Abstract
Mast cells are difficult to purify from heterogeneous cell populations and to preserve, especially for pre-embedding immunostaining at the ultrastructural level. We have developed a technique that permits the isolation of a pure population of mast cells suitable for immunocytochemical studies. A rat mast cell-specific monoclonal antibody (MAb AA4) conjugated to tosylactivated Dynabeads 450 was used to immunomagnetically separate mast cells from rat bone marrow and peritoneal cell suspensions. Approximately 85% of the mast cells were recovered in the positive population that comprised virtually pure mast cells. After microwave fixation, morphological examination showed that the cells were intact and retained their ultrastructural detail. Mast cells in all stages of maturation were immunolabeled with a panel of antibodies after immunomagnetic separation. The combination of immunomagnetic separation followed by immunostaining should prove useful for the study of mast cell maturation and for the characterization of other specific cell types that are present in tissues in only limited numbers.
Keywords
T
The present study extends the use of one of these mast cell-specific antibodies, MAb AA4, to the separation of mast cells in various stages of maturation from a mixed population of cells. Mast cells comprise only a small proportion of the cells in rat bone marrow and are present in all stages of maturation. Although the majority of peritoneal mast cells in the adult rat are mature, repopulation of the peritoneal cavity following distilled water injection is a commonly used model for mast cell maturation (Fawcett 1955; Mendonça et al. 1986). The heterogeneity of the maturing mast cells makes them impossible to separate from other cells on the basis of their density.
Previously, a panel of mouse MAbs was raised against the cell surface of RBL-2H3 cells, a rat basophilic leukemia cell line (Basciano et al. 1986; Kitani et al. 1991). Many of these antibodies, including MAb AA4 (Oliver et al. 1992; Jamur et al. 1995; Faraco et al. 1997) recognize cell surface antigens that are unique to rat mast cells. The present study utilizes MAb AA4 for immunomagnetic separation of rat bone marrow-derived and peritoneal mast cells. This antibody binds approximately 5 × 106 molecules on the surface of RBL-2H3 cells, about 15 times the number of IgE receptors on these cells, without inducing histamine release (Basciano et al. 1986). It binds close to the high-affinity IgE receptor, and inhibits IgE binding as well as receptor-mediated histamine release. MAb AA4, an IgGγ1 isotype, recognizes novel α-galactosyl derivatives of the ganglioside G
Since the introduction of immunomagnetic separation to deplete malignant neuroblastoma cells from bone marrow before autologous transplantation (Treleaven et al. 1984), this technique has proved to be a reliable method of separating target cells from a heterogeneous cell population. Immunomagnetic separation has been used to isolate a wide variety of cell types, including biliary epithelial cells (Joplin et al. 1989), epithelial cells from solid tumors (Yaremko et al. 1996), trophoblasts from peripheral blood (Durrant et al. 1994), CD4+ and CD8+ T-cells from human blood (Stanciu et al. 1996), natural killer cells from spleen (Ellison and Gartner 1995), macrophages from synovial tissue (Kossovsky et al. 1993), and CD34+ progenitor cells from bone marrow (Kirshenbaum et al. 1991).
In this study, a method was developed that combines immunomagnetic separation of rat bone marrow-derived and peritoneal mast cells with subsequent immunoelectron microscopic characterization of the isolated cells. This method facilitates the isolation and characterization of mast cells in all stages of maturation.
Materials and Methods
Antibodies
Monoclonal antibodies AA4 (Guo et al. 1989), BC4 (Basciano et al. 1986), and AD1 (Kitani et al. 1991) were raised in our laboratory in mice against the surface of RBL-2H3 cells, a rat basophilic leukemia cell line. They were purified from ascites fluid by ammonium sulfate precipitation, followed either by ion exchange chromatography on a DE-52 column, MAbs BC4 and AD1, or HPLC, MAb AA4. An affinity-purified mouse anti-rat IgE MAb was purchased from Biosource International (Camarillo, CA). All the antibodies used are IgG71.
