Abstract
Neuropeptide Y (NPY) and peptide YY (PYY) are structurally related peptides that are considered to mediate inhibitory actions on gastrointestinal motility, secretion, and blood flow. Several receptor subtypes for these peptides have been identified and the Y1, Y2, Y4/PP1, Y5, and Y5/PP2/Y2b receptors have been cloned. In this article we report the immunocytochemical localization of the Y1 receptor to myenteric and submucosal nerve cell bodies, endothelial cells, and scattered endocrine-like cells of rat intestinal tract. Moreover, double immunofluorescence demonstrates that subpopulations of the Y1 receptor-positive nerve cell bodies are immunopositive for NPY, vasoactive intestinal polypeptide, and nitric oxide synthase. In part, such co-localizations were made possible by use of peroxidase-mediated deposition of tyramide, which permitted use of antisera derived from the same species. Our observations suggest the existence of multiple neuronal, endothelial, and endocrine target sites for NPY and PYY and that some of the actions of these regulatory peptides can be mediated by vasoactive intestinal peptide and nitric oxide synthase.
NPY and PYY have similar biological effects on the gastrointestinal system. These include effects on pancreatic and gastrointestinal secretion, blood pressure, and motility of the gastrointestinal tract, which all are of inhibitory nature (Sheikh 1991). NPY and PYY exert their actions through several receptor subtypes, of which five have been cloned. All of these contain seven transmembrane domains and are members of the G-protein-coupled superfamily of receptors. The first receptor to be cloned was the Y1 receptor (Y1-R) (Eva et al. 1990, 1992; Herzog et al. 1992; Larhammar et al. 1992). Subsequently, the Y2 receptor (Gerald et al. 1995; Rose et al. 1995; Gehlert et al. 1996; Nakamura et al. 1996), the Y4/PP1 receptor (Bard et al. 1995; Lundell et al. 1995; Gregor et al. 1996b; Yan et al. 1996), the Y5 receptor (Gerald et al. 1996; Hu et al. 1996), and the Y5/PP2/Y2b receptor (Gregor et al. 1996a; Matsumoto et al. 1996; Weinberg et al. 1996) were cloned. These receptors show low sequence similarity and differ in binding properties and tissue distribution. Y1-R binds NPY and PYY as well as the Y1-R-selective analogue [Leu31, Pro34]-NPY with similar affinities (Herzog et al. 1992; Larhammar et al. 1992) and is expressed in the brain and in several peripheral organs (Eva et al. 1990; Wharton et al. 1993).
Earlier studies have localized NPY and PYY binding sites to submucosal and myenteric plexi in the gastrointestinal tract using receptor autoradiography (Walsh et al. 1993; Mao et al. 1996). The identity of these binding sites was further analyzed using the NPY analogues [Leu31, Pro34]-NPY and NPY[13–36], which preferentially bind to the Y1-R and the Y2 receptor, respectively. In light of the recent cloning of new subtypes of PP family receptors, these binding studies have to be reevaluated, because [Leu31, Pro34]-NPY binds to the Y1-R as well as to the Y4/PP1, Y5, and Y5/PP2/Y2b receptors.
In the present study we raised an antibody against the N-terminal portion from the extracellular region of the rat Y1-R and employed this antibody to study the possible co-localization of the Y1-R with various neuronal and endocrine markers in the gastrointestinal tract. In addition, we demonstrate that horseradish peroxidase (HRP)-catalyzed deposition of tyramide is a useful tool for detection of cellular colocalization of antigens recognized by antisera raised in the same species.
