Abstract
The breast and ovarian cancer susceptibility gene BRCA1 encodes a phosphoprotein of 1863 amino acids containing a highly conserved N-terminal RING finger domain and a C-terminal acidic region typical of several transcription factors. BRCA1 acts as a tumor suppressor that may inhibit the proliferation of breast and ovarian cancer cells. To gain knowledge and to further understand the biological function of BRCA1, we examined its localization and expression in various tissues from 20-year-old male and female cynomolgus monkeys (
Keywords
R
Development of breast and/or ovarian tumors in linked families is associated with loss of the wild-type BRCA1 allele, which supports its predicted role as a tumor suppressor gene (Smith et al. 1992; Kelsell et al. 1993; Neuhausen and Marshall 1994; Kelsell et al. 1996). The nuclear localization of this 220-kD phosphoprotein (Chen et al. 1995,1996a,b; Rao et al. 1996; Scully et al. 1996; Thomas et al. 1996), and the existence of an amino terminal highly conserved RING finger domain (Szabo et al. 1996; Wu et al. 1996) and a carboxy terminal negatively charged transactivation domain (Chapman and Verma 1996; Monteiro et al. 1996) are consistent with its involvement in transcription activation. Moreover, BRCA1 expression is significantly lower in invasive cancer cells than in normal breast tissue or in pre-invasive duct carcinoma in situ (Thompson et al. 1995). Furthermore, inhibition of BRCA1 expression with anti-sense oligonucleotides results in accelerated growth of normal mammary epithelial cells in primary culture and of MCF-7 breast cancer cells (Thompson et al. 1995), and the anti-sense BRCA1-expressing NIH3T3 cells showed increased growth rate, anchorage-independent growth, and tumorigenicity in nude mice in contrast to the parental and sense transfectants, thus providing direct biological evidence that BRCA1 acts as a tumor suppressor (Rao et al. 1996). In fact, overexpression of the wild-type but not of the mutant BRCA1 specifically inhibits growth of breast and ovarian cancer cell lines (Holt et al. 1996), whereas other reports showed death of cells that acutely overproduced BRCA1 (Shao et al. 1996; Wilson et al. 1997). Taken together, these data are consistent with the function of BRCA1 as a tumor suppressor, inferred from human genetic analysis.
In mouse mammary gland, BRCA1 expression is induced during puberty, pregnancy, and after treatment of ovariectomized animals with estrogen and progesterone (Lane et al. 1995; Marquis et al. 1995). It is noteworthy that, in estrogen receptor-positive breast cancer cells, the increase in BRCA1 expression after estrogen treatment is not coordinated with the early induction of an estrogen-dependent pS2 gene but closely paralleled the increase of the S-phase-dependent marker cyclin A, thus suggesting that its expression is modulated by the proliferative status of the cells (Gudas et al. 1995; Spillman and Bowcock 1996; Marks et al. 1997). In agreement with this observation, BRCA1 expression and phosphorylation, which are regulated by cyclin-dependent kinase 2 and other kinases associated with cyclin D and A, are shown to be cell cycle-dependent, maximal levels occurring in S- and M-phases (Chen et al. 1996a). In fact, BRCA1 mRNA expression is tightly regulated during mammary epithelial proliferation and differentiation, and this regulation occurs coordinately with BRCA2 (Vaughn et al. 1996; Rajan et al. 1996; Spillman and Bowcock 1996).
In situ hybridization analysis suggests that mouse BRCA1 may play a critical role in cell growth and differentiation because its transcript is expressed at 9.5 days and later stages and appears to be generally expressed throughout developing mouse embryos, primarily in rapidly proliferating cell types undergoing differentiation (Lane et al. 1995; Marquis et al. 1995). These findings are consistent with the observation that homozygous deletion of BRCA1 in mice is lethal in early embryogenesis (Gowen et al. 1996; Hakem et al. 1996, Liu et al. 1996). The death of mutant BRCA15–6 mice before gastrulation may result from a failure of the proliferative burst required for development of the different germ layers (Hakem et al. 1996). However, the report of a breast cancer patient homozygous for a non-sense mutation in both BRCA1 alleles (Boyd et al. 1995) suggests that the loss of BRCA1 function has different repercussions in humans than in mice.
