an ACF (Pretlow 1994; Pretlow et al. 1992a; Pretlow and Pretlow 1997) is further evidence for the role of ACF in colon tumorigenesis. Because ACF are observed as early as 2 weeks after a single dose of carcinogen and the entire colon can be scanned for these lesions in 1–2 hr, ACF have been used widely as intermediate biomarkers to assess the ability of agents to protect against or promote colon cancer (Pretlow et al. 1992b; Magnuson et al. 1993; Pereira et al. 1994; Wargovich et al. 1995,1996); reviewed in Bird (1995), Pretlow (1994), Pretlow et al. (1992a). Molecular analysis of these microscopic lesions from animals (Stopera et al. 1992; Shivapurkar et al. 1994; Singh et al. 1994; Zaidi et al. 1995) has allowed the molecular characterization of some of the early events of colon carcinogenesis.
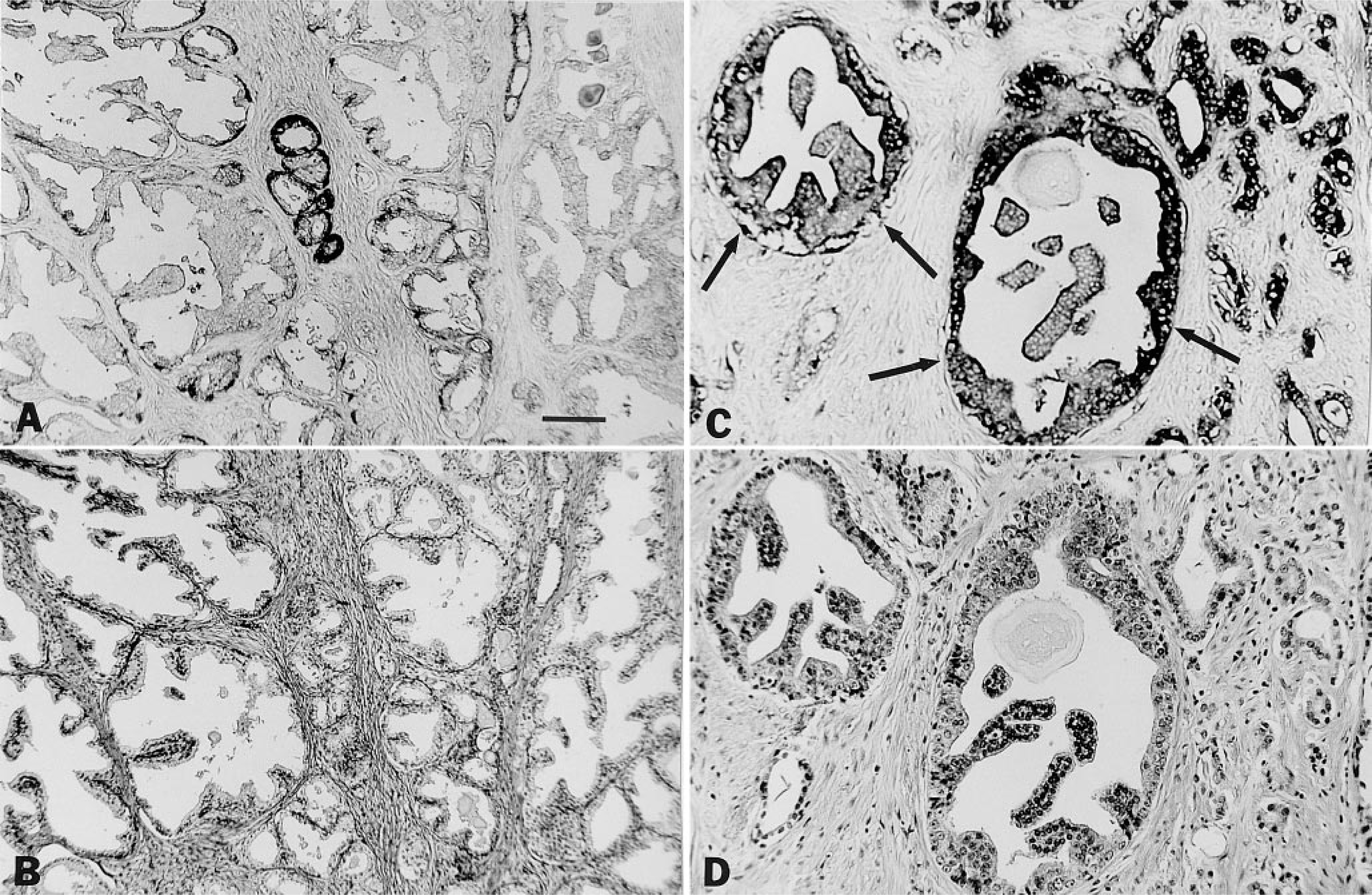
Sections of human prostate. Immunohistochemical demonstration of increased expression of nm23 antigen in the epithelium of some benign prostate glands (
Aberrant Crypt Foci in Humans
Our laboratory was the first to report ACF in whole-mount preparations of grossly normal human colon mucosa (O'Riordan et al. 1990; Pretlow et al. 1991). The ACF in humans (Figures 1E and 1F) clearly resemble those seen in animals in morphology and histological appearance (Pretlow et al. 1991, 1994c; Roncucci et al. 1991a, b; Pretlow 1994; Konstantakos et al. 1996; Siu et al. 1997). Although ACF from humans are enzyme-altered (O'Riordan et al. 1990; Pretlow et al. 1991), the enzyme alterations studied to date are not as marked or consistent as those observed in F344 rats. The finding that 93% of human ACF exhibit increased expression of carcinoembryonic antigen (Pretlow et al. 1994c) provides an important corroborative marker for ACF in histological sections and possible insights regarding the role of this molecule in colon tumorigenesis (Augenlicht 1994). Siu et al. (1997) identified ACF in whole-mount human colons, embedded ACF in paraffin, and used the increased expression of carcinoembryonic antigen to identify these human ACF in histological sections for their evaluation of dysplasia. These ACF were sectioned longitudinally to permit visualization of the entire lengths of crypts. It was found that 54% of 50 ACF from 28 patients displayed dysplasia when serial sections of ACF were evaluated (Siu et al. 1997). This is a much higher proportion of human ACF with dysplasia than reported previously in patients with sporadic colon cancer for which only single sections were evaluated (Jen et al. 1994; Yamashita et al. 