Materials and Methods
Production of Antisera
An N-terminal peptide (amino acids 20–31) of the rat Y1-R (Genbank accession number Z11504) without any consensus sites for N-linked glycosylations was synthesized (Ross-Petersen; Hørsholm, Denmark), coupled to keyhole limpet hemocyanin (Boehringer Mannheim; Mannheim, Germany) using glutaraldehyde, and was used for sc immunization of four rabbits. This peptide represented a region of the Y1-R with no homology to other cloned NPY receptor subtypes, indicating that crossreactivity of the antisera to NPY receptor subtypes different from Y1-R is unlikely. Sera were obtained after repeated boosters. To remove antibodies directed against keyhole limpet hemocyanin, antisera, diluted 1:1000 in Tris-buffered saline, were absorbed overnight at 4C with 200 μg/ml keyhole limpet hemocyanin coupled to CNBr-activated Sepharose beads (Pharmacia Biotech; Uppsala, Sweden) according to the manufacturer's instructions. Preliminary immunocytochemical stainings of rat brain sections showed that of the four antisera, only antisera 307 and 308 were able to stain neurons. This staining was eliminated after preabsorption of the antisera with the synthetic Y1-R peptide.
Animals
Three adult Wistar rats were perfused intracardially with saline, followed by 4% paraformaldehyde in 0.1 M sodium phosphate buffer, pH 7.4. Subsequently, specimens from the intestines (duodenum, mid-jejunum, mid-ileum, proximal and distal colon) were postfixed in the same fixative overnight, cryoprotected in 30% sucrose, embedded in Tissue-Tek (Miles; Elkhart, IN), and frozen in n-hexane (Merck; Darmstadt, Germany) cooled by liquid nitrogen. In addition, specimens from the colon muscle wall were carefully separated from the rest of the gut wall using a dissecting microscope and fine tweezers, frozen in liquid nitrogen, and stored at −80C.
Immunocytochemistry
Five-μm cryostat sections were placed on slides, airdried, dehydrated, and pretreated for 30 min with methanol containing 0.03% H2O2 to inhibit endogenous peroxidase activity (Streefkerk and van der Ploeg 1976). After incubation for 30 min in 10% normal goat serum, the sections were exposed overnight at 4C to rabbit Y1-R antiserum (307 diluted 1:4000; 308 diluted 1:8000). Subsequently, biotin-labeled goat anti-rabbit Ig (Dako; Glostrup, Denmark) diluted 1:500 was applied. The biotin-labeled sites were visualized by HRP-catalyzed deposition of fluorochrome-conjugated tyramide using the Renaissance TSA-Direct kit (DuPont NEN; Boston, MA), which involved application of HRP-conjugated streptavidin followed by deposition of either tetramethyl rhodamine-tyramide or fluorescein-tyramide according to the manufacturer's instructions (cf. Bobrow et al. 1989). This principle for biotin amplification has been noted to enhance the immunocytochemical signal (Adam 1992).
For double immunofluorescence for Y1-R and either vasoactive intestinal polypeptide (VIP) or nitric oxide synthase (NOS), the sections were pretreated with methanol/H2O2 and 10% normal serum from the species donating the secondary antisera as above, followed by exposure to the Y1-R antiserum together with either polyclonal guinea pig antiserum to synthetic human VIP (Milab; Malmö, Sweden) diluted 1:500 or mouse monoclonal IgG2a antibody to brain NOS 10 μg/ml (Transduction Laboratories; Lexington, KY). Subsequently, the site of antigen-antibody reaction was revealed with fluorescein isothiocyanate (FITC)-conjugated donkey anti-guinea pig Ig (Jackson ImmunoResearch Laboratories; West Grove, PA) diluted 1:100, FITC-labeled goat anti-mouse Ig (Dako) diluted 1:30, and biotin-labeled goat anti-rabbit Ig (Dako) diluted 1:500. To reduce unspecific staining and ensure species specificity, the secondary antibodies were preabsorbed overnight at 4C with 10 μl/ml normal rat serum and 10 μl/ml normal serum from the species recognized by the other secondary antibodies. As above, the biotin-labeled sites were revealed using the Renaissance TSA-Direct kit.