The lack of well-characterized antibodies not only significantly delayed characterization of the BRCA1 function at the molecular level but also hindered analysis of its tissue- and cell-specific expression (Chen et al. 1996a; Thomas et al. 1996; Wilson et al. 1996). Although the spatial pattern of BRCA1 expression has been reported in adult mouse tissues by in situ analysis (Marquis et al. 1995), the only data available concerning the cell-specific localization of BRCA1 mRNA expression in human tissues, as revealed by either in situ hybridization (Kainu et al. 1996) or immunoperoxidase (Chen et al. 1995) techniques, are restricted to normal and tumoral breast tissues. The aim of the present study was to gain knowledge about the localization and tissue distribution of the BRCA1 gene expression as revealed by in situ hybridization in adult primate cynomolgus (
Materials and Methods
Animals
The 20-year-old male and 20-year-old female
Histological Techniques
For in situ hybridization, the different tissues were rapidly excised without any fat and frozen on dry ice in support medium (OCT) and then kept at −8C. Ten-μm serial sections were then cut at − 20C and mounted on Superfrost/Plus Microscope slides (Fisher Scientific; Montréal, PQ, Canada) and were thereafter fixed in 4% paraformaldehyde in 0.1 M phosphate buffer (pH 7.4) for 10 min at 20C.
Preparation of Human BRCA1 DNA Probes
Three different DNA fragments derived from exon 11 of the BRCA1 gene were obtained after PCR amplification of human genomic DNA using the following pairs of primers: (a) 5′-CTAAGTGTTCAAATACCAGTGAAC-3′ and 5′-CTG-AGTGCCATAATCAGT-ACCAGG-3′; (b) 5′-GCAGAAG-AGGAATGTGCAACA-3′ and 5′-GCCTCTGAACTGAGA-TGATAG-3′; and (c) 5′-TCTATCATCTCAGTTCAGAGG-3′ and 5′-ACTGGAGCC-CACTTCATTAGTAC-3′. Fragments a, b, and c correspond to nucleotides 2248–2459 (211
In Situ Hybridization
In situ hybridization of frozen tissue sections with human BRCA1 DNA probe was adapted from previously described methods (Pelletier et al. 1988; Tong et al. 1989). Briefly, the sections were prehybridized for 1 hr at 20C in 50% (v/v) formamide, 5 × SSPE (1 × SSPE being 0.18 M NaCl, 10 mM NaH2PO4, pH 7.4, 1 mM EDTA), 0.1% (w/v) sodium dodecyl sulfate (SDS), 0.1% (w/v) BSA, 0.1% (w/v) Ficoll, 0.1% (w/v) polyvinylpyrrolidone, 200 μg denatured salmon testis DNA/ml, 200 μg yeast tRNA/ml, 20 μg poly(A)+ RNA/ml (Boehringer Mannheim; Laval, PQ, Canada), and 4% dextran sulfate. Hybridization was carried out for 18 hr at 42C in the same buffer containing 2 × 106 cpm heat-denatured [α-35S]-labeled human BRCA1 DNA fragment b/section. After hybridization, the sections were rinsed once for 1 hr with 2 × SSC (1 × SSC being 0.15 M NaCl and 0.015M Na-cit-rate, pH 7.0) and then processed through washes with increasing stringency to minimize the nonspecific binding as follows: 1 × SSC at 20C for 1 hr; 0.5 × SSC at 20C for 1 hr; 0.5 × SSC at 42C for 1hr; 0.5 × SSC at 20C for 1 hr; 0.1 × SSC at 20C for 30 min. In parallel, the control adjacent sections from each tissue were treated with pancreatic RNAse A (100 μg/ml; Boehringer Mannheim) for 45 min at 37C before hybridization. After these procedures the sections were dehydrated and exposed for autoradiography with Kodak X-Omat film before being coated with liquid phototographic emulsion (Kodak NTB-2; Intersciences, Markham, Ontario, Canada). The times of exposure for the tissue sections was as follows: stomach 2 days; salivary glands 3 days; ovaries and oviduct 4 days; testes, epididymis, seminal vesicles, pituitary gland, pancreas, liver, and large intestine 5 days; mammary gland, skin, uterus (body), and vagina 7 days; kidneys, small intestine, heart and thyroid gland 8 days; urinary bladder 16 days; cervix, aorta, and trachea 21 days. After indicated exposure times, the slides were developed and the sections stained with hematoxylin–eosin.
Histological Examination
Several bright- and darkfields of all tissue sections were examined by a pathologist and independently by a second observer. The measurement was done twice. Tissues were scored for BRCA1 expression and distribution on a qualitative scale as follows: -, absence of specific signal; ∘, heterogeneous expression with less than one quarter of cells showing a specific signal; ∘, homogeneous expression with one to three quarters of cells showing positive signal; •, homogeneous expression with most cells showing strong positivity or grain density observed in more than three quarters. The same scoring system was applied to all tissue sections.
Results
To select organs exhibiting a hybridization signal, we first observed the autoradiographic reaction occurring on X-ray films. All the organs showing a specific reaction were then processed for light microscopic autoradiography. As indicated in Table 1 and further detailed below, our findings clearly show widespread tissue distribution of the BRCA1 mRNA, which not only is strongly expressed in various epithelial cell types but is also specifically detectable, in general, to a weaker extent in several other cell types.