Coupling of Antibody to Magnetic Beads
Equal volumes of tosylactivated Dynabeads M-450 (4 × 108 beads/ml, 30 mg beads/ml) (Dynal; Lake Success, NY) and MAb AA4 (150 μg/ml dissolved in 0.05 M borate buffer, pH 9.5) (antibody/bead ratio 75 μg/15 mg) were mixed. For most preparations, 2 ml of tosylactivated Dynabeads was mixed with 2 ml of antibody solution in 15-ml Falcon 2095 tubes (Becton Dickinson; Lincoln Park, NY). The solution was incubated for 24 hr at 22C with slow end-over-end rotation. After incubation the Dynabeads were collected with the Magnetic Particle Concentrator (MPC; Dynal). The tube was placed in the MPC for 5 min to allow the beads to be attracted to the magnet in the MPC and then the supernatant was discarded while the tube was in the MPC. The coated beads were then washed, using the MPC, three times for 10 min each, once for 30 min, and then overnight at 4C with 5 ml PBS containing 0.1% BSA. The coated beads were collected with the MPC, the supernatant discarded, and the beads resuspended in PBS + 0.1% BSA to a concentration of 30 mg/ml and stored at 4C until use. Normal mouse IgG (Jackson ImmunoResearch; West Grove PA) was coupled to tosylactivated Dynabeads using the protocol for cell separation applications as recommended by the manufacturer.
Cells
Bone marrow from both femurs of young (150 g) male and female Wistar rats was removed by flushing the marrow cavity with Dulbecco's PBS containing 2% bovine serum albumin (BSA), heparin (1000 U/ml) (Produtos Roche Quimicas e Farmaceuticos SA; Rio de Janeiro, Brazil), and DNase (1000 U/ml) (Sigma Chemical; St Louis, MO), and the cells from each animal were pooled. The bone marrow was dissociated by aspiration with a Pasteur pipette. The cells were then rinsed five times by centrifugation (1000 rpm; 27 x g) in PBS + 2% BSA. Peritoneal mast cells were collected by lavage of the peritoneal cavity from adult (250 g) Wistar rats with approximately 15 ml of PBS. The peritoneal cells were washed twice by centrifugation in PBS + 2% BSA.
Cell Separation
The cells were incubated with various concentrations of coated beads (3 beads/target cell being optimal) in PBS containing 2% BSA for 5–10 min in Falcon 2095 tubes at room temperature (RT) with slow end-over-end rotation. After incubation the mast cells were isolated by washing four times with PBS containing 2% BSA and once in PBS using the MPC.
Cell Counts
The number of mast cells in the total cell population from the peritoneal cavity was determined before immunomagnetic isolation and the number of mast cells recovered after immunomagnetic isolation was determined by counting the number of metachromatic cells after toluidine blue staining (4% formaldehyde, 0.1% toluidine blue, and 1% acetic acid, pH 3.5) using a Neubauer camera.
Because many of the mast cells in the bone marrow are immature and are not metachromatic with toluidine blue, the percentage of mast cells present in the bone marrow was determined before isolation by counting, in a Neubauer Camera, the positive and negative cells after immunostaining with either MAb AA4 or anti-IgE. After immunomagnetic isolation, the number of mast cells was determined by counting the number of cells that were either free or attached to MAb AA4-conjugated beads with and without immunostaining.
Light Microscopy
Cells were placed on coverslips coated with CELL TAK (Becton Dickinson Labware; Collaborative Biomedical Products, Bedford, MA), fixed, and stained with toluidine blue for 10 min.
Electron Microscopy
Microwave Fixation. Cells were suspended in 5 ml of fixative containing 2% formaldehyde, 0.05% glutaraldehyde, 0.025% CaCl2 in 0.1 M cacodylate buffer, pH 7.4, and microwave-irradiated for 4 sec at 100% power (Jamur et al. 1995). Immediately after irradiation, 10 ml of PBS was added to the suspension and the cells were centrifuged for 2 min at 1000 rpm. The cells were then rinsed twice in PBS, PBS containing 0.1 M glycine, PBS, and then immunostained.
Immunostaining. After microwave fixation the cells were incubated with primary antibody (MAb BC4 0.5 μg/ml; AD1 10 μg/ml) diluted in PBS +1% BSA for 1 hr at RT. After incubation the cells were rinsed sequentially in PBS + 1% BSA, PBS, PBS +1% BSA, and then incubated for 1 hr with donkey anti-mouse F(ab)′2 conjugated to horseradish peroxidase diluted (25 μg/ml final concentration) in PBS + 1% BSA. The cells were then rinsed in PBS +1% BSA, PBS and 0.1 M cacodylate buffer (pH 7.4) and immersed in diaminobenzidine (DAB) (Kirkegard & Perry; Gaithersburg, MD) incubation medium (12.5 ml cacodylate buffer, 12.5 mg DAB, and 250 μl 1% H2O2) for 30 min at RT. The cells were then rinsed 10 times in cacodylate buffer. For light microscopy, endogenous peroxidase was blocked by incubating the samples for 30 min in 3% H2O2 in methanol at RT. For electron microscopy, some samples were packed by centrifugation in 2% agar. Cells incubated without primary antibody, or normal mouse IgG (5 μg/ml; Jackson ImmunoResearch) in place of primary antibody served as controls. No immunostaining was observed in any of the control preparations. Furthermore, no cells other than mast cells were stained in peritoneal or bone marrow cell suspensions.
