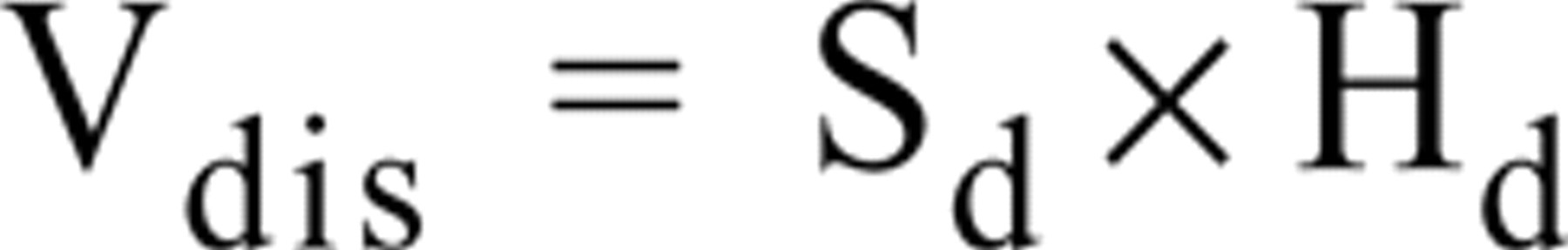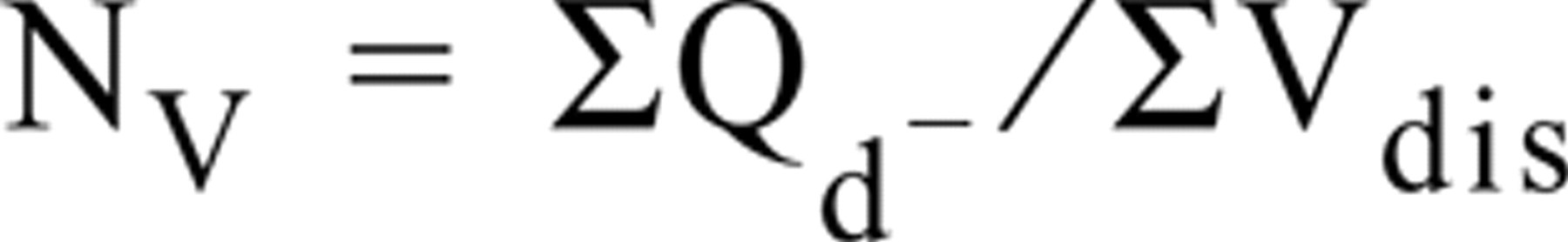Abstract
This study was designed to investigate (a) the presence of protein gene product 9.5 (PGP 9.5), ubiquitin, and neuropeptide Y (NPY) in the neuroendocrine and secretory epithelium of the human normal prostate and its secretions, and (b) the changes in immunoreactivity to these proteins in men with benign prostatic hyperplasia. Western blotting and light microscopic immunohistochemistry techniques were used and the numerical density of immunoreactive neuroendocrine cells, and the volume fractions of immunostained secretory epithelium were evaluated. Western blotting revealed the presence of the three antigens in both tissue homogenates and prostate secretion. Some neuroendocrine cells immunoreacted to PGP 9.5 and NPY in all the prostate regions of control specimens. Ubiquitin immunoreactivity was detected in the nuclei from both basal cells and secretory epithelial cells. The cytoplasm of the secretory cells and the glandular lumen also showed immunostaining for the three proteins. The numerical densities of both PGP 9.5 and NPY neuroendocrine cells were lower in hyperplasia than in controls. No differences in the volume fraction occupied by epithelial immunostaining to both proteins was found between hyperplastic and control prostates. We concluded that (a) PGP 9.5 and NPY, but not ubiquitin, are common antigens in both neuroendocrine and secretory prostate cells, (b) the three immunoreactive proteins contribute to the prostate secretions, and (c) the secretion of ubiquitin is markedly diminished in the hyperplastic epithelium. (J Histochem Cytochem 48:1121–1130, 2000)
Protein gene product 9.5 (PGP 9.5), an ubiquitin carboxyl-terminal hydrolase (Wilkinson et al. 1989) described in neurons and neuroendocrine cells (Doran et al. 1983; Jackson et al. 1985; Rode et al. 1985; Kent and Rowe 1992; Properzi et al. 1992), was also detected by Western blotting in testicular tissues (Wilkinson et al. 1992). On the other hand, ubiquitin, a protein involved in extralysosomal protein degradation, modulation of some membrane receptors, response to heat shock, and turnover of cytoskeletal elements (Fried et al. 1987; Murti et al. 1988; Crooke et al. 1990), has been detected in human seminal plasma (Lippert et al. 1993). Recently, PGP 9.5 and ubiquitin immunoreactivities were reported in the epididymal epithelium of both rat and human (Santamaráa et al. 1993; Martán et al. 1995; Fraile et al. 1996). These findings suggest that both proteins play a role in spermatozoon maturation because they are secreted by epithelial cells from specific regions of the epididymis and become linked to spermatozoa (Lippert et al. 1993; Santamaráa et al. 1993; Martán et al. 1995). It is also possible that immunoreactivity to PGP 9.5 and ubiquitin could be detected in prostate glandular epithelia, as was demonstrated for other neural glycoproteins (Kujat et al. 1990).
Neuropeptide Y (NPY) is a small polypeptide expressed in neurons of the central and peripheral nervous systems (Tatemoto et al. 1982). Abundant NPY fibers run parallel to muscle fibers to innervate smooth muscle fibers and vessels in the prostate (Chapple et al. 1991; Properzi et al. 1992; Iwasa 1993; Jen and Dixon 1995). Some authors (Kanzaki et al. 1996) have detected intense NPY immunoreactivity in cultured Leydig cells, and NPY transcripts have been detected in Leydig cells, Sertoli cells, epididymis, and vas deferens of the rat. Therefore, extraneuronal members of the family of NPY-related peptides appear to be involved in the physiology of male reproductive tract. Although many neuropeptides and related substances have been investigated in studies on innervation and neuroendocrine cell interactions in normal and pathological prostates (Chapple et al. 1991; Properzi et al. 1992; Gkonos et al. 1995; Jen and Dixon 1995; Tainio 1995), few studies on the presence of neuropeptide-like substances in epithelial non-neuroendo-crine prostate cells or in prostate secretions have been developed.
In the present work, we hypothesized that some peptides that have been described to perform only endocrine–paracrine activity might also have an exocrine function, and that the secretion of these proteins into the prostate fluid could be relevant for the composition and properties of the seminal plasma (Di-Sant'Agnese et al. 1989; Iwamura et al. 1994). In addition, changes in the expression of these substances in disorders such as benign prostatic hyperplasia (BPH) could add more information about epithelial alterations produced in abnormal prostate growth.
The aims of this study were (a) to examine the distribution of PGP 9.5, ubiquitin, and NPY immunore-activities in both neuroendocrine and secretory epithelium of the human prostate and in the prostate secretion and (b) to investigate the changes in immunoreactivity to those proteins in normal prostate and in BPH.
Materials and Methods
Materials
The prostates from 45 men (aged 60–80 years) who were clinically and histopathologically diagnosed with BPH were obtained by surgery (adenomectomy). In addition, the prostates of five men (aged 60–70 years) with BPH and eight men (aged 20–40 years) without BPH (histologically normal prostates) were obtained from autopsies between 8 and 10 hr after death. The condition for selection of these specimens was the absence of reproductive, endocrine, and related diseases in the patient histories. In addition, prostate secretions from the five men with BPH and from eight normal, young men (controls) were obtained during urologic examination. The age ranges of these patients were the same as indicated for histological and immunocytochemical studies. The procedures followed for obtaining all these materials were in accordance with the guidelines of the ethical committee of the La Princesa Hospital.
