Abstract
A smooth membrane system consisting of subsurface cisternae (SSC) underlies the lateral plasmalemma of auditory outer hair cells (OHCs). The SSC contain Ca-ATPase and are regarded as an intracellular Ca2+ reservoir like the sarcoplasmic reticulum of myocytes. Recently, it has been demonstrated that Ca-ATPase activity in sarcoplasmic reticulum is regulated by Ca2+/calmodulin-dependent protein kinases (CaM kinases). Here we investigated the presence of CaM kinases in OHCs and their possible association with the SSC. Inner ears collected from adult gerbils and from neonates at 2-day intervals between 0 and 20 days after birth were immunostained with antibodies specific for different CaM kinases. A polyclonal antiserum against CaM kinase IV yielded a strong immunostaining reaction along the lateral wall of OHCs. The staining appeared after the tenth postnatal day and continued into adulthood. No other site in the inner ear, including cochlear inner hair cells and vestibular hair cells, was reactive. The kinase's apparent association with the SSC strongly supports its involvement in intracellular Ca2+ homeostasis and suggests a role in regulating the OHCs' slow motile responses.
Free cytosolic calcium (Ca2+) acts widely as an intracellular second messenger. A variety of stimuli can increase cytosolic Ca2+ levels by promoting an influx of extracellular Ca2+ through voltage- or ligand-gated Ca channels or by releasing the ion from intracellular stores sequestered in specialized elements of smooth endoplasmic reticulum. In the opposite direction, CaATPases in the plasma membrane (PMCAs) and smooth endoplasmic reticulum (SERCAs) pump the ion out of the cytosol, returning its concentration to resting levels (Carafoli and Stauffer 1994; Carafoli et al. 1996; Martonosi 1996; Mintz and Guillain 1997).
The signaling effect of intracellular Ca2+ can be mediated through activation of Ca2+/calmodulin-dependent protein kinases (CaM kinases). The four known isoforms of CaM kinase have a tissue- and cell type-specific distribution pattern and mediate diverse physiological activities (Sikela and Hahn 1987; Ohmstede et al. 1989; Schulman 1993). Of these, CaM kinase IV has been implicated in the regulation of gene transcription based, in part, on its localization in nuclei of cerebellar granular cells (Ohmstede et al. 1989; Jensen et al. 1991; Sakagami et al. 1994; Nakamura et al. 1995) and in neurotransmitter release based on its capacity to phosphorylate synapsin I (Miyano et al. 1992). In addition, CaM kinases have been found to act by phosphorylating and thereby increasing the activity of SERCAs in sarcoplasmic reticulum (Leddy et al. 1993; Mattiazzi et al. 1994; Baltas et al. 1995; Damiani et al. 1995; de Alfonzo et al. 1996; Grover et al. 1996; Narayanan and Xu 1997).
In sensory hair cells, Ca2+ plays an important role in mechanoelectrical transduction (Hudspeth 1985; Ohmori 1985, 1988; Pickles and Corey 1992) and also in mediating the slow motile responses of auditory outer hair cells (OHCs) (Flock 1988; Zenner et al. 1988; Corwin and Warchol 1991). OHCs contain under their lateral plasmalemma a well-developed subsurface cisternal system (SSC) which sequesters Ca2+ by means of SERCAs (Schulte 1993). The SSC has been considered analogous to the sarcoplasmic reticulum of muscle and involved with motility (Flock et al. 1986; Lim 1986; Flock 1988). The question arises then concerning the presence and possible role of CaM kinases in regulating OHC activity. The presence of calmodulin in OHCs (Slepecky and Ulfendahl 1993; Pack and Slepecky 1995; Imamura and Adams 1996) and a recent study documenting the inhibition of Ca2+-induced slow motility by CaM kinase inhibitors support the enzymes' activity in OHCs (Puschner and Schacht 1997). This study sought further evidence for this by undertaking to localize CaM kinases in the inner ear immunohistochemically. The findings demonstrate CaM kinase IV exclusively along the lateral wall of cochlear OHCs, where its activity could participate in the regulation of Ca2+ levels in SSC and cell motility.
