Abstract
From a practical point of view, one of the most difficult issues in the standardization of IHC for FFPE tissue is the adverse influence of formalin upon antigenicity, as well as the great variation in fixation/processing procedures. Based on previous study, an additional study using four markers demonstrated the potential for obtaining equivalent IHC staining among FFPE tissue sections with periods of formalin fixation ranging from 6 hr to 30 days. On this basis, the following hypothesis is proposed. “The use of optimized AR protocols permits retrieval of specific proteins (antigens) from FFPE tissues to a defined and reproducible degree (expressed as R%), with reference to the amount of protein present in the original fresh/unfixed tissue”. This hypothesis may also be presented mathematically: the protein amount in a fresh cell/tissue, expressed as Pf, produces an IHC signal in fresh tissue of ∫ (Pf). When the identical IHC staining plus AR treatment is applied to a FFPE tissue section, the IHC signal may be represented as ∫ (Pffpe). The degree of retrieval after AR (R%) is calculated as follows: R% = ∫ (Pffpe)/ ∫ (Pf) × 100%. The amount of protein in the FFPE tissue may then be derived as follows: Pffpe = Pf × R%. In a situation where optimized AR is 100% effective, the IHC signal would then be of equal strength in fresh tissue and FFPE tissue, and Pffpe = Pf. Further studies are designed to test the limitations of the proposed hypothesis.
Keywords
S
A ‘total test’ approach was advocated in 1992 (Taylor 1992). In essence, the ‘total test’ embraces all procedures performed to accomplish an IHC stain, from sample collection to writing a final report. From a practical point of view, one of the most difficult issues in the standardization of IHC on FFPE tissues is the adverse influence of formalin, a major uncontrollable intrinsic factor. One proposal seeking to address this difficult issue was the Quicgel method, using a breast cancer cell line embedded in agar gel, processed, and incorporated into the FFPE tissue block side-by-side with the tissue specimen under exactly the same conditions to establish an artificial internal control. It was claimed that the Quicgel method allowed accurate calculation of the amount of protein (estrogen receptor) in the tested sample tissue based on biochemical quantitative analysis (Riera et al. 1999). The Quicgel method, however, has proven not to be practical for routine use due to logistical issues; in addition it is not applicable for retrospective studies on archival tissue. Leong (2004) postulated that internal controls were required for IHC to optimize variable influences due to intrinsic factors. The desired internal control should be some tissue component that exists in the same tissue section as the target antigen when tested by IHC. To date, it has proven difficult to identify a quantifiable internal control in FFPE tissue sections for the following reasons: (1) there have been no systematic attempts to identify and quantify non-lesional tissue components that may also be found within the test sample; (2) although ubiquitous candidate proteins do exist, distribution of proteins (antigens) in different tissues may be variable (Pusztaszeri et al. 2006), and their response to processing may be variable; (3) IHC staining methods used must be strictly controlled to yield a reproducible intensity of staining; and (4) computer-assisted image analysis will be necessary to measure and compare intensity of the reference standard vs the test antigen.
Based on numerous reports that antigen retrieval (AR)-IHC gives excellent results for many of the markers used in diagnostic pathology (Shi et al. 1997), the possibility of improving the standardization of IHC through the use of AR technique has been suggested (Taylor 1994,2006). Furthermore, the use of a ‘test battery’ approach has been advocated to find an optimal protocol of AR-IHC (Shi et al. 1996), based on monitoring the heating condition (temperature and duration of heating) and the pH value of the AR solution, which are the two major factors that influence the effectiveness of AR-IHC. In our experience, a consistent ‘maximal retrieval’ level, showing the strongest intensity of AR-IHC, may be obtained for many antibody/antigen pairings by using this ‘test battery’ approach (Shi et al. 1996).
We conducted an experiment using AR-IHC on FFPE tissues fixed in formalin for different periods ranging from 4 hr to 30 days to explore the possibility of obtaining equivalent IHC staining following the ‘maximal retrieval’ for selected antigen/antibody combinations. In one early study, five antibodies were tested with results that support the notion that it is possible to achieve ‘equalized’ maximal immunostaining levels in FFPE tissue sections fixed in formalin for variable times, as long as 1 month (Shi et al. 1998).
