Abstract
SPARC (secreted protein, acidic and rich in cysteine)/osteonectin is a matricellular, counteradhesive glycoprotein that disrupts cell-matrix interactions, interacts with growth factors and components of extracellular matrix, and modulates the cell cycle, but appears to subserve only minor structural roles. SPARC is expressed in a variety of tissues during embryogenesis and remodeling and is believed to regulate vascular morphogenesis and cellular differentiation. Although usually limited in normal adult tissues, SPARC is expressed at significant levels in the adult central nervous system. Using a monoclonal antibody against bovine bone osteonectin, we have determined the localization of SPARC in newborn (3-day-old) and adult (4–8-year-old) normal bovine retinas. SPARC was present in the soma of ganglion cells and strong reactivity was found in ganglion cell axons. Muller cells displayed no immunoreactivity, but SPARC was present in retinal astrocytes that were identified by the astrocyte marker glial fibrillary acidic protein (GFAP). Newborn calf retina showed a staining pattern similar to that of adult retina but exhibited significantly reduced levels of SPARC. Minimal levels of SPARC protein were also detected in some capillaries of the inner retina of both newborn and adult animals, whereas large vessels were negative. The presence of SPARC in the retina was confirmed by Western blotting of retinal extracts. These data indicate that SPARC originating from both neurons and glia of the inner retina may be an important modulator of retinal angiogenesis. The increased expression of SPARC in adult relative to newborn retinal tissue also indicates that SPARC has an ongoing role in the maintainance of retinal functions.
SPARC (secreted protein acidic and rich in cysteine), also termed osteonectin, is a highly conserved extracellular glycoprotein. Widespread expression of SPARC in tissues undergoing developmental or injury-related remodeling and morphogenesis has been reported (Sage et al. 1986,1989; Sage and Bornstein 1991; Lane and Sage 1994). Unlike adhesive extracellular proteins such as fibronectin, laminin, vitronectin, or fibrillar collagens, SPARC belongs to a group of matricellular proteins defined as secreted components that do not serve structural roles but interact with growth factors, specific components of the extracellular matrix, and various cell surface receptors (Bornstein 1995). SPARC exerts a counteradhesive activity that modulates cell shape through interference with molecules that support cell adhesion. This counteradhesive function allows cell movement or shape changes required for development or remodeling. Microinjection of anti-SPARC antibodies into the blastocoel of Xenopus embryos resulted in deformed eyes and embryonic axes (Purcell et al. 1993), and in the nematode Caenorhabditis elegans altered morphology and viability have also been shown to be coincident with inappropriate expression of SPARC (Schwarzbauer and Spencer 1993). These functional studies in embryos and the widespread expression of SPARC during development (Holland et al. 1987; Sage et al. 1989; Mundlos et al. 1992) indicate that SPARC plays an important (but ill-defined) role in the development of many tissues, including the eye. In contrast, the expression of SPARC in adult tissues is usually more limited (Lane and Sage 1994), e.g., SPARC has been identified principally in tumors (Schulz et al. 1988; Porter et al. 1995) and in tissues undergoing re-modeling (Sage et al. 1989; Raines et al. 1992; Reed et al. 1993). Because SPARC mRNA and protein appear to be expressed at significant levels in normal adult brain (Mendis et al. 1994a; 1995), SPARC might have an ongoing role in the adult central nervous system.
Two SPARC family-related genes have recently been identified. SC1 was isolated from a rat brain library (Johnston et al. 1990) and QR-1 from embryonic quail retina (Guermah et al. 1991). SC1 is highly enriched in neural tissue and is localized within neurons in the central nervous system in both developing and adult animals (McKinnon and Margolskee 1996; Mendis et al. 1996a; b; Soderling et al. 1997). QR-1 is a retina-specific gene and encodes an extracellular matrix protein that plays an important role during retinal differentiation (Casado et al. 1996). To our knowledge, there is no detailed report of SPARC expression in retina. We therefore investigated the immunocytochemical localization of SPARC in the normal bovine retina, a widely accepted model for studies of microvascular functions in vitro.
Materials and Methods
Animals and Preparation of Tissue
Bovine eyes from four normal adult cows (4–8 years old) and two calves (3 days old) were obtained from a local slaughterhouse and were transported to the laboratory on ice within 2 hr after death. The anterior segment, lens, and vitreous were removed and the posterior eyecup was fixed by immersion in methyl Carnoy solution (60% methanol, 30% chloroform, and 10% glacial acidic acid) overnight at 4C. Small pieces of retinas were dehydrated in ethanol solutions (70, 80, 95, and 100%) and were embedded in paraffin.
Retinal tissue for immunoblotting was dissected from fresh adult eyes, rinsed with sterile PBS containing a complete protease inhibitor cocktail (Boehringer Mannheim Biochemica; Mannheim, Germany), and homogenized in lysis buffer [0.5% NP-40, 10% glycerol, 1 mM DTT, 2.5 mM EGTA, 5 mM EDTA, 150 mM NaC1, 50 mM Hepes (pH 7.4), 1 mM NaF, and 0.5 mM Na-vanadate] with complete protease inhibitor cocktail (same as above) with a Teflonglass homogenizer. After incubation for 20 min on ice, the homogenized sample was centrifuged at 13,000 × g for 10 min at 4C. The protein concentration of the supernatant was assayed with the Bradford reagent (Pierce; Rockford, IL), and the supernatant was aliquotted and stored at −70C.
