Abstract
MP78/70 is a matrix protein, with 78-kD and 70-kD isoforms, which was initially identified in bovine tissue extracts designed to solubilize elastin-associated microfibrils. Peptide analysis has shown that MP78/70 is closely related to the human protein, βig-h3. In the present study an antibody raised to a synthetic βig-h3 peptide was shown specifically to identify MP78/70 in purified form and in bovine tissue extracts. This is consistent with MP78/70 and βig-h3 being the bovine and human forms, respectively, of the same protein. The antibody was further affinity-purified on MP78/70 bound to Sepharose and used to localize the protein in a range of bovine tissues. Immunofluorescence showed that MP78/70 was localized to collagen fibers in tissues such as developing nuchal ligament, aorta and lung, and mature cornea; to reticular fibers in fetal spleen; and to capsule and tubule basement membranes in developing kidney. No general localization to elastic fibers was observed. The staining pattern in most tissues more closely resembled that of Type VI collagen, which occurs as collagen fiber-associated microfibrils, than that of fibrillin-1, a component of elastin-associated microfibrils. However, MP78/70 appeared to be less widely distributed than Type VI collagen. Immunoelectron microscopy showed that MP78/70 was predominantly found in loose association with collagen fibers in most tissues examined and was also located on the surface of the capsule basement membrane in developing kidney. Double labeling experiments indicated that MP78/70 is co-distributed with Type VI collagen microfibrils located in these regions. In some elastic tissues significant immunolabel was detected in regions of interface between collagen fibers and fibrillin-containing microfibrils of adjacent elastic fibers, and at the outer margins of the latter structures. Overall, the evidence points to MP78/70 having a bridging function, perhaps in association with Type VI collagen microfibrils, linking or stabilizing the interaction between interstitial collagen fibrils and other matrix structures, including some basement membranes and elastin-associated microfibrils.
Keywords
W
Materials and Methods
Antibody Production
A synthetic peptide TQLYTDRTEKLRPEMEG(C) was commercially prepared (Chiron Mimotopes; Melbourne, Australia) that corresponds to residues 118–134 of the proteins encoded by mouse and human βig-H3 cDNAs. This region of the molecule lies between the cysteine-rich N-terminal domain and the four regions of internal homology that have similarity to the insect adhesion molecules, the fasciclins (Elkins et al, 1990). Database searches showed that the sequence is unique to βig-h3, with little similarity to other known proteins. The peptide was also selected to have no homology with the MP78/70 peptides sequenced previously (Gibson et al. 1996). The peptide was coupled via its carboxyl-terminal cysteine to diphtheria toxoid (Commonwealth Serum Laboratories; Melbourne, Australia) activated by N-(epsilon-maleimidocaproyloxy) succinamide (MCS) (Sigma; St Louis, MO) (Maeji et al. 1992). Briefly, 2 mg of MCS was dissolved in 10 μl of dimethylformamide and slowly added to 6 mg of diphtheria toxoid in nitrogen-flushed 0.1 M phosphate buffer, pH 7.0 (600 μl). The mixture was then shaken gently for 1 hr and passed through a column (4 ml) of Biogel P6, equilibrated in phosphate buffer, to remove unbound MCS. Peptide (2 mg) that had been dissolved in 20 μl of dimethylformamide was slowly added to the protein eluate and the mixture was stirred gently for 18 hr under nitrogen. The peptide-diphtheria toxoid conjugate was recovered from unbound peptide by gel filtration on Biogel P6 and stored at −20C. Antibodies were then raised in rabbits by regular sc injection of the conjugate (1 mg/injection) in Freund's adjuvant. The resulting antibodies were purified by affinity chromatography on columns of MP78/70 coupled to CNBr-activated Sepharose CL-4B using the method of Kohn and Wilcheck (1982). Antibodies were eluted with 0.2 M glycine, pH 2.8, and recovered as described previously (Gibson and Cleary 1987). Monoclonal anti fibrillin-1 antibody A5 and polyclonal anti-tropoelastin antibodies have been described previously (Gibson and Cleary 1987; Gibson et al. 1995). Polyclonal antibodies to purified pepsin-treated Type VI collagen were prepared in rabbits and affinity-purified on columns of the protein coupled to Sepharose CL-4B. Residual activity to other collagen types was removed by passage of the antibody over Sepharose columns coupled with collagens I, III, and V.
Assessment of Antibody Specificity
The affinity-purified anti-MP78/70 antibodies were assessed by ELISA and immunoblotting for crossreactivity with other matrix proteins (Gibson and Cleary 1985). Purified bovine proteins and proteoglycans to be tested were MAGP-2, MAGP-1, fibrillin-1, MP78/70, tropoelastin, collagen Types 1, III, V, and VI (native and pepsin-treated), decorin, and biglycan. MAGP-2, MAGP-1, fibrillin-1, and MP78/70 were prepared from reductive saline extracts of fetal nuchal ligament (Gibson et al. 1989). Tropoelastin was prepared using the method of Brown et al. (1992), and collagens were prepared from fetal calf skin or nuchal ligament as described previously (Gibson and Cleary 1985). Decorin and biglycan were prepared from fetal calf skin using the method of Choi et al. (1989). The specificity of the affinity-purified anti-peptide antibodies was also confirmed by immunoblotting against extracts of nuchal ligament from a 210-day-old fetal calf. Extracts were generated by sequential treatment of tissue homogenate with saline, 1 M NaCl, 6 M guanidinium chloride (GuHCl), saline plus dithiothreitol (DTT), and 6 M GuHCl plus DTT as previously described (Gibson et al. 1989).
Immunofluorescence Microscopy
Tissue blocks were taken from adult cattle or 210-day-old fetal calves within 1 hr of maternal death and snap-frozen in OCT compound. Sections (4 μm thick) were cut using a cryostat and incubated with primary antibody, followed by an appropriate secondary antibody coupled to fluorescein as described previously (Gibson and Cleary 1987). For optimal activity the anti-MP78/70 antibodies and the anti-fibrillin-1 monoclonal antibody A5 required pretreatment of the tissue sections with 6 M GuHCl and 6 M GuHCl/DTT, respectively, indicating that the epitopes are masked. In these instances the tissue was prefixed with paraformaldehyde (Gibson et al. 1989).
Antibodies to Type VI collagen and tropoelastin were applied to unfixed tissues. Affinity-purified rabbit antibodies were applied at a concentration of 20 μg/ml and the monoclonal antibodies used were ascites fluid diluted 1:100. Control sections were incubated with IgG (20 μg/ml) purified from preimmune rabbit serum and diluted ascites containing a monoclonal antibody to the parasite Giardia lamblia (Henderson et al. 1996). Sections were examined and photographed with automatic exposure using a Nikon microphot FX-A microscope with a 100-W high-pressure mercury lamp light source, a 470-nm/490-nm excitation filter, a 520-nm/560-nm barrier filter (filter block B1E), and CF epifluorescence fluor objectives. Control sections were photographed using manual exposure times matching those of the appropriate antibodies.