Breast and Skin Tissues
As a control, we first analyzed by in situ hybridization the BRCA1 expression in normal breast tissues from an adult woman. In human mammary gland, BRCA1 mRNA was clearly expressed by acinar epithelial cells and duct epithelial cells, whereas few stromal cells were weakly labeled (data not shown). No signal was observed in sections treated with RNAse before hybridization. In close agreement with these observations, in cynomolgus monkey mammary glands the signal was almost exclusively located in the duct and acinar epithelial cells, whereas heterogeneous weak expression was found in stromal cells (Figure 1).
BRCA1 expression in cynomolgus monkey tissuesa,b
In breast skin, the basal cells of the epidermis as well as keratinocytes showed a specific signal (data not shown). In the dermis, the sebaceous glands were weakly labeled (data not shown).
Female Genital System
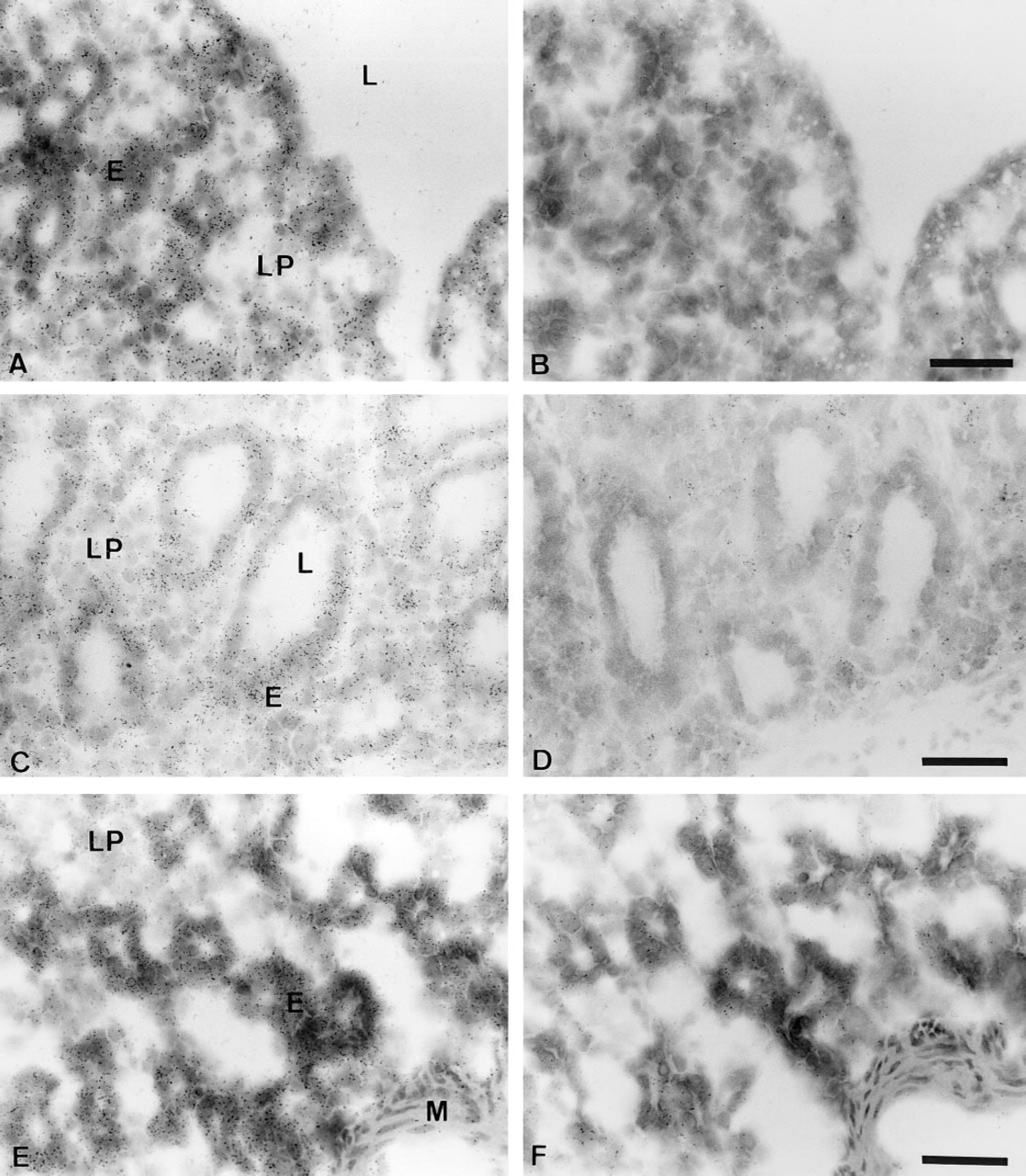
In the ovary, strong specific hybridization signal was detected in the granulosa cells of the primary small and large secondary follicles as well as in the luteal cells of the corpus luteum (Figure 2; and data not shown). The interstitial glandular cells of the ovarian stroma were also specifically labeled, but to a lesser extent. In maturing follicles, the theca interna appeared weakly labeled (data not shown). We also noted relatively strong labeling in the epithelial cells overlying the tunica albuginea, and the ovarian cortical connective tissue was diffusely covered by silver grains (Figure 2C). As shown in Figure 2E, the epithelial cells of the oviduct exhibited a positive signal comparable to that observed over the ovarian surface epithelial cells.
