Abstract
Nidogen-1, a key component of basement membranes, is considered to function as a link between laminin and collagen Type IV networks and is expressed by mesenchymal cells during embryonic and fetal development. It is not clear which cells produce nidogen-1 in early developmental stages when no mesenchyme is present. We therefore localized nidogen-1 and its corresponding mRNA at the light and electron microscopic level in Day 7 mouse embryos during the onset of mesoderm formation by in situ hybridization, light microscopic immunostaining, and immunogold histochemistry. Nidogen-1 mRNA was found not only in the cells of the ectoderm-derived mesoderm but also in the cytoplasm of the endoderm and ectoderm, indicating that all three germ layers express it. Nidogen-1 was localized only in fully developed basement membranes of the ectoderm and was not seen in the developing endodermal basement membrane or in membranes disrupted during mesoderm formation. In contrast, laminin-1 and collagen Type IV were present in all basement membrane types at this developmental stage. The results indicate that, in the early embryo, nidogen-1 may be expressed by epithelial and mesenchymal cells, that both cell types contribute to embryonic basement membrane formation, and that nidogen-1 might serve to stabilize basement membranes in vivo.
Keywords
B
Nidogen-1 (150 kD), also referred to as entactin, is a small rod-shaped molecule with three globular domains (G1, G2, G3) connected by a flexible link and a rod (Fox et al. 1991; Mayer and Timpl 1994). It shows high in vitro binding activity for laminin-1 (Fox et al. 1991), and the binding site has been localized to a single laminin-type epidermal growth factor-like module γ1III4 on the laminin γ1-chain (Mayer et al. 1993). Antibodies against this laminin module were shown to inhibit nidogen-1 binding (Mayer et al. 1993) and to interfere with basement membrane formation in organ cultures (Ekblom et al. 1994; Kadoya et al. 1997). Nidogen-1 also binds to collagen Type IV (Aumailley et al. 1989) and is capable of forming ternary complexes with laminin-1 and collagen Type IV (Fox et al. 1991; Brown et al. 1994; Mayer et al. 1995). Nidogen-1 could therefore serve as a crucial link protein stabilizing network formation in basement membranes (Timpl and Brown 1996). It has recently been shown by double labeling that nidogen-1 and laminin-1 are indeed colocalized in adult and fetal kidney basement membranes, indicating that nidogen-1 could connect the laminin-1 and collagen Type IV networks in vivo (Miosge et al. 1999). This has raised the question of whether nidogen-1 is already involved in the stabilization of basement membranes in the early mouse embryo.
Only a few ultrastructural studies have been performed thus far, and these were in adult tissues (Grant and Leblond 1988; Schittny et al. 1988; Desjardins and Bendayan 1989; Katz et al. 1991; Miosge et al. 1999). As yet, nidogen-1 has not been localized in the early mouse embryo at the ultrastructural level. Nidogen-1 mRNA has been detected by in situ hybridization in a variety of tissues (Thomas and Dziadek 1993; Ekblom et al. 1994; Sephel et al. 1996; Kadoya et al. 1997) but not in the early mouse embryo nor at the ultrastructural level.
The early mouse embryo Day 7 is a suitable model for the investigation of basement membrane formation. The first well structured inner embryonic basement membrane appears between the endo- and ectoderm at a site in the embryo proper at which a mesoderm is not yet present (Herken and Barrach 1985). On the other hand, in another region, the mesoderm develops from the ectoderm. This process is paralleled by the destruction and re-formation of the ectodermal basement membrane. At the same developmental stage, the formation of the endodermal basement membrane starts. This opens up an opportunity to determine which cell types produce nidogen-1 during mesoderm formation and to elucidate the molecular composition of three differing developmental stages of basement membrane maturation.
Therefore, we localized nidogen-1 and its binding partners, laminin-1 and collagen Type IV, in a fully developed basement membrane, a disintegrated membrane, and a membrane that was just starting to develop. Moreover, we identified the cells that express nidogen-1 at this developmental stage.