Immunoelectron Microscopy
For single immunolabeling experiments, blocks approximately 1 mm3 were cut from a range of tissues of a 230-day-old fetal calf and an adult cow within 30 min of death. The tissue blocks were fixed and embedded in either LR White or Lowicryl resin as described previously (White et al. 1988; Gibson et al. 1989). Thin sections from these blocks were mounted on Celloidon-coated nickel grids, treated with 6 M GuHCl, 50 mM Tris buffer, pH 8.0, for 5 min, and washed vigorously in 50 mM Tris-buffered saline (pH 8). The sections were then incubated with 20 mM glycine prepared in 6 mM PBS, pH 7.4, for 50 min (five 10-min changes) to block free aldehyde groups. Immunolabeling was carried out as described previously (White et al. 1988) using anti-peptide antibody (10 μg/ml) that had been affinity purified on MP78/70-Sepharose. Duplicate sections were treated with IgG (10 μg/ml) from preimmune rabbit serum as negative controls. Antibody binding was detected by protein A coupled to 20-nm gold particles (Sigma) diluted 1:40.
For double labeling experiments, the method was again based on that described by White et al. (1988). Briefly, blocks of nuchal ligament from a 260-day-old fetal calf were lightly fixed in PBS containing paraformaldehyde (4%), polyvinylpyrrolidone (
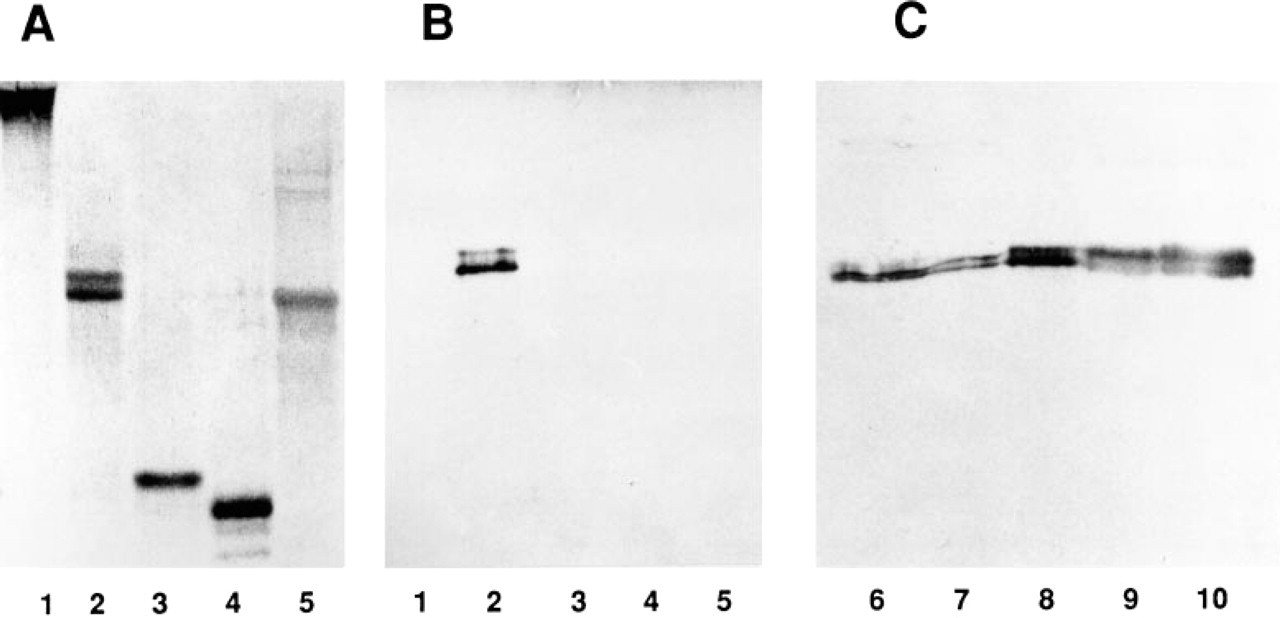
Immunoblotting of MP78/ 70 with anti-[βig-h3 peptide] antibody. Purified ligament proteins were analyzed by SDS-PAGE on 10% gels (
Results
Antibody Specificity
The affinity-purified anti-[βig-h3 peptide] antibody reacted strongly with MP78/70 by ELISA, with a titer of 1:320. No crossreactivity of the antibody was detected with elastic fiber-associated proteins, fibrillin-1, MAGPs 1 and 2, and tropoelastin, or with collagen Types I, III, V and VI (not shown). Immunoblotting showed that the antibody reacted with both the 78-kD and the 70-kD isoforms of MP78/70 (Figure 1B, Lane 2). This indicated that a sequence similar to that of the peptide was present in MP78/70. Again, no crossreaction was detected with fibrillin-1, MAGP-1, MAGP-2, or tropoelastin (Figures 1A and 1B). Purified decorin, biglycan, and collagen Types I, III, V, and VI were also shown to be unreactive with the antibody using the immunoblotting technique (data not shown). The anti-[βig-h3 peptide] antibody was also tested for crossreactivity with other tissue antigens by immunoblotting against a series of sequential extracts from fetal nuchal ligament. The antibody detected only MP78/70 in each of the extracts (Figure 1C). A major portion of MP78/ 70 was extracted from the tissue by treatment with 6 M GuHCl in the absence of a reducing agent (Figure 1C, Lane 8). However, significant amounts of MP78/70 were extracted only after treatment of the homogenate with a reducing agent, indicating that a proportion of the MP78/70 was disulfide-bonded to an insoluble component of the tissue, consistent with previous observations (Gibson et al. 1989). Overall, the evidence confirmed that the anti-peptide antibody specifically reacted with MP78/70 in bovine tissue extracts.
Immunofluorescence
The distribution of MP78/70 was determined in a range of elastic and nonelastic tissues from a 210-day-old fetal calf and in some adult bovine tissues. In each tissue the MP78/70 distribution was compared to those of Type VI collagen and fibrillin-1. Unless otherwise stated, photographs of control sections incubated with preimmune rabbit IgG or mouse ascites fluid showed no fluorescence signal when the appropriate exposure times were used (not shown).