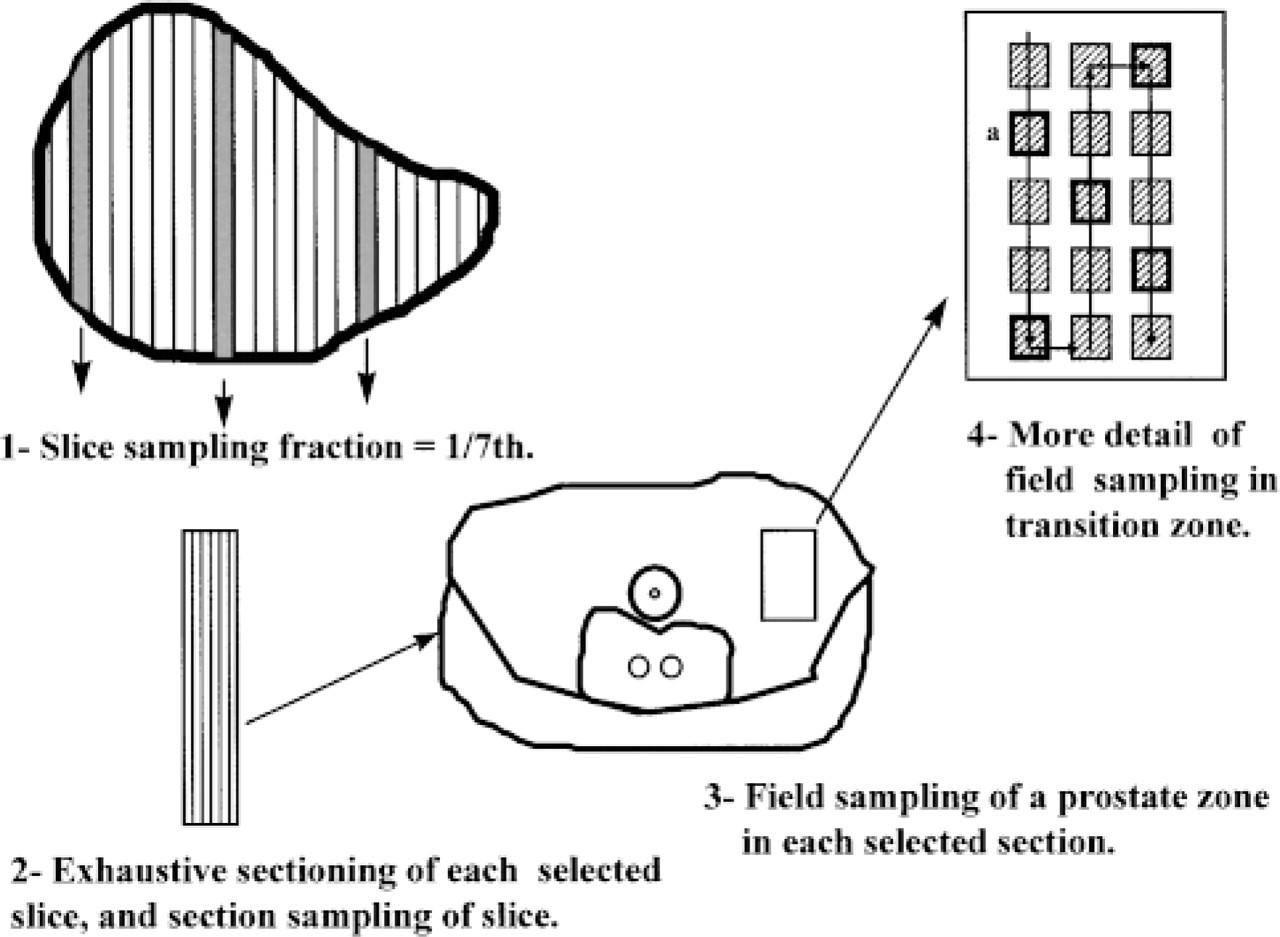
Schematic showing the sampling scheme for slices, sections, and fields in control prostate. (1) The prostate is cut into 2-mm thick slices and a known fraction of the slices is taken. In this example, one-seventh has been taken, the first slice sampled was chosen randomly between 1 and 7 (in the diagram, section 3), and then every seventh slice (systematic random sampling method). (2) Each sampled slice is embedded in paraffin and systematically randomly sampled 15-μm-thick sections were chosen. (3) In each section, systematic random sampling was again performed to select the fields of measurement. The diagram shows the selection of fields in a portion of the transition zone (dotted rectangle). (4) More detail of field sampling. For each sampled zone, an automatic scanning of the microscope stage was performed, with known step lengths in the × and Y directions, starting from a random position, to select a first field (for example, a), and then every three fields until all the selected zone was completely scanned.
Processing of Materials
For morphometric and light microscopic immunohistochemical studies, three 2-mm-wide slices sectioned perpendicularly to the prostate urethra were systematic randomly sampled from each specimen (Gundersen and Osterby 1981) (see diagram of the sampling procedure in Figure 1), fixed for 10 hr in 4% paraformaldehyde in PBS, pH 7.4, and embedded in paraffin.
Western Blotting Analysis
For Western blotting analysis, prostate secretions, unfixed fragments of the three prostate zones (central, transition, and peripheral zones; McNeal 1992) from each control specimen, and unfixed fragments from each BPH specimen were homogenized in 0.5 M Tris-HCl buffer (ph 7.4) containing 1 mM ethylene diamino tetraacetic (EDTA), 12 mM 2-mercaptoethanol, and 1 mM phenylmethyl sulfonylfluoride (PMSF). The homogenates were centrifuged at 10,000 × g for 30 min. After boiling for 2 min at 98C, 25-μg aliquots of protein extracts were separated in sodium dodecyl sulfate (SDS)-polyacrylamide (15% w/v) slab minigels according to the procedure of Laemmli (1970). Electrophoretic blotting onto nitrocellulose and immunological protein detection were carried out as described by Towbin et al. (1979), using either rabbit polyclonal anti-PGP 9.5 (code number RA95103; Ultraclone, Cambridge, UK), rabbit polyclonal anti-ubiquitin (code number Z458; Dako, Glostrup, Denmark), or rabbit polyclonal anti-NPY (code number BH3; Hammersmith Hospital, London, UK) as first antibody, and the rabbit extra-avidin peroxidase staining kit (Sigma Chemicals; Barcelona, Spain) as the second antibody. The purity and specificity of primary antibodies had been tested by the manufacturers and corroborated in previous studies (Jackson et al. 1985; Rode et al. 1985; Santamaráa et al. 1993). The filters were developed by an enhanced chemiluminescence Western blotting analysis according to the procedure described by the manufacturer (Amersham; Poole, UK).
Immunohistochemical Methods
Five-μm-thick serial sections from each slice were immunostained by the avidin-biotin-peroxidase method. Before incubation with the primary antibody, endogenous peroxidase activity of the tissues was blocked with 0.3% hydrogen peroxide in PBS. The tissues were incubated overnight at 4C with the same primary antibodies used for Western blotting: rabbit polyclonal antibody against PGP 9.5 at 1:1000 dilution; rabbit polyclonal antibody to ubiquitin at 1:400 dilution; and rabbit polyclonal antibody to NPY at 1:2000 dilution. To compare the immunoreactivity of PGP 9.5 and NPY with that of the neuroendocrine marker chromogranin A (Cetin 1992), which is a sensitive marker for most of the neuroendocrine cells of the prostate (Speights et al. 1994; Noordzij et al. 1995), some pairs of adjacent serial sections were consecutively immunostained with a mouse monoclonal chromogranin A antibody (code number AM126–5M; Biogenex, San Ramon, CA) at 1:50 dilution and either PGP 9.5 or NPY. Incubations were carried out for 30 min with either a goat anti-mouse (for chromogranin A) or a goat anti-rabbit (for PGP 9.5 or NPY) biotinylated immunoglobulin (Dako). The sections were subsequently treated with an avidin-biotin-peroxidase complex (Dako) and developed with diaminobenzidine (DAB) plus hydrogen peroxide. The sections were then counterstained with methyl green or Harris' hematoxylin, dehydrated in ethanol, and mounted in Depex (Serva; Heidelberg, Germany).
To assess the immunostaining specificity, negative controls obtained by either omitting primary antibodies or using these antibodies preabsorbed with a tenfold excess (by weight) of purified antigens were also analyzed. The antibody–antigen complexes were incubated in PBS, pH 7.4, overnight at 4C and centrifuged for 15 min before use.
Morphometric Methods
Estimation of the amount of neuroendocrine cells that were chromogranin-, PGP 9.5-, or NPY-immunoreactive was performed using the technique of the optical disector, an unbiased stereological method (Bjugn and Gundersen 1993; Wreford 1995; Mayhew and Gundersen 1996). Briefly, the procedure used was as follows. Groups of three 15-μm-thick sections for each immunostaining type were chosen by systematic random sampling from each tissue slice selected in each specimen. Such section thickness is obligatory for performance of the optical disector method (Mayhew and Gundersen 1996). To improve the penetration of the antibodies in the sections, 0.03% Triton-100 was added to the diluents in the immunohistochemical technique.