Materials and Methods
Animals and Tissue Processing
Cochleas from 42 Mongolian gerbils (
Gerbils were anesthetized by IP injection of urethane (1.5 g/kg body weight). Exsanguination was performed by transcardial perfusion with warm physiological saline containing 0.1% sodium nitrate, followed by 10 ml of a 10% formalin solution containing 0.5% zinc dichromate, pH 5.0, at room temperature (RT). The bulla was opened rapidly, the round window was perforated, and 1.0 ml of fixative was infused gently through the oval window. The inner ears were dissected free and immersed in fixative for 15 min, rinsed with saline, and decalcified by immersion in 0.12 M EDTA (pH 7.0) for 48-72 hr at RT with gentle stirring. The EDTA solution was changed daily. The decalcified inner ears were dehydrated in a graded series of ethanols, cleared in Histoclear (National Diagnostics; Manville, NJ), and embedded in Paraplast Plus (Curtin Matheson; Marietta, GA). Serial midmodiolar sections were cut at 5 μm thickness and mounted on glass slides.
For surface preparations, inner ears from three young adults were fixed and processed as described above. After decalcification, the cochleas were bisected with a sharp razor blade and then sliced into half-turns. The tectorial membrane and lateral wall were removed, leaving the organ of Corti attached to the modiolus. The above procedures were performed in 0.1 M PBS, pH 7.2, at RT and the resultant specimens were held in PBS at 4C until further processing.
Immunohistochemistry
Immunostaining of tissue sections was performed as described previously (Schulte and Adams 1989; Schulte and Schmiedt 1992). Deparaffinized and rehydrated sections were immersed for 15 min in 3% H2O2 to block endogenous peroxidase. After washing with distilled water, the sections were treated with 0.1% Triton X-100 in PBS for 30 min, rinsed with PBS, and flooded for 30 min with PBS containing a 1% concentration of appropriate normal serum. Sections were then incubated overnight at 4C with one of the primary antibodies diluted 1:100 with PBS. The primary antibodies tested included: goat anti-mouse CaM kinase IV, goat anti-rat CaM kinase I (Santa Cruz Biotechnology; Santa Cruz, CA), and mouse anti-CaM kinase II α-subunit (Affinity BioReagents; Golden, CO). The sections were then rinsed in PBS and incubated with appropriate biotinylated secondary antibodies diluted 1:200. After rinsing with PBS, the sections were flooded with avidin-biotin-horseradish peroxidase complex (Vectastain ABC kit; Vector Laboratories, Burlingame, CA) for 30 min. The sections were again rinsed with PBS and reacted for 10 min in substrate medium containing 3,3′-diaminobenzidine HCl (DAB; Sigma Chemical, St Louis, MO) before dehydration and mounting. To provide accurate comparisons among cochleas at different developmental stages, sections representing all time points were stained together in the same protocol under identical conditions.
Nonimmune goat serum was substituted for the CaM kinase IV antiserum as a methods control procedure. Sections from composite blocks containing a wide range of gerbil, rat, and mouse organs processed in parallel with the cochlea sections provided positive and negative tissue controls for assessing specificity of the CaM kinase IV antiserum.
Immunostaining of Surface Preparations
The dissected specimens of cochlea were equilibrated in PBS for 20 min, immersed in 3% H2O2 for 30 min, and rinsed again with PBS for 20 min. The pieces were then immersed in 1% Triton X-100 in PBS for 1 hr and then in 0.01% Triton X-100 in PBS for 30 min. This was followed by incubation for 1 hr with 5% normal rabbit serum in 0.01% Triton-PBS and immersion in a 1:100 dilution of anti-CaM kinase IV, overnight at 4C. After washing in PBS for 20 min, the pieces were incubated with biotinylated rabbit anti-goat IgG diluted 1:300 in 0.01% Triton-PBS for 1 hr, rinsed in PBS, immersed in ABC reagent for 2 hr, rinsed again with PBS for 20 min, and reacted with DAB for 10 min. The stained specimens were mounted organ of Corti side up in 50% glycerol in thin wells constructed on a slide glass with cut coverglasses.