On this basis, the following hypothesis is proposed.
Hypothesis
“The use of an optimized AR protocol permits retrieval of specific proteins (antigens) from FFPE tissues to a defined and reproducible degree (the retrieved rate of AR, expressed as R%), with reference to the amount of protein present in the original fresh/unfixed tissue.” This hypothesis may also be presented mathematically: the protein amount in a fresh cell/tissue, expressed as Pf, produces an IHC signal in fresh tissue of ∫ (Pf). When the identical IHC staining plus AR treatment is applied to a FFPE tissue section, the IHC signal is ∫ (Pffpe). The degree of retrieval after AR (R%) is calculated as follows: R% = ∫ (Pffpe)/ ∫ (Pf) × 100%. The amount of protein in the FFPE tissue may then be derived as follows: Pffpe = Pf × R%. In a situation where optimized AR is 100% effective, the IHC signal would then be of equal strength in fresh tissue and FFPE tissue, and Pffpe = Pf.
Based on this hypothesis, it is possible to measure the adverse influence of formalin fixation and tissue-embedding processing for certain ubiquitous antigens. Having derived these data experimentally, such antigens may then serve as quantifiable internal reference standards for other test antigens where data are not available with respect to loss or degree of retrieval when compared with fresh frozen tissue.
Preliminary Test
A preliminary test of this hypothesis was performed using routinely processed FFPE tissue/cell sections of human breast cancer obtained from the Norris Cancer Hospital and Research Institute, Los Angeles, CA and cultured cell pellets of human breast cancer cell line MCF-7 with variable periods of fixation in 10% neutral-buffered formalin (NBF) ranging from 6 hr to 30 days. This study of human archival tissue specimens was exempted under 45 CFR 46.101 (b) and was approved by the Institutional Review Board (IRB #009071) at the University of Southern California. All cell/tissue sections were routinely processed for AR-IHC using 0.05% citraconic anhydride (Sigma Chemical Co.; St Louis, MO), pH 7.5, as the AR solution with a plastic pressure cooker heated in a microwave oven (1100 W, 60 Hz; Sharp Carousel, Bangkok, Thailand) as previously reported (Shi et al. 2000; Namimatsu et al. 2005). To more accurately compare the results of IHC, all staining procedures were performed identically in the same side-by-side run. Four monoclonal antibodies for estrogen receptor (ER, 1:100; NeoMarkers, Fremont, CA), MIB-1 (1:500; Dako, Glostrup, Denmark), cytokeratin (AE-1 cocktail, 1:500; Signet Laboratories, Dedham, MA and CAM 5.2, 1:50; Becton Dickinson, San Jose, CA), and Her-2/neu (1:200; BioGenex Laboratories, San Ramon, CA) were used as the primary antibodies. The Vectastain Elite ABC kit (Vector Laboratories; Burlingame, CA) was used for IHC staining following the manufacturer's instruction. 3,3′-DAB was used as chromogen, and hematoxylin was used as counterstain. Positive and negative controls were routinely applied. Evaluation of IHC staining results was conducted by two independent observers by light microscopy. Intensity of positive immunostaining was graded as strong (+++), moderate (++), weak (+) or negative (-).
Results
All four markers showed positive IHC staining results among FFPE tissue sections fixed for various periods ranging from 6 hr to 30 days, although the immuno-staining intensity of 30-day fixed FFPE tissue sections for Her-2/neu and ER was slightly weaker than that obtained in FFPE tissue sections fixed for shorter periods. Both intensity and percentage of positively stained cells in all FFPE tissue sections achieved a strong (+++) level (Figure 1). This preliminary test supports the potential feasibility of standardization of IHC staining results in tissues undergoing variable periods of formalin fixation, as indicated by our previous study (Shi et al. 1998).
Further Experiments Designed for Testing Hypothesis
To examine this hypothesis, further studies are necessary, beginning with the use of a well-defined cell/tissue model system in which the amount of selected antigen can be measured accurately on a cell-to-cell basis in fresh and FFPE specimens.