Double immunocytochemistry for Y1-R and NPY was performed by visualizing the Y1-R-immunoreactive sites with tetramethyl rhodamine-tyramide as above, followed by incubation for 30 min in 10% normal swine serum and exposure at 4C overnight to polyclonal rabbit antiserum to CPON (C-terminal peptide of NPY) (Affiniti; Nottingham, UK) diluted 1:500. Finally, the site of antigen-antibody reaction was revealed with FITC-conjugated swine anti-rabbit Ig (Dako) diluted 1:30. For controls, parallel sections were similarly processed omitting the CPON primary antibody, and other sections were stained only for CPON immunoreactivity. Double immunofluorescence for Y1-R and either PYY, substance P, serotonin, neurotensin, or motilin was performed by substituting the CPON antiserum with polyclonal antiserum to either PYY (Milab), substance P (#K25) (Nilsson et al. 1975), serotonin (IncStar; Stillwater, MN), neurotensin (Affiniti), or motilin (Affiniti).
Controls included absorption of the Y1-R antisera with 100 μg/ml synthetic Y1-R peptide. In addition, the co-localization of NPY Y1-R and brain neuronal NOS was controlled by detecting NPY Y1-R-immunoreactive sites using the Y1-R primary antiserum and Texas Red-labeled donkey anti-rabbit Ig (Jackson ImmunoResearch Laboratories) diluted 1:100, followed by restaining for NADPH diaphorase activity. The NADPH diaphorase activity known to be present in neurons (Thomas and Pearse 1961) has been suggested to be due to the presence of NOS in neurons (Dawson et al. 1991; Hope et al. 1991). NADPH diaphorase was detected by incubating sections in a freshly prepared solution of 0.1 M Tris-HCl buffer, pH 7.2, containing 0.2% Triton X-100, 0.1 mM nitroblue tetrazolium (Sigma; St Louis, MO), 0.01% sodium azide, and 1 mM β-NADPH (Sigma) at 37C for 45 min. For controls, the NADPH substrate was omitted from the incubation medium.
The stainings were examined with a Multiprobe 2001 inverted confocal laser scan microscope system (Molecular Dynamics; Stockholm, Sweden) equipped with an argon-krypton laser and a x 100, NA 1.40 Nikon objective. To eliminate signals in the FITC channel from tetramethyl rhodamine fluorescence, a fraction of the tetramethyl rhodamine channel signal was subtracted from the FITC channel signal using the “enhance separation” function. The value of this fraction was set using a section that was only tetramethyl rhodamine-labeled. Other specimens were examined by conventional transillumination or by fluorescent epi-illumination using selective and aligned FITC, Texas Red, and double FITC/Texas Red filters.
Western Blotting
Samples of colon muscle wall were homogenized in sample buffer (62.5 mM Tris-HCl, pH 6.8, 10% glycerol, 2% SDS) and insoluble material was removed by centrifugation. The protein concentration was determined using the DC protein assay (Bio-Rad Laboratories; Hercules, CA). Subsequently, β-mercaptoethanol (Bio-Rad) and bromophenol blue (Bio-Rad) were added to 5% and 0.0025%, respectively, and samples were heated at 100C for 3 min.
Samples of 150 μg protein per lane were separated on an SDS-polyacrylamide gel (11%) and electroblotted onto nitrocellulose membrane. The nitrocellulose filter was incubated overnight in PBS containing 3% skimmed milk powder, rinsed in PBS containing 0.1% Tween-20 (Merck), and exposed overnight to Y1-R 308 antiserum diluted 1:8000 in PBS, 3% skimmed milk powder. Control nitrocellulose filter was incubated in Y1-R 308 antiserum 1:8000 preabsorbed overnight with 100 μg/ml of the peptide antigen. After washing in PBS containing 0.1% Tween-20, the site of antigen-antibody reaction was revealed with alkaline phosphatase-labeled swine anti-rabbit Ig (Dako) diluted 1:1000 and development in bromochloroin-dolylphosphate nitroblue tetrazolium medium as described (Larsson 1988).