Results
An immunosurvey of organs from adult rats and mice confirmed the presence of CaM kinase IV in brain, where the enzyme has previously been found (Ohmstede et al. 1989; Jensen et al. 1991; Sakagami et al. 1994; Nakamura et al. 1995). Sites showing a strong positive reaction included nuclei of granule cells in the cerebellum (Figure 1) and neurons in layers II-IV of the cerebral cortex (Figure 2). A similar pattern was seen in the gerbil brain, although staining was less intense than that observed in the mouse and rat. No other sites among the many organs surveyed, including heart, lung, liver, spleen, pancreas, salivary glands, small and large bowel, lymph nodes, ovary, and skeletal muscle, stained positively with antiserum against CaM kinase IV. Control sections of inner ear and brain in which nonimmune goat serum was substituted for the primary antiserum showed no immunoreactivity (data not shown). The antibodies against CaM kinase I and CaM kinase II showed no positive staining in the cochlea or any other tissue surveyed.
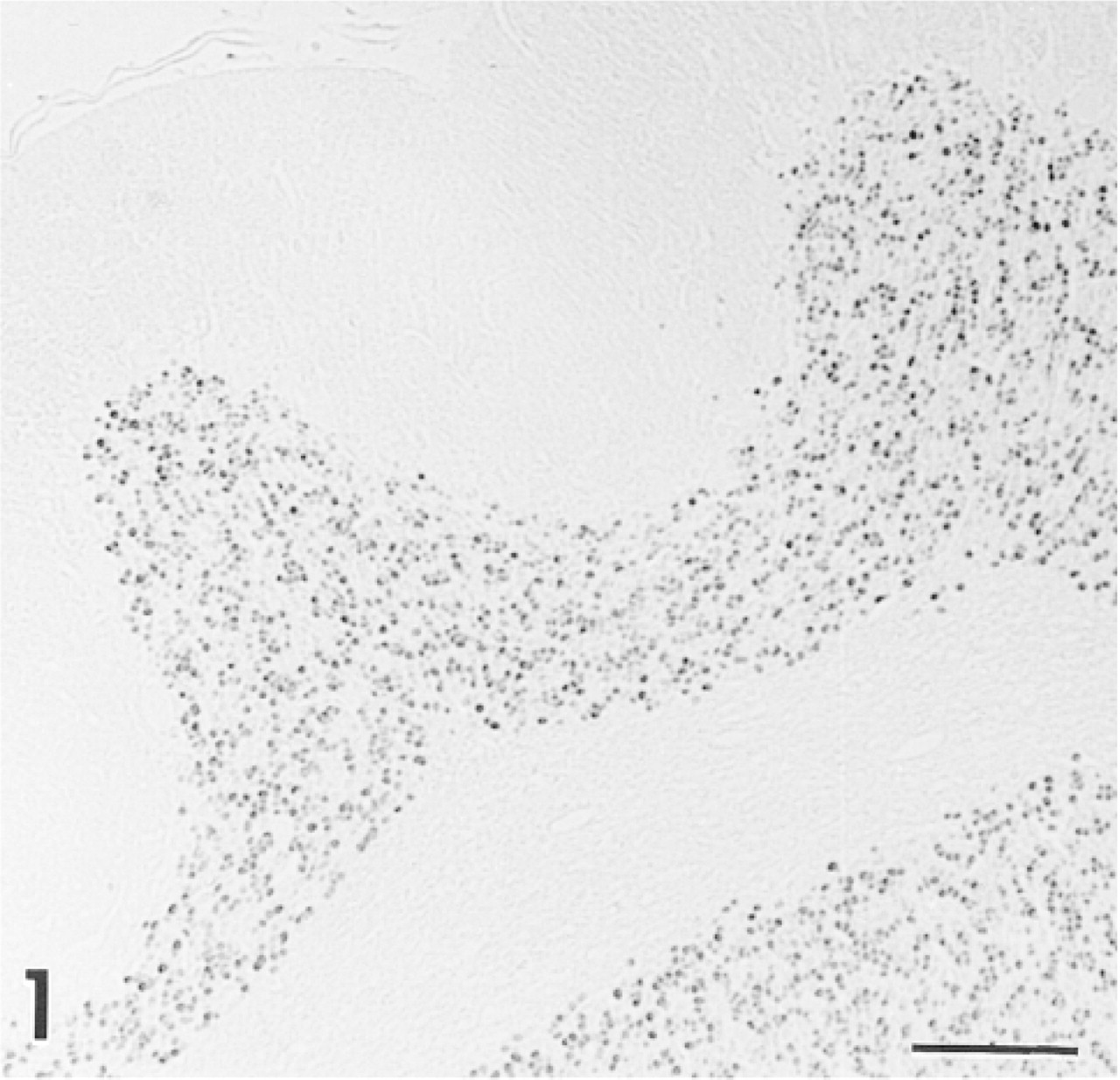
Positive control sections from rat brain show strong staining with anti-CaM kinase IV in nuclei of cerebellar neurons. Similar staining was seen in the mouse and gerbil. Bar = 100 μm.
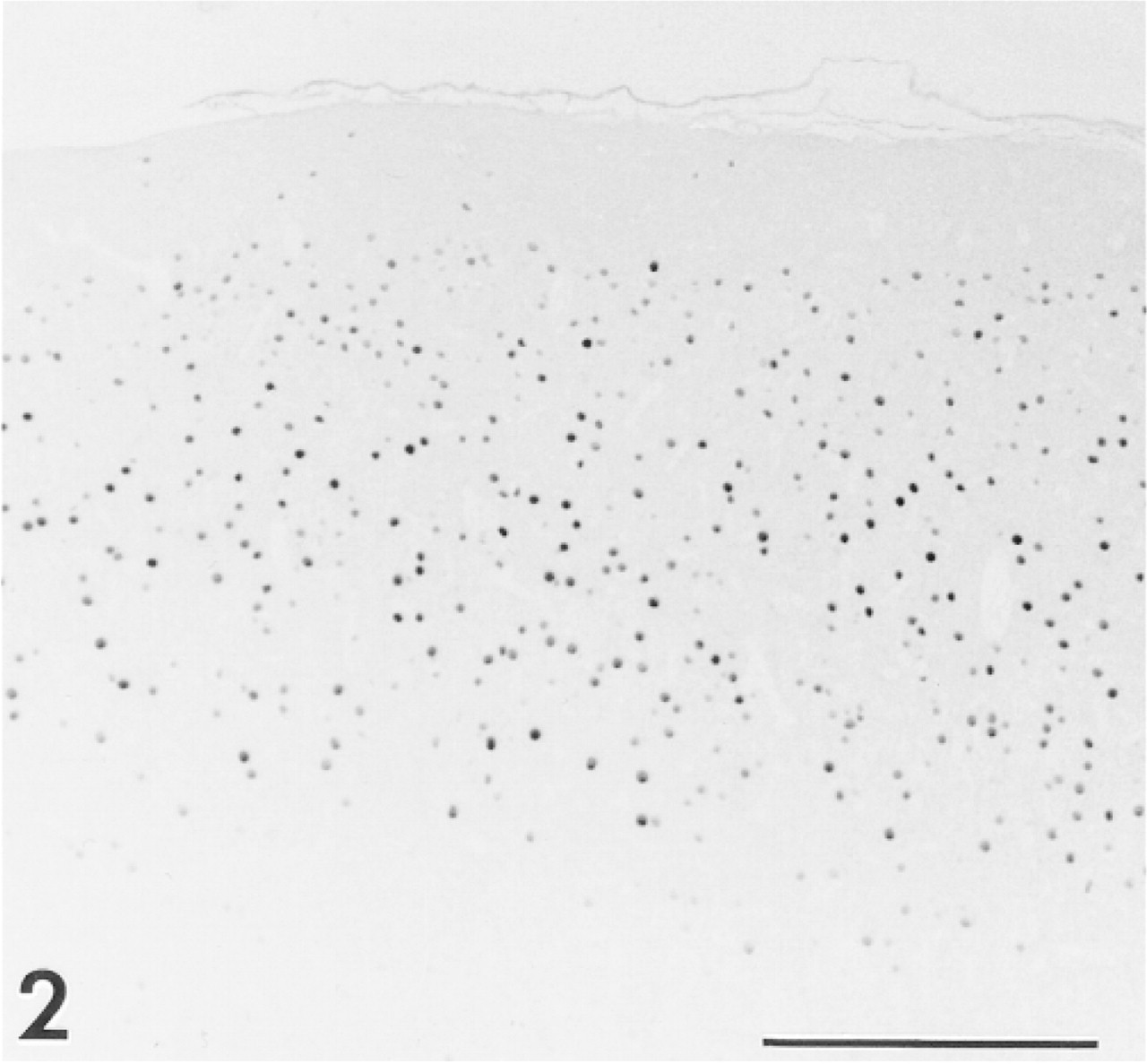