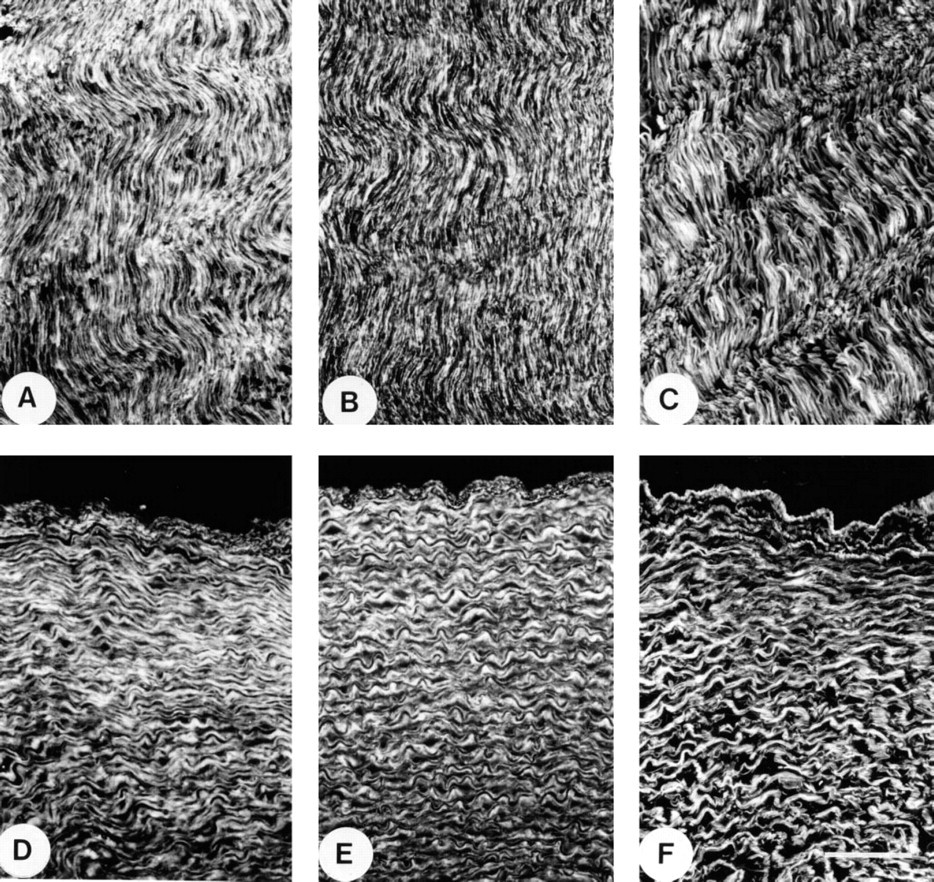
Immunofluorescence localization in fetal nuchal ligament and thoracic aorta. (
Elastic Tissues. In developing nuchal ligament, MP78/ 70 was localized extensively throughout the extracellular matrix (Figure 2A). The staining pattern had extensive overlap with that of Type VI collagen (Figure 2B), which is associated with collagen fibers between cells and elastic fibers. However, some MP78/70 also appeared to be co-localized with fibrillin-1, which is located only on elastic fibers (Figure 2C). In aorta, MP78/70 was localized most strongly in the inner media to the matrix between elastic fibers (Figure 2D). In the outer media, immunostaining was also evident around the surface of smooth muscle cells in the vicinity of basement membranes. The adventitia showed weak staining that appeared to co-distribute with collagen fibers (not shown). A similar distribution was found for Type VI collagen (Figure 2E), which contrasted with the elastic fiber staining of the anti-fibrillin 1 antibody (Figure 2F).
Immunofluorescence localization in fetal lung and skin. (
In lung, MP78/70 was localized as a continuous network throughout the parenchymal tissue. The protein was also identified surrounding bronchioles and in the walls of blood vessels (Figure 3A). This distribution was similar to that of Type VI collagen (Figure 3B). In contrast, anti-fibrillin-1 monoclonal antibody A5 localized in the parenchyma only to elastic fibers, which have a fragmented appearance. Fibrillin-1 was also identified around bronchioles and in the elastic fibers of blood vessel walls (Figure 3C).
In skin, MP78/70 exhibited a diffuse distribution in the matrix between the elastic fibers throughout the tissue and was particularly prominent in the upper papillary dermis (Figure 3D). Staining of the epidermis and around hair follicles was also evident but this appears to be nonspecific because these structures were also stained in control sections treated with preimmune IgG and photographed with the same exposure time (Figure 3G). The anti-Type VI collagen antibodies gave a strong uniform but diffuse staining pattern throughout the dermal matrix (Figure 3E), whereas anti-fibrillin-1 antibodies localized only to discrete elastic fibers and fine fibrils (Figure 3F). In elastic ear cartilage, MP78/70 was strongly immunolocalized to fibers, presumably collagenous, within the perichondrium and to the outer margins of the cartilage. No staining of the elastic fibers in the cartilage matrix was detected. Under the same experimental conditions these structures stained very strongly with anti-tropoelastin antibodies (results not shown).
Other Tissues. In spleen, MP78/70, Type VI collagen, and fibrillin-1 were all strongly localized in a reticular network that extended throughout the red and white pulp (Figures 4A–4C). However, in the fibroelastic septa, MP78/70 and Type VI collagen were distributed in the matrix around the elastic fibers, whereas fibrillin-1 was located only on these structures. MP78/70 was particularly prominent at the interface between the septa and the surrounding pulp (Figure 4A). The most marked difference between the localization of MP78/70 and Type VI collagen occurred in developing skeletal muscle. The anti-MP78/70 antibody gave only weak perimysial staining (Figure 4D) in contrast to antibodies to Type VI collagen and fibrillin-1, both of which gave strong staining of the perimysium and endomysium (Figures 4E and 4F).