Sample Processing
Cells were postfixed in 2% OsO4 for 1 hr at RT, rinsed in distilled water, dehydrated through a graded series of ethanols, rinsed in acetone, and embedded in Embed 812 (EM Sciences; Fort Washington, PA). Thin sections were cut with a diamond knife and stained for 10 minutes each in Reynolds' lead citrate (1963) and uranyl acetate.
Results
Using a mast cell-specific antibody, MAb AA4, conjugated to magnetic beads, it was possible to isolate pure populations of bone marrow derived and peritoneal mast cells in all stages of maturation (Figures 1–3). When the antibody-conjugated beads were mixed with bone marrow or peritoneal cell suspensions, the beads bound specifically to the mast cells (Figures 1A, 1B, and 2). If uncoated beads or beads coated with normal mouse IgG were used instead of MAb AA4–conjugated beads, no cells were bound. After immunomagnetic separation, two populations of cells were obtained. The positive population, composed of MAb AA4-conjugated magnetic beads and attached cells, contained over 99% mast cells (Figures 1C and 1D; Table 1). The negative population, the total cell suspension minus the cells removed by the immunomagnetic beads, contained virtually no mast cells (Figures 1E and 1F). The method was highly efficient, and over 85% of the mast cells were recovered from the initial cell suspensions from either the bone marrow or the peritoneal cavity. The efficiency of recovery was independent of the number of mast cells in the starting material but was dependent on the ratio of magnetic beads to mast cells. Although mast cells represent only 2.4 ± 0.6% of the cells in the bone marrow and 25 ± 4.5% of the cells in peritoneal washings (Table 2), both were recovered with equal efficiency and purity using a target bead:cell ratio of 3:1 (Table 1). If a lower number of beads, 2:1, was used, the recovery decreased significantly. When the bead:cell ratio was increased to 4:1, the mast cell recovery was not increased, but more non-mast cells were trapped in cell-bead aggregates.
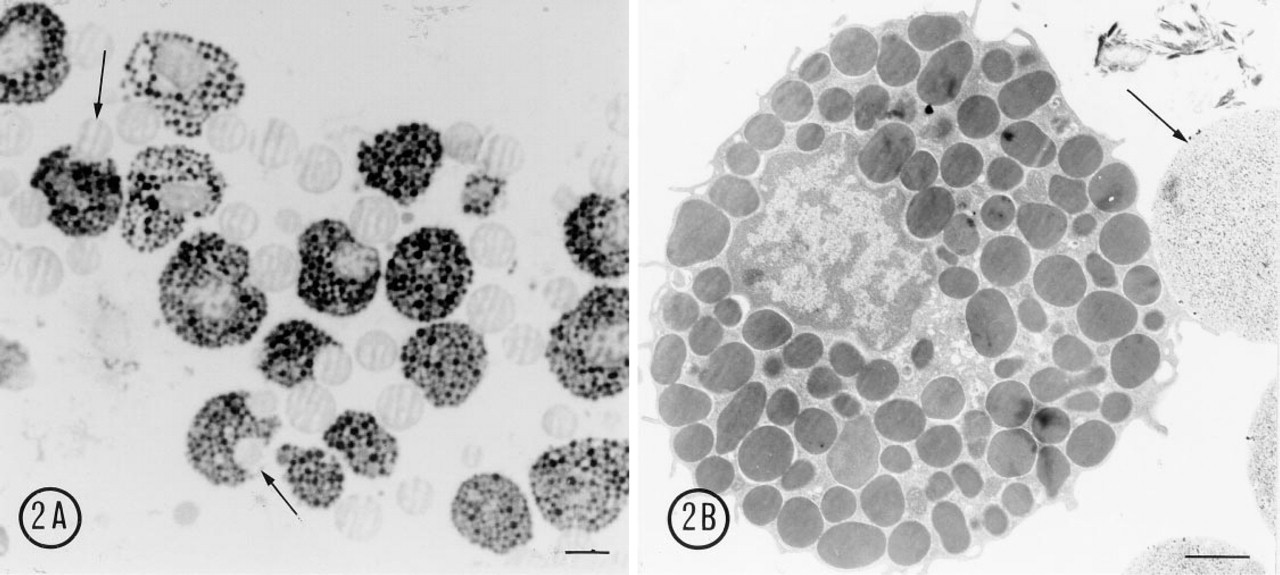
Immunomagnetic separation with MAb AA4-coated beads did not induce degranulation (Figure 2), even though the surface of the cells was bound to the beads and in some cases, if fixation was delayed, the mast cells partially engulfed the beads (Figure 2A). By electron microscopy, the isolated mast cells showed no ultrastructural alterations. The cytoplasmic granules were intact and electron dense (Figure 2B).