Neurons of rat cerebral cortex show strong nuclear staining. Many immunoreactive cells are located in layers II through IV. Anti-CaM kinase IV. Bar = 200 μm.
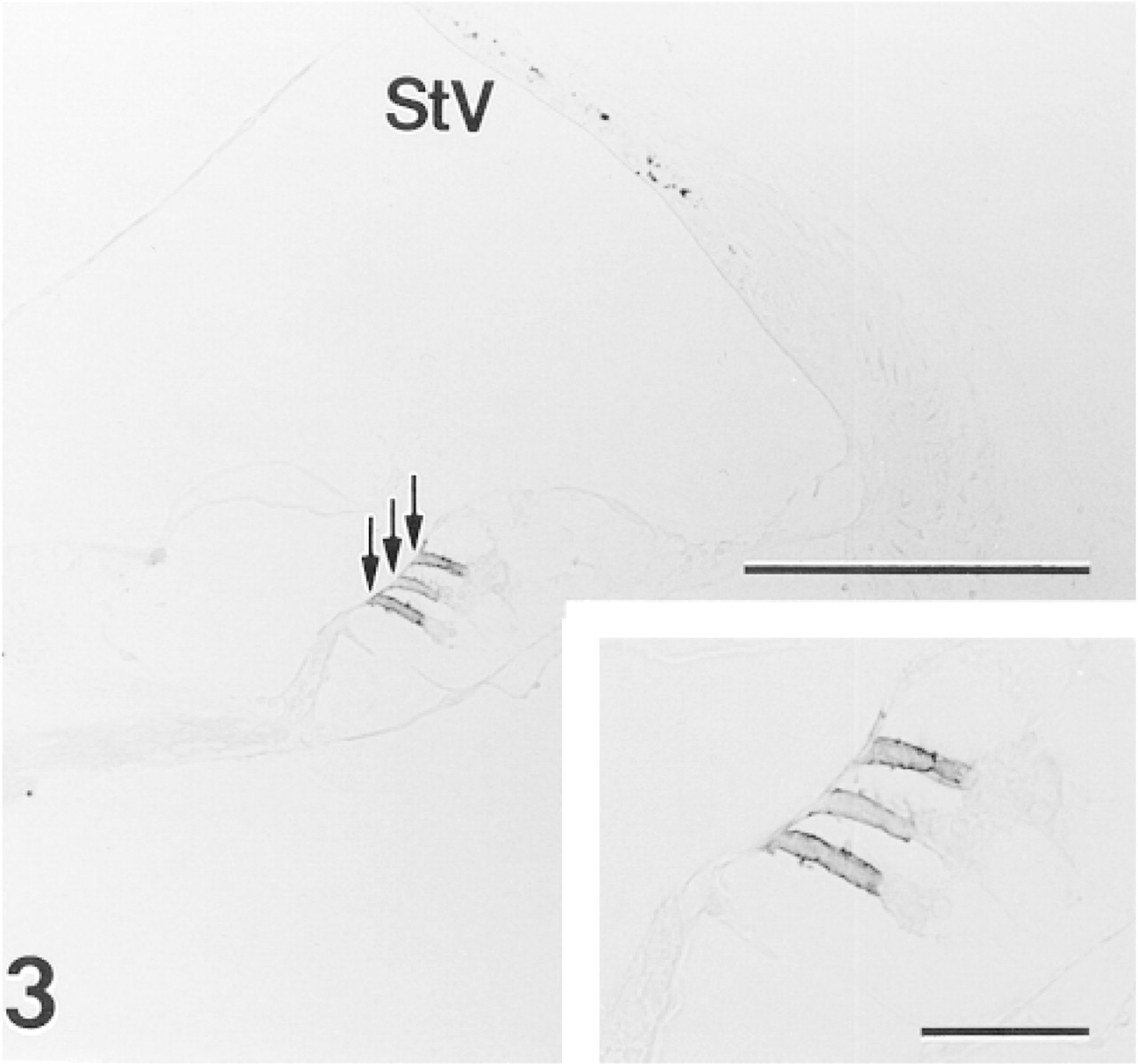
Intense immunoreactivity is present along the lateral border of OHCs (arrows) in middle turn of a 3-month-old gerbil cochlea. Punctate staining in the stria vascularis (StV) is due to endogenous melanin pigment in intermediate cells. Anti-CaM kinase IV. Bar = 100 μm;
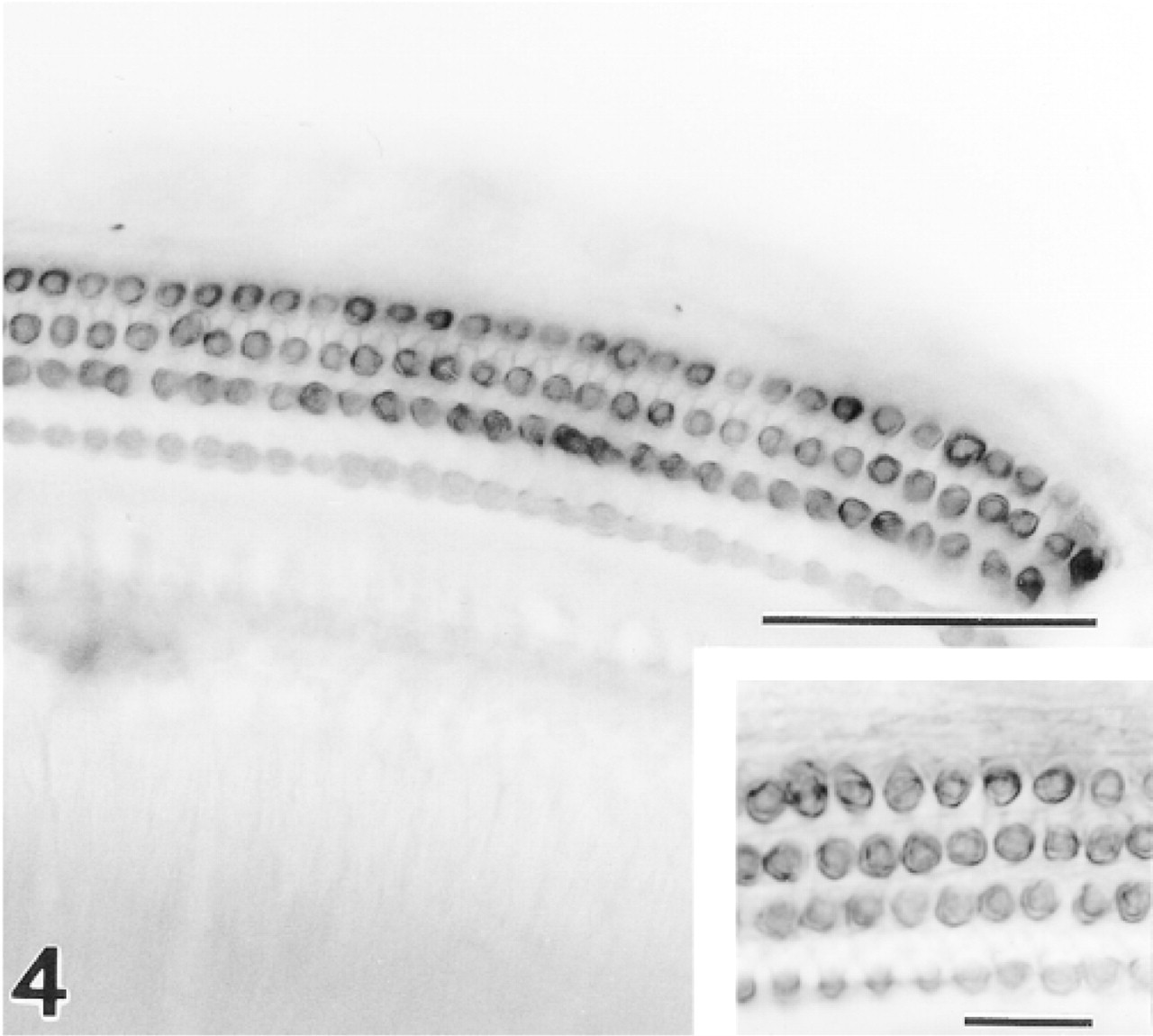
All three rows of OHCs show strong positive staining in a surface preparation from the upper basal turn. Anti-CaM kinase IV. Bar = 50 μm.
In sections of the adult gerbil inner ear, moderate to strong immunoreactivity for CaM kinase IV was present along the lateral borders of OHCs (Figure 3). Taller OHCs in all three rows from the apical and middle turns of the cochlea showed consistent immunopositivity, whereas the shorter OHCs in the lower basal turn and hook varied from weakly reactive to unstained. In the neonatal series, positive immunostaining first appeared in OHCs between 10 and 12 days after birth and reached adult levels by 20 days after birth (data not shown). No other site in the inner ear, including inner hair cells and vestibular hair cells, evidenced affinity for the CaM kinase IV antiserum.
In surface preparations, all three rows of OHCs showed intense staining with the CaM kinase IV antiserum (Figure 4). The strongest staining was in a rim just under the plasmalemma, although the staining appeared to extend into the cytoplasm and nuclear envelope in some cells. Such cytoplasmic and nuclear reactivity was never observed in sectioned materials and possibly represents a diffusion artifact owing to the extensive detergent permeabilization of the whole-mount preparations.
Discussion
OHCs possess prominent SSC that consist of smooth endoplasmic reticulum (Santi 1988), and vary among genera in the extent of layering (Smith 1968; Engström and Engström 1972; Kimura 1975). Among other proposed functions, the SSC has been considered analogous to the sarcoplasmic reticulum of muscle cells.