Materials and Methods
Animals
Female NMRI mice were kept on a normal day/night cycle and received Altromin commercial food and water ad libitum. The day on which at 1100 hr a vaginal plug was detected after a mating period of 3 hr was designated as Day 0 of gestation.
Tissue Processing
On Day 7 of gestation, the pregnant animals were sacrificed by cervical dislocation and the embryos were removed from the uterus. For light microscopy, 10 specimens were fixed in 4% paraformaldehyde in phosphate buffer, dehydrated, and embedded in paraffin (Miosge et al. 1996). For immunogold histochemistry and in situ hybridization, five specimens were fixed for 30 min at 4C in 4% paraformaldehyde and 0.5% glutaraldehyde in 0.1 M PBS, treated with 10 mM ammonium chloride in 0.1 M PBS for 45 min, dehydrated in a graded ethanol series up to 70% ethanol, and embedded in the acrylic resin LR Gold (London Resin; Reading, UK). The resin was hardened at −25C with the addition of 0.8% of the light-sensitive initiator benzil and the light of a halogen lamp. As control specimens, three Day 17 mouse fetuses were dissected and halves of the kidneys, as well as three tissue pieces of EHS tumor, were processed as described above.
Paraffin sections (5 μm) and semithin (1 μm) and ultrathin (0.08 μm) sections were cut according to procedures previously described in detail (Herken and Miosge 1991; Miosge et al. 1996).
Sources of Antibodies
An affinity-purified monoclonal antibody JF4 (Fox et al. 1991) (2.2 mg/ml) against murine recombinant nidogen-1 was applied (Chemicon; Hofheim, Germany). A monoclonal rat anti-mouse antibody was prepared against murine laminin-1 and reacted exclusively with the laminin α1-chain in immunoblots. Ammonium sulfate precipitation resulted in IgG which was diluted in PBS to a concentration of 1 mg/ml (Miosge et al. 1995). Collagen Type IV polyclonal antibody against murine collagen Type IV (Andujar et al. 1985) was purchased from Quartett (Berlin, Germany).
In Situ Hybridization at the Light and Electron Microscopic Levels
An Acc I/Bgl II fragment of mouse nidogen-1 (X14480; Mann et al. 1989), corresponding to bp 621-1066, and coding for amino acids 201-348, was used for hybridization. Nonradioactively labeled sense and antisense RNAs were produced in vitro with the digoxigenin (DIG) labeling kit (Boehringer; Mannheim, Germany).
Semithin sections for the final light microscopic silver enhancement procedures were pretreated in Lugol's solution and 4% sodium thiosulfate, then incubated with the hybridization solution (50% formamide, 5 times; SSC, 0.1 mg/μl yeast tRNA) for 16 hr at 50C. The RNA concentration for nidogen-1 was 100 ng digoxigenin-labeled antisense probe in 100 μl hybridization solution per section. In control sections, an equivalently labeled amount of sense RNA was used instead of antisense RNA. Posthybridization treatment included washes with 1 times; SSC (twice for 15 min at 50C), 1 times; SSC (twice for 15 min), 0.1 times; SSC (four times for 15 min), both at 60C, and finally PBS for 15 min at room temperature (RT). Specimens were then incubated with a 1:60 dilution of anti-DIG (sheep IgG; Quartett) gold-labeled antibody in PBS for 1 hr at RT (Miosge et al. 1998). As controls, the procedure described above was carried out either without any probes or using only colloidal gold not coupled to antibody. None of them showed any staining.
For electron microscopic in situ hybridization, the grids were pretreated with PBS for 15 min at RT and then incubated with the hybridization solution for 16 hr at 45C. The RNA concentration for nidogen-1 was 200 ng digoxigeninlabeled antisense probe in 20 μl hybridization solution per section. Posthybridization treatment was the same as described above. Specimens were then incubated for 40 min with a 1:60 dilution of gold-labeled anti-DIG antibody at RT (Drabent et al. 1997; Miosge et al. 1998). Again, each hybridization was accompanied by a negative control with a sense probe. We also incubated the sections with a goat anti-rabbit IgG (Medac; Hamburg, Germany) coupled to colloidal gold or with colloidal gold alone. None of these controls showed any staining.