Results
Localization of Y1-R Immunoreactivity in Rat Intestines
Throughout the rat intestine, Y1-R immunoreactivity was detected in endothelial cells of blood vessels (Figure 1A) and in the cytoplasm of a few endocrine-like cells situated in the intestinal villi (Figure 1B) using either of the two Y1-R antisera. The Y1-R-positive endocrine-like cells were most numerous in the small intestine. Double immunofluorescence showed that the Y1-R-positive endocrine-like cells did not co-store either peptide YY, substance P, serotonin, neurotensin, or motilin. In addition, both Y1-R antisera stained the cytoplasm of many nerve cell bodies in the myenteric and submucosal plexi (Figure 2), the muscularis mucosae and scattered nerve fibers in the circular muscle, and in the lamina propria in the intestinal tract. After preabsorption with the peptide used to raise the antisera, the reactivity of the Y1-R 307 antiserum was totally eliminated, whereas a weak staining of the neurons remained when the Y1-R 308 antiserum was used.
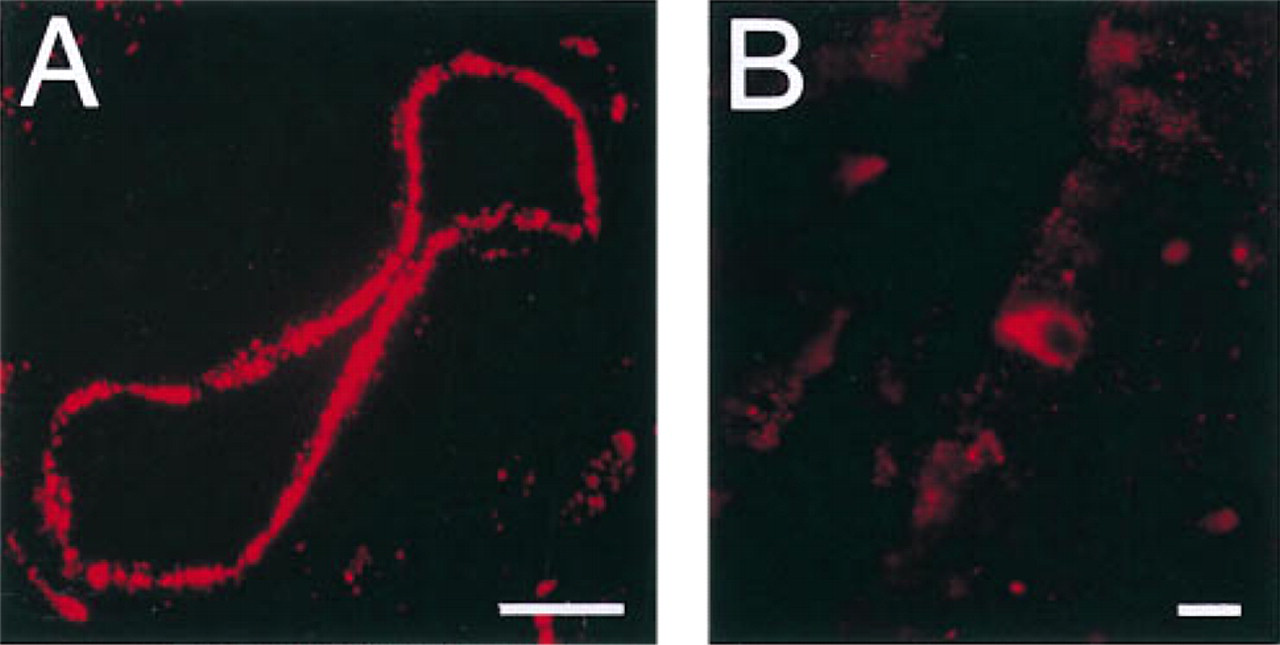
Localization of Y1-R immunoreactivity in endothelial cells of a blood vessel in the ileum (
A new method was applied to study the co-localization of Y1-R and NPY (CPON), because the antisera directed against these antigens were raised in the same species. After immunocytochemical detection of Y1-R using tetramethyl rhodamine-tyramide deposition, the sections were sequentially exposed to the CPON anti-serum and to FITC-conjugated secondary antiserum. By this method, a subpopulation of the Y1-R-positive nerve cell bodies situated in the submucosal plexus in the small intestine could be shown to co-store CPON immunoreactivity (Figures 2A–2C). In the submucosal and myenteric plexi throughout the intestinal tract, immunoreactive CPON could also be localized to Y1-R-negative nerve terminals close to Y1-R-displaying nerve cell bodies. CPON-stained sections that were not prestained for Y1-R revealed the same distribution of CPON immunoreactivity. In addition, control sections prestained for Y1-R using the tyramide deposition followed by exposure to FITC-conjugated secondary antiserum alone showed no staining with the latter secondary antiserum. This demonstrates that, on the one hand, the tyramide deposits prevent the first cycle of primary and secondary antisera from reacting with the second cycle of antisera. On the other hand, the antigenicity of the second epitope is not impaired by the deposition of tetramethyl rhodamine-tyramide, because cells co-storing both antigens could be detected.