The cell/tissue model system is established based on quantitatively comparable cell lines that are processed in fresh frozen and FFPE cell/tissue blocks under variable conditions, including the period of formalin fixation, delayed times of fixation, and varying storage conditions, as well as other technical issues such as thickness of each tissue section. The experiment is designed to simulate fixation and processing schedules in general use in histopathology laboratories. By using this model system, serial experiments may be carried out to examine the hypothesis based upon a multi-dimensional study such as depicted in the ‘vector-gram’ (Figure 2).
IHC must be performed in a side-by-side fashion for accurate comparison of immunostaining intensity and evaluated by appropriate standard positive controls. It is also necessary to evaluate the intensity of IHC based on both manual and computer-assisted image analysis. Use of a tissue microarray method is proposed to simplify these tests using either a series of algorithms called AQUA technology for quantitative assessment (Cregger et al. 2006) or comparative spectral imaging in double-or triple-stained slides. A selected antibody panel including cytoplasmic, nuclear, and surface markers will be tested to confirm results. Accurate biochemical quantification of proteins in cell/tissue model will be required for validation of IHC findings.
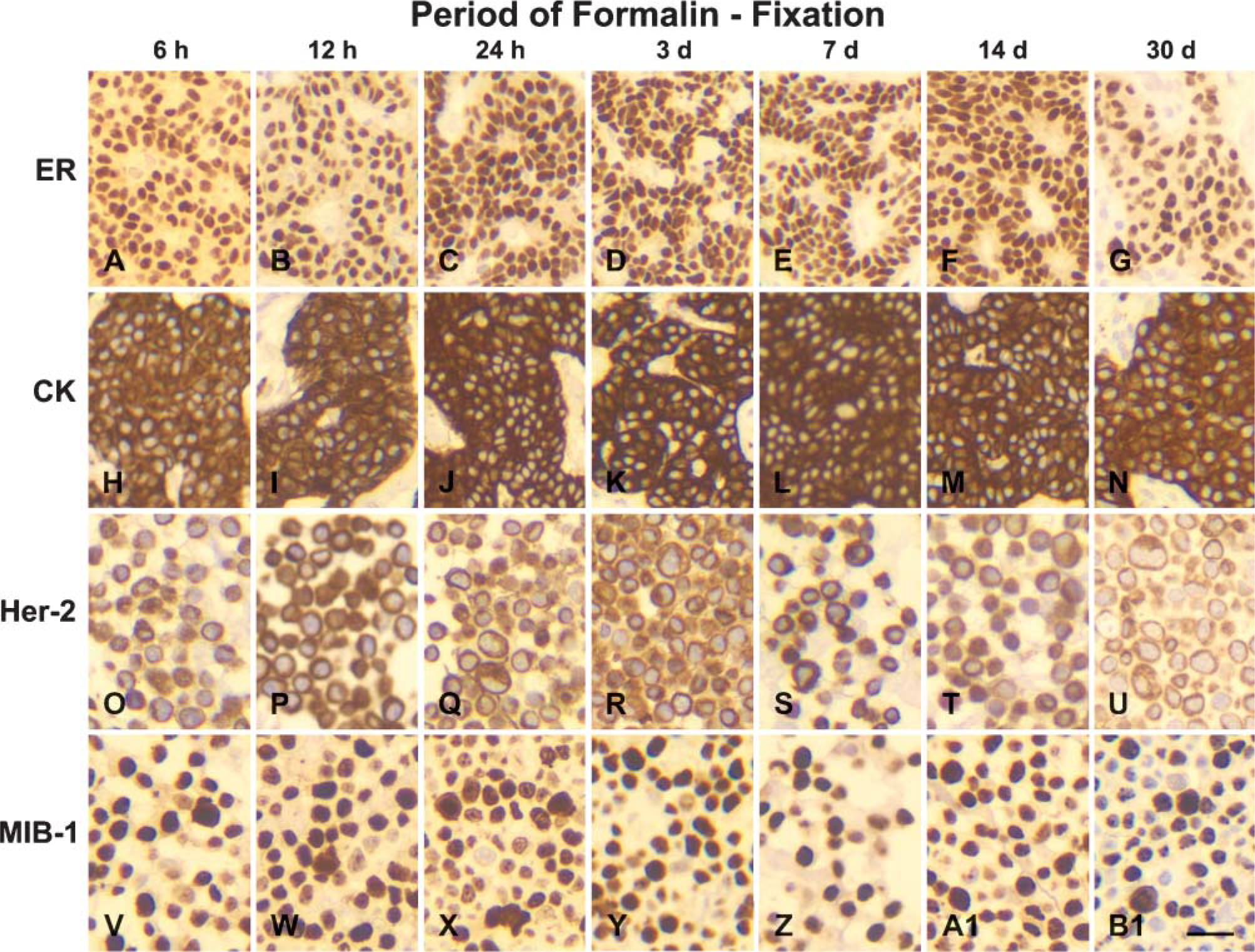
Comparison of immunohistochemical staining results among variable periods, 6 hr (h) to 30 days (d), of formalin-fixed, paraffin-embedded human breast cancer tissue
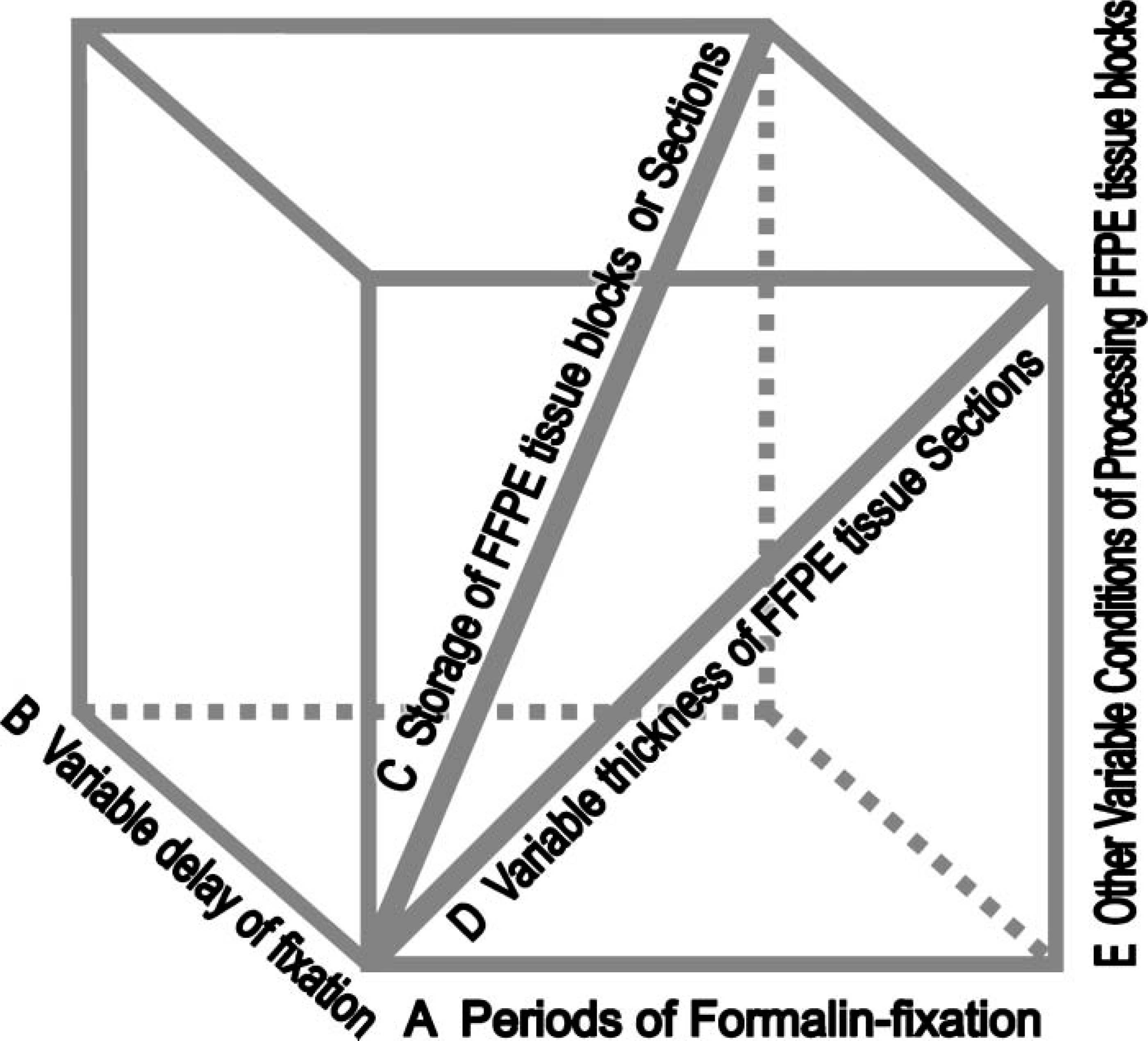
Diagram depicts the further-designed studies to test our hypothesis with respect to standardization of immunohistochemistry based on the antigen-retrieval technique exemplified in a multiple direction to draw a conclusion.