In the uterus, silver grains were observed in high concentrations over both the glandular and the luminal epithelial cells of the endometrium (Figure 3A). The connective tissue of the lamina propria–submucosa was also labeled. The smooth muscle nuclei of myometrium were weakly labeled (data not shown). In the cervix and vagina, specific hybridization signal was detected in the stratified squamous epithelial cells, and a weaker reaction was detected in the connective tissue (Figures 3C and 3E).
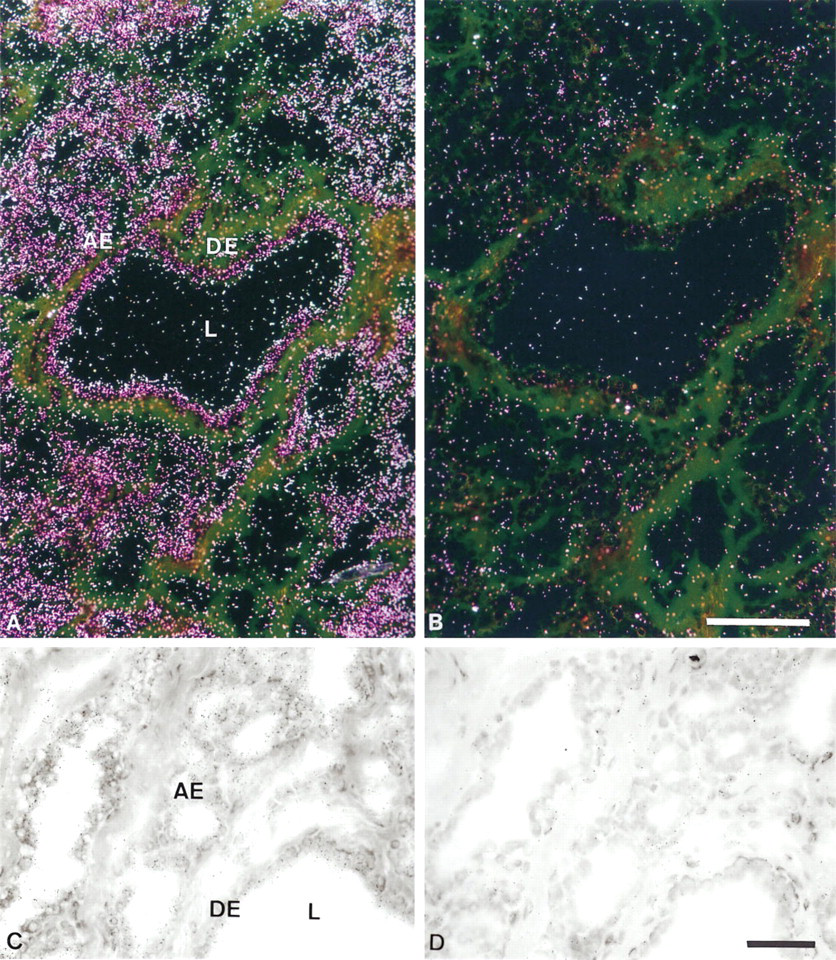
In situ hybridization analysis of BRCA1 mRNA expression in cynomolgus monkey mammary gland. (
Male Genital System
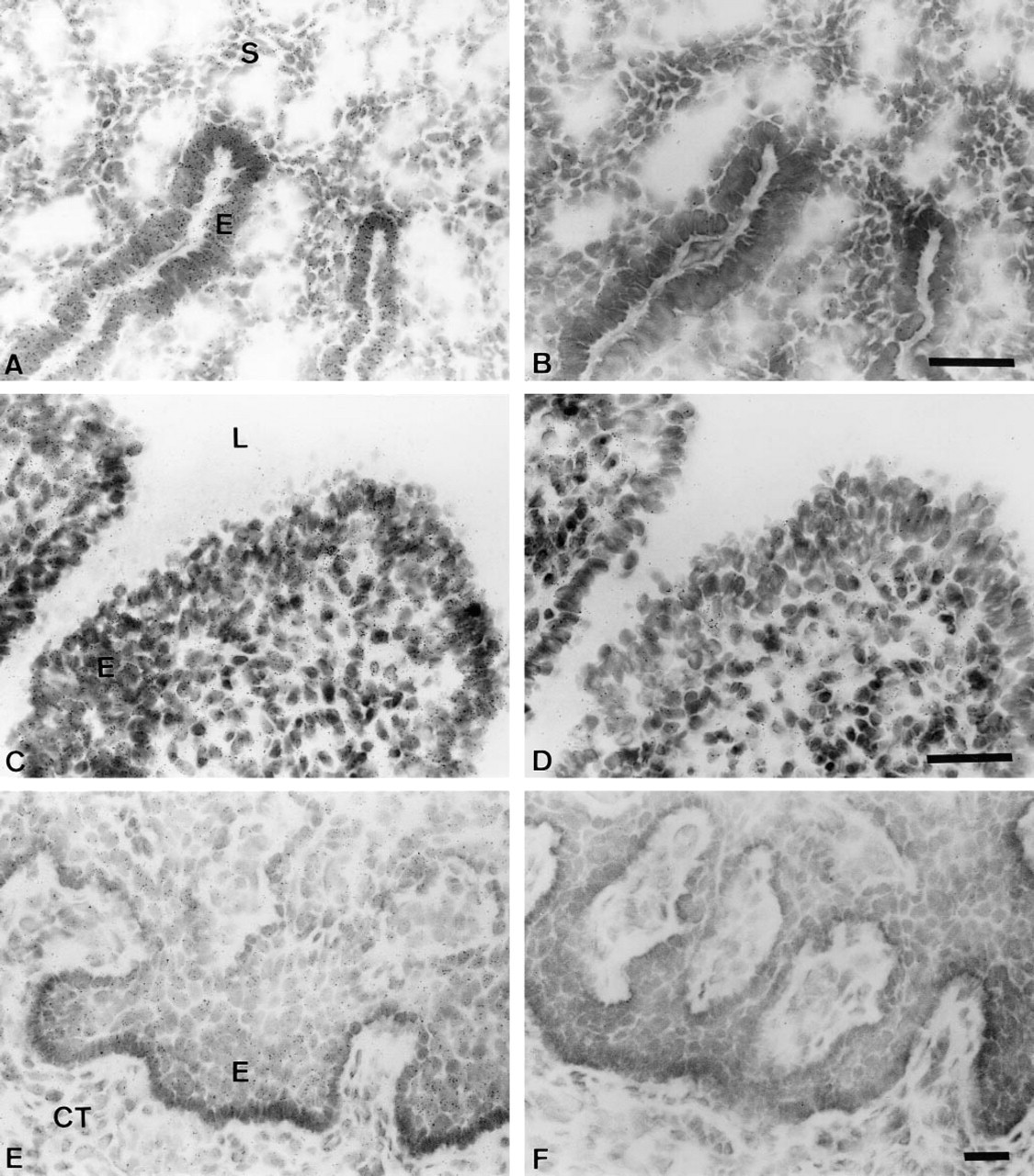
In the testis, strong specific reaction could be detected in the germinal epithelium of the seminiferous tubules (Figure 4A). Similar data were also obtained in the rat testis using a rat BRCA1 exon 11 cDNA probe (Durocher et al., unpublished data). Very strong BRCA1 expression was primarily observed in the epithelium of the epididymis, whereas only a few silver grains could be detected over the smooth muscle cells. The spermatozoa found in the lumen of the ducts did not show any specific reaction (Figure 4C). In the seminal vesicles the positive reaction, which was relatively weaker than that observed in the epididymis, was also mostly restricted to the epithelial cells bordering the glandular lumen (Figure 4E).
Endocrine Organs
In the pituitary gland, all the cells of the anterior lobe appeared to express BRCA1 mRNA. In the posterior lobe, a lower number of dispersed cells, most likely corresponding to pituicytes, were covered with silver grains (Figure 5A). In the thyroid gland, the hybridization signal was detected in the follicular epithelium and the C-cells (Figure 5C). In the endocrine pancreas, most of the islets showed strong labeling (data not shown). In the adrenal glands, strong homogeneous BRCA1 expression was observed in the zona glomerulosa of the cortex (Figure 5G). Moreover, cells of the two other cortical zones, i.e., zona fasciculata and zona reticularis, were also specifically labeled, although the signal was weaker than in the zona glomerulosa. Medullary cells were poorly but specifically labeled (data not shown).
Digestive System
In the stomach, strong BRCA1 mRNA expression was observed over the epithelial cells of the mucosal gastric glands, whereas the lamina propria did not appear specifically labeled (Figure 6A). In the small intestine, the glandular epithelium of the mucosa was strongly labeled, whereas the lamina propria and smooth muscle exhibited a weak reaction (Figure 6C).