Another advantage of immunomagnetic isolation was the ability to isolate mast cells in all stages of maturation and to characterize the isolated cells by immunomicroscopy. Three distinct stages of mast cell maturation could be distinguished: mature, immature, and very immature. The mature mast cells had a centrally located nucleus, a cytoplasm filled with dense granules, and few organelles (Figures 3A and 3B). The immature mast cells had a concave nucleus, a well-developed Golgi apparatus, and variable numbers of electron-dense cytoplasmic granules (Figures 3C and 3D). The very immature mast cells had a large lobated nucleus, virtually no cytoplasmic granules, and few organelles other than mitochondria (Figures 3E and 3F). To confirm that the cells isolated with the MAb AA4 beads were indeed mast cells, they were immunolabeled with antibodies against mast cell surface components and IgE. MAb AD1 is directed against a rat mast cell-specific variant of CD63, a 50–60-kD glycoprotein present on the granule membrane and surface of mast cells. MAb BC4 is specific for the α-subunit of the high-affinity IgE receptor, and the anti-IgE reacts with the IgE bound to the FcεRI receptor on the cells. Because all of antibodies used were directed against glutaraldehyde-sensitive antigens, the mast cells were fixed by microwave fixation after immunomagnetic isolation. All of the cells isolated with MAb AA4-coated magnetic beads from either bone marrow or the peritoneal cavity immunolabeled with all three antibodies (Figure 3) regardless of their stage of maturation, thus confirming their identity as mast cells.
(A) Light microscopy, 1-μm semithin section, toluidine blue staining. After immunomagnetic separation with MAb AA4-conjugated magnetic beads, the mature peritoneal mast cells remain intact. Some cells have partially engulfed the magnetic beads (arrows). Bar = 10 μm. (
Discussion
The feasibility of immunomagnetic separation of lung mast cells was previously demonstrated (Okayama et al. 1994). However, the present study is the first to immunomagnetically isolate mast cells in all stages of maturation and to subsequently characterize them by immunoelectron microscopy. This investigation has shown that immunomagnetic separation is a rapid, gentle method for purification of a small population of difficult to handle cells. Mature mast cells are readily induced to degranulate and are often difficult to preserve morphologically (Lawson et al. 1977; Chen and Enerback 1994). This technique readily permits isolation and subsequent immunoelectron microscopic characterization of these cells.