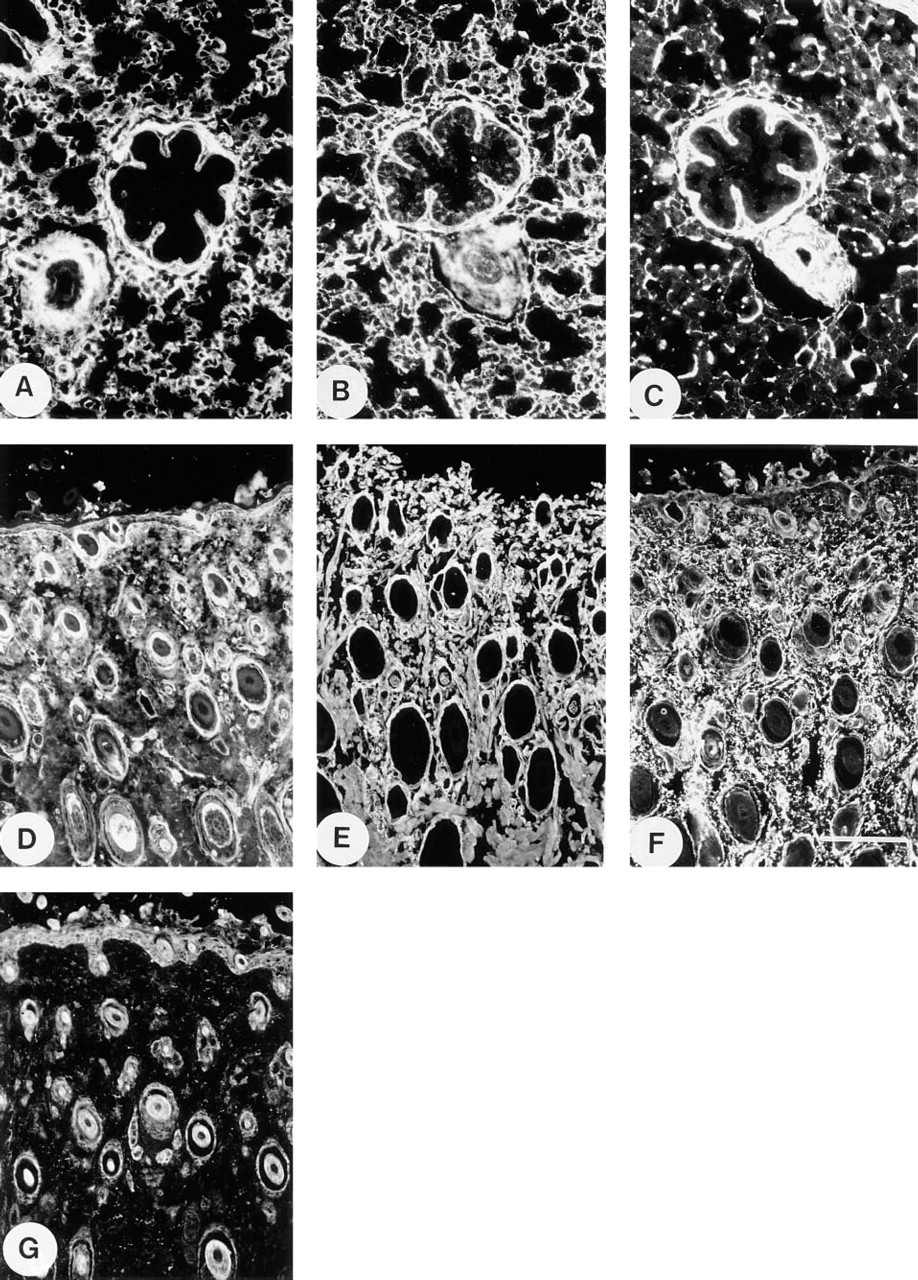
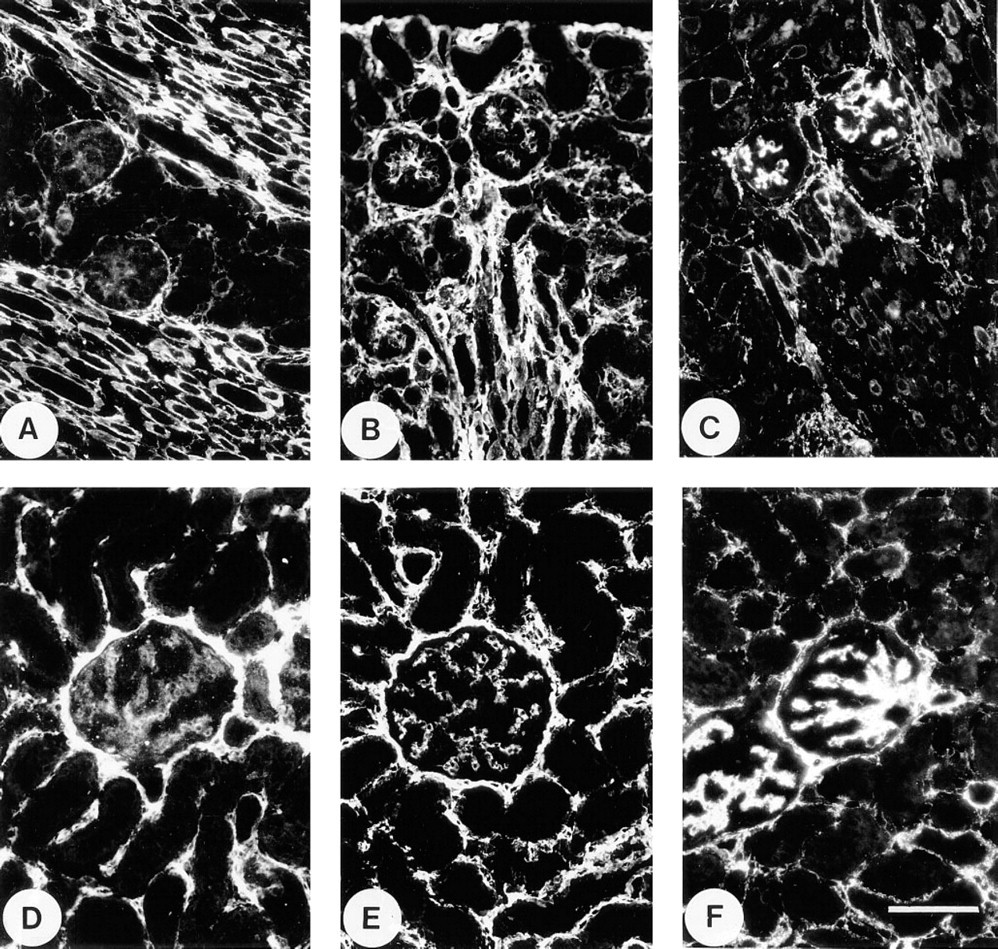
In developing kidney, MP78/70 was identified predominantly in close association with the basement membranes of Bowman's capsule and the proximal tubules. Weak immunostaining of the glomerular mesangium was also evident (Figure 5A). In contrast, Type VI collagen had strong localization throughout the peritubular matrix as well as in the mesangium (Figure 5B). Fibrillin-1 was identified mainly in the mesangium, although some intermittent staining around tubules was evident (Figure 5C). In adult kidney, all three proteins had extensive distributions in the peritubular matrix (Figures 5D–5F). The staining intensity of the mesangium relative to the peritubule region varied between the three antibodies. Anti-MP78/70 antibody stained the mesangium weakly (Figure 5D), antiType VI collagen antibody stained both regions with comparable intensity (Figure 5E), and anti-fibrillin-1 antibody gave a much stronger signal in the mesangium (Figure 5F).
In the adult eye, the suspensory ligament of the lens did not stain with anti-MP78/70 or anti-Type VI collagen antibodies (Figures 6A and 6B), in contrast to anti-fibrillin antibodies, which stained this structure intensely (Figure 6C). All three antibodies showed strong staining of the matrix of the ciliary body to which the zonule is attached. In cornea, the anti-MP78/70 and anti-Type VI collagen antibodies both gave intense, diffuse staining throughout the matrix (Figures 6D and 6E), whereas anti-fibrillin-1 antibody localized to discrete fibrils (Figure 6F). No staining of the epithelium was detected with any of the antibodies. Overall, MP78/70 had a distribution that was similar but not identical to that of Type VI collagen in most of the tissues examined, and which was distinct from that of fibrillin-1.
