Abstract
Serine protease inhibitors (serpins) are a superfamily of proteins involved in many important biological processes, including inflammation. Serpins dysfunction-related diseases are mainly treated by augmentation therapy using serpins purified from human plasma. Pnserpin from hyperthermophilic archaeon
Introduction
Serine protease inhibitors (serpins) are a superfamily of proteins with inhibitory activity toward serine or cysteine proteases, widely distributed in eukaryotes, bacteria, and archea.1,2 Serpins, such as antithrombin 3 and α1-antitrypsin,4,5 are involved in many important biological processes including inflammation, immunity, cancer, coagulation, fibrinolysis, complement activation, angiogenesis, apoptosis, neoplasia, and viral pathogenesis.6–11 The primary function of most members of the serpin family is to neutralize overexpressed serine proteinase activity. But there is some evidence that serpins can exert anti-inflammatory and tissue-protective effects independent of their protease inhibition activity, such as inhibition of pro-inflammatory cytokines production and activity.12,13 Alterations of a serpin which affect its structure and/or secretion and thus reduce its functional levels may result in pathology, especially for lung, liver, and pancreatic destructive diseases.14–16 At present, serpins dysfunction-related diseases are mainly treated by augmentation therapy. Currently, serpins for augmentation therapy is mainly the serpins protein purified from human plasma, such as α1-antitrypsin.17–19 Due to the huge demand, providing adequate supply of serpins from plasma purification will be challenging.
The serpin genes have also been found in thermophiles in recent years. Some of these thermophilic serpins have been biochemically and structurally characterized, for instance, thermopin from the moderate thermophilic bacterium
In this study, the anti-inflammatory activity of Pnserpin was tested using xylene-induced acute inflammatory model of mouse ear swelling and lipopolysaccharide (LPS)-induced murine RAW 264.7 macrophages cellular model. The inhibition of mouse ear swelling and the production of pro-inflammatory cytokines in mouse serum or in macrophages cell were used to evaluate the anti-inflammatory effect of Pnserpin.
Materials and methods
Materials
The RAW264.7 cell line was purchased from the Type Culture Collection of the Chinese Academy of Sciences (Shanghai, China). Dulbecco’s modified Eagle’s medium (DMEM) was obtained from Gibco (Carlsbad, USA). LPS were purchased from Sigma (Missouri, USA). A Cell Counting Kit-8 (CCK-8) and enzyme-linked immunosorbent assay (ELISA) kit were obtained from R&D (Minneapolis, USA).
Animals
Kunming mice of clean grade (18–22 g) were purchased from Animal Experiment Center, Jilin University (number of animal license SYXK 2013-0005). Before the start of all experiments, animals were acclimatized to their living environment for at least 3 days and were used only once in each assay. All studies were approved by the Animal Welfare and Ethics Group of the Laboratory Animal Science Department, Jilin University (Changchun, China).
Xylene-induced ear swelling
A total of 60 mice were randomly divided into six groups (A–F). Groups C–E were tail intravenous injected with Pnserpin at one of three different doses (1.2, 2.4, or 4.8 mg/kg) for 6 days. The mice in A and B groups were used as negative control and xylene-induced model controls, respectively, and were given the same volume of physiological saline. The mice in F group were used as positive controls and were given the same volume of dexamethasone (1.25 mg/kg). One hour after the sixth day of administration, animals received a 50-μL smearing of xylene on both the anterior and posterior surfaces of the right ear lobe. Two hours later, blood was taken from the eyeballs of mice and the mice were euthanized by cervical dislocation. The serum was collected by centrifugation at 1510 × g, 10 min. 9-mm sections were taken from both ears with a cork borer and tissue subsequently weighed. The thickness of the mouse ear was measured with a Vernier caliper. The degree of ear swelling was calculated according to the following formula

where A and B denote ear swelling of the negative group and ear swelling of the drug groups, respectively.
Histological evaluation
For histological analysis, ear samples were fixed in 4% paraformaldehyde, embedded in paraffin, and sectioned at a thickness of 5 μm. Sliced sections were stained with hematoxylin and eosin (H&E).
Cell culture and treatment
RAW264.7 mouse macrophage cells were cultured in DMEM medium supplemented with 10% fetal bovine serum (FBS), glutamine, and antibiotics at 37°C under 5% CO2. Cells at 80%–90% confluency were centrifuged at 120 × g at 4°C for 10 min and cell concentration was adjusted to (2 × 106) cells/mL, where the cell viability was always more than 90%. A total of 50 μL of cell suspension was seeded into a tissue culture grade 96-well plate (4 × 105 cells/well) and incubated for 2 h at 37°C, 5% CO2 for cell attachment. Then, cells were stimulated by 1 μg/mL of LPS with or without the presence of Pnserpin or dexamethasone tested at the final volume of 100 μL/well. Cells were further incubated at 37°C, 5% CO2 for 24 h for use.