An average of 100 fields per section in each prostate zone from controls and the same number of fields per section in each BPH specimen were systematically randomly sampled (Figure 1) and used to count the number of immunoreactive cells. All measurements were performed using an Olympus microscope with a × 100 objective (numerical aperture 1.4) at a final magnification of ×1200. The microscope was connected to a videocamera and supplied with a motorized stage connected to a Commodore Amiga 2000 computer. The software used (Stereologic Software Package, GRID; Interactivision, Silkeborg, Denmark) (Martán et al. 1997) commands the XY movement of the stage and allows the automatic selection of microscopic fields. The program generates the disector grid that was superimposed to the microscopic image captured by the videocamera and projected to the monitor. The disector volume (Vdis) was calculated with the formula
where Sd = 1300 μm2 (area of the disector grid), Hd = 10 μm [distance between the two focal planes chosen for determining the disector volume into the tissue section; this distance was measured by means of a microcator (Heidenhain; Traunreut, Germany) connected to the Z displacement of the microscope stage]. Because the total thickness of the section was 15 μm, a safeguard space of 2.5 μm above and below the optical section was maintained to avoid artifacts produced in the physical surface of the section. All these calculations lead to Vdis = 13,000 μm3.
The neuroendocrine cell nuclei eligible to be counted were determined by using Sterio's convention (Sterio 1984); only those nuclei that appeared in the upper focal plane and were not observed in the lower focal plane were counted (Qd −). The numerical density of cells (nuclei) or number of cells per unit of epithelial volume (NV) was then obtained by the formula
where ΣVdis = total number of disectors applied in each selected section multiplied by Vdis. NV was calculated as number of cells per mm3 of epithelial volume in each prostate zone or BPH specimen.
To quantify the immunostaining of epithelial non-neuroendocrine cells for PGP 9.5, ubiquitin, and NPY, their respective volume fractions were measured, i.e., the ratio between immunostained epithelial area and the reference area of prostate epithelium (excluding the glandular lumen). These volume fractions were estimated in an average of 25 systematically randomly sampled light microscopic fields (an average of 194,000 μm2 per field) in five systematically randomly selected sections of each control prostate area or BPH specimen. These measurements were performed counting the points hitting either immunostained epithelial area or the reference area, using the GRID software package which provides a counting point grid with a point associate area, A(p) = 45 μm2. The final magnification used for these measurements was × 500.