Immunohistochemistry
Serial 5-μm-thick paraffin sections were cut in transverse or tangential planes, deparaffinized, and rehydrated through a series of ethanol solutions before washing in PBS. Nonspecific binding sites were blocked by incubation in 10% normal goat serum in PBS for 2 hr at room temperature (RT). Sections were immunostained with mouse anti-bovine osteonectin monoclonal antibody (MAb) (mouse 1gG1, 1:250 dilution of a 3.7 mg/ml stock solution; Haematologic Technologies, Essex Junction, VT) for 2 hr at RT in a humid chamber with agitation. Adjacent serial sections or the same section (for double labeling) was incubated with polyclonal rabbit anti-human glial fibrillary acidic protein (GFAP; 1:1000 dilution of an 8.7 mg/ml stock solution; BioMakor, Rehovot, Israel). Negative controls included replacement of primary antibodies by normal mouse IgG1 or PBS. Sections were washed three times for 10 min each in PBS and were incubated in fluorescein isothiocyanate-conjugated goat anti-rabbit IgG or Texas red-conjugated goat anti-mouse IgG (1:200) for 1 hr at RT. After two washes with PBS, cell nuclei were counterstained for 30 min with Hoechst 33258 fluorochrome (0.1 μg/ml in distilled water; Flow Laboratories, McLean, VA). After a brief rinse with PBS, sections were mounted in 80% glycerol and were photographed with a Nikon fluorescence microscope with wavelength-specific filters. Osteonectin antibody binding was also visualized with a mouse IgG-avidin-biotinylated peroxidase complex (Vector Labs; Burlingame, CA) developed with diaminobenzidine and hydrogen peroxide.
Whole-mount Immunohistochemistry
A portion of the adult bovine retina extending from disc to periphery was removed from the eyecup without contamination of pigment epithelium or other tissues. The retina was washed with PBS and placed in 30% sucrose to freeze on dry ice. Subsequently, tissue was thawed at RT and rinsed with PBS several times after removal of sucrose. The tissue was then fixed for 1 hr in methanol at RT, washed in PBS, and incubated with 5% normal goat serum with 0.2% Triton X-100 overnight at 4C. Primary antibodies (mouse anti-osteonectin and rabbit anti-GFAP, at the same concentration as above) were incubated with the tissue for 2 days at 4C. After a thorough washing (24 hr), secondary antibodies (fluorescein isothiocyanate-conjugated goat anti-rabbit IgG or Texas red-conjugated goat anti-mouse IgG) were incubated with the tissue for 5 hr on a shaker at RT. After an overnight wash in PBS, a glass slide was inserted below the retina tissue and a brush was used to move and spread the retina flat on the slide (nerve fiber layer face up). Excess solution was drained off and the retina was coverslipped with glycerol.
Western Blot
Retinal tissue was assayed for SPARC by SDS-PAGE, followed by immunoblotting. The frozen aliquots were thawed, solubilized in SDS-PAGE sample buffer (Laemmli 1970), and heated for 5 min at 100C with 50 mM dithiothreitol (DTT). Proteins (40 and 80 μg/lane) were analyzed by SDS-PAGE on 4/10% polyacrylamide minigels. Proteins were fractionated and were electrotransfered to nitrocellulose membranes (0.45-μm pore size). The blots were stained with 0.1% amido black (in 10% acetic acid and 20% methanol) and were photographed. Subsequently, the blot was blocked overnight at 4C with 1% nonfat milk in PBS containing 0.05% Tween 20 and was incubated sequentially with mouse anti-bovine osteonectin (1:1000) (2 hr) and rabbit anti-mouse IgG. The bound protein-immunoglobulin complexes were detected with 125I-labeled protein A at 0.5 μCi/ml (New England Nuclear; Boston, MA) and were visualized by autoradiography.
Results
In adult bovine retina, staining with anti-SPARC IgG was restricted to the ganglion cell layer (GCL), nerve fiber layer (NFL), and to some of the retinal capillaries (Figures 1B and 1C). Control retinas showed no staining in the corresponding areas (Figure 1A). Although SPARC is believed to function in the vasculature (Sage and Vernon 1994), there were only low levels of immunoreactivity in some of the capillaries in the inner capillary beds within the NFL and GCL and in the two capillary beds along the inner and outer borders of the inner nuclear layer (Figure1C and inset). Medium and large blood vessels in the inner retina did not show any immunoreactivity with the MAb. In contrast, many cell bodies in the GCL and all axon bundles in the NFL were stained for SPARC (Figures 1B and 1C).