Light Microscopic Immunohistochemistry
Sections were deparaffinized and rehydrated, and endogenous peroxidase was blocked. They were then treated with 0.01% protease XXIV (Sigma; Deisenhofen, Germany) for 5 min at RT. Each step was followed by a rinse in PBS. Primary antibodies (anti-laminin-1 and anti-nidogen-1 diluted 1:50 in PBS; anti-collagen Type IV 1:100) were incubated for 1 hr at RT. Bridge antibody (anti-rabbit or anti-rat swine IgG; Dakopatts, Hamburg, Germany) was applied (diluted 1:50 in PBS), followed by the PAP complex (diluted 1:150 in PBS), each for 30 min at RT. Finally, the sections were treated with 3,3′-diaminobenzidine and counterstained with hematoxylin. Negative controls were performed by applying normal rabbit or rat serum instead of the primary antibodies, and these remained unstained.
Electron Microscopic Immunohistochemistry
Sixteen-nanometer gold particles were prepared according to Frens (1973) and Slot and Geuze (1981). The gold particles were used to label antibodies according to DeMey et al. (1981) and Herken et al. (1987). Tissue sections on nickel grids were incubated for 15 min at RT with PBS and then incubated with anti-nidogen-1 (diluted 1:200 with PBS), anti-laminin-1 (diluted 1:100 with PBS), or anti-collagen Type IV (diluted 1:200 in PBS) antibodies for 1 hr at RT. The sections were rinsed with PBS and incubated for 20 min with the secondary gold-coupled antibody (affinity-purified goat anti-rat IgG or anti-rabbit IgG; Medac) diluted 1:300 in PBS. Sections were rinsed with water, stained with uranyl acetate for 15 min and lead citrate for 5 min, and then examined with a Zeiss EM 109 electron microscope. To exclude nonspecific binding, we incubated control sections with the pure gold solution under the same conditions as described above. We also incubated the tissue sections with goat anti-rabbit or anti-rat antibodies coupled to colloidal gold. All controls were negative.
Results
Light Microscopic Immunohistochemistry for Nidogen-1, Laminin-1, and Collagen Type IV
All three components were detected in the mouse embryo Day 7 in basement membrane zones of the maternal blood vessels, Reichert's membrane, extraembryonic, and basement membrane zones of the embryo proper, shown here for collagen Type IV (Figure 1A).
At a higher magnification of all three germ layers, linear staining for collagen Type IV was seen in basement membrane zones between the ectoderm and the mesoderm and less intensely between the mesoderm and the entoderm (Figure 1B). A similar staining pattern was also found for nidogen-1 (Figure 1C) and lam-inin-1 (Figure 1D).
Ultrastructural Localization of Nidogen-1, Laminin-1, and Collagen Type IV
For the localization of the proteins in the basement membranes, the ultrastructural method was chosen to allow an unequivocal identification of deposits in these structures. Again, staining for nidogen-1 was seen in Reichert's membrane (Figure 2A) still surrounding the mouse embryo at this early developmental stage.
In the extraembryonic part of the egg cylinder, a basement membrane had developed between the extraembryonic ectoderm and the yolk sac cells. Staining for nidogen-1 revealed diffusely distributed gold labeling over the entire extraembryonic basement membranes (Figure 2B). Within the embryo proper, basement membrane development had started in the embryonic part of the egg cylinder. At this stage of development, the embryonic basement membrane was characterized by the appearance of electron-dense extracellular matrix between the ectodermal and endodermal cell layers, giving the impression of a thick basement membrane that was positive for nidogen-1 (Figure 2C). In the caudal (primitive streak) and the lateral regions of the transition zone between the extraembryonic and the embryonic part of the egg cylinder, the mesodermal layer had started to develop between the ectoderm and the endoderm. In this area, the basement membrane, which is later disrupted by mesodermal cells that migrate from the embryonic ectoderm into a cavity formed between the ectodermal and endodermal layer (mesodermal space), was also stained for nidogen-1. In contrast, the basement membrane remnants of the disrupted basement membrane carried along the surface of the migrating mesodermal cells (Miosge et al. 1993) did not stain for nidogen-1. However, the basement membrane of the ectoderm, which seals the mesoderm cavity with its mesodermal cells after migration has ceased, were again positive for nidogen-1 (Figure 2D). No staining for nidogen-1 was seen in the region at which the endodermal basement membrane starts to develop (Figure 2E).