Finally, to test the accuracy of protein quantification in FFPE tissue sections by this approach, it will be essential to perform a parallel study on selected human tissue sections with known amounts of certain proteins (e.g., ER) in a double-blind fashion. The initial research design using the cell/tissue model is presented to examine the limitations of this hypothesis based on correlated accurate quantitative biochemical measurements and IHC staining results.
Publications Relevant to the Hypothesis
In addition to the preliminary studies described here, there is a body of literature that supports the notion that effective optimal AR may be a useful approach to ‘neutralizing’ the adverse and variable effects of fixation. Boenisch (2005) documented a recent IHC study using human tonsil tissue fixed in 10% NBF for 12 hr and 1, 2, 4, and 8 days to determine whether AR could be applied to ‘equalize’ variable immunostaining results resulting from inconsistent formalin fixation. Among 30 antibodies tested in his experiment, 26 showed consistent optimal staining by using one single AR protocol (0.01 M citrate buffer, pH 6.0, with heating at 97C for 20 to 60 min). Boenisch concluded that “Application of a given method for heat retrieval can compensate for variable formalin-induced damages resulting from inconsistencies in the length of formalin fixation and thus equally restore the immunoreactivities on a wider scale of antigens.”
Other articles demonstrate consistent restoration of immunoreactivity (AR rate = 100%) for many antigens (proteins), exemplified by ER, progesterone receptor (PR), HER-2/neu, Ki-67 (MIB-1), etc. for FFPE tissues, indicating high concordance between IHC and positive biochemical results (MacGrogan et al. 1996; Pertschuk and Axiotis 2000). More than a dozen articles have demonstrated comparable IHC staining results between frozen and FFPE tissue sections following AR. For example, Von Boguslawsky (1994) performed IHC detection of PR for 25 paired frozen and FFPE tissue sections of breast cancer to compare the percentage of positively stained nuclei between frozen and FFPE tissue sections and demonstrated that with the AR treatment, 84% (21/25) of FFPE tissue sections showed identical positive nuclear staining to that obtained in frozen tissue sections. Among the remaining four cases (4/25), compared to the frozen tissue sections only one case showed a lower percentage of positive cells in the FFPE tissue section. Higher percentages of positive staining were found in the other three cases for FFPE tissue sections.
Reliable ‘inter-laboratory’ IHC staining for Her-2/neu, ER, etc. also can be achieved based on optimal AR-IHC protocols and stringent quality control using standard reference materials (Jacobs et al. 2000; Rhodes et al. 2000,2002).
In conclusion, broad-based experimental data from multiple investigators support the notion that carefully performed AR-IHC has the potential to greatly improve the reliability of IHC staining. In addition, using this approach it may be possible to identify certain ubiquitous antigens that show a consistent degree of restoration of antigenicity following optimized AR. Such antigens, when accurately quantified by controlled experiments, may serve as the basis for developing a panel of ‘quantifiable internal reference standards’ for IHC. Further studies are required, but the initial findings are encouraging for a positive and useful outcome.
Footnotes
Acknowledgements
This study was supported by National Institutes of Health, Grant 1 R33 CA-103455-01.
We thank Henry Lin, PhD student at the Department of Pathology, University of Southern California, for his kind help in preparing figures.