Statistical Analysis
For each parameter studied, the means ± SD were calculated. The differences among prostate zones and between transition zone of controls and BPH specimens were evaluated by ANOVA. Comparison between each pair of means was performed using the Fisher and Behren's test.
Results
Comparison of the prostates obtained during surgery with those from autopsies showed neither histological nor immunohistochemical changes.
Western Blotting Results
In the extracts of all prostate specimens studied, either normal or hyperplastic, Western blotting analysis revealed three immunoreactive bands, located at approximately 7 kD (NPY), 27 kD (PGP 9.5), and 60 kD (ubiquitin) (Figure 2a). In the prostate secretions, Western blotting analysis revealed two ubiquitinimmunoreactive bands located at approximately 66 and 97 kD, one band PGP 9.5-immunoreactive at 27 kD, and two NPY-immunoreactive bands located at 7 and 97 kD (Figure 2b).
Immunohistochemical Qualitative Results
PGP 9.5 immunoreactivity was observed around the nucleus and apical cytoplasm in most epithelial secretory cells throughout all prostate zones (Figure 3). In several tubuloacini, PGP 9.5 immunostaining was also found in the glandular lumen (Figures 3 and 4). Cell debris was occasionally observed in the glandular lumen (Figure 3). Chromogranin-positive neuroendo-crine cells were detected scattered among the epithelial secretory cells (Figure 5a). Some of these cells were also immunoreactive for PGP 9.5 (Figure 5b). Many PGP 9.5-immunoreactive nerves were detected in the connective tissue of the capsule, around the prostate vessels, and in the vicinity of tubuloacini (Figure 5b).
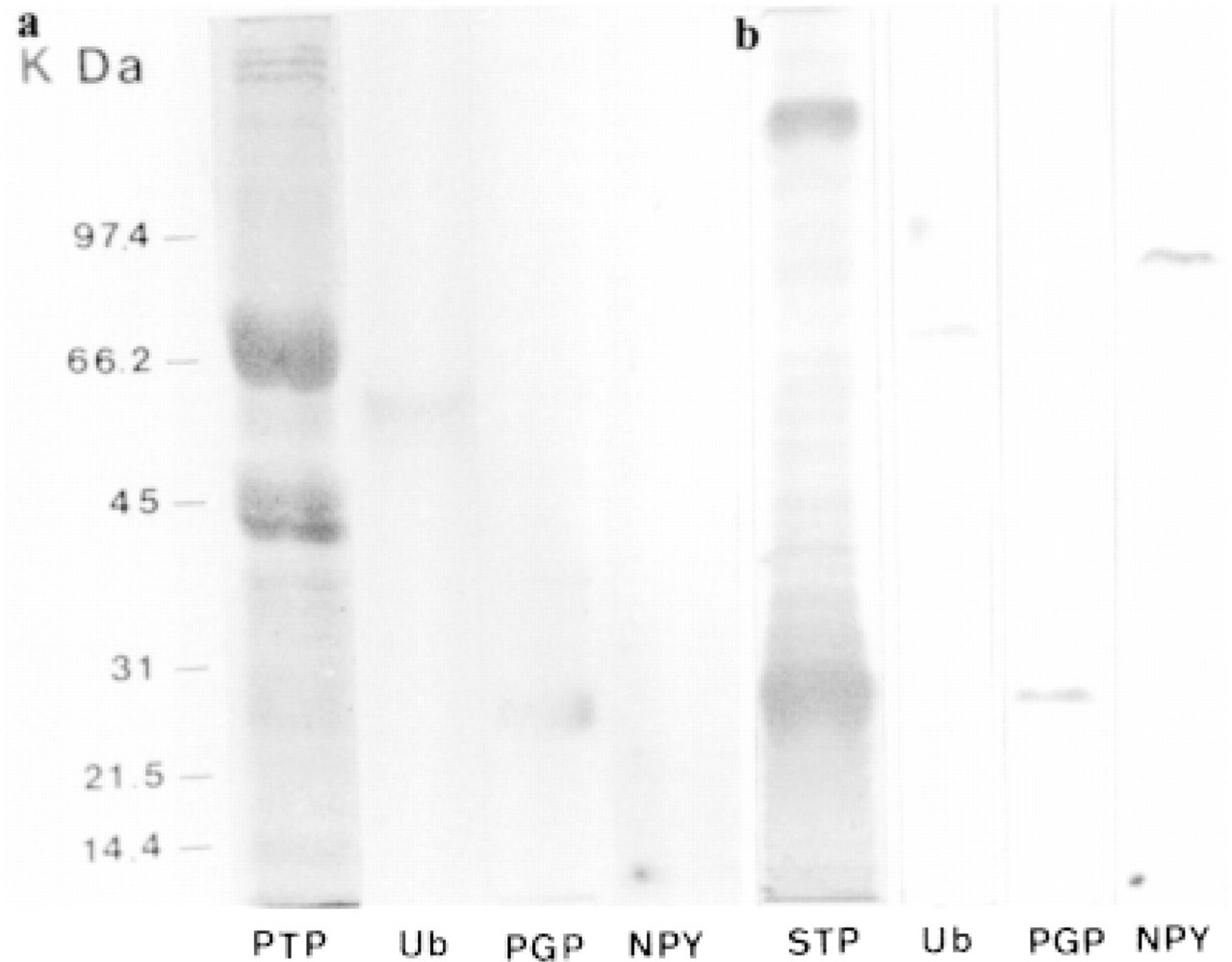
Western blotting analysis in extracts from normal prostate (
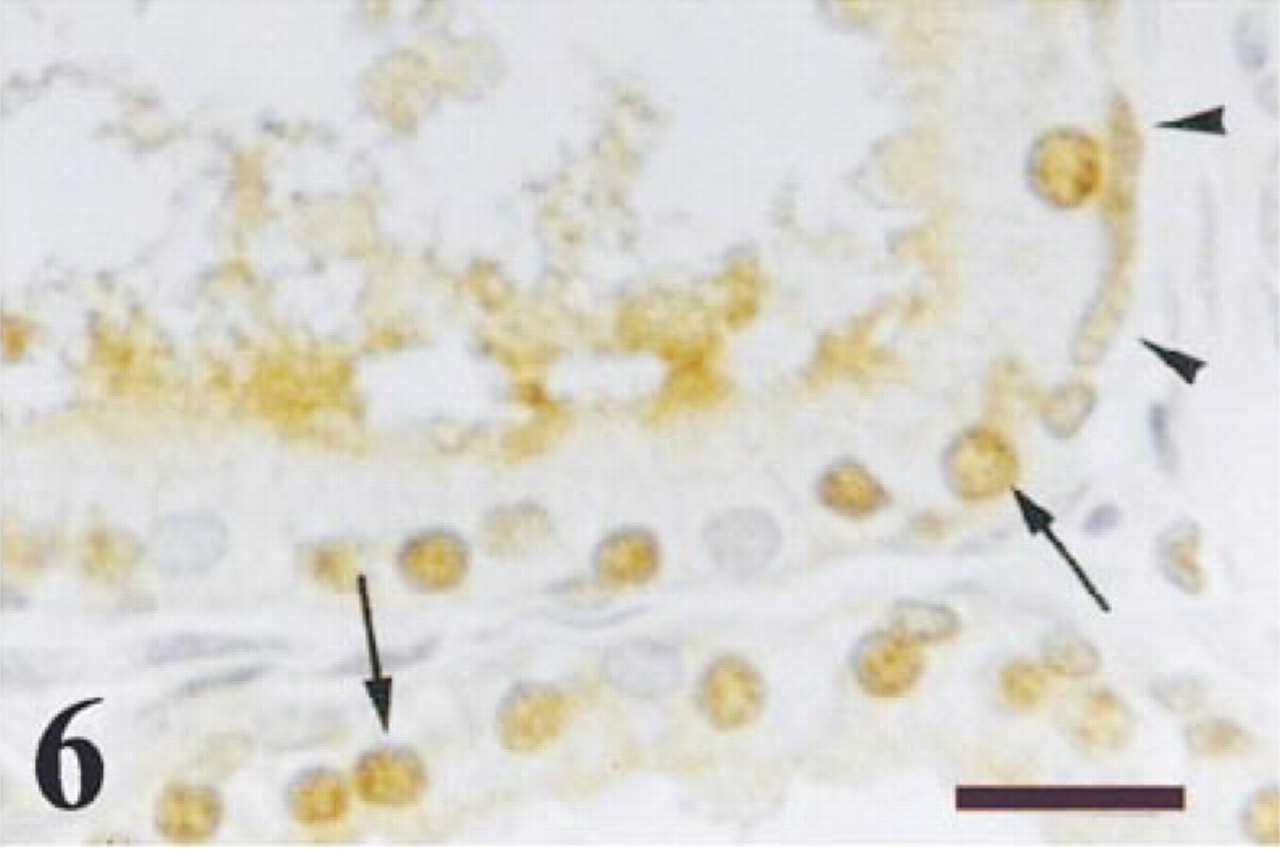
Ubiquitin immunoreactivity was detected in both basal and secretory epithelial cells in all prostate zones (Figure 6). This immunostaining was observed in the nuclei and the cytoplasm in both cell types. In addition, in many areas the glandular lumen and the apical border of the secretory epithelial cells showed intense ubiquitin immunoreaction (Figure 6). Weak ubiquitin immunoreactivity was detected in some cells from the fibromuscular stroma.
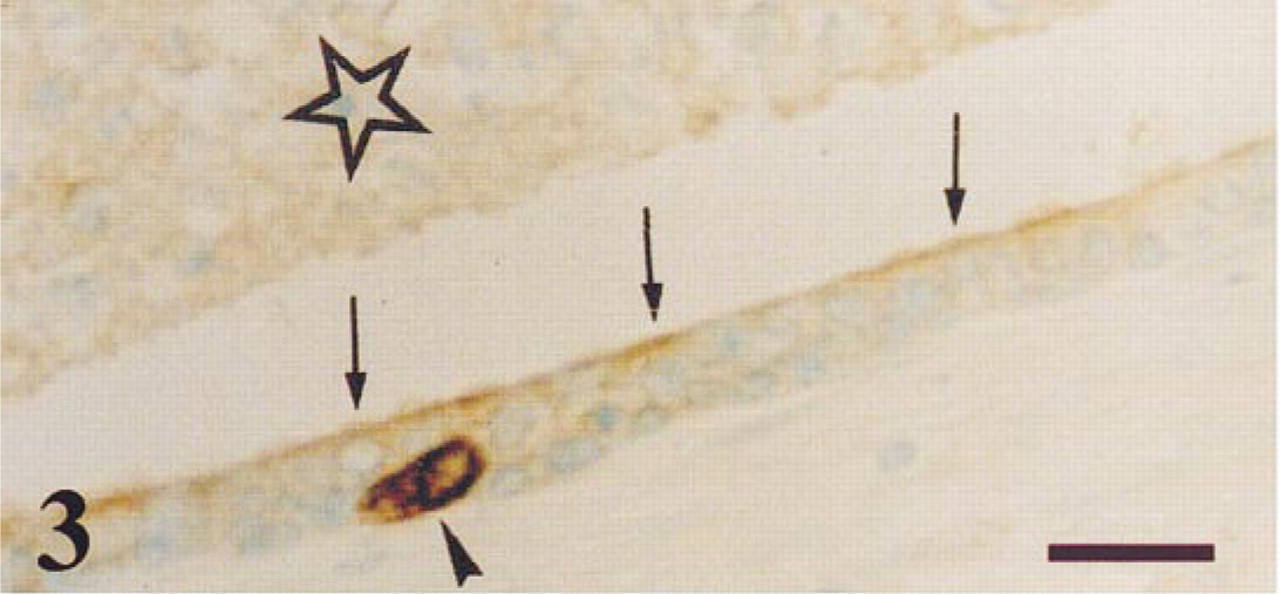
Peripheral zone of a normal prostate. PGP 9.5 immunoreaction is observed in the apical border (arrows) of epithelial secretory cells, in the glandular lumen (star), and in a neuroendocrine cell (arrowhead). Some non-immunostained cell debris is observed in the lumen. Bar = 14 μm.
BPH specimen immunostained for PGP 9.5. Immunoreaction was observed in the epithelial cells and in the glandular lumen (star) of a hyperplastic nodule. Bar = 14 μm.