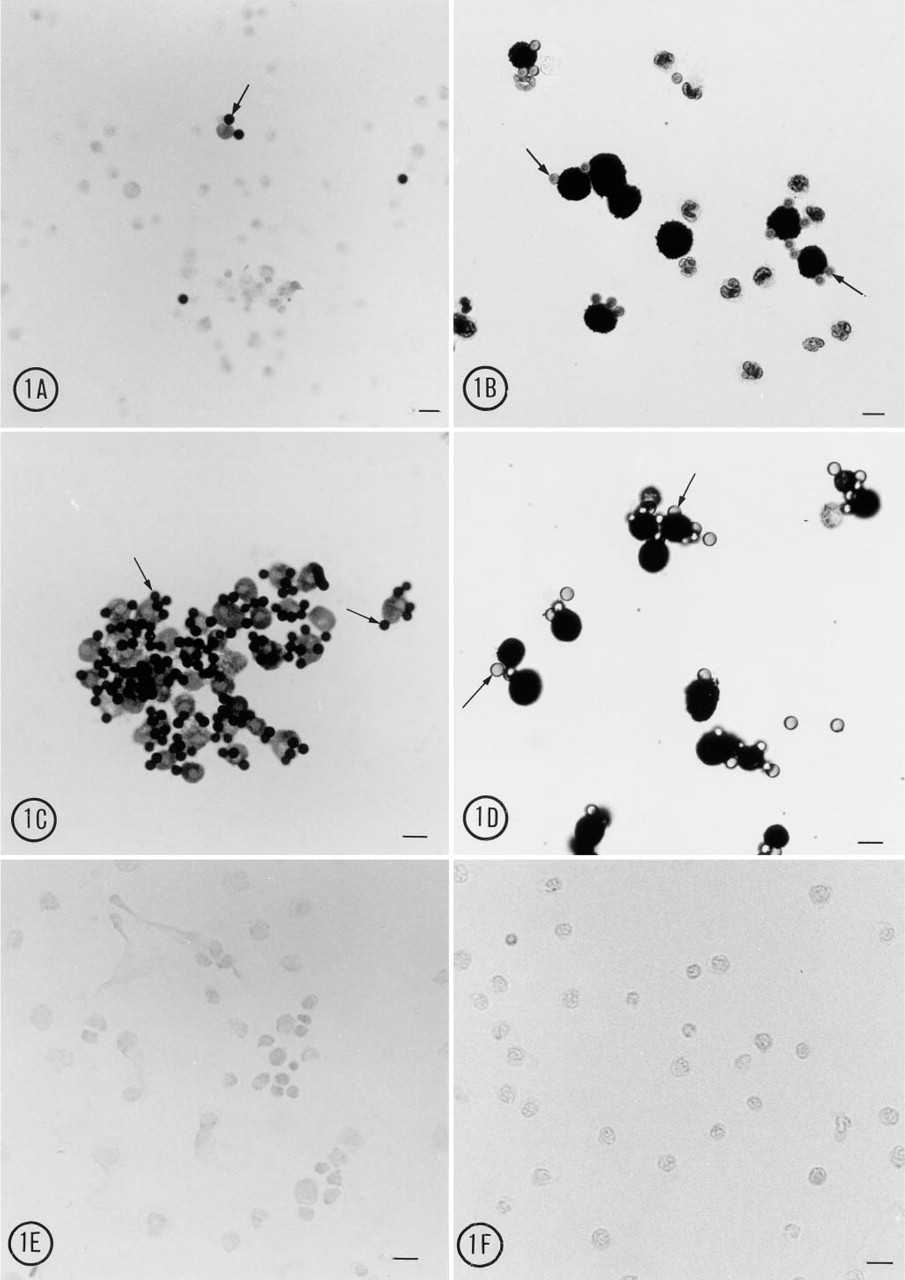
Light microscopy, toluidine blue staining. In the total cell population before immunomagnetic isolation, the MAb AA4-conjugated magnetic beads (arrows) are attached only to small immature mast cells and larger mature mast cells. (
Although the technique is simple and rapid, several steps in the method require particular attention. For immunomagnetic isolation of mast cells, tosylactivated magnetic beads were selected over anti-mouse IgG-coated beads available from Dynal for the conjugation of MAb AA4. Because mast cells have Fcγ receptors, it was of concern that using IgG-coated beads as the starting bead would increase nonspecific binding of the beads to the mast cell surface. In addition, with the tosylactivated beads the primary antibody is covalently bound to the magnetic beads, presumably resulting in a more stable preparation. The method presented here for conjugating the antibody to tosylactivated Dynabeads differs from the one currently recommended by Dynal. With MAb AA4 it was necessary to incubate the preparation at RT without additional BSA and to substitute overnight incubation in PBS + 0.1% BSA instead of Tris buffer to block free tosyl groups. With normal mouse IgG and other antibodies (unpublished observations), the coupling could be successfully performed at 37C in the presence of BSA and Tris could be used to block unreacted tosyl groups. Therefore, coupling conditions may need to be individualized for a given ligand. Addition of DNase to the solution for isolation of bone marrow cells was essential to reduce adhesion of the cells to each other. It was also critical to wash the mast cell population extensively after isolation to remove any contaminating cells that might be nonspecifically trapped with the mast cells. As with other secretory cells, it was necessary to perform the isolation procedure at RT rather than at 4C to avoid stimulating secretion by cold exposure during rotation.