In the tubular glands of the colon, the epithelial cells showed a strong signal but the lamina propria and the muscularis mucosa exhibited a very weak reaction (Figure 6E). In the liver, all the hepatocytes were covered with a few silver grains, whereas the biliary duct cells were poorly labeled (data not shown). In the exocrine pancreas, the acinar cells showed a strong signal and the ductal cells were covered by only a few silver grains (Figure 5E). In the salivary glands, both acinar and duct epithelial cells were moderately labeled (data not shown).
Urinary System
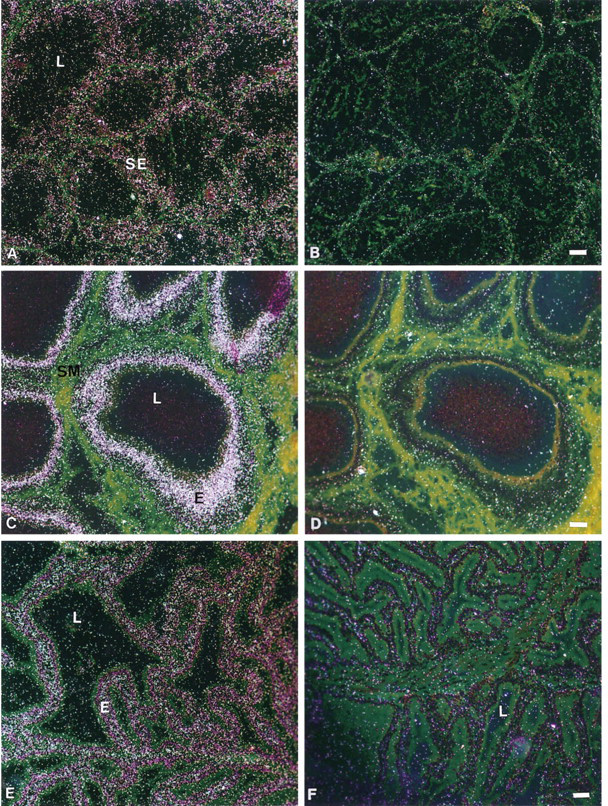
In the bladder, the transitional urinary epithelium was homogeneously labeled, and a weak reaction was detected over smooth muscle cells (Figure 7A). The connective tissue did not show any specific reaction (Figure 7A). In the kidney, the tubule epithelial cells, as well as the glomeruli, were specifically labeled with the same intensity (Figure 7C).
Other Tissues
In the heart muscle, the nuclei of the cardiac myocytes were strongly covered by silver grains, whereas the muscle fibers exhibited no specific labeling (Figure 7F). The endocardial and aortic endothelial cells exhibited weak specific staining (data not shown).
In the trachea, the respiratory epithelium in contact with the airway was strongly labeled (Figure 7G). The glandular epithelium also exhibited specific labeling, which appeared weaker than that observed over the respiratory epithelium.
Discussion
The present study demonstrates widespread tissue-and cell-specific expression of the BRCA1 transcript in adult male and female cynomolgus monkey tissues. BRCA1 mRNA is primarily expressed in a large variety of epithelia in tissues derived from the ectoderm, endoderm, and mesoderm. Nevertheless, a weak but specific signal was also observed in various other cell types. Our data are therefore in close agreement with the tissue distribution of BRCA1 mRNA expression in adult mouse as measured by Northern analysis (Lane et al. 1995) and by RNAse protection as well as in situ hybridization analyses (Marquis et al. 1995). Although BRCA1 has been shown to have tumor-suppressive properties in breast and ovarian cells, its broad distribution and its crucial role in the development of different germ layers during embryogenesis (Gowen et al. 1996; Hakem et al. 1996; Liu et al. 1996), also suggest a more generalized role.
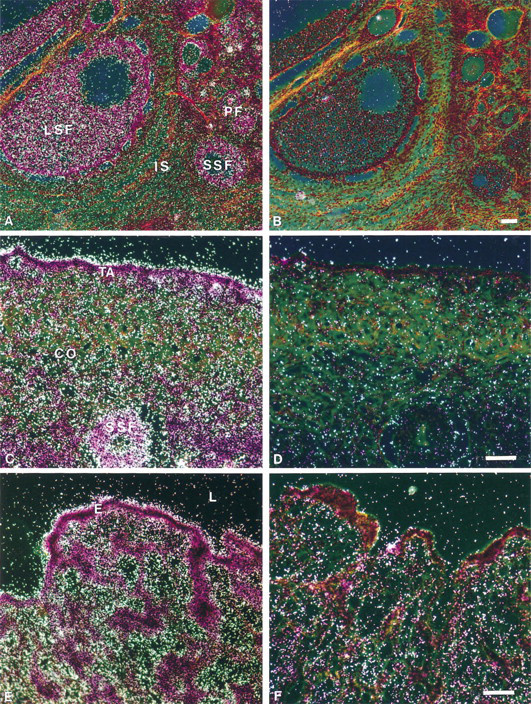
In situ hybridization analysis of BRCA1 mRNA expression in cynomolgus monkey ovary and oviduct. (
In situ hybridization analysis of BRCA1 mRNA expression in cynomolgus monkey endometrium, uterine cervix, and vagina. (
In situ hybridization analysis of BRCA1 mRNA expression in cynomolgus monkey male genital system. (
In this regard, there is evidence that BRCA1 may have a role in the control of recombination and in the maintenance of genome integrity, thus suggesting that a loss of its function might result in genomic errors and the subsequent activation of checkpoint genomic guardian functions, the outcome of which might be cell-cycle arrest (Scully et al. 1997). If this hypothesis is true, then the loss of BRCA1 function may not initiate tumorigenesis but rather may accelerate its progression in cells that have already sustained damage to such a checkpoint function (Scully et al. 1997). However, a wide variety of human malignancies, including breast and ovarian cancers, exhibit decreased ability to undergo apoptosis. This might be, at least in part, due to lack of or decreased levels of BRCA1 expression, considering the recent demonstration that BRCA1 may induce apoptosis (Shao et al. 1996).