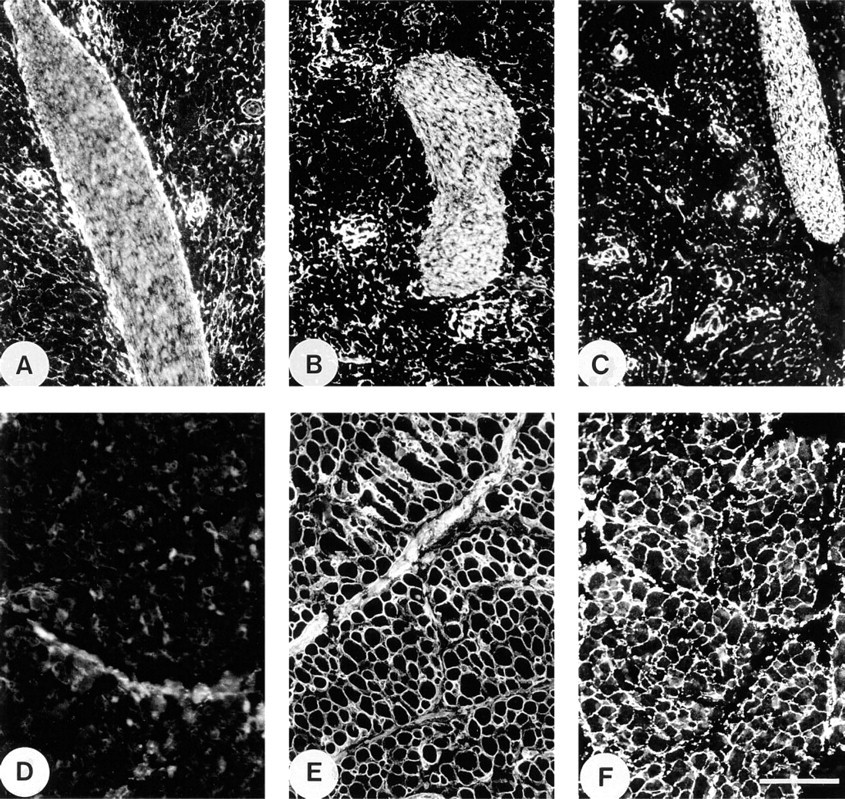
Immunofluorescence localization in fetal spleen and skeletal muscle. (
Immunoelectron Microscopy
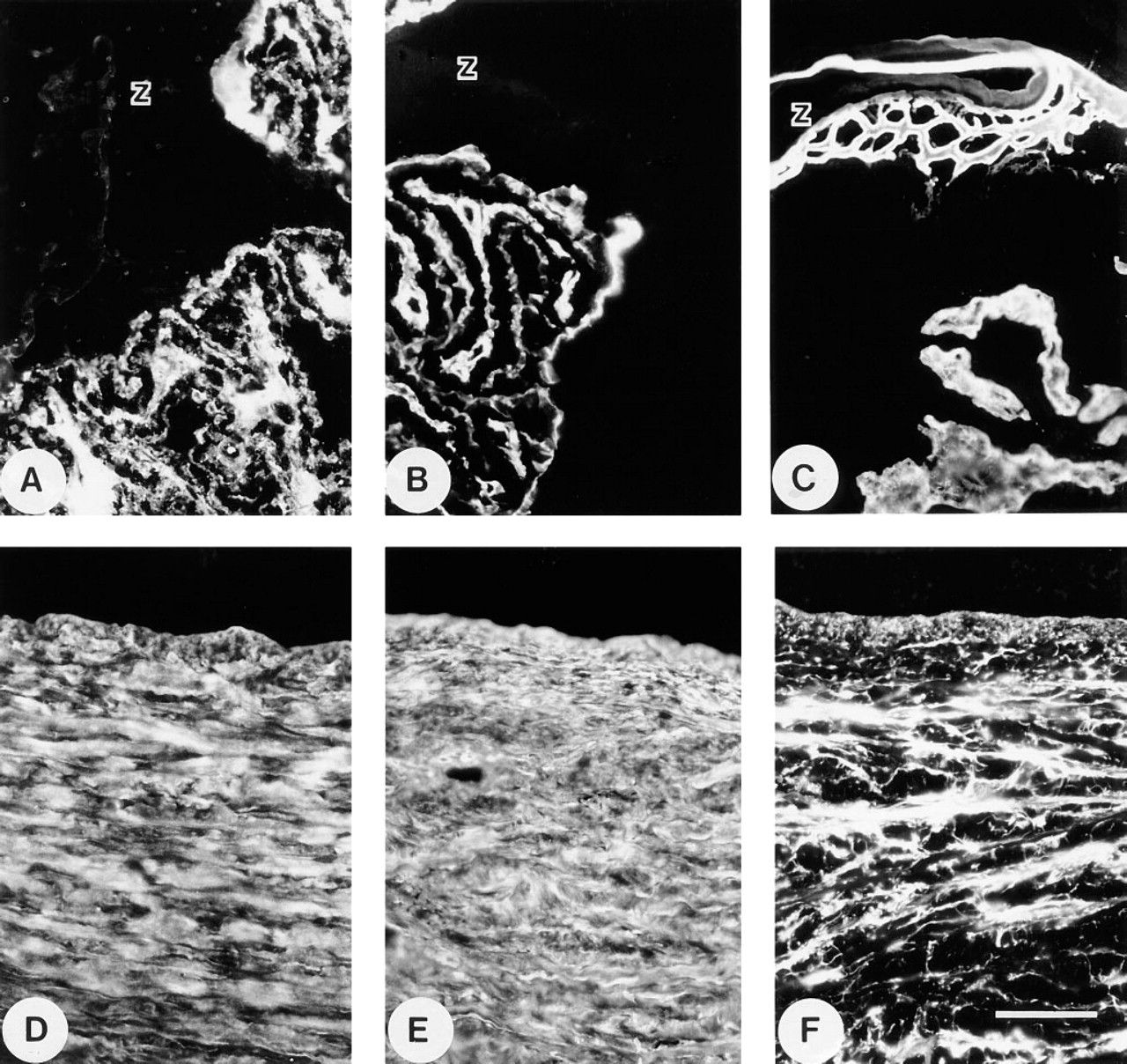
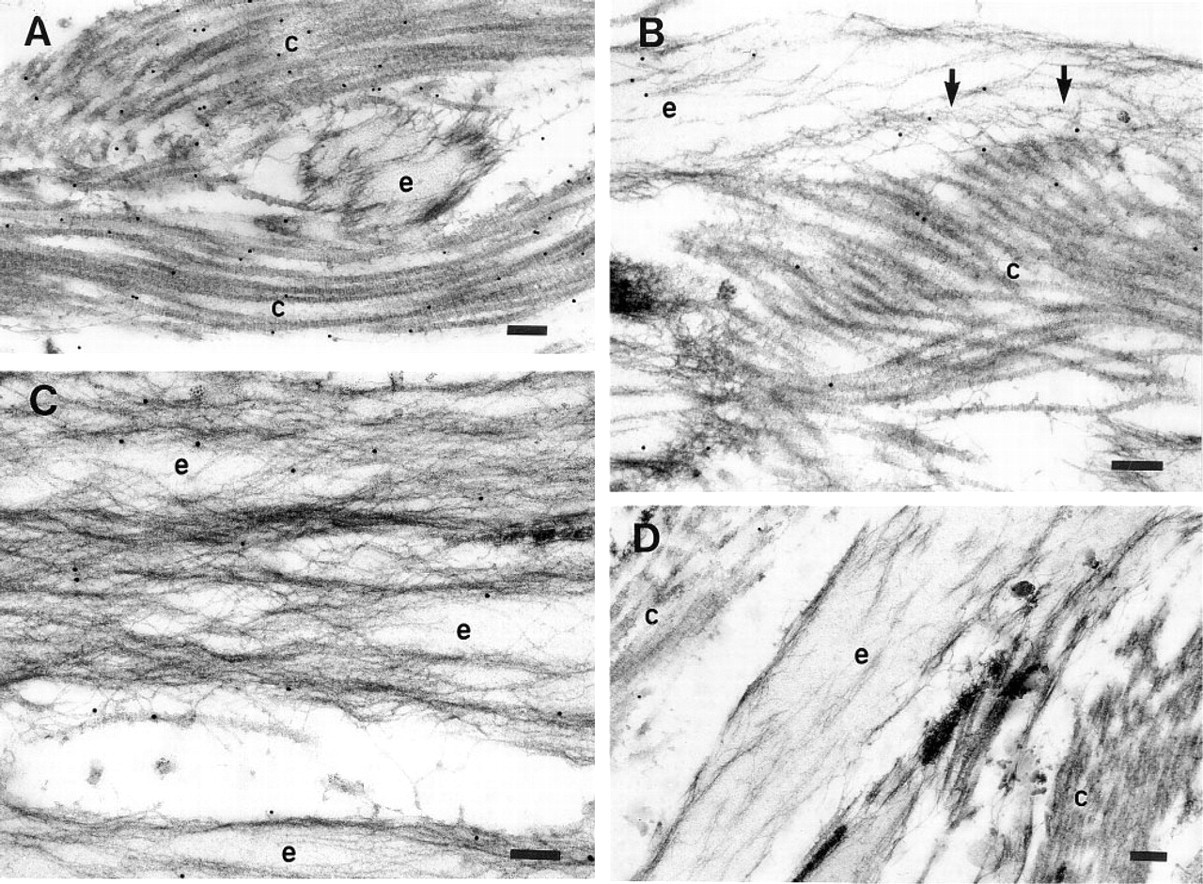
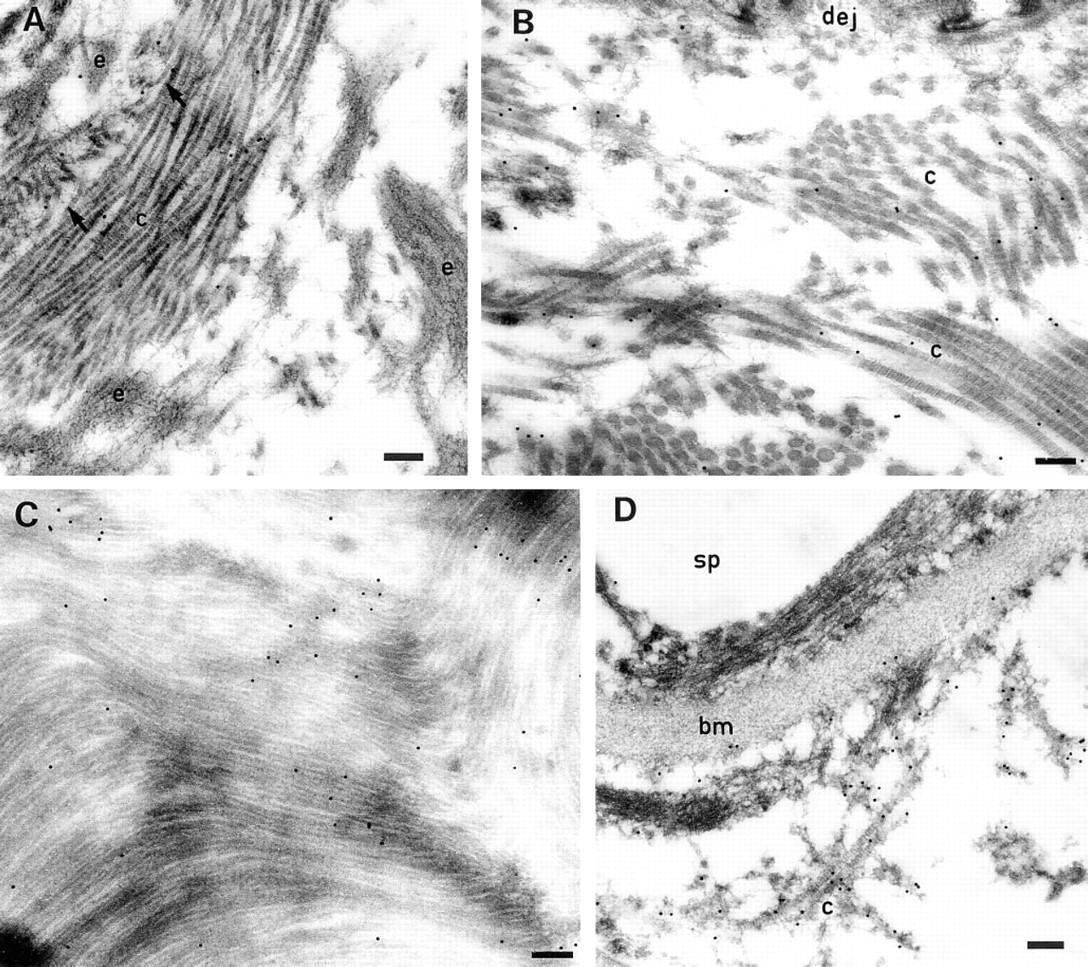
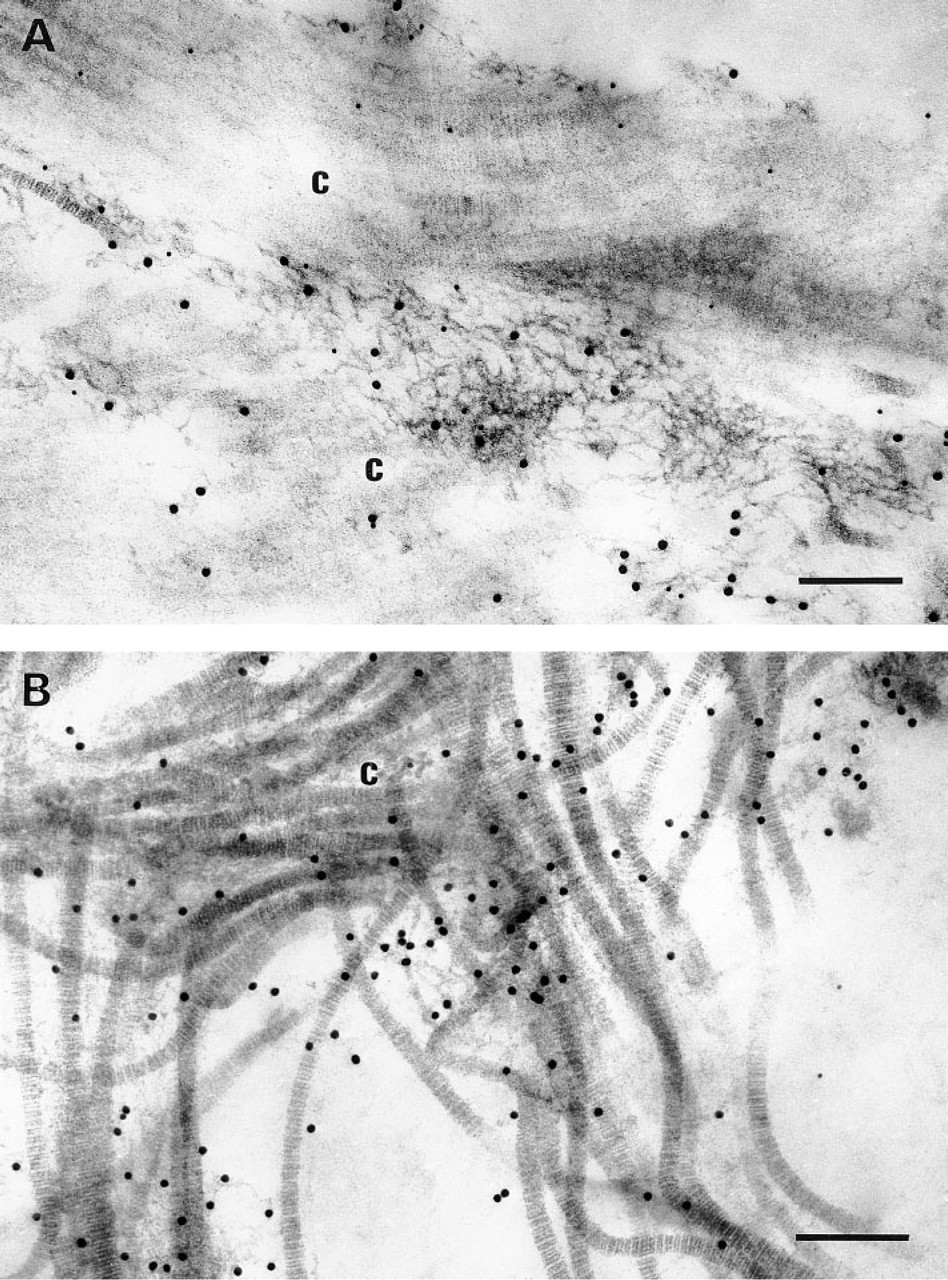
The ultrastructural location of MP78/70 was determined in several fetal tissues and adult cornea using the immunogold labeling technique (Figures 7 and Figures 8). In developing nuchal ligament, MP78/70 was predominantly localized in loose association with collagen fibrils (Figure 7A). The localization appeared to be to fine filaments surrounding the collagen fibrils rather than to the fibrils themselves. Significant immunogold labeling was also found in regions of interface between collagen fibers and fibrillin-containing microfibrils of adjacent elastic fibers (Figure 7B). Occasionally immunolabel was detected on fibrillin-containing microfibrils at the outer margins of developing elastic fibers, with no collagen fibrils in close proximity (Figure 7C), suggesting that some MP78/70 was bound to the microfibrils. In control sections incubated with IgG from preimmune rabbit serum as primary antibody, very few immunogold particles were detected and these appeared to be randomly scattered throughout the tissue (Figure 7D). Anti-MP78/70 antibody also localized predominantly around collagen fibers in developing aorta and skin and in mature cornea (Figure 8). Figure 8A is a section of inner aortic media showing extensive labeling around a collagen fiber. Again, no general staining of elastic fibers is evident, although a significant number of immunogold particles are located at the interface with an adjacent elastic fiber. Figure 8B shows a section of upper papillary dermis, the region of skin that was stained most significantly by immunofluorescence. Here, too, MP78/70 is located around bundles of collagen fibrils. Similarly, in adult cornea MP78/70 is immunolocalized in widespread association with the dense collagen fibers that make up the bulk of the