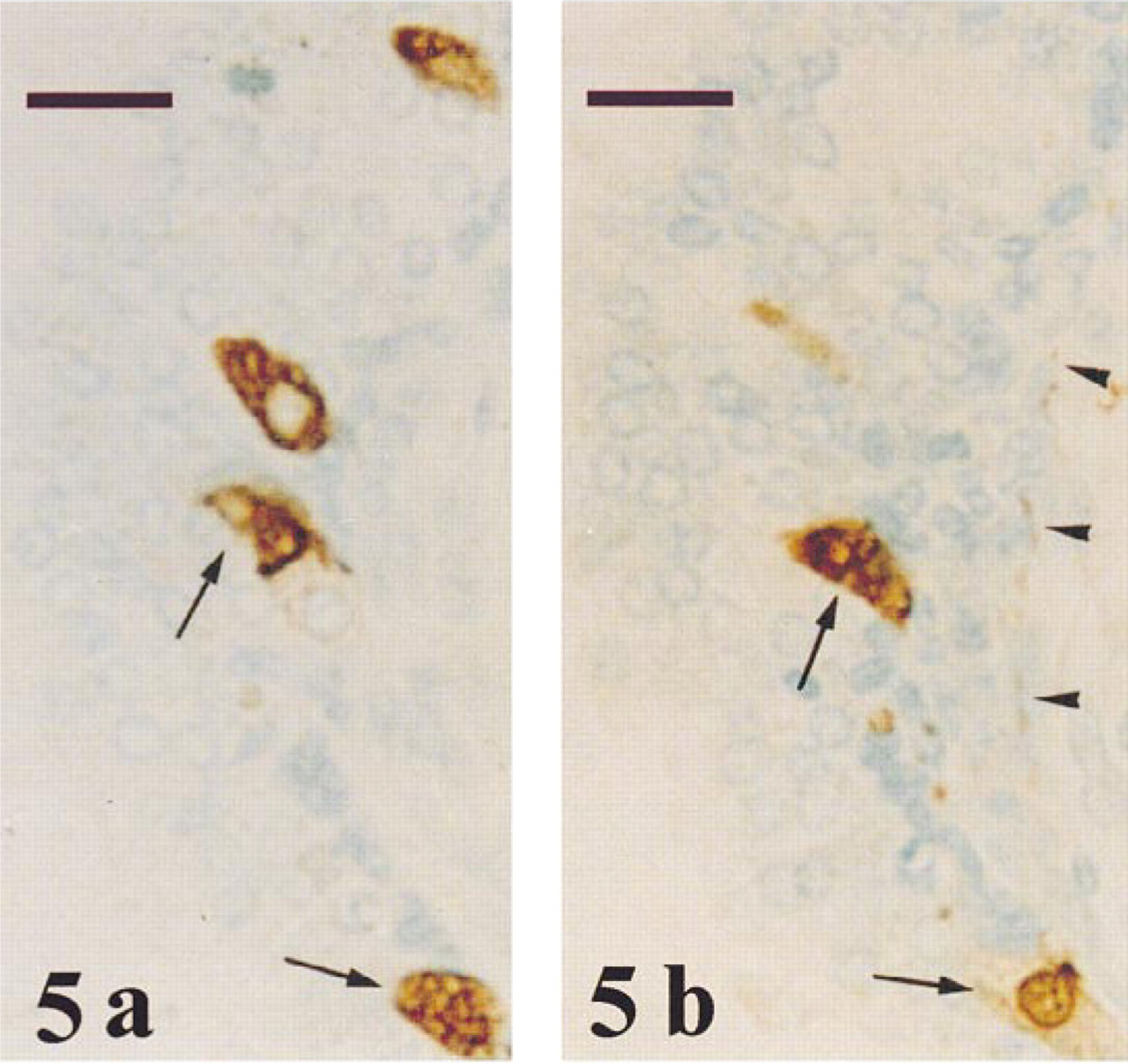
Transition zone of a normal prostate after immunostaining of two adjacent sections for chromogranin (
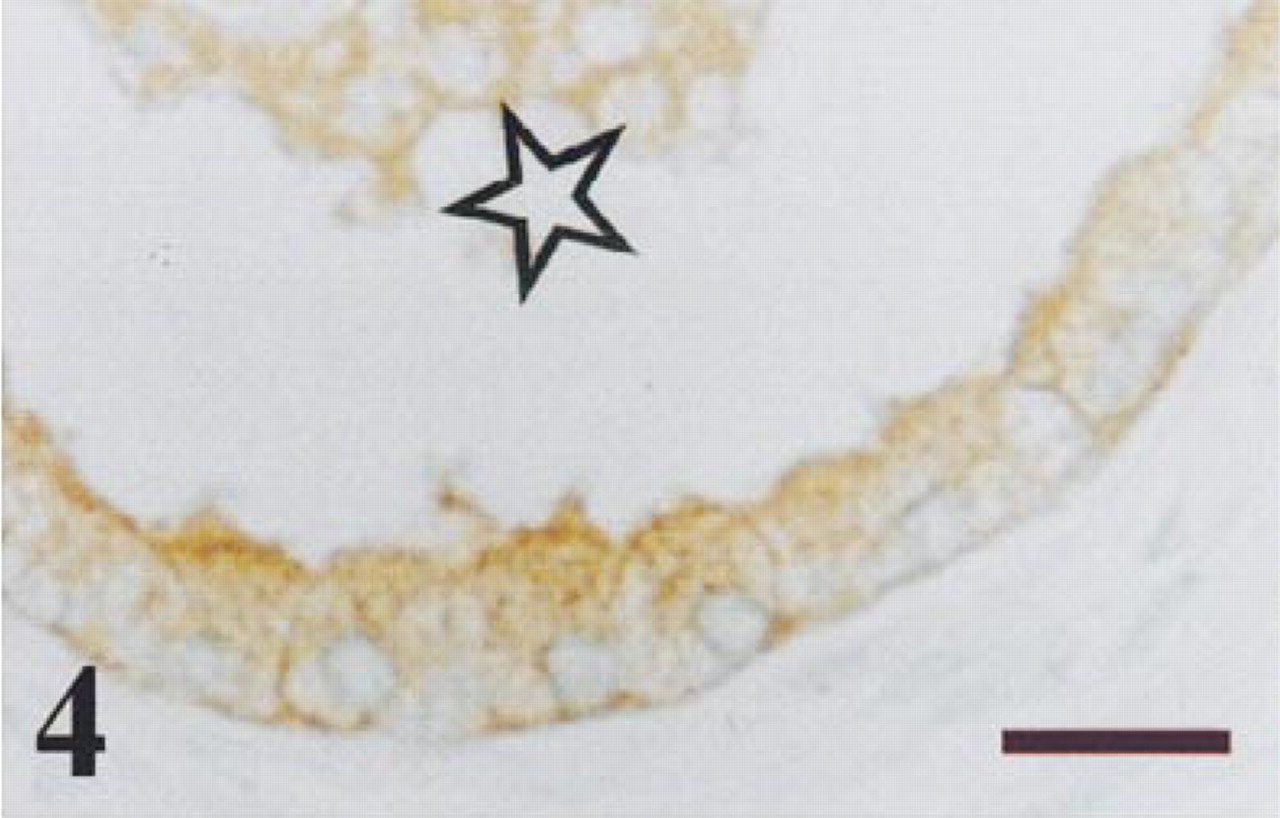
Transition zone of a normal prostate immunostained for ubiquitin. Immunoreaction was detected in the nuclei of both basal cells (arrowheads) and secretory epithelial cells (arrows), as well as in the apical cytoplasm and luminal secretion of secretory cells. Bar =14 μm.
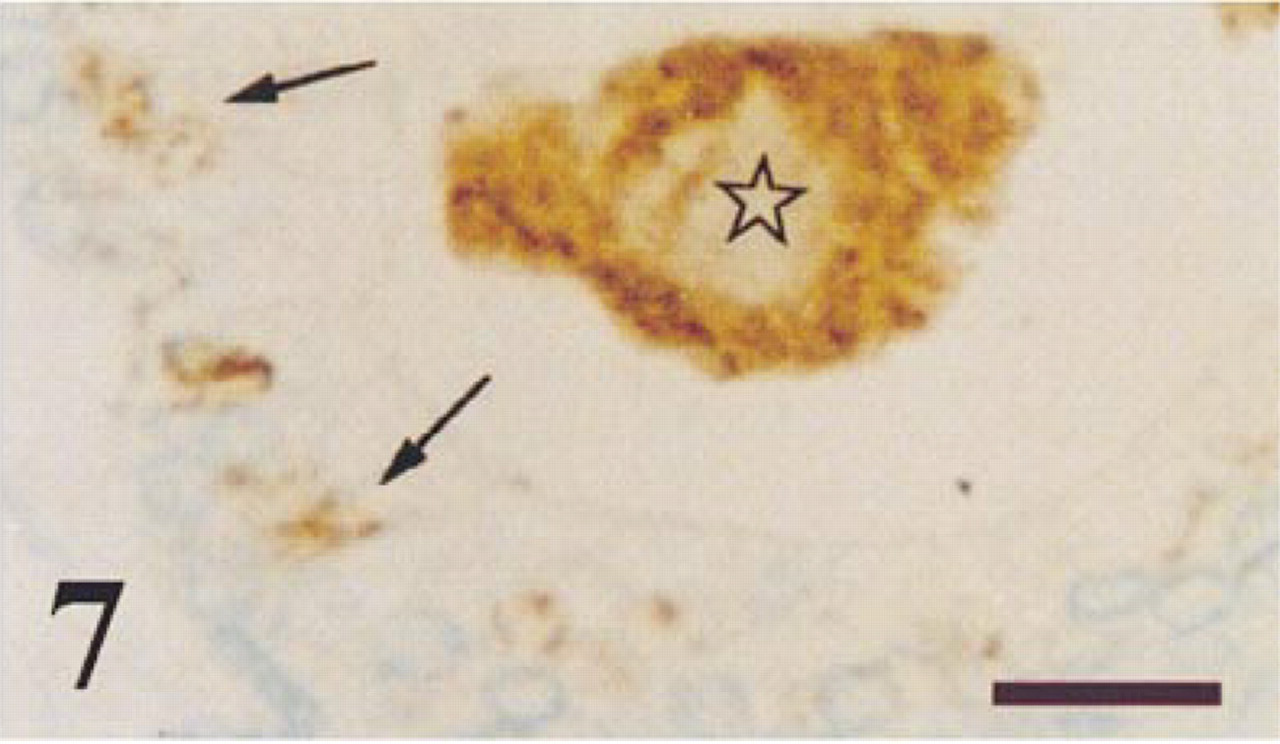
Prostate from a man with BPH immunostained for NPY. Granular immunoreaction was observed in the secretory cells of some tubuloacini (arrows) and in the glandular lumen (star). Bar = 14 μm.