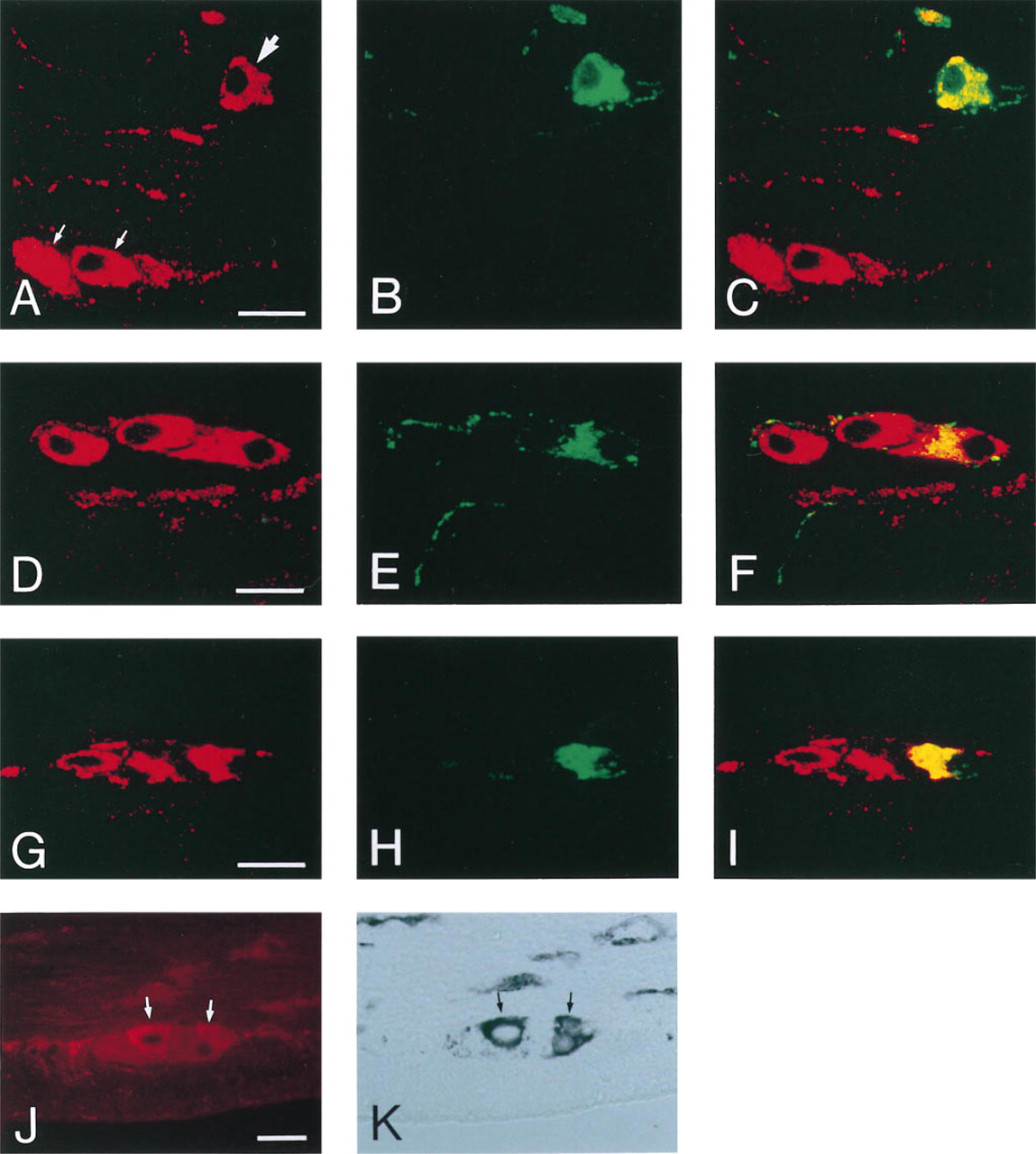
Immunocytochemical localization of Y1-R in submucosal and myenteric nerve cell bodies using the Y1-R 308 antiserum. Confocal images of ileum double stained for Y1-R and CPON (
Throughout the intestinal tract, a fraction of the Y1-R-positive nerve cell bodies present in the submucosa also displayed VIP immunoreactivity (Figures 2D–2F). These Y1-R and VIP co-storing cells were most numerous in the small intestine. In addition, VIP-positive nerve fibers were present adjacent to Y1-R-positive neurons in the submucosal and the myenteric plexus in all intestinal regions. Double immunofluorescence for CPON and VIP in the ileum further demonstrated that all immunopositive nerve cell bodies of the submucosal plexus co-stored both peptides, as previously described by Ekblad et al. (1987).
Double immunofluorescence for Y1-R and NOS in all intestinal regions showed that a fraction of the Y1-R-positive nerve cell bodies in the myenteric plexus co-stored NOS (Figures 2G–2I). In the small intestine, immunoreactive NOS was also detected in very few Y1-R-positive submucosal nerve cell bodies. The co-localization of Y1-R and NOS was further confirmed by substituting immunocytochemical detection for NOS with histochemical staining for NADPH-diaphorase activity (Figures 2J–2K).
Western Blotting of Colon Muscle Protein
The presence of Y1-R in the intestines was confirmed by Western blotting of extracts from the colon muscle layers, followed by detection with either the Y1-R 308 antiserum or the Y1-R 308 antiserum preabsorbed with the peptide used to raise the antiserum. Figure 3 shows that the antiserum recognizes a 70-kD protein, which was not detected after antigen preabsorption of the antiserum. This size exceeds the size of the protein predicted from the cDNA (44 kD), indicating that the Y1-R is glycosylated. In addition, other bands appeared, that did not disappear after preabsorption of the antiserum with antigen.