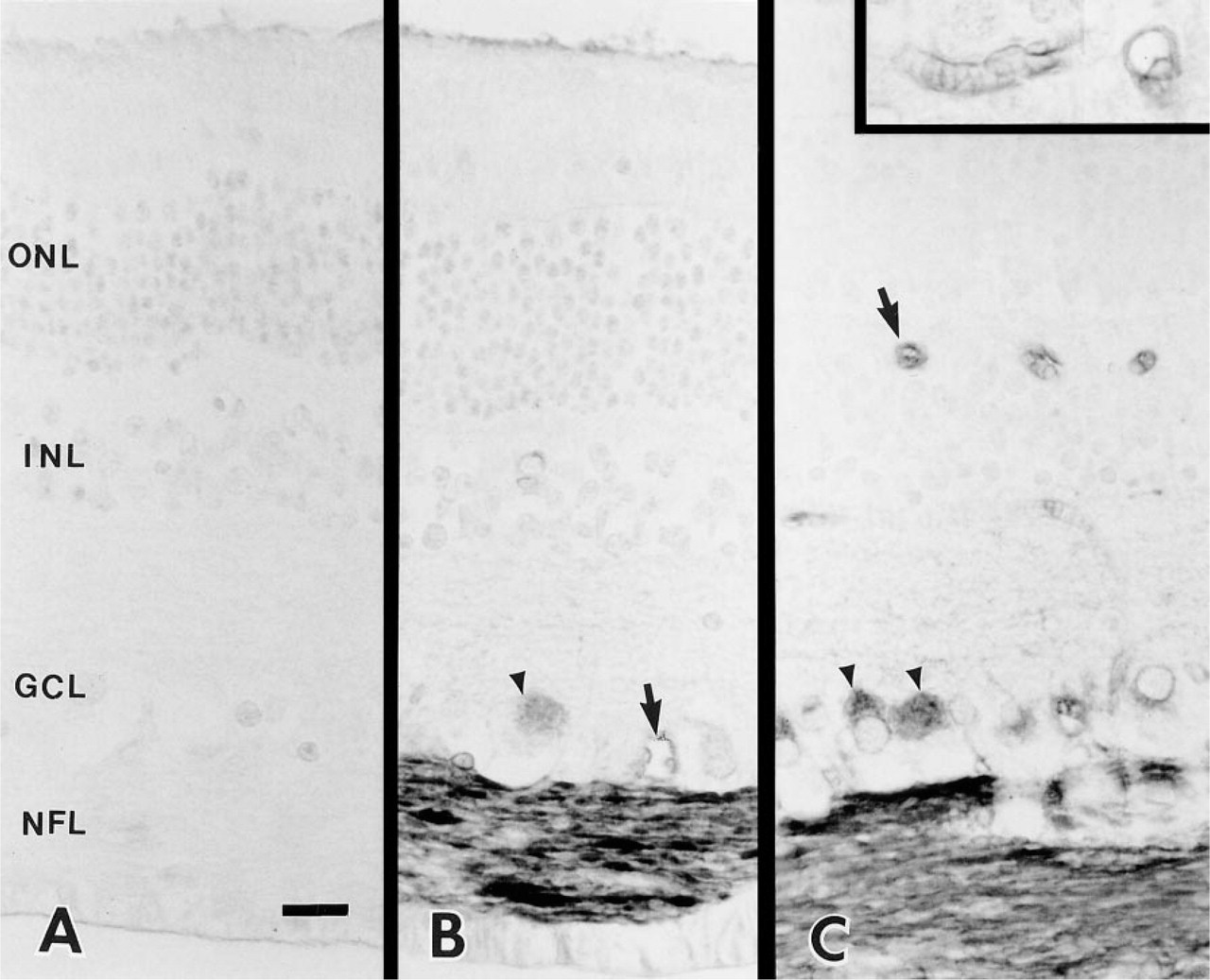
Immunohistochemical localization of SPARC protein in the adult bovine retina. (A) Control section of bovine retina exposed to mouse IgG1 instead of primary anti-SPARC IgG. (B,C) Longitudinal sections of bovine retina exposed to anti-SPARC IgG. Immunohistochemistry was performed with an avidin-biotin-peroxidase method (A-C). Axon bundles in the nerve fiber layer and associated ganglion cell bodies (arrowheads) were specifically labeled by the antibody. Some of the capillaries in the inner retina were also reactive (arrows). (Inset) Higher magnification of capillaries labeled by anti-SPARC IgG. ONL, outer nuclear layer; INL, inner nuclear layer; GCL, ganglion cell layer; NFL, nerve fiber layer. Bar = 10 μm.
Hoechst 33258 (the DNA binding fluorochrome bis-trihydrochloride) binds to nuclear chromatin of all retinal nuclei. Figure 2A is a double exposure of a section double labeled with Hoechst 33258 and anti-SPARC IgG. All the nuclei of the cells of the outer nuclear layer, inner nuclear layer, and GCL were labeled by Hoechst 33258. The bovine GCL contains large ganglion cell bodies (arrowheads) and neurons which could be either smaller ganglion cells or displaced amacrine cells (small arrow). Astrocyte cell bodies (large arrow) lie within the axon bundles in the NFL. In all animals, SPARC protein was detected in large ganglion cell somas (Figure 2A, arrowheads; and Figure 2B). Figure 2B shows a single large ganglion cell in which the cytoplasm and a large primary dendrite were heavily labeled by the antibody, but the nucleus was devoid of reactivity. Without a specific amacrine cell marker, it is difficult to distinguish displaced amacrine cells from small ganglion cells, but none of the smaller neurons was labeled by the anti-SPARC antibody. High levels of SPARC protein were also present in most of the ganglion cell axons within the NFL throughout the retina (Figures 1B, 1C, 2A, and 2B). In Figure 2B, two small axon bundles are cut in cross-section and show large and small labeled axons.