tissue (Figure 8C). The Bowman's capsule of developing kidney is shown in Figure 8D. In this region, MP78/70 is localized to fine filaments attached to the outer surface of the capsule basement membrane as well as filaments that surround collagen fibers in the pericapsular connective tissue. No staining of the basement membrane itself is evident. Control sections from the above tissues that had been treated with IgG from preimmune rabbit serum in place of primary antibody showed no significant immunogold labeling (not shown).
Immunofluorescence localization in fetal and adult kidney. (
Double labeling experiments were conducted on sections of developing nuchal ligament tissue using pre-embedding labeling with anti-Type VI collagen antibodies followed by postembedding labeling with anti-MP78/70 antibodies. Extensive co-localization of Type VI collagen, identified by protein A–20-nm gold particles, and MP78/70, identified by protein A–10-nm gold particles, was observed to fine microfibrils, characteristic of Type VI collagen, surrounding bundles of collagen fibrils (Figure 9A). Controls sections incubated with IgG from preimmune serum instead of the two antibodies showed no localization of gold particles to these structures (not shown). An additional control was conducted in which the blocks were pre-embedding-labeled for Type VI collagen but the anti-MP78/70 antibodies were omitted from the postembedding treatment (Figure 9B). In this control, Type VI collagen microfibrils were identified by 20-nm gold particles but no labeling of these structures with 10-nm gold was evident. This finding indicates that the protein A–10-nm gold, applied after embedding, is not binding to the anti-Type VI collagen antibodies embedded in the tissue. Overall, the results confirmed that MP78/70 is closely associated with Type VI collagen microfibrils in nuchal ligament tissue.
Immunofluorescence localization in adult ocular tissues. (
Discussion