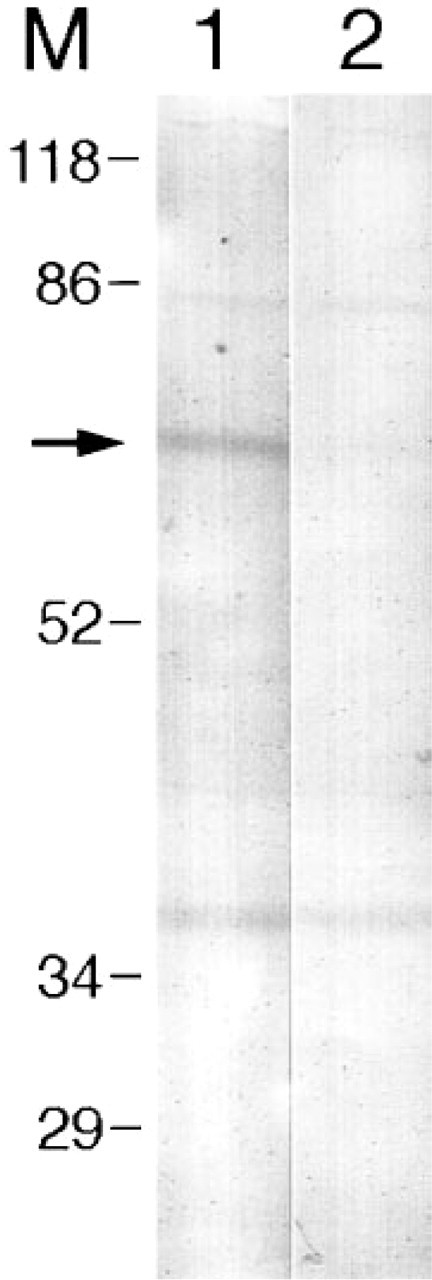
Western blot analysis of extracts from colon external muscle. Lane 1 is detected with Y1-R 308 antiserum. Lane 2 is detected with Y1-R 308 preabsorbed with synthetic Y1-R peptide. Arrow indicates the Y1-R-specific band of 70 kD. M, molecular weight markers expressed in kD.
Discussion
The present study describes the localization of Y1-R in the rat intestine using immunocytochemistry. The presence of Y1-R in the colon was confirmed by identification of a Y1-R-specific protein of 70 kD, corresponding to the previous published size of Y1-R (Sheikh and Williams 1990; Inui et al. 1992; Mao et al. 1996). Throughout the rat intestine, specific Y1-R immunoreactivity could be localized to many nerve cell bodies of the submucosal and myenteric plexi. Previous studies have also demonstrated the presence of NPY/PYY binding sites in submucosal and myenteric neurons by receptor autoradiography (Walsh et al. 1993; Mao et al. 1996). These studies, however, were not able to differentiate between NPY/PYY receptor subtypes. Our present results suggest that Y1 receptors are responsible for at least a part of the NPY/PYY binding observed. Our data further agree with a study demonstrating expression of Y1-R in human colon submucosal and myenteric nerve cell bodies by in situ hybridization (Wharton et al. 1993).
To characterize the Y1-R-positive neurons, we studied the co-localization of Y1-R with the neuronal markers NPY, VIP, and NOS. Interestingly, Y1-R was found to be present in populations of NPY-, VIP-, and NOS-immunoreactive neurons. The presence of NPY-containing nerve fibers adjacent to Y1-R-positive nerve cell bodies suggests that NPY acts as a neurotransmitter by interacting with Y1-R in enteric neurons. The co-localization of NPY and Y1-R in submucosal neurons further indicates a possible autocrine role of NPY in these neurons. In addition, Y1-R-positive enteric neurons may respond to PYY released from endocrine cells situated in the intestinal epithelium. PYY has been suggested to be the mediator of the phenomenon termed “ileal brake,” which results in decreased intestinal transit in the presence of unabsorbed nutrients in the intestinal lumen (Mannon and Tailor 1994). It has further been suggested that the inhibitory effects of NPY/PYY on colon motility are mediated in part by inhibition of excitatory enteric neurons (Hellström 1987). Therefore, it is possible that NPY/PYY indirectly affect intestinal motility through interaction with Y1-R in the myenteric neurons. The action of PYY may additionally or alternatively be transmitted through Y1-R-positive sensory neurons, which have been postulated to reside in the submucosa (Gershon et al. 1994).
The co-localization of Y1-R and NOS in myenteric neurons indicates the possibility that NPY/PYY-binding to Y1-R is followed by regulation of NO release from the neurons. NO is known to be released from enteric neurons on electric stimulation, thereby inducing nonadrenergic, noncholinergic relaxation of smooth muscle (Gershon et al. 1994). This proposed mechanism for NPY/PYY action is supported by a study demonstrating that the motor excitation induced by PYY is reduced by inhibition of NOS (Fox-Threlkeld et al. 1993). However, other mechanisms underlying the inhibitory effects of NPY/PYY on intestinal motility have also been suggested (Wiley and Owyang 1987; Sawa et al. 1995).