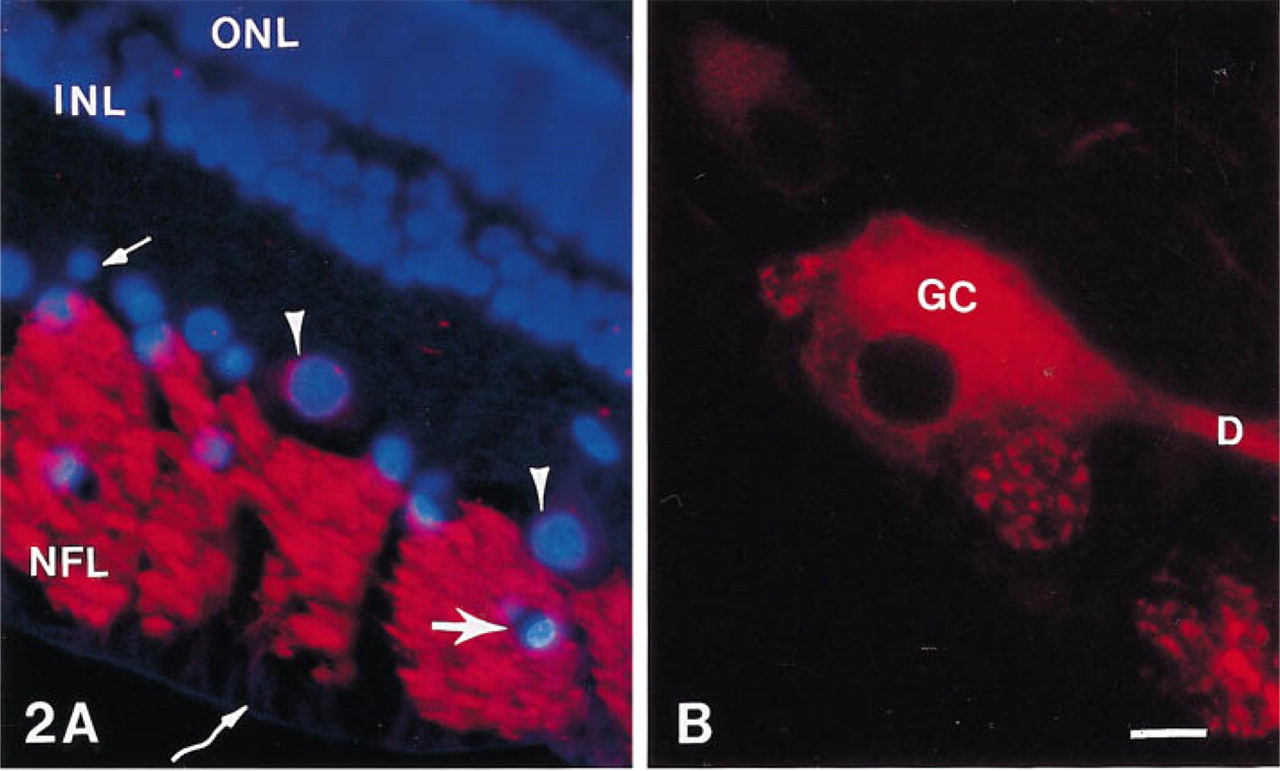
Epifluorescence micrographs of bovine retina illustrating the immunolabeling patterns revealed with anti-SPARC IgG. (
Muller cells run perpendicularly through all retinal layers, with end-feet forming the inner border of the retina. These cells showed no immunoreactivity with the anti-SPARC antibody (Figures 2A, curved arrow, and Figure 1B), but were reactive with anti-GFAP antibody (data not shown). The other large cell population in the retina that exhibited SPARC immunoreactivity was the astrocyte glia. Astrocyte cell bodies are confined to the GCL and NFL of the bovine retina. Their long processes surround ganglion cell axons and extend within and along the NFL axon bundles. Figure 3A shows a tangential section of a calf retina through an NFL axon bundle that was double labeled for Hoechst 33258 and anti-SPARC IgG. Figure 3B shows the same bundle labeled for anti-GFAP IgG. All of the cell bodies except the small vessels in Figure 3A are astrocytes because they contain GFAP (Figure 3B). These astrocytes are labeled for SPARC (Figure 3A, arrowheads). A single astrocyte cell body shown at higher power contains SPARC (Figure 3C, arrowhead) and GFAP (Figure 3D, arrowhead). In comparision to the strong staining reaction of ganglion axons with the anti-SPARC antibody, the labeling of astrocyte cell soma and processes was relatively weak. Because the thin astrocyte processes wrap around the axons, which contain high levels of SPARC, it is difficult to identify SPARC in the astrocyte processes in sections.