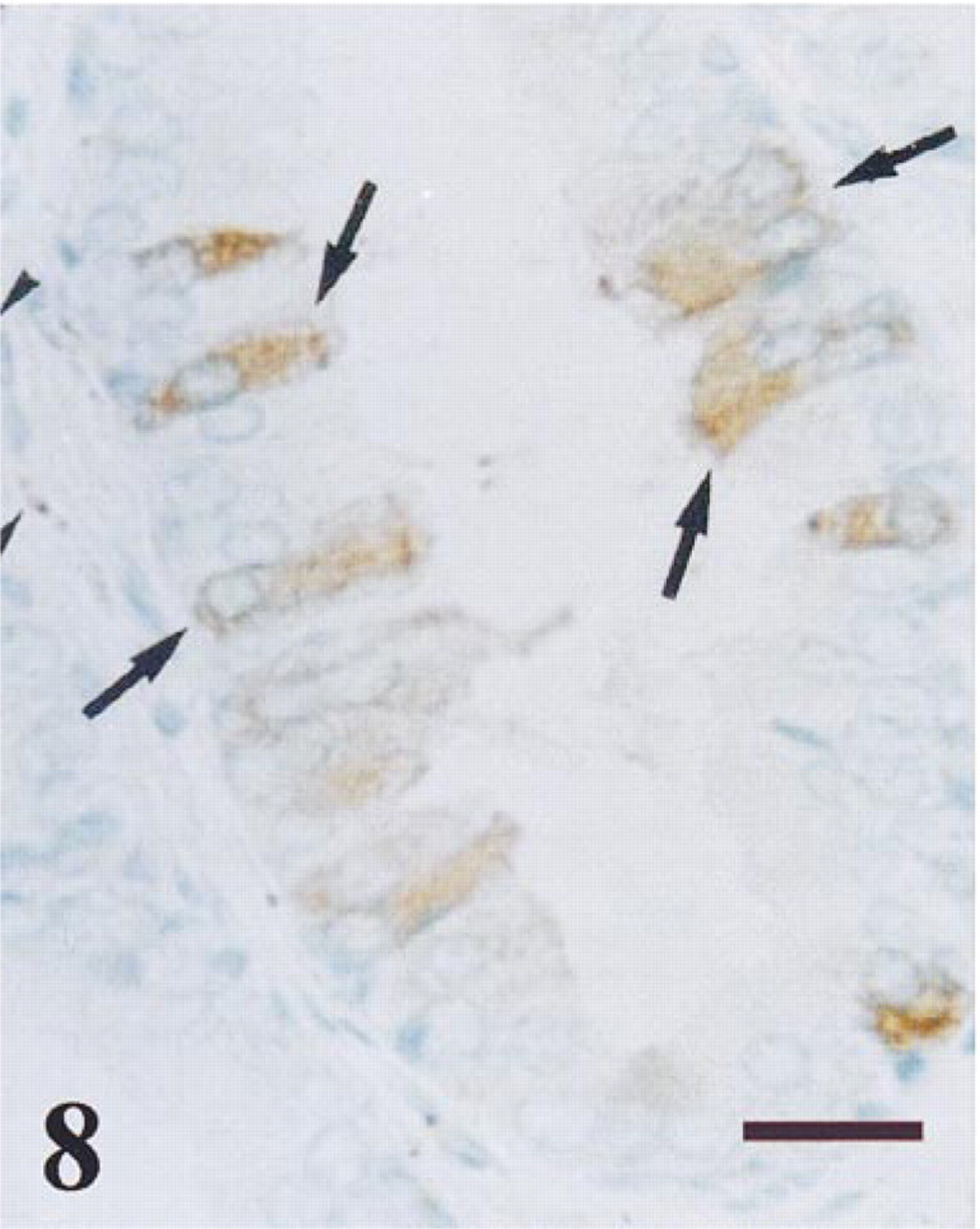
Transition zone of a normal prostate immunostained for NPY. Granular immunoreactivity was observed in several secretory cells (arrows). NPY-positive nerves (arrowheads) were also seen. Bar = 14 μm.
Central zone of a normal prostate after immunostaining of two adjacent sections for chromogranin (
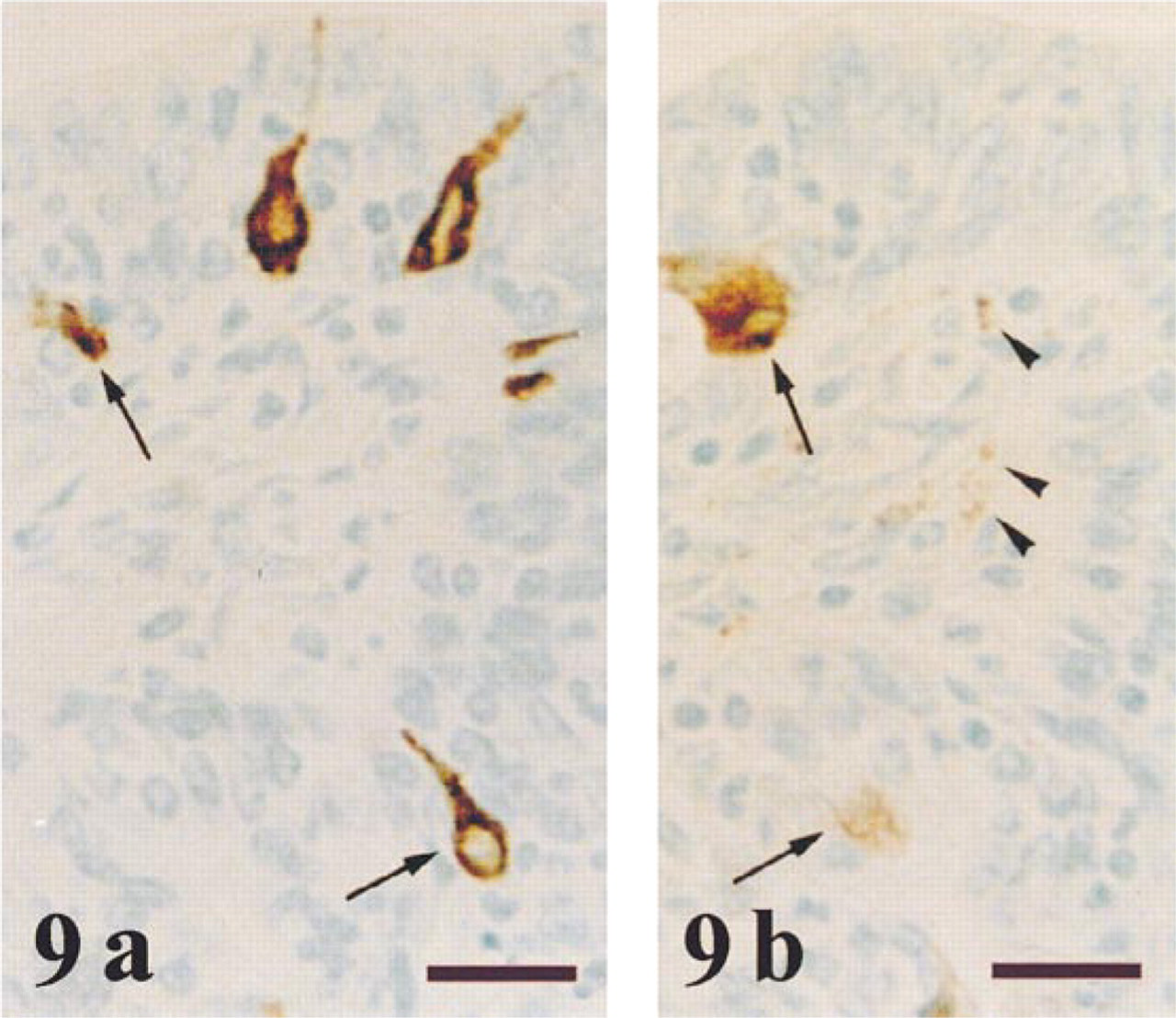
NPY immunoreaction was detected in some dispersed areas, mainly in the central and transition prostate zones. This immunoreaction was granular and was localized in the cytoplasm of many secretory epithelial cells (Figures 7 and 8). Occasionally, the glandular lumen also immunoreacted to NPY (Figure 7). In some chromogranin-positive neuroendocrine cells that were scattered throughout the prostate (Figure 9a), NPY immunoreaction was also detected (Figure 9b). NPY-immunoreactive nerve fibers were observed around tubuloacini in all the prostate zones (Figures 8 and 9b). No stromal immunostaining for NPY was seen.
In both control and BPH specimens, the sections incubated either omitting primary antibodies or using each of the antibodies employed in this study preabsorbed with the corresponding antigen showed no immunostaining in any tissue component (Figures 10–15).