In addition to inhibiting gastrointestinal motility, PYY and NPY represent modulators of intestinal ion transport (Saria and Beubler 1985; Cox et al. 1988). The fact that submucosal neurons co-storing Y1-R and VIP innervate the mucosa suggests the possibility that the antisecretory effects of NPY/PYY are mediated in part by VIP released from Y1-R-positive neurons. VIP is a polypeptide considered to play a role in the regulation of intestinal ion transport (Waldman et al. 1977). In addition, a recent report suggests that PYY inhibits colon electrolyte transport via presynaptic as well as postsynaptic sites (Nakanishi et al. 1996), whereas other studies demonstrated that neuronal blockade did not interfere with the NPY/PYY response (Cox et al. 1988; Ballantyne et al. 1993). The postsynaptic sites for NPY/PYY action may be the enterocytes themselves, because studies have demonstrated the existence of PYY-preferring receptors in the intestinal epithelium (Laburthe et al. 1986; Mannon et al. 1993). The fact that immunoreactive Y1-R was localized only to very few endocrine-like cells in the intestinal epithelium in our study indicates that the majority of the PYY binding sites in the mucosa belong to another receptor subtype.
Our results further demonstrate that the Y1-R is present in the cytoplasm as well as at the surface of the immunopositive nerve cell bodies and endocrine cells. A cytoplasmic localization of Y1-R immunoreactivity has previously been shown in neurons in the dorsal root ganglia and in the medulla oblongata (Zhang et al. 1994; Yang et al. 1996). This subcellular localization could be explained by a high rate of synthesis of the receptor.
The presence of Y1-R immunoreactivity in endothelial cells confirms a previous study localizing Y1-R mRNA to the intima and the media of human colon blood vessels (Wharton et al. 1993). From the figure included by Wharton et al., it appears that the highest concentration of silver grains was localized over the intima. However, these authors reported a size of the Y1-R mRNA transcript different from the sizes reported by other groups (Eva et al. 1990; Larhammar et al. 1992; Nakamura et al. 1995), indicating that the specificity of the probe must be questioned. In addition, recent studies have indicated that the NPY-induced vasoconstriction in human cerebral and subcutaneous resistance arteries is mediated by locally expressed Y1-R (Nilsson et al. 1996a,b). The complete role of the endothelium in NPY-mediated vascular events has yet to be established, because both endothelium-dependent and endothelium-independent mechanisms have been described (Daly and Hieble 1987; Pernow and Lundberg 1988; Budai et al. 1989).
Finally, the present study describes a double staining method employing antibodies from the same species. Double immunocytochemistry can easily be performed when the primary antisera are derived from different species. However, it is often desirable to evaluate cellular co-localization of two different antigens that are recognized by antisera raised in the same species (Larsson 1988). By detecting the sites for the first antigen-antibody reaction by deposition of tyramide-conjugated fluorochrome, the reagents used in the first immunocytochemical staining cycle were protected against reactivity with reagents used in the second staining cycle. While this study was in progress, Hunyady et al. (1996) published results in agreement with this observation. In addition, we now describe that the tyramide deposition does not prevent co-detection of two different antigens co-localized in the same cells.
In summary, our present data demonstrate the localization of Y1-R immunoreactivity in myenteric and submucosal nerve cell bodies co-storing NPY, VIP, or NOS, in endothelial cells, and in a few endocrine-like cells in the rat intestine, thereby indicating potential sites for NPY and PYY action. Similar localization studies of the other NPY/PYY/PP receptor subtypes will shed additional light on the mechanisms underlying the NPY/PYY/PP-induced actions on the gastrointestinal tract.
Footnotes
Acknowledgements
Supported by a grant from the Danish National Research Fund (Center for Gene Regulation and Plasticity in the Neuroendocrine Network) and by the Danish Biotechnology Program (Center for Medical Biotechnology).