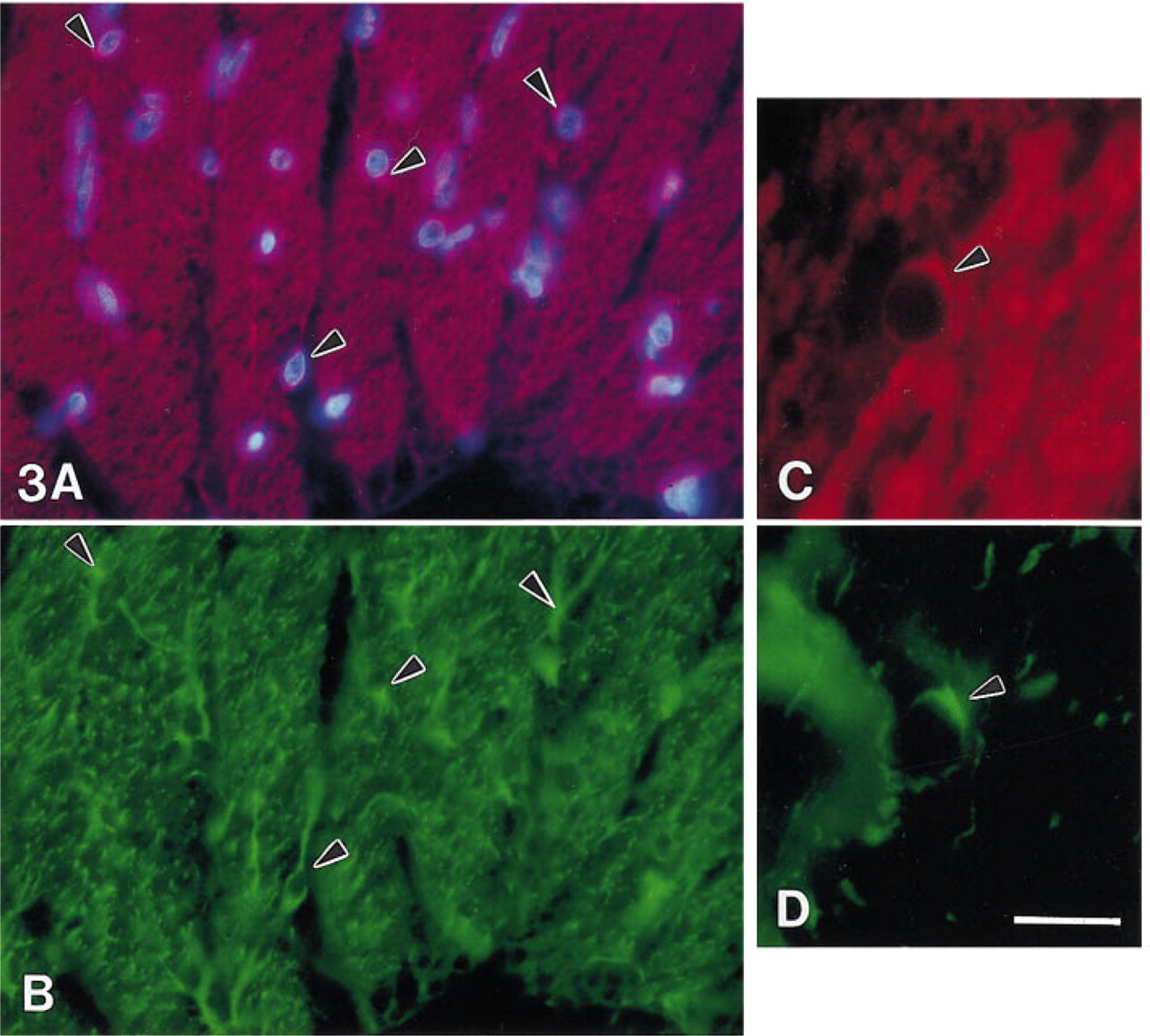
Double antibody staining of SPARC and GFAP in the same section (
Whole-mount immunohistochemistry was performed to provide a three-dimensional view of the NFL axon bundles. Double labeling with the focal plane at the level of the NFL showed that the axons were heavily stained by anti-SPARC IgG (Figure 4A) and that the astrocyte processes associated with the same axon bundles were labeled with the anti-GFAP antibody (Figure 4B). Data from the whole-mount further confirmed the observation in the retinal sections for axonal labeling with anti-SPARC IgG (Figure 1). By comparing the double labeling for SPARC and GFAP in the whole-mounts and by careful focusing through the axon bundles, we confirmed the staining pattern seen in sections, i.e., that the astrocyte cytoplasm and its attenuated long processes were stained with anti-SPARC IgG but at a significantly reduced level compared to that of the ganglion neurons.