Immunocytochemical demonstration of SERCA in the SSC (Schulte 1993; Zine and Schweitzer 1996) supports this view and testifies to the role of the organelle as a Ca2+ reservoir. SERCA activity in sarcoplasmic reticulum of myocytes is known to be modulated by CaM kinases (Leddy et al. 1993; Mattiazzi et al. 1994; Baltas et al. 1995; Damiani et al. 1995; de Alfonzo et al. 1996; Grover et al. 1996; Narayanan and Xu 1997). Therefore, the question addressed in the present study concerned whether SSC in OHCs are associated with CaM kinases as is sarcoplasmic reticulum.
The results documented the expression of CaM kinase IV in the region underlying the lateral plasmalemma of OHCs, a distribution corresponding with that of SSC. Whether the enzyme resides in membranes of SSC could not be determined with certainty at the light microscopic level of resolution. However, the appearance of CaM kinase IV immunoreactivity at 12 days after birth coincided with the development of SSC (Weaver and Schweitzer 1994) and the onset of cochlear function on the twelfth postnatal day in the gerbil (Finck et al. 1972; Woolf and Ryan 1984; McGuirt et al. 1995). SSC are not present at 6 days after birth. Interrupted segments of cisternae appear by 8 days after birth, and the first full single layer is complete at 10 days after birth (Souter et al. 1995). The layers then multiply and reach a maximum of six in the mature gerbil cochlea (Weaver and Schweitzer 1994). The advent of staining in the lateral wall for CaM kinase IV at 12 days after birth also coincided with the first expression of SERCA between 12 and 14 days after birth (Schulte 1993; Zine and Schweitzer 1996), again indicating close association of the enzyme with the cisternae.
Immunopositivity for CaM kinase IV also correlated well with the relative abundance of SSC along the gerbil's place-frequency map. The immunostaining appeared weakest and was inconsistent in the short OHCs of the lower basal turn and hook, where the SSC are less prevalent and less well developed (Lutz and Schweitzer 1995). Furthermore, the weaker reactivity for CaM kinase IV in these higher-frequency regions corresponded with weaker basal staining for SERCA, which also is SSC-associated (Schulte 1993). Notably, inner hair cells and vestibular hair cells that lack multilayered SSC failed to stain for CaM kinase IV.
CaM kinase IV has heretofore been primarily regarded as a regulator of gene transcription (Enslen et al. 1994; Antoine et al. 1996; Ho et al. 1996; Sun et al. 1996; Wayman et al. 1996; Williams et al. 1996; Alevizopoulos et al. 1997). The present observations, however, differ from prior findings in showing absence of nuclear localization, and attest to a role other than regulation of gene expression for CaM kinase IV in OHCs. Recently, it was shown that KN-62, an inhibitor of CaM kinases II and IV (Enslen et al. 1994; Williams et al. 1996), suppresses the Ca2+-induced motility of OHCs (Puschner and Schacht 1997). This finding and our data showing the enzyme in close association with the SSC strongly suggest that the function of CaM kinase IV relates to the OHC's slow contractile response. CaM kinase IV's influence on motility may depend on the enzyme increasing the activity of SERCA in SSC as do other CaM kinases in sarcoplasmic reticulum (Leddy et al. 1993; Mattiazzi et al. 1994; Baltas et al. 1995; Damiani et al. 1995; de Alfonzo et al. 1996; Grover et al. 1996; Narayanan and Xu 1997). In this case, the enzyme would be expected to exert a brief self-regulating effect which, by lowering cytosolic Ca2+, would end in terminating its own Ca2+-dependent activity. Alternatively, the capacity of CaM kinase IV to phosphorylate myosin light chains (Miyano et al. 1992) that may be present in the OHC's cortical lattice suggests another pathway via which the kinase could influence Ca2+-mediated OHC contractility. Defining the precise mechanism by which CaM kinase IV regulates OHC responses remains a challenge for future investigations.
Footnotes
Acknowledgments
Supported by Research Grants R01 DC00713 and P01 DC00422 from the National Institute on Deafness and Other Communication Disorders, National Institutes of Health.
We thank Ms Leslie Harrelson for editorial and Ms Barbara Schmiedt for technical assistance.