Mast cell recovery after immunomagnetic isolation a
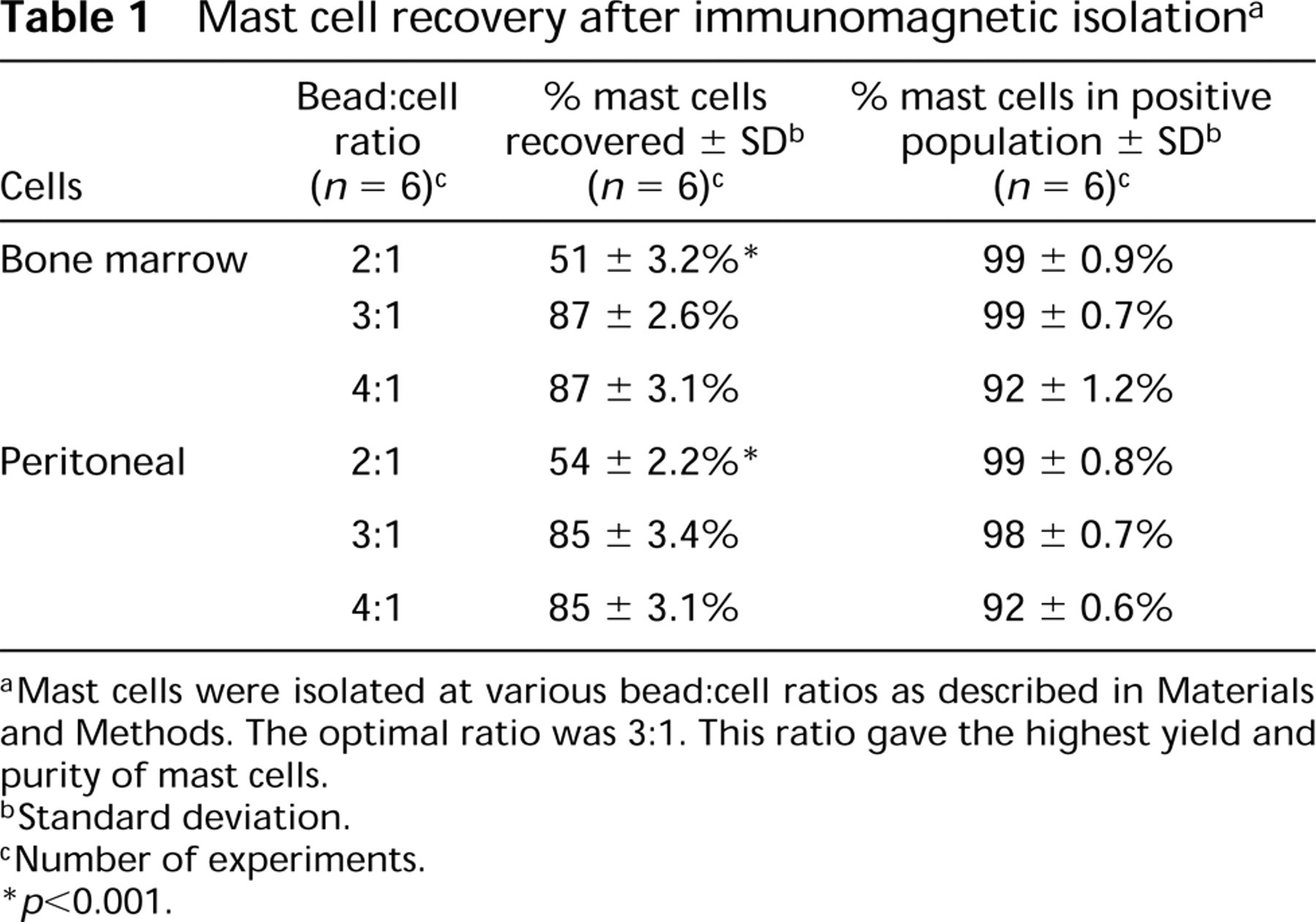
a Mast cells were isolated at various bead:cell ratios as described in Materials and Methods. The optimal ratio was 3:1. This ratio gave the highest yield and purity of mast cells.
b Standard deviation.
c Number of experiments.
∗ p > 0.001.