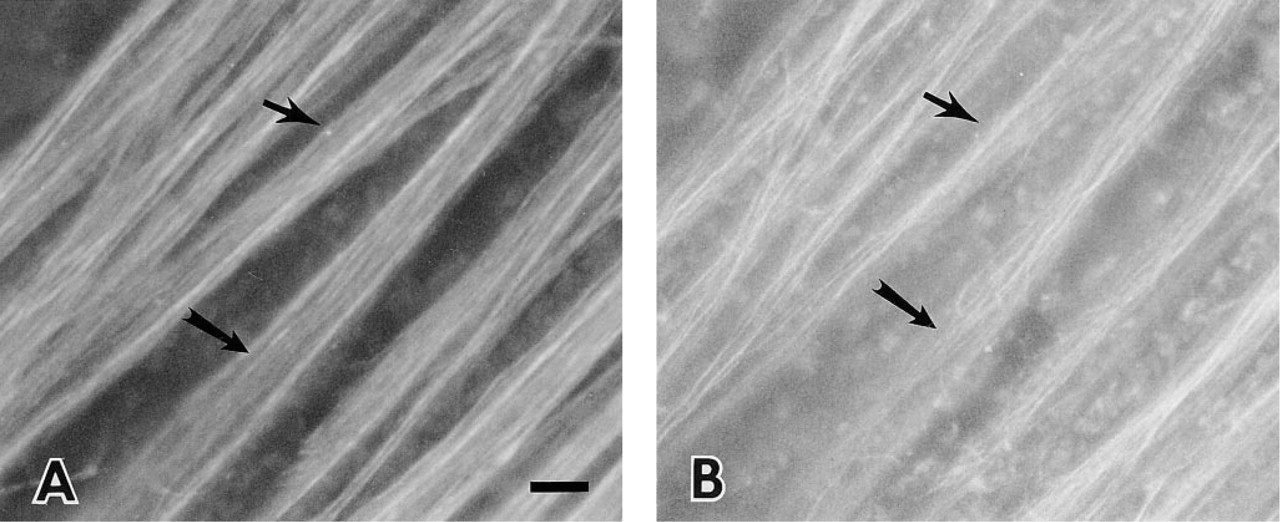
Whole-mount of adult bovine retina double stained with (A) anti-SPARC IgG and (B) anti-GFAP IgG. The nerve fiber layer was the superficial layer. Astrocyte processes are shown (arrows) along with axons that were labeled by anti-SPARC IgG (arrows). Bar = 10 μm.
The 3-day-old calf retinas showed a neuronal and glial staining pattern similar to that of the adult bovine retina, but the apparent level of SPARC protein in the calf retina was significantly lower. This result indicates that SPARC increases with age in the retina, although a change in epitope accessibility can not be excluded in young tissue. Figure 5 illustrates the difference in intensity of staining between bovine (Figure 5A) and calf retina (Figure 5B). The ganglion cell bodies (not seen in Figure 5B) were only lightly labeled compared to axon staining in calf retina. Compared to adult retina, only some of the axons were heavily labeled in the calf NFL, and these appeared to be large axons. The large vessels in both adult and calf retinas revealed no SPARC immunoreactivity (Figure 5, arrows).
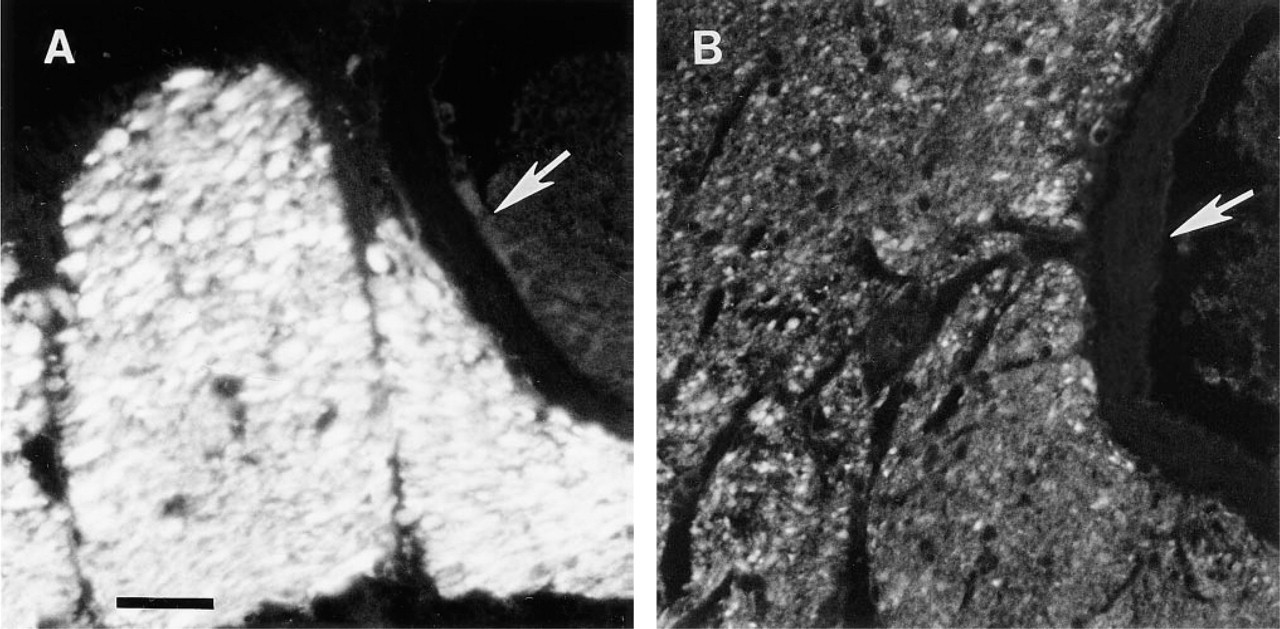
Expression of SPARC protein in adult and calf retina; tangential sections of nerve fiber layer. (A) Adult bovine; (B) 3-day-old calf. Large nerve bundles accompany a large vessel (arrows). Sections exposed to anti-SPARC IgG followed by Texas red-conjugated goat anti-mouse IgG. Bar = 10 μm.