Staining for laminin-1 was seen in Reichert's membrane (Figure 2F) and in all of the basement membranes described above, shown here for the basement membrane between the endo- and ectoderm (Figure 2G) and the ecto- and mesoderm (Figure 2H). Lami-nin-1 was detected in the basement membrane starting to develop adjacent to the endoderm. The same staining pattern was found for collagen Type IV. Staining was again seen in Reichert's membrane (Figure 2I) and in the basement membranes of the embryo proper, shown here for the ectodermal basement membrane (Figure 2J).
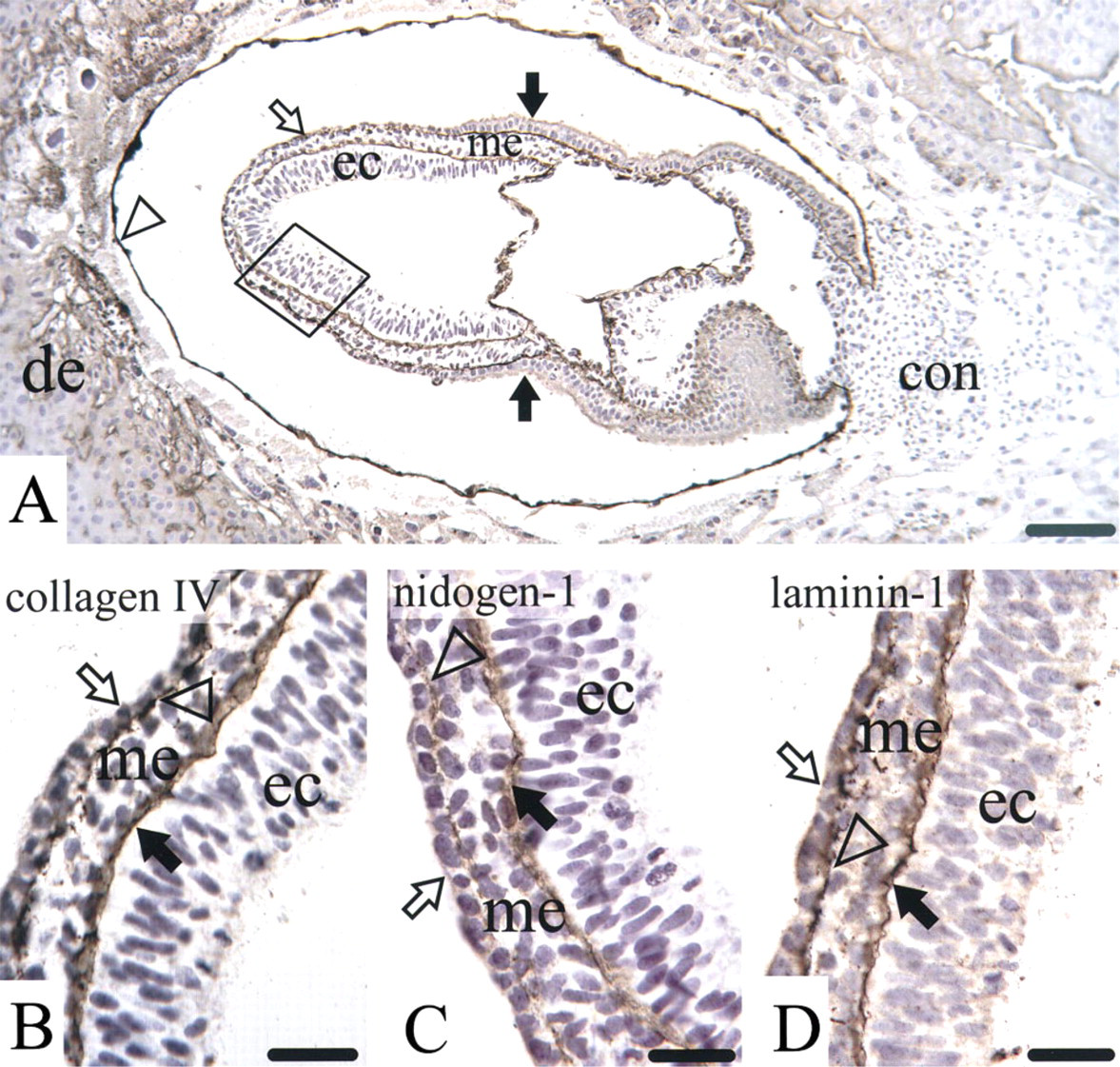
Light microscopic immunostaining for collagen Type IV (
Nidogen-1 mRNA Expression and Localization
To reveal which cells in the early mouse embryo produce nidogen-1, we performed in situ hybridization at the light and electron microscopic levels.