Cell viability
The viability of cells was measured using CCK-8 assay. Briefly, 4 × 104 cells/well RAW264.7 cells were plated in 96-well plates, and incubated for 2 h, then treated with different concentrations (0.05, 0.1, and 0.2 mg/mL) of Pnserpin for 24 h. Equal volume of medium was used as blank control. After treatment, cells were incubated with 10 μL of CCK-8 for another 4 h in dark. The absorbance at 450 nm was recorded using a PR 4100 microplate reader (Bio-Rad Inc., Hercules, USA).
Analysis of cytokines using ELISA assay
TNF-α, IL-6, and IL-1β concentrations in the cell culture supernatant or mouse serum were detected using the ELISA kit according to manufacturer’s instruction, and the results were expressed in pg/mL of protein. All of the analyses were performed in triplicate.
Measurement of NO production
NO production was analyzed using Griess reagent as described previously. 27 Briefly, 50 μL of culture supernatant were transferred to wells of a 96-well plate, and 50 μL of Griess reagent (containing 0.5% sulfanilic acid, 0.002% N-1-naphtyl-ethylenediamine dihydrochloride, and 14% glacial acetic acid) was then added. The absorbance at 550 nm wavelength was measured using a BioTek microplate reader. Sample nitrite concentrations were estimated from a sodium nitrite standard calibration curve.
Statistical analysis
Statistical analyses were carried out with SPSS 13.0 software (SPSS Inc., Chicago, USA). Data were analyzed by Student’s two-tailed t-test for comparison between two groups, and data were presented as mean ± standard deviation (SD).
Results
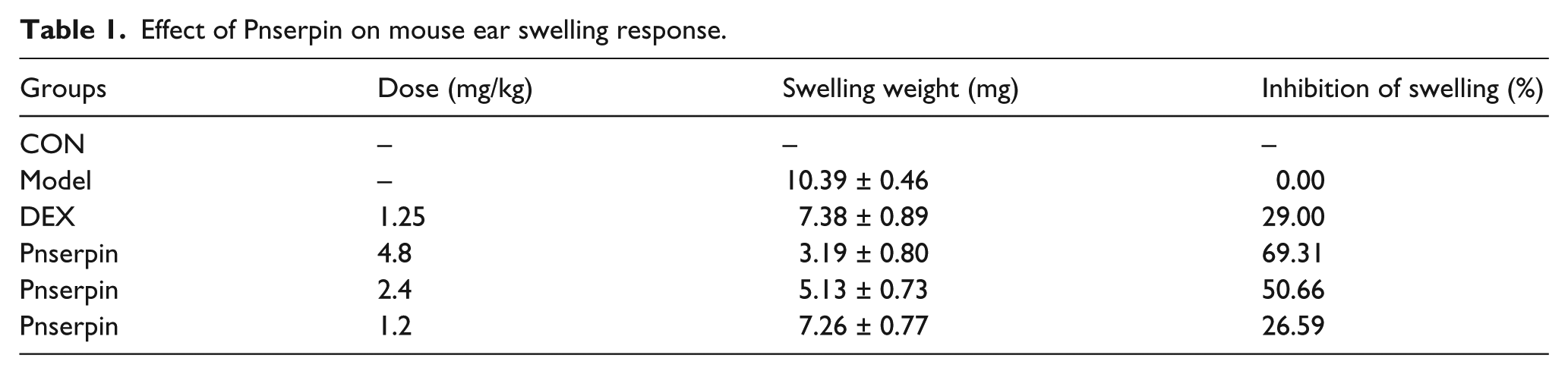
Inhibition effect of Pnserpin on xylene-induced mouse ear swelling
In our previous work, gene of Pnserpin from thermopile

Effect of Pnserpin on mouse ear swelling response. (a) mouse ear swelling weight and (b) mouse ear thickness.
Effect of Pnserpin on mouse ear swelling response.
Effect of Pnserpin on xylene-induced mouse ear histomorphology
The histological structures of the punch ears were observed by H&E staining. The xylene-induced model group showed the maximum thickness compared to the negative control group and the groups pretreated with Pnserpin (4.8, 2.4, or 1.2 mg/kg) and dexamethasone (1.25 mg/kg). There are broadening tissue space, discrete and fractured striated muscle fiber, and capillary congestion, with inflammatory cell infiltration in the ear tissue of xylene-induced model group (Figure 2). In the groups pretreated with dexamethasone (1.25 mg/kg) or Pnserpin (4.8, 2.4, or 1.2 mg/kg), these phenomena were reduced to different degree. Especially the group pretreated with 4.8 mg/kg Pnserpin, the morphology of the mouse ear was close to that of the negative control group (Figure 2(A-d) and (B-j)). These results indicated that Pnserpin have anti-inflammatory activity in xylene-induced mouse ear swelling model.