The existence of mRNA splice variants of BRCA1 expressed in normal tissues, cell lines, and tumors from various tissues, including breast, ovary, lung, and colon, was recently demonstrated (Lu et al. 1996; Wilson et al. 1997). Among these splice variants, the BRCA1Δ11b lacking most of exon 11 (positions 908–4215) was localized in the cytoplasm instead of the nucleus and appears to have a distinct putative role because its overexpression is not toxic to the cells, in contrast to the full-length BRCA1 protein (Wilson et al. 1997). In the present analysis, this shorter transcript and the other, lacking not only this region of exon 11 but also exons 9 and 10, were not detectable with our probe, which corresponds to nucleotides 2751–2999 in exon 11 of the human BRCA1 cDNA.
In cynomolgus monkey mammary glands, BRCA1 mRNA expression was almost exclusively located in the duct and acinar epithelial cells, whereas heterogeneous weak expression was found in stromal cells. This observation is in agreement with the findings that BRCA1 is primarily expressed in human and mouse normal mammary epithelium, as revealed by in situ hybridization (Lane et al. 1995; Marquis et al. 1995; Kainu et al. 1996). However, in the mouse mammary gland, BRCA1 expression is sharply increased by pregnancy and continues during lactation, and in ovariectomized animals treatment with 17β-estradiol and progesterone causes a synergistic increase in its expression in developing alveolar epithelium (Lane et al. 1995; Marquis et al. 1995). Moreover, it has been demonstrated that BRCA1 is expressed at higher levels in normal human mammary cells than in breast cancer cells and that decreased expression of this gene increases the proliferative rate of both normal and malignant breast epithelial cells (Thompson et al. 1995; Kainu et al. 1996). More recently, it has been demonstrated that BRCA1 expression is induced in rapidly proliferating mammary cells and is downregulated in quiescent cells (Rajan et al. 1996; Vaughn et al. 1996). It has been speculated that induction of BRCA1 expression in actively proliferating mammary cells suggests the existence of a homeostatic loop implying that the proliferation-induced upregulation of BRCA1 constitutes a protective response tending to decrease breast cancer risk (Rajan et al. 1996). These findings are consistent with the observation that patients with BRCA1-linked hereditary breast cancers have highly proliferative tumors that are associated with increased aggressiveness of the malignancy (Marcus et al. 1994; Eisinger et al. 1996; Sobol et al. 1996). Taken together, these data strongly suggest that BRCA1 is involved in the maintenance of negative growth regulatory processes in mammary epithelial cells.
The pattern of BRCA1 expression in the monkey ovary, showing a predominant expression in granulosa cells in developing follicles and relatively lower levels in stromal cells, is in agreement with the data obtained in adult mouse ovary. Moreover, we have also shown in maturing follicles that the theca interna is weakly labeled, whereas relatively strong BRCA1 expression is found in luteal cells of the corpus luteum. It will be of interest to study whether the expression of BRCA1 in these cell types may be hormonally regulated.
Our original observation of relatively strong labeling found in the epithelial cells overlying the tunica albuginea of the ovary is in contrast to the lack of BRCA1 mRNA expression described in murine ovarian epithelium (Marquis et al. 1995) but is consistent with the finding that mutations in the BRCA1 gene markedly increase the risk for development of epithelial ovarian cancer (Narod et al. 1994; Claus and Schwartz 1995). Embryologically, the surface epithelium of the human ovary has the potential to differentiate into epithelium that closely resembles that of the fallopian tubes, the endometrial lining, or the endocervical glands. In support of this, our analysis also clearly demonstrates the predominant expression of BRCA1 in luminal epithelial cells of the endometrium as well as in epithelial cells of the oviduct, cervix, and vagina of the female cynomolgus monkey. In agreement with our data, the BRCA1 expression was almost exclusively observed in the glandular portions of the endometrium in the mouse (Marquis et al. 1995). Although we have found a BRCA1 mutation segregating in a family with cancers of the breast and fallopian tube (Simard et al. 1994), no BRCA1 mutation was detected in two other families affected with both of these cancers (Durocher et al. 1996b). In this latter study, no BRCA1 mutation was found in several families with a history of cancer of the breast, ovary, uterus and/or cervix (Durocher et al. 1996b), thus indicating that additional analysis will be required to establish the potential role, if any, of a BRCA1 mutation in cancers at these sites.