At the light microscopic level, nidogen-1 mRNA was found in the mouse embryo Day 7 in the cytoplasm of the cells of the mesoderm (Figure 3A). As expected, the nuclei of the cells were not stained. Furthermore, the cytoplasm of the cells of the other two germ layers, the endoderm and the ectoderm, exhibited labeling for nidogen-1 mRNA (Figure 3B). In addition, the cytoplasm of the parietal endoderm cells secreting Reichert's membrane were positive for nidogen-1 mRNA (Figure 3B). The control hybridizations with sense probes for nidogen-1 revealed no labeling in any of the described structures (Figure 3C).
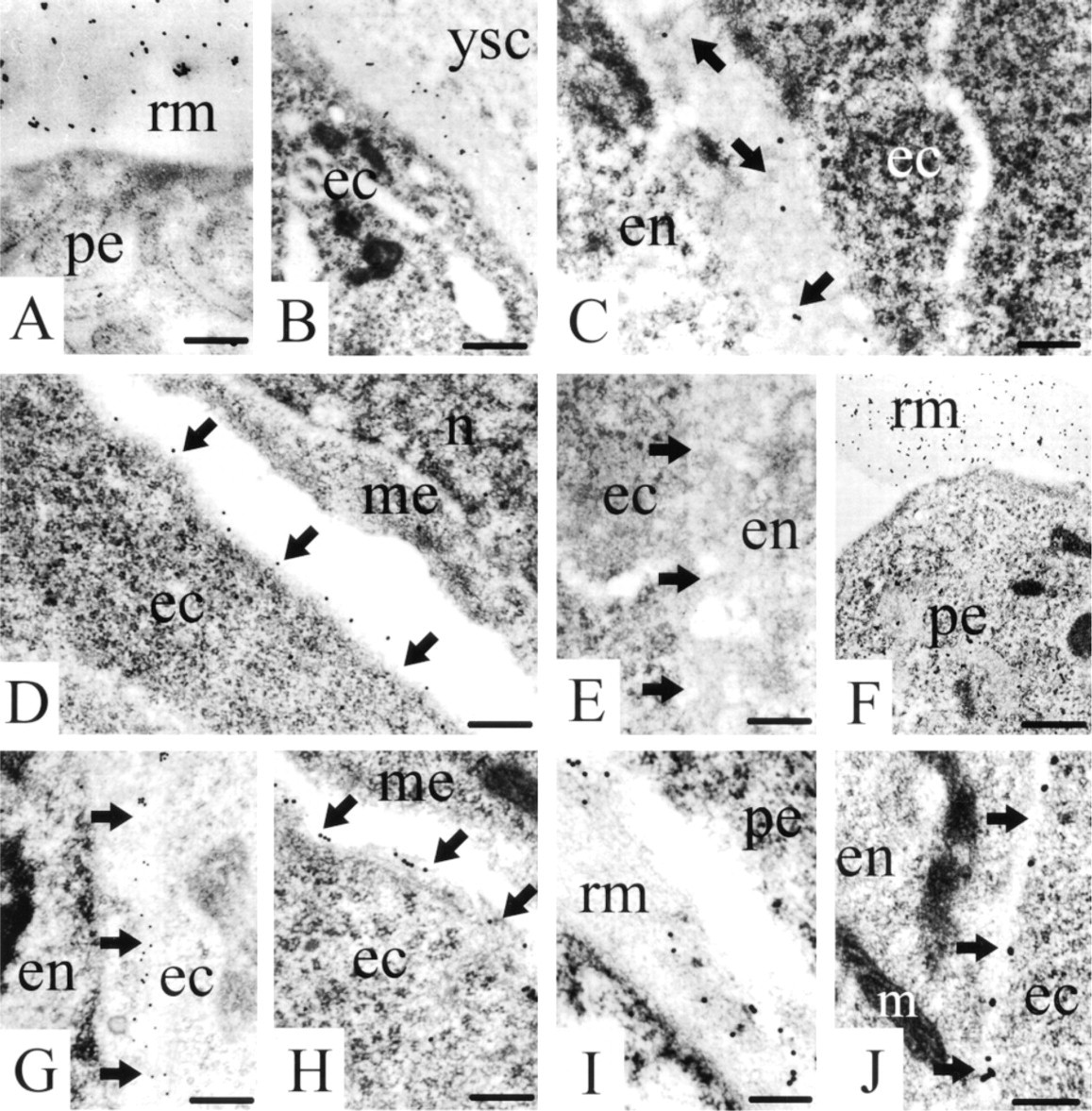
Immunogold histochemistry for nidogen-1 (
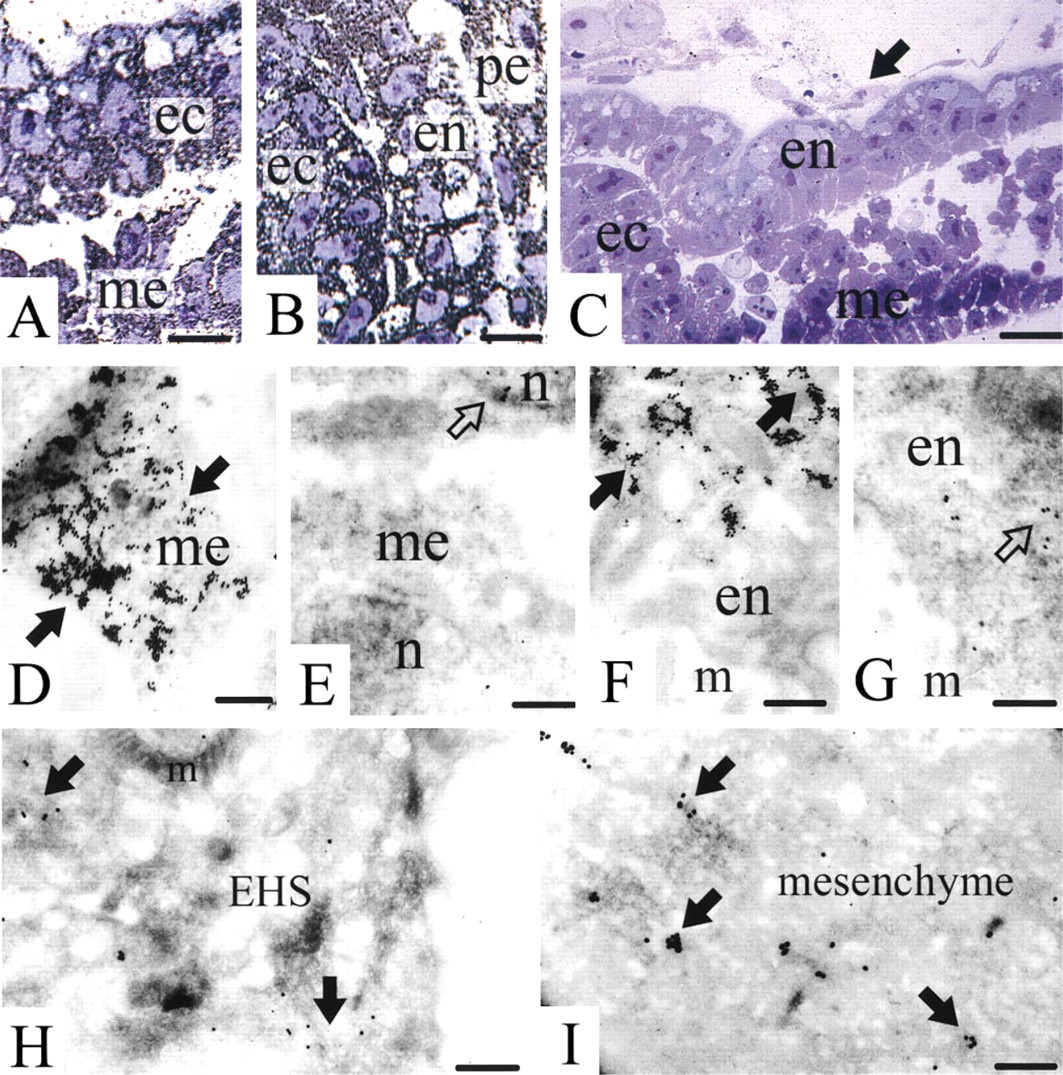
Light microscopic (
Electron microscopic in situ hybridization confirmed the staining of the cytoplasm of the cells of the three germ layers and revealed the strongest labeling in the cytoplasm of the mesoderm (Figure 3D). As for all hybridizations performed, the corresponding sense probe revealed only sparse labeling, regarded as nonspecific background (Figure 3E). In addition, the cytoplasm of the ecto- and endoderm (Figure 3F) was positive for nidogen-1 mRNA. Again, the corresponding hybridization with the sense probe revealed a low labeling intensity, regarded as nonspecific background (Figure 3G).