Histopathological sections showing ear swelling induced by xylene treatment. (A) HE × 100; (B) HE × 200. (a, g) Normal ear; (b, h) xylene-induced model control group; (c, i) treatment with Dexamethasone (1.25 mg/kg); (d, j) treatment with Pnserpin (4.8 mg/kg); (e, k) treatment with Pnserpin (2.4 mg/kg); and (f, l) treatment with Pnserpin (1.2 mg/kg).
Inhibition effect of Pnserpin on serum TNF-α and IL-6
Compared to negative control group (TNF-α: 112 pg/mL, IL-6: 37 pg/mL), the serum production of TNF-α and IL-6 significantly increased (TNF-α: 812 pg/mL, IL-6: 340 pg/mL) after treatment with xylene in the model group. Pretreatment with dexamethasone (1.25 mg/kg) or Pnserpin (4.8, 2.4, or 1.2 mg/kg) could markedly reduce the production of these cytokines induced by xylene. In the group pretreated with 4.8 mg/kg Pnserpin, the production of TNF-α and IL-6 reduced to 223 and 40 pg/mL, respectively, which are close to the negative control group. These data suggested that Pnserpin could inhibit the serum production of TNF-α and IL-6 induced by xylene in a dose-dependent manner, and the anti-inflammatory activity of Pnserpin correlated with the suppression of pro-inflammatory cytokines (Figure 3).
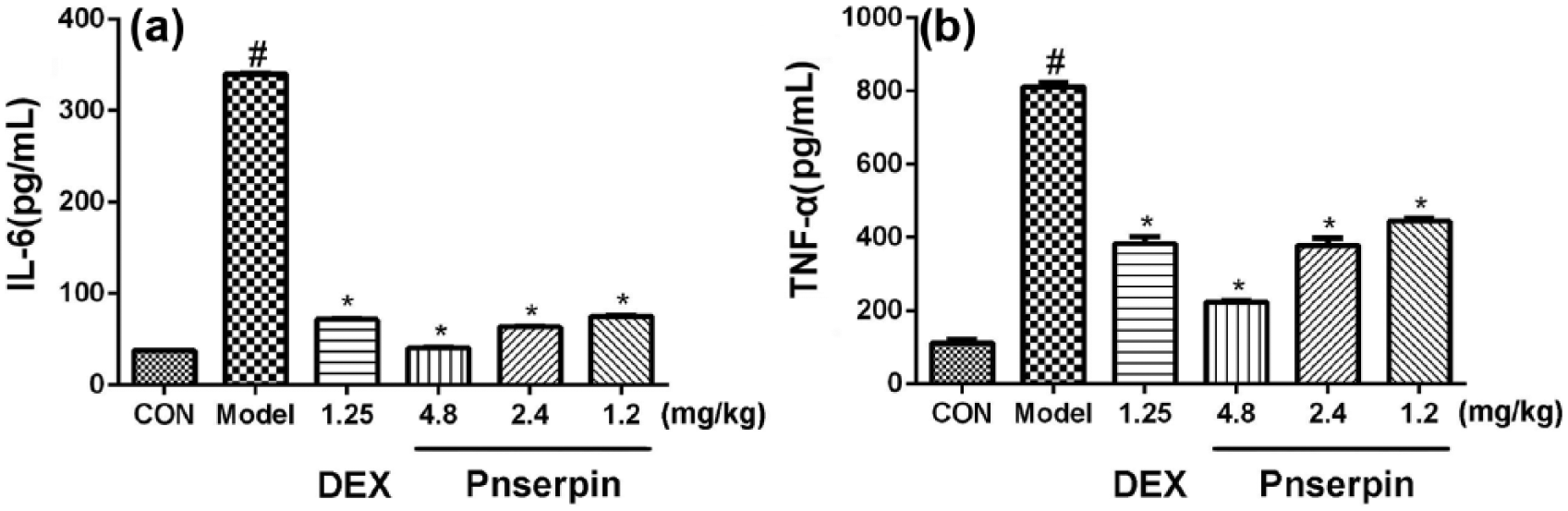
Inhibition effect of Pnserpin on serum TNF-α and IL-6. (a) production of IL-6 and (b) production of TNF-α.
Inhibition effect of Pnserpin on LPS-stimulated RAW264.7
To further confirm the anti-inflammatory activity of Pnserpin at the cellular level, LPS-stimulated RAW264.7 macrophages were used to investigate the inhibition effect of Pnserpin on the expression levels of representative pro-inflammatory cytokines. First, the cell viability of mouse macrophage treated with a series of concentrations of Pnserpin and 1-μg/mL LPS was examined. As shown in Figure 4, Pnserpin at concentrations of 0.05–0.2 mg/mL did not affect cell viability.