Neuroendocrine Cells: Quantitative Results
The NV of chromogranin, PGP 9.5, and NPY neuroendocrine-immunoreactive cells was significantly higher (p < 0.05) in the transition zone of control prostates than in the prostates of BPH cases. In control cases, the NV of chromogranin-, PGP 9.5-, and NPY-immunostained cells was more elevated (p < 0.05) in the transition zone than in both central and peripheral zones. These results are shown in Table 1.
In transition zone of controls, 67% of the neuroendocrine cells that were immunoreactive to chromogranin also showed immunoreaction to PGP 9.5, whereas 15% of those chromogranin-positive cells were immunostained for NPY. In BPH specimens, 6% of chromogranin-immunoreactive neuroendocrine cells were immunostained for PGP 9.5, and 12% of chromogranin-immunostained cells were also positive for NPY.
Numerical densities of neuroendocrine prostate cells, and evaluation of epithelial secretory cell immunostaining a
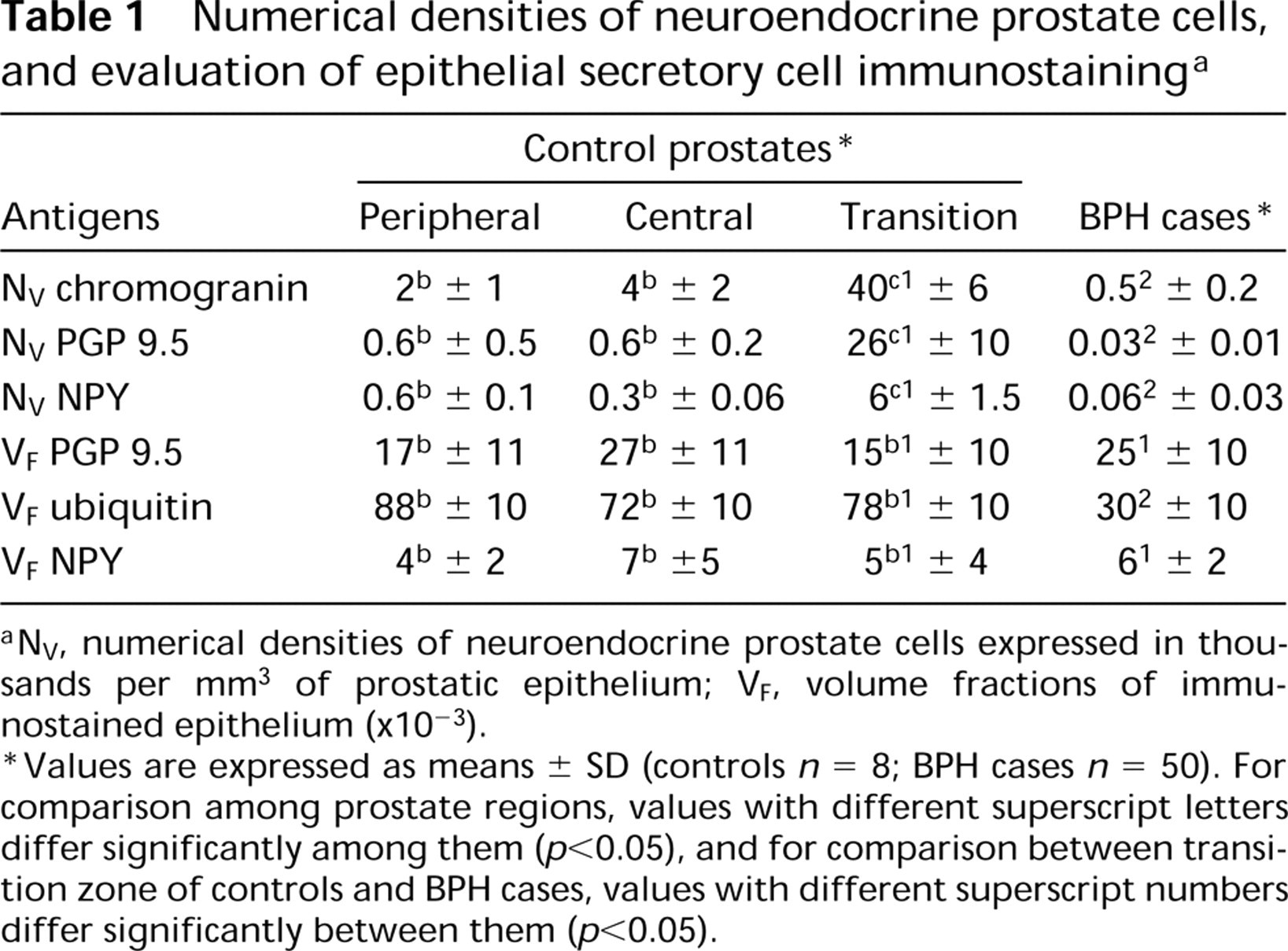
NV, numerical densities of neuroendocrine prostate cells expressed in thousands per mm3 of prostatic epithelium; VF, volume fractions of immunostained epithelium (×10−3).
Values are expressed as means ± SD (controls n = 8; BPH cases n = 50). For comparison among prostate regions, values with different superscript letters differ significantly among them (p < 0.05), and for comparison between transition zone of controls and BPH cases, values with different superscript numbers differ significantly between them (p < 0.05).
Epithelial Immunostaining: Quantitative Results
The quantitative results for epithelial non-neuroendocrine immunostaining are indicated in Table 1. Volume fraction of PGP 9.5 epithelial immunostaining showed no significant differences among prostate zones in controls and between transition zone of controls and BPH cases. Volume fraction for ubiquitin immunostaining did not differ significantly among prostate zones in controls. Nevertheless, this volume fraction was significantly lower in BPH than in the transition zone of control cases (p < 0.05).
Volume fraction for NPY immunostaining did not show significant differences among prostate zones in control cases. No significant differences in this fraction were found between BPH and the transition zone of control cases.
Discussion
The specificities of all immunoreactions reported in the present study were confirmed by Western blotting methods. These methods also demonstrated the presence of PGP 9.5, NPY, and ubiquitin immunoreactivities in the prostate secretions.
Transition zone of a control prostate after immunostaining for PGP 9.5. The immunoreactivity is observed in luminal secretions (star), apical border of epithelium (arrowheads), and nerve fibbers surrounding the acini (arrows). Bar =14 μm.
Adjacent section to that observed in Figure 10, immunostained with antibody against PGP 9.5 absorbed with the antigen. No immunoreaction is detected. Bar = 14 μm.
Peripheral zone of a control prostate after immunostaining for ubiquitin. The immunoreactivity is observed in some epithelial nuclei (arrowheads) and in the apical border of epithelium (arrows). Bar =14 μm.
Adjacent section to that observed in Figure 12, immunostained with antibody against ubiquitin absorbed with the antigen. A remarkable decrease of immunoreaction is detected in both nuclei and cytoplasm. Bar =14 μm.