The presence of SPARC protein in the adult retina was confirmed by SDS-PAGE of adult retinal extracts (Figure 6, Lanes 1 and 2) and by subsequent immuno-blotting with the anti-SPARC antibody (Figure 6, Lanes 3 and 4). The molecular weight of SPARC on SDS-PAGE is 43 kD (Sage et al. 1984). The anti-SPARC monoclonal antibody reacts with a sequence that is not common to other SPARC-like proteins, e.g., SC1 and QR1. Because it recognizes only one band at 43 kD, coincident with the molecular weight of SPARC, this blot excludes the possibility that the antibody also recognizes other SPARC-like proteins such as SC1 or QR1, which have greater apparent molecular weights by SDS-PAGE analysis.
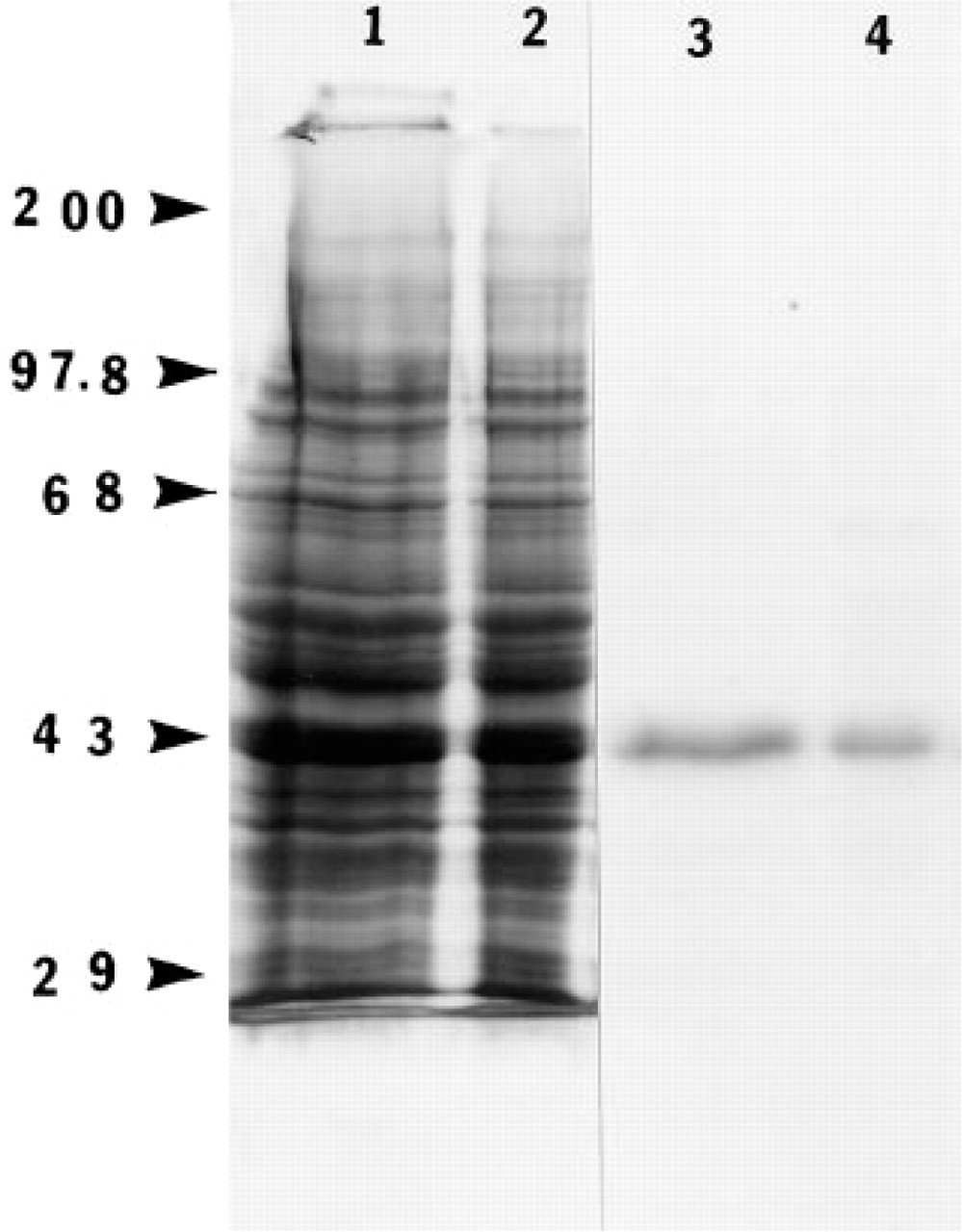
Immunoblotting analysis of SPARC protein in the bovine adult retina. Protein isolated from bovine adult retina were separated on polyacrylamide gels (4/10%) in the presence of DTT and were transferred electrophoretically to a nitrocellulose membrane. Lanes 1 and 2 represent aliquots of 80 μg and 40 μg, respectively, that were transferred to the blot and stained with amido black. Lanes 3 and 4 represent an autoradiograph of Lanes 1 and 2 after exposure to anti-SPARC IgG, followed by [125I]-protein A, respectively. Molecular weight standards (kD × 10-3) are indicated at left.