Most mast cell isolation procedures rely on cell density to purify mast cells. Immunomagnetic isolation appears to be superior to these other methods of isolating mast cells, especially immature mast cells. A major disadvantage of density-based purification is that mast cells can sediment with other cell types of similar density (Lagunoff 1972). This is especially true of immature mast cells (WoldeMussie and Beaven 1986), making their purification difficult if not impossible. Another disadvantage of density gradient techniques is the time that may be required to run a gradient separation. For example, to purify rat intestinal mast cells it was necessary to employ sequential Percoll and BSA gradients (Befus et al. 1986). Although the recovery (84%) and purity (<95%) were excellent, the method required 4 hr for the BSA gradient alone. Immunomagnetic separation can be accomplished much more readily.
Mast cell numbers a

a The total number of cells per animal and the number of mast cells obtained from the bone marrow and peritoneal cavity per animal before immunomagnetic isolation was determined as described in Materials and Methods.
b Standard deviation.
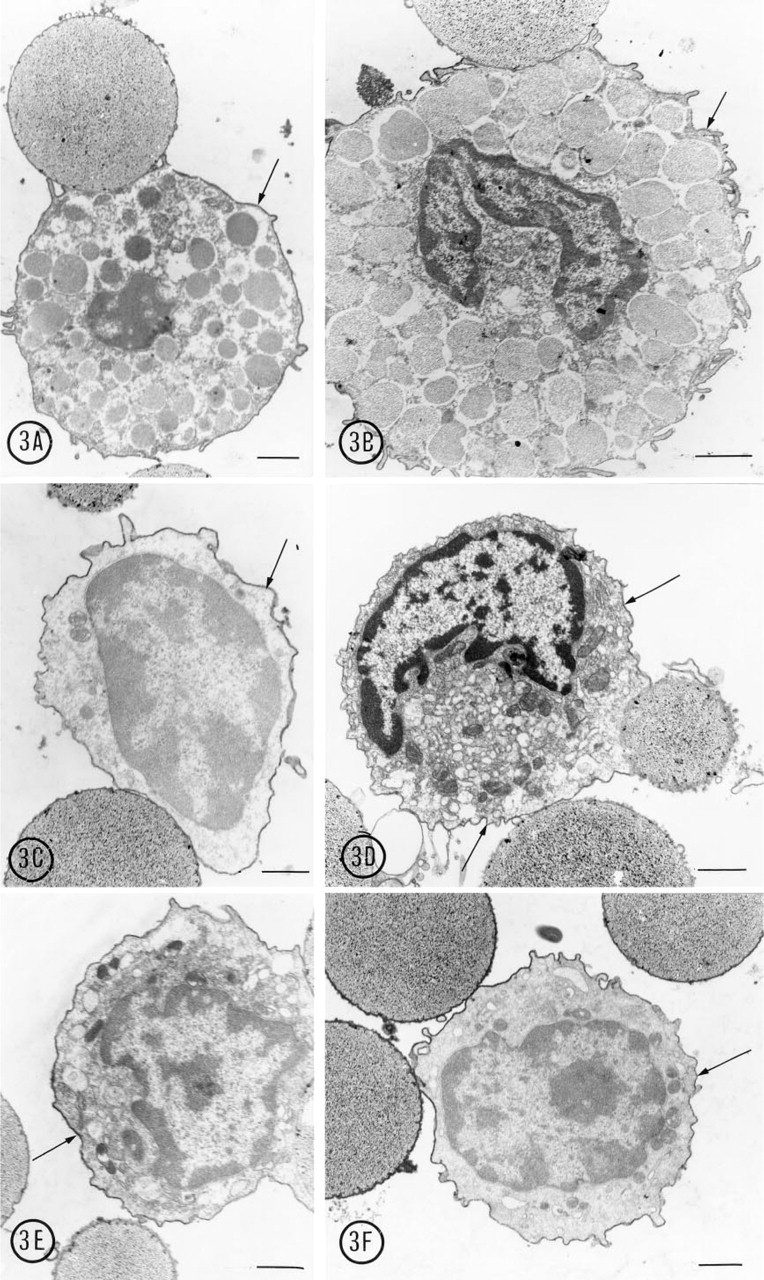
Mast cells in all stages of maturation were isolated with MAb AA4-conjugated beads and immunostained with mast cell-specific antibodies. (
Footnotes
Acknowledgements
Supported in part by FUNPAR and CAPES.
We thank Maria Tereza Picinoto Maglia, Jose Augusto Maulin for technical assistance, and Vera Regina Fontana Pioteke for preparation of the micrographs. We also thank the Department of Morphology, Laboratory of Electron Microscopy, Faculdade de Medicina de Riberao Preto-USP for the use of their facilities.