Because of the very high labeling intensity seen for nidogen-1 mRNA in all three germ layers and the fact that nidogen-1 mRNA has previously been detected mainly in mesenchymal cells, we performed several control experiments. Hybridization for nidogen-1 was carried out on EHS tissue (an epithelial tumor), the previous primary source of nidogen-1 (Timpl et al. 1983). Here the cytoplasm of the tumor cells exhibited a lower labeling intensity for nidogen-1 mRNA (Figure 3H). In agreement with the previous investigations (Ekblom et al. 1994; Dziadek 1995), hybridization of Day 17 mouse embryo kidney exhibited staining of the cytoplasm of mesenchymal cells for nidogen-1 mRNA (Figure 3I).
Discussion
Nidogen-1 is produced by mesenchymal cells during fetal mouse development (Thomas and Dziadek 1993, 1994) and is believed to serve as a link molecule to connect the laminin-1 and collagen Type IV networks already in fetal basement membranes in vivo (Miosge et al. 1999). On the other hand, basement membranes are seen at sites in the early embryo at which no mesoderm or mesenchyme is to be found (Salamat et al. 1995). Furthermore, nidogen-1 has already been detected in the preimplantation embryo (Wu et al. 1988), where there is no mesoderm present at all that could have produced it. Therefore, the question arises as to which cells in the early embryo express nidogen-1.
We found strong expression of nidogen-1 mRNA in the developing mesodermal cells, and the cells of the endo- and ectoderm also produced nidogen-1 mRNA. This supports the emerging concept that although nidogen-1 is predominantly a product of mesenchymal origin (Ekblom et al. 1994), it can also be generated by other cell types. The mRNA levels found in the cells of all three germ layers of the early mouse embryo were much higher than those in EHS tumor cells or in the fetal mesenchymal cells of the developing kidney employed as controls. We assume that the extensive production of nidogen-1 is maintained in the early embryo to allow a rapid turnover of basement membranes in the quickly developing tissue. The low amount of mesoderm then present might not be sufficient to maintain the turnover needed at this point.