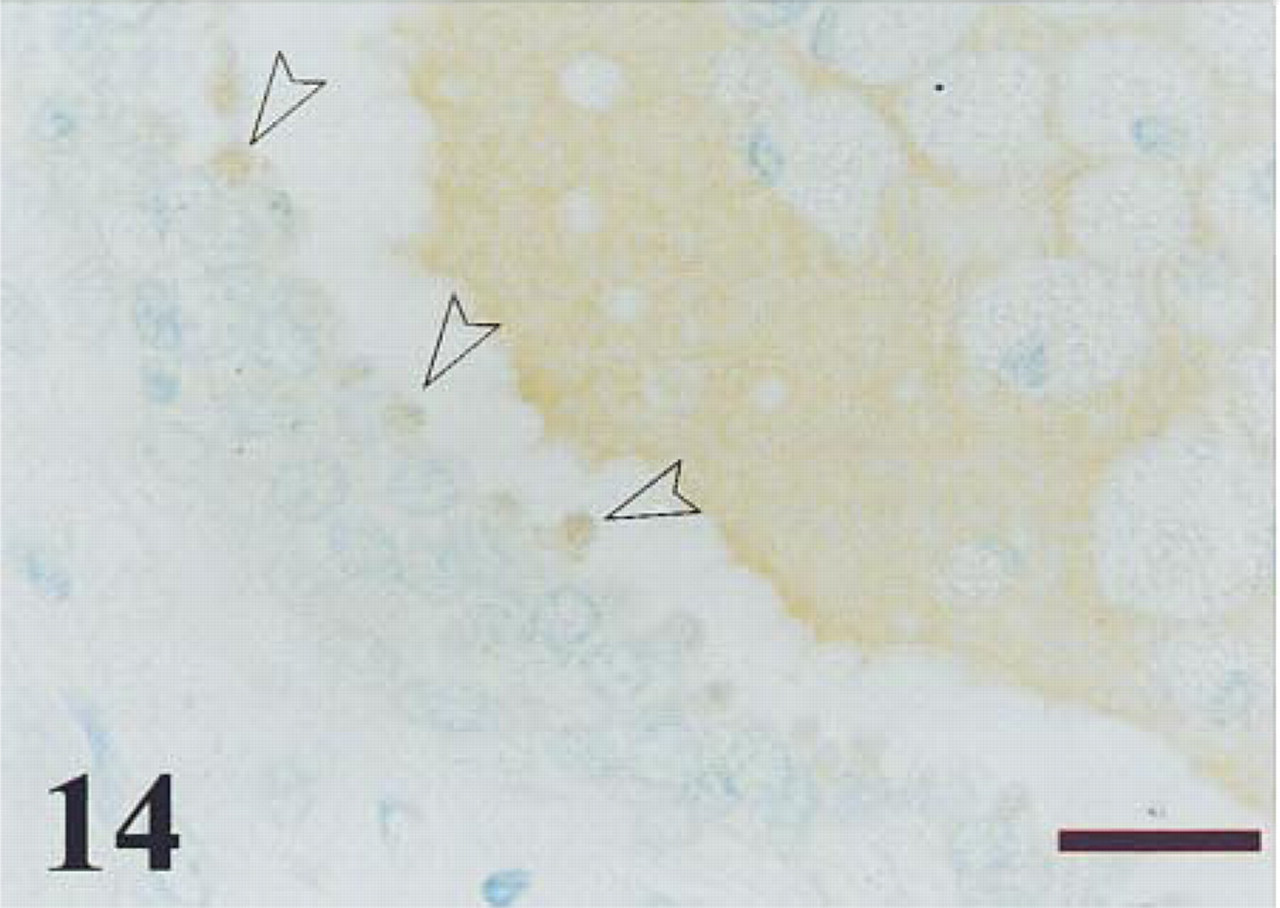
BPH specimen after immunostaining for NPY. Immunoreactivity is observed in the apical border of the epithelium (arrowheads) and in the luminal secretions. Non-immunoreactive cell debris is also observed in the lumen. Bar = 14 μm.
Adjacent section to that observed in Figure 14, immunostained with antibody against NPY absorbed with the antigen. No immunoreaction is detected. Bar = 14 μm.
The estimations of relative amounts of neuroendo-crine cells that were immunoreactive for chromogranin, PGP 9.5, and NPY agree with previous observations (Cohen et al. 1993; Noordzij et al. 1995) showing a predominance of neuroendocrine cells in the transition zone of the normal prostate.
A significant decrease in the global population of chromogranin-immunoreactive neuroendocrine cells and in those cells that co-localized chromogranin and either PGP 9.5 or NPY was found in BPH specimens. This finding agrees with previous studies of large hyperplastic prostate nodules (Cockett et al. 1993; Noordzij et al. 1995), although a relative increase of neuroendocrine cells was also reported in small, early hyperplastic nodules (Cockett et al. 1993). The specimens analyzed in the present study were obtained from patients with long standing BPH and therefore without small, early hyperplastic nodules.
Not all neuroendocrine cell subpopulations undergo the same rate of decrease in BPH. Therefore, the percentage of chromogranin-positive cells that expressed NPY shows little change in BPH specimens, whereas the proportion of chromogranin-positive cells that co-localized PGP 9.5 was significantly decreased in BPH.
PGP 9.5 and ubiquitin immunoreactivities were also observed in the cytoplasm of epithelial secretory cells and in the glandular lumen. Cytoplasmic localization of ubiquitin has been reported in other cell types, such as hepatoma cells (Schwartz et al. 1984) and epithelial columnar cells from human epididymis (Fraile et al. 1996). In addition, high levels of ubiquitin have been found in prostate tissue homogenates (Lombardo et al. 1996). The localization of both proteins in the glandular lumen suggests that they are secreted by the prostate, as occurs in the epididymis of the rat and humans (Martán et al. 1995; Fraile et al. 1996) and explains the presence of ubiquitin in seminal plasma (Lippert et al. 1993). On the other hand, the many ubiquitin-immunoreactive nuclei observed in normal and hyperplastic prostates might be related to stages of the cell cycle, because ubiquitinated histones 2A and 2B appear in anaphase and disappear either in metaphase or during induction of apoptosis (Mueller et al. 1985; Glotzer et al. 1991; Marushige and Marushige 1995; Yu et al. 1996).
It has been proposed that the family of NPY generelated peptides might play a role in the physiology of the male reproductive tract (Chapple et al. 1991; Gregor et al. 1996). NPY immunoreactivity in the prostate has been described only in nerve bundles and nerve terminals (Properzi et al. 1992; Iwasa 1993; Gkonos et al. 1995; Tainio 1995), with a similar distribution to that observed for vasoactive intestinal polypeptide (VIP) and nitric oxide synthase (NOS) fibers (Hedlund et al. 1997). In the present study, NPY immunoreactivity was also found in epithelial secretory cells and neuroendocrine cells.
The localization of NPY immunostaining in the apical cytoplasm of prostate secretory cells and glandular lumen suggests that the proteins synthesized and secreted by these cells also contain NPY-immunoreactive substances. The present finding agrees with the detection in bull semen of a basic protein called seminal plasmin, which has an extensive homology to the NPY family. This peptide has a role in calcium transport regulation in sperm and in modulation of the zona pellucida-induced acrosome reaction (Herzog et al. 1995). Interestingly, NPY also appears to have remarkable antibacterial activity, which could be important in maintaining a germ-free environment during fertilization (Sitaram et al. 1997). The observation of NPY immunoreactivity in epithelial prostate cells and their secretions is also in agreement with the detection of NPY in prostasomes (prostate-derived organelles present in human seminal plasma) (Stridsberg et al. 1996; Arienti et al. 1998).
As was previously hypothesized, the proteins analyzed in this study are present in the epithelial secretory cells and in luminal prostate secretions. The finding of two ubiquitin bands in the Western blot of prostate secretion might be due to the presence of ubiquitinated proteins with diverse molecular weight, and the migration differences detected between tissue extracts and prostate secretion for the NPY band might be explained because the prostasomic NPY may be similar to that detected in the tissue (7-kD migration band) or linked to other proteins (97-kD migration band). It has been observed (Berthold and Bartfai 1997) that a G-protein frequently binds NPY, and it is interesting to note that the molecular weight of this protein was similar (about 100 kD) (Gimpl et al. 1991) to that observed in the present study for the 97-kD NPY band.
Some of the findings described in the present work could have relevance to normal or pathological function of the prostate. For example, (a) although some authors (Aumüller et al. 1999) have recently postulated a neurogenic origin of human prostate neuroen-docrine cells, based on histogenetic studies in human embryos, other authors (Xue et al. 1997, 1998) have reported that neuroendocrine and epithelial (basal and secretory) cells in the human prostate share a common keratin phenotype and that both probably originate from a common epithelial precursor. In addition, the frequent observation of neuroendocrine differentiation in prostate adenocarcinoma represents the differentiation repertoire of local stem cells (Bonkhoff 1998). The common expression of NPY and PGP 9.5 in both neuroendocrine and epithelial secretory cells observed in the present work also supports the hypothesis of a common origin for both cell lineages. (b) No regional differences in volume fractions of epithelial PGP 9.5, ubiquitin, and NPY immunoreactivities were seen in controls. However, the epithelial volume fraction that immunostained for ubiquitin in BPH specimens was decreased in comparison with the value obtained for the transition zone of controls. This suggests a selective epithelial impairment in the expression of this substance. (c) Some studies have suggested that PGP 9.5 can induce enhancement of cell proliferation (Giambanco et al. 1991; Walker et al. 1995). Therefore, this substance might be involved in the abnormal growth of the prostate.
Summarizing, we can conclude that (a) PGP 9.5, and NPY, but not ubiquitin, are antigens common to both neuroendocrine and secretory prostate epithelium, (b) these three immunoreactive proteins appear to have an exocrine function in the prostate, and (c) BPH is associated with a selective impairment of ubiquitin secretion.
Footnotes
Acknowledgment
We are indebted to Prof J.M. Polak (Department of Histochemistry, Hammersmith Hospital; London, UK) who supplied the NPY antibody and the PGP 9.5 antibody manufactured by Ultraclone.