Discussion
Our data demonstrate for the first time that SPARC protein is present in bovine retina and is confined to the ganglion cell soma and axon, to astrocytes, and to some capillaries. Although the function of SPARC in the retina and elsewhere in the central nervous system remains obscure, our identification of the cell types that serve as the most likely source of the protein in the retina represents a first step in the characterization of SPARC as a regulatory protein in this tissue.
Immunohistochemical labeling patterns are known to vary under different conditions of fixation and tissue processing and in different species. We have found that methyl Carnoy solution is the best fixative for preservation of the antigenicity of SPARC. The MAb used in our study was produced against bovine bone osteonectin and reacts specifically with SPARC protein of bovine tissues. Our immunoblotting assay confirmed that the antibody recognizes specifically the 43-kD band, which is coincident with the molecular weight of SPARC on SDS-PAGE.
SPARC is a well-characterized matricellular glycoprotein that modulates cell-matrix interactions. Because SPARC can disrupt cell contacts with extracellular matrix and therefore alter cell shape, its major function has been referred to as counteradhesion, which leads to cell rounding. It also has been shown to modulate the expression and interaction of a variety of matrix components and growth factors (Lane and Sage 1994). The limited expression of SPARC in adult tissues, particularly in quiescent tissues and cells, and the increased expression in tissues undergoing repair or remodeling are consistent with these functions. However, in the central nervous system there appears to be a constitutive, relatively robust expression of SPARC. In fact, studies have reported low levels of SPARC protein and mRNA in the embryonic nervous system (Nomura et al. 1989; Bassuk et al. 1993) and significant levels of SPARC in the normal adult brain in regions enriched in synapses (Mendis et al. 1995). In the adult brain, some counteradhesive glycoproteins, such as SPARC and SC1, are associated with the extracellular matrix and are believed to regulate adhesive events at synaptic areas and to prevent further neurite outgrowth (Mendis et al. 1994b). We have demonstrated that SPARC is expressed predominantly in ganglion cell soma, and especially in axons, in the bovine retina. This distribution suggests an ongoing association with axon transport, axon growth, and maintenance of cell shape. In the retinal plexiform layers in which synapses are concentrated, SPARC was not detected; an active role for SPARC related to retinal synapses therefore tends to be excluded.
Because SPARC contains a follistatin domain capable of binding growth factors (Hemmati-Brivanlou and Melton 1994), modulation of growth factors and matrix components represent two possible roles for SPARC in retinal tissue. During retinal development or in the ischemic retina, astrocytes, Muller cells, and ganglion cells have been shown to synthesize vascular endothelial growth factor (VEGF) in response to hypoxia (Stone et al. 1995; Shima et al. 1996). The Muller cells and ganglion cells also express transforming growth factor-β (Anderson et al. 1995). It is possible that the SPARC produced in the GCL/NFL could interact with these growth factors and regulate retinal angiogenesis. This hypothesis is supported by studies in vitro showing that SPARC regulates endothelial cell proliferation in part through an interaction with VEGF (C. Kupprion, K. Motamed, and E. H. Sage, submitted for publication). Retinal neovascularization is regulated by various molecular cues, such as growth factors and extracellular matrix components, and by environmental factors, such as hypoxia. Nothing is known concerning the regulation of SPARC itself in the retina and its potential interaction with other molecules in the retina. However, the localization of SPARC and VEGF in the retina and the close relation to the superficial layer of retinal vessels provide strong evidence that SPARC could directly or indirectly (e.g., through VEGF or other factors) regulate retinal angiogenesis. In addition, some of the capillaries (endothelial cells) were labeled by anti-SPARC IgG, although large vessels in the inner retina were unreactive. Because not all the capillaries were stained with the anti-SPARC anti-body, it is likely that the amounts of SPARC in some of the vessels were too minimal to be detected by immunochemistry. In addition, the attenuated endothelial cell cytoplasm is often difficult to visualize by immunocytochemical methods (Lane and Sage 1994).
Our identification of SPARC in the adult and newborn bovine retina, together with the demonstration of SPARC family proteins SC1 and QR1 in the central nervous system (Casado et al. 1996; Mendis et al. 1996b), suggests that SPARC and SPARC-like proteins are important not only in development of the retina but also in maintenance of its normal functions in the mature retina. More detailed study will be required to elucidate the precise functions of these proteins in both the developing and the mature retina.
Footnotes
Acknowledgements
We thank Dr Yi Li for assistance in this project. QY was supported by PHS NRSA T32 GM07270 from NIGMS and T32 EY07031 from NEI. This work was also supported by GM40711 and EY04536.