Previous studies have shown that MP78/70 is a significant component of and is strongly bound to the extracellular matrix in developing elastin-rich tissues (Gibson et al. 1989). Experiments have shown that a proportion of the protein was resistant to extraction from nuchal ligament by the strong chaotrope 6 M GuHCl but was readily solubilized by saline treatment if a reducing agent was present (reductive saline extraction). This indicated that some MP78/70 had become disulfide-bonded to an insoluble element of the matrix. The finding that the reductive saline extracts contained only three other major proteins, fibrillin-1, MAGP-1, and MAGP-2, each of which is a component of elastin-associated microfibrils, suggested that MP78/70 was also derived from elastic fibers. This possibility was supported by observations that polyclonal antibodies to MP78/70 preparations localized to elastin-associated microfibrils. However, these antibodies strongly crossreacted with fibrillin-1 (Gibson et al. 1989), precluding their use for further immunohistochemistry. Analysis of MP78/70 peptides had revealed that all five that were sequenced had close similarities (including two peptides of 11 and 12 amino acids that were totally homologous) with the recently cloned human protein βig-h3. Therefore, it was apparent that MP78/70 and βig-h3 represent the bovine and human forms, respectively, of the same protein (Gibson et al. 1996). βig-h3 was identified as a TGF-β-inducible gene in a lung adenocarcinoma cell line (Skonier et al. 1992). The cDNA sequence encoded a 76-kD protein containing four regions of internal homology, 11 cysteine residues, and an RGD motif. Evidence indicated that the protein was processed by cleavage of the carboxyl terminus to a 68-kD form that had an anti-adhesive effect on a range of cell types (Skonier et al. 1994). More recently, βig-h3 was found to be deposited into the extracellular matrix by cultured skin fibroblasts and to promote the attachment and spreading of these cells on plastic. Moreover, pig h3 was identified immunohistochemically in human skin, suggesting that the protein was a normal constituent of the extracellular matrix (LeBaron et al. 1995).