1995). Molecular analyses of human ACF have identified multiple genetic alterations in these microscopic lesions, including frequent mutations of K-ras (Pretlow et al. 1993a; Jen et al. 1994; Smith et al. 1994; Yamashita et al. 1995), less frequent mutations of the APC gene (Jen et al. 1994; Smith et al. 1994), and genomic instability (Augenlicht et al. 1996; Heinen et al. 1996). It is hoped that continued studies of human ACF will provide further insights into the earliest genetic alterations that initiate and promote the development of this very common human cancer.
Enzyme-altered or Antigen-altered Foci in Human Prostate
The studies of putative premalignant lesions in human prostate in our laboratory are much more preliminary. Monger et al. (1994), using sections of whole prostate embedded in glycol methacrylate, was the first to demonstrate focal areas of benign prostatic epithelium with a marked decrease of acid phosphatase and 5′nucleotidase activities. It is interesting that prostate intraepithelial neoplasia (PIN), a putative precursor of invasive prostate cancer, similarly expresses decreased acid phosphatase and 5′-nucleotidase activities compared with both prostate cancer and benign prostate (Monger et al. 1994). Aberrant expression of CD44, transforming growth factor-α, and nm23 (Figures 2A and 2B) also has been observed in a small number of benign prostate epithelial glands (Pretlow et al. 1994a, 1995) after staining of paraffin-embedded prostate tissues with monoclonal antibodies specific for these antigens. The increased expression of nm23 in the epithelium of some benign prostate glands (Figures 2A and 2B) parallels the increased expression of this antigen in PIN (Figures 2C and 2D) (Pretlow et al. 1995). Perhaps in the future these putative premalignant lesions will be used as biomarkers to evaluate chemopreventive agents for human prostate cancer.
In conclusion, histochemistry, including enzyme histochemistry of glycol methacrylate-embedded tissues, enzyme histochemistry of segments of intact tissue, and immunohistochemistry, has provided invaluable tools to identify and study putative premalignant lesions from their earliest identification through their progression to cancer.
Footnotes
Acknowledgements
Supported in part by NIH grants CA66725, CA48032, CA54031, CA57179, DK45770, DK51347, and CA43703.
We thank the many students, research associates, and research assistants who contributed to this work, including Betty Barrow, Christopher Cheyer, Leon Hudson, Lawrence Monger, M. Nagabhushan, Mary Ann O'Riordan, and I-Mei Siu.