Effect of Pnserpin on cell viability of RAW264.7. (a) Treatment with LPS or Pnserpin alone and (b) treatment with both LPS and Pnserpin.
The release of pro-inflammatory cytokines TNF-α, IL-6, IL-1β, and NO from RAW264.7 macrophages induced by LPS were detected. Stimulation of the cells with LPS for 24 h increased the levels of the detected pro-inflammatory cytokines (IL-6: 165 pg/mL; IL-1β: 192 pg/mL; TNF-α: 1652 pg/mL; NO: 54 μM), while pretreatment of RAW264.7 cells with Pnserpin (0.05, 0.1, and 0.2 mg/mL) significantly attenuated the ability of LPS to increase the levels of cytokines in a concentration-dependent manner (Figure 5). A volume of 0.2 mg/mL Pnserpin reduced the production of IL-6, IL-1β, TNF-α, and NO to 100 pg/mL, 96 pg/mL, 846 pg/mL, and 23 μM, respectively. To exclude the possibility that the inhibitory effects of Pnserpin on LPS-induced cytokine release were due to the binding of Pnserpin to LPS, the interaction between Pnserpin and LPS was analyzed via native PAGE. The electrophoresis results showed that there were no difference between the incubated Pnserpin/LPS mixture and Pnserpin alone, indicating that the inhibitory effects of Pnserpin do not involve a direct interaction with LPS. These results indicated that Pnserpin may have anti-inflammatory activity at the cellular level.

Effects of Pnserpin on IL-6, IL-1β, TNF-α, and NO productions in RAW264.7 cells induced by LPS. (a) IL-6 production, (b) IL-1β production, (c) TNF-α production, and (d) NO production.
Discussion
Serpins have been identified in higher multicellular eukaryotes and viruses. Until 2002, Irving et al. reported 12 serpin-like sequences in the genomes of prokaryotic organisms, extending this protein family to all major branches of life. 28 In recent years, the serpin genes have also been found in thermophiles. In our previous work, through sequence alignment with other serpins, including eukaryotic and thermophilic serpins, we found that Pnserpin had characteristic sequence features of the serpins superfamily. 26 Experimental results showed Pnserpin had inhibitory activity against chymotrypsin-, trypsin-, elastase-, and subtilisin-like proteases. Therefore, we speculate that Pnserpin may have a function similar to α1-antitrypsin and other eukaryotic serpins.
To verify our speculation, we investigated the anti-inflammatory effect of Pnserpin using xylene-induced mouse ear swelling model and LPS-stimulated RAW264.7 macrophages cell model, which are well-known and widely used animal model and cell model for inflammation studies.29–32 In mouse ear swelling model, Pnserpin could inhibit ear swelling induced by xylene in a concentration-dependent manner. In the group that pretreated with 4.8 mg/kg Pnserpin, the swelling inhibition rate reached as high as 69% and the morphology of the mouse ear was similar to that of the negative control group. Compared to the model group, pretreatment with Pnserpin can significantly inhibit the production of pro-inflammatory cytokines TNF-α and IL-6 in mouse serum. Pnserpin also suppress the production of TNF-α, IL-6, IL-1β, and NO in LPS-stimulated RAW264.7 macrophages. These results indicated that Pnserpin might have anti-inflammatory effects in vivo and in vitro.
Recent studies indicated that α1-antitrypsin has suppressive activity in inflammatory pathway through both enzyme inhibitory and non-inhibitory mechanisms.33–35 Based on the sequence alignment of Pnserpin and α1-antitrypsin in the RCL (Reactive Center Loop) region, and the serine protease inhibition activities of pnserpin, 26 we speculated that the anti-inflammatory activity of Pnserpin might depend on its serine protease inhibition activity. However, the mechanism of α1-antitrypsin’s non-inhibitory anti-inflammatory activity remains unclear; we cannot exclude that Pnserpin plays an anti-inflammatory activity through non-inhibitory mechanism. In the future, we will further test the anti-inflammatory effect, mechanism, and the safety of Pnserpin in more different animals and cellular models to expand the source of serpins for augmentation therapy.
In conclusion, this study demonstrates that Pnserpin, a serine protease inhibitor from thermophilic archaeon
Footnotes
Acknowledgements
Rui Fei and Huan Zhang contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was supported by the National Natural Science Foundation of China (no. 31400681), the Science and Technology Development Program of Jilin Province (no. 20160520142JH), the Norman Bethune Program of Jilin University (no. 2015424), and the Science and Technology Program of Health and Family Planning Commission of Jilin Province (no. 2015Q016).