Ultrastructural localization of MP78/70 in nuchal ligament of 230-day-old fetal calf. (
Ultrastructural localization of MP78/70 in fetal aorta, skin, and kidney, and in adult cornea. (
In the present study, antibodies prepared to a synthetic βig-h3 peptide were used to confirm that MP78/70 is the bovine form of the same protein and to determine its tissue distribution and ultrastructural location within the matrix. MP78/70 was the only protein from a range of purified bovine matrix macromolecules that reacted to the peptide antibody on ELISA and immunoblotting. Moreover, MP78/70 was also the only protein identified by the antibody in extracts of nuchal ligament tissue (Figure 1). Immunofluorescence localization studies showed that MP78/70 was extensively present in the extracellular matrix of a wide range of developing and mature tissues (Figures 2–6). The protein was identified predominantly in the region of interstitial collagen fibers and in association with some basement membranes, including those around smooth muscle cells in developing aorta and those of Bowman's capsule and tubules in fetal kidney. No general co-localization with elastic fibers was observed. MP78/70 had extensive co-distribution with Type VI collagen in tissues such as developing nuchal ligament, aorta and lung, and mature cornea. However, there were differences in distribution in some tissues, such as developing kidney, skin, and muscle. The most marked difference was observed in fetal skeletal muscle, in which little MP78/70 was detected, in contrast to Type VI collagen, which was found throughout the perimysium and endomysium. Type VI collagen has previously been reported to be a constituent of perimysium and endomysium in skeletal muscle from adult chickens (Swasdison and Mayne 1989). The distribution of Type VI collagen found in the other tissues was also in agreement with previous studies (Gibson and Cleary 1983; Zimmermann et al. 1986; Timpl and Engel 1987; Amenta et al. 1988; Rittig et al. 1990; Zhu et al. 1994). The distribution of MP78/70 in fetal bovine skin (Figure 3D) appeared to be similar to that described for βig-h3 in human tissue (LeBaron et al. 1995), with the highest signal being in the matrix close to the dermal/epidermal junction. In adult bovine cornea, MP78/70 was strongly detected throughout the stroma but not in the epithelium (Figure 6D). This finding is consistent with the recent study of Hirano et al. (1996), which immunochemically identified βig-h3 throughout the stroma of normal adult cornea. In contrast, Escribano et al. (1994) have described βig-h3 as being confined to the epithelial layer in normal rabbit cornea, although it should be noted that Northern blotting showed a high steady-state level of its mRNA throughout the tissue. It is interesting that MP78/70 was found throughout the developing aortic wall, suggesting that the protein is a significant normal component of the extracellular matrix of arteries (Figure 2D). This contrasts with the recent findings of O'Brien et al. (1996), which indicate that βig-h3 is absent from the walls of normal human coronary arteries and that the protein is immunodetectable only in the intima of atherosclerotic vessels. The reasons for the differences between the present study and those of Escribano et al. and O'Brien et al. are unclear. They may be due to variations between species, the age of the tissues, and to distinctions in structure between aorta and coronary arteries. However, it is interesting that the anti-peptide antibody described in the present study required pretreatment of tissue with 6 M GuHCl to identify MP78/70 in the extracellular matrix of most tissues. This suggests that the peptide epitopes (and indeed the protein itself) are heavily masked within the matrix.
Co-localization of MP78/70 and Type VI collagen to fine microfibrils surrounding collagen fibers. The tissue is nuchal ligament from a 260–day-old fetal calf. Type VI collagen microfibrils were identified by preembedding immunolabeling and detection with protein A–20-nm gold particles. MP78/70 was localized using subsequent postembedding immunolabeling and detection with protein A–10-nm gold particles (
Immunoelectron microscopy revealed that MP78/70 was predominantly localized in loose association with collagen fibril bundles in a number of developing tissues and in mature cornea (Figures 7 and Figures 8). In addition, localization of MP78/70 was confirmed at the outer margin of the basement membrane of Bowman's capsule in developing kidney. In fetal nuchal ligament and aorta, the protein was also detected at regions of interface between collagen fibers and the microfibrillar component of developing elastic fibers. In nuchal ligament, some immunostaining for MP78/70 was present at the outer margins of the elastin-associated microfibrils, often in regions in which collagen fibrils were not evident. This indicates that some MP78/70 may be attached to these fibrillin-containing microfibrils.
Double labeling experiments (Figure 9) confirmed that the MP78/70 localization around collagen fibers corresponds to the region containing the fine (5-nm diameter) Type VI collagen microfibrils that surround interstitial collagen fibrils in the extracellular matrix of most tissues (Keene et al. 1988; Timpl and Chu 1994). Type VI collagen microfibrils are also found in association with the cell surface and attached to certain basement membranes (Maier and Mayne 1987; Rittig et al. 1990; Zhu et al. 1994). Recently Hirano et al. (1996) have presented immunohistochemical evidence for the co-localization of βig-h3 with Type VI collagen microfibrils in the cornea of patients with Fuchs' dystrophy. The results from the present study suggest that MP78/70 (βig-h3) may associate with Type VI collagen in a range of normal tissues. Preliminary biochemical evidence supporting this idea is that a proportion of MP78/70 can be co-purified, in aggregate form, with nonreduced native Type VI collagen from GuHCl extracts of nuchal ligament. In addition, purified MP78/70 will bind in vitro to the pepsin-resistant triple-helical region of Type VI collagen using a modified ELISA (unpublished observations).
Overall, the evidence suggests that MP78/70 (βig-h3) functions as a bridging molecule, possibly in association with Type VI collagen microfibrils, linking interstitial collagen fibers with other structural elements of the extracellular matrix, including certain basement membranes and elastin-associated microfibrils. Such a role may be important for anchoring elastin-associated microfibrils to collagen fibers during stretching of elastic fibers.
Footnotes
Acknowledgements
Supported by the National Health and Medical Research Council of Australia.
We are indebted to Denise Yeats and Betty Reinboth for skilled technical assistance.