One of the major functions of nidogen-1 is its strong interaction with laminins, giving rise to further interaction sites for basement membrane assembly (Timpl and Brown 1996). This concept was underlined by the recent generation of knockout mice lacking the γ1-chain of laminin. These mice develop no basement membranes at all and die early during embryogenesis (Smyth et al. 1999). At the cellular level, basement membrane assembly appears to be controlled in at least two different ways, as shown by several biosynthetic and tissue studies (Dziadek 1995). In some cases, biosynthesis of laminins and nidogen-1 occurs in the same cell and is very likely followed by rapid intracellular formation and secretion of the laminin-nidogen complex (Wu et al. 1988). This also enhances stability against endogenous proteolysis (Dziadek 1995). Typical examples are the formation of Reichert's membrane by parietal endoderm (Thomas and Dziadek 1993) and of the lens capsule (Dong and Chung 1991). The alternative pathway involves cellular cooperation, where laminin is usually derived from an epithelial and nidogen from a mesenchymal compartment, as shown for the branching morphogenesis of kidney, lung, and salivary glands (Thomas and Dziadek 1993, 1994; Ekblom et al. 1994), for mammary glands (Warburton et al. 1984), and skin formation (Fleischmajer et al. 1995). Antibodies that block the laminin-nidogen interaction interfere with morphogenesis (Ekblom et al. 1994; Kadoya et al. 1997), and this strongly indicates that complex formation occurs in the extracellular space. This appears to be different during kidney development, which depends on mesenchymal-epithelial cooperation (Ekblom 1993) and the production of nidogen-1 by the mesenchyme (Ekblom et al. 1994). Which pathway is active in the early mouse embryo remains to be elucidated. Recently, a new nidogen isoform, nidogen-2, has been described (Kohfeldt et al. 1998). We found this protein to be colocalized with laminin-1 in murine kidney basement membranes in vivo, where it could fulfill functions similar to those of nidogen-1 (unpublished data). Whether this is also true for nidogen-2 in the early mouse embryo remains unclear.
Only a few authors have investigated basement membrane components in the early mouse embryo (Leivo et al. 1980; Herken and Barrach 1985). Laminin synthesis apparently starts in the 4-8-cell embryo (Cooper and MacQueen 1982). Laminin was found before nidogen in the preimplantation embryo, while both proteins were simultaneously present in extraembryonic basement membranes (Wu et al. 1988), a region within the embryo at which basement membrane formation starts (Herken and Barrach 1985; Salamat et al. 1995). We have previously shown that laminin-1 is lined up between the endo- and ectodermal cells before a proper basement membrane is seen (Miosge et al. 1993). This was again seen for laminin-1 and collagen Type IV but not for nidogen-1 in the present study. Nidogen-1 was present only when a fully structured basement membrane, such as the ectodermal basement membrane, was seen. In contrast, the developing endodermal basement membrane lacked nidogen-1, although its mRNA was already present in the adjacent cell layers. This indicates that all three major basement membrane components must be present before a morphologically distinct basement membrane is detectable. Our results clearly demonstrate that nidogen-1 is produced in all cells of the three germ layers and contributes to the formation of the basement membrane of the ectoderm during the onset of mesoderm formation. No nidogen-1 can be found in the disintegrating basement membrane during mesoderm formation. Because nidogen-1 protects laminin-1 against proteolytic cleavage (Dziadek 1995), its removal might be the initializing step towards basement membrane disintegration. Moreover, nidogen-1 was not seen in the very early stages of basement membrane formation. Viewed together, these results back the current basement membrane models proposing two networks of laminin-1 and collagen Type IV interconnected by nidogen-1 (Timpl and Brown 1996).
Footnotes
Acknowledgments
Supported by a grant from the German Research Council (DFG) to N. Miosge (Mi 573/1-2).
We wish to thank Dr Rupert Timpl, MPI for Biochemistry, Martinsried, for the kind gifts of cDNA, and Cyrilla Maelicke, BSc, for editorial correction of the manuscript. Parts of the work were taken from the doctoral thesis of Tobias Fahl.