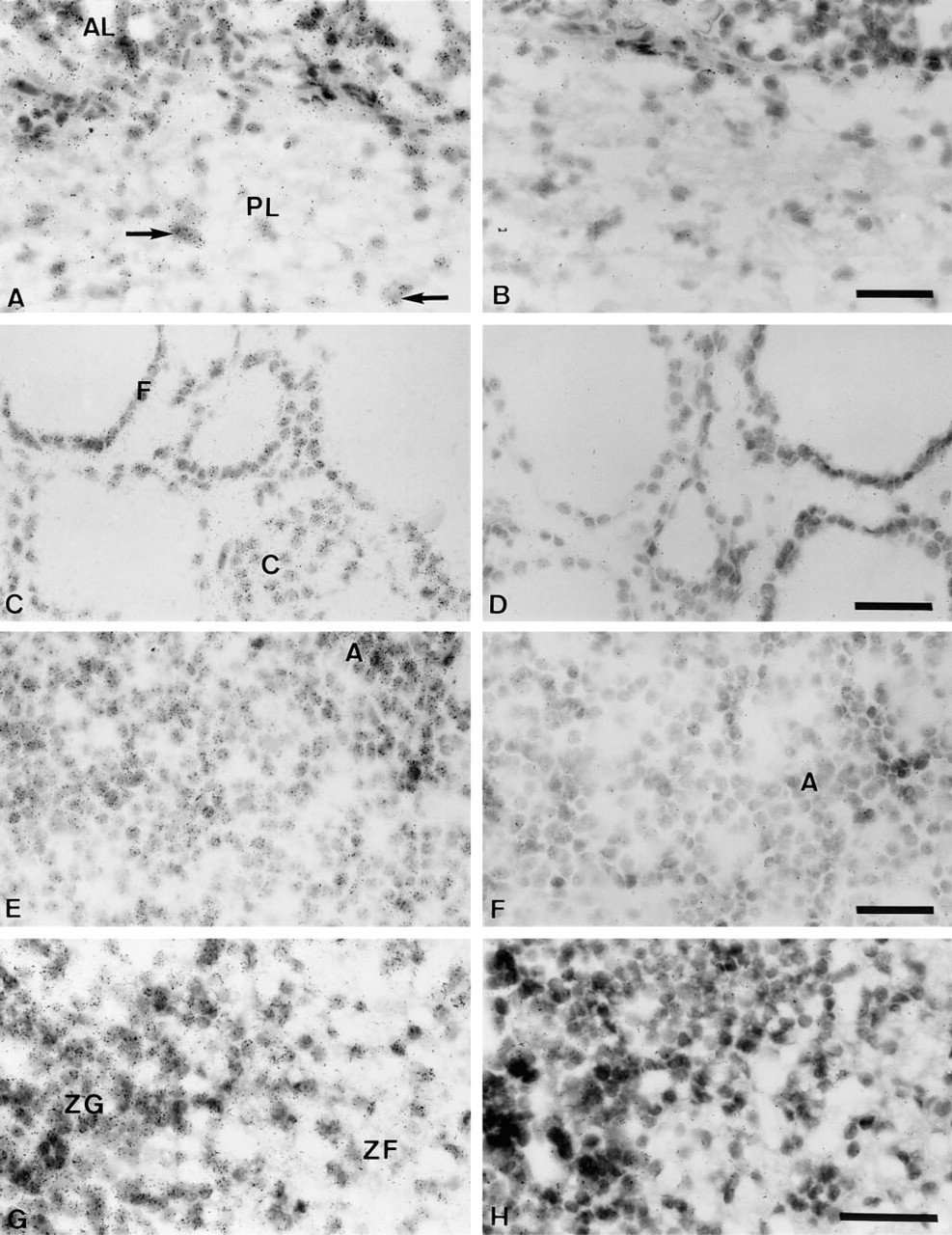
In situ hybridization analysis of BRCA1 mRNA expression in cynomolgus monkey pituitary, thyroid, pancreas, and adrenal cortex. (
In situ hybridization analysis of BRCA1 mRNA expression in cynomolgus monkey gastrointestinal tract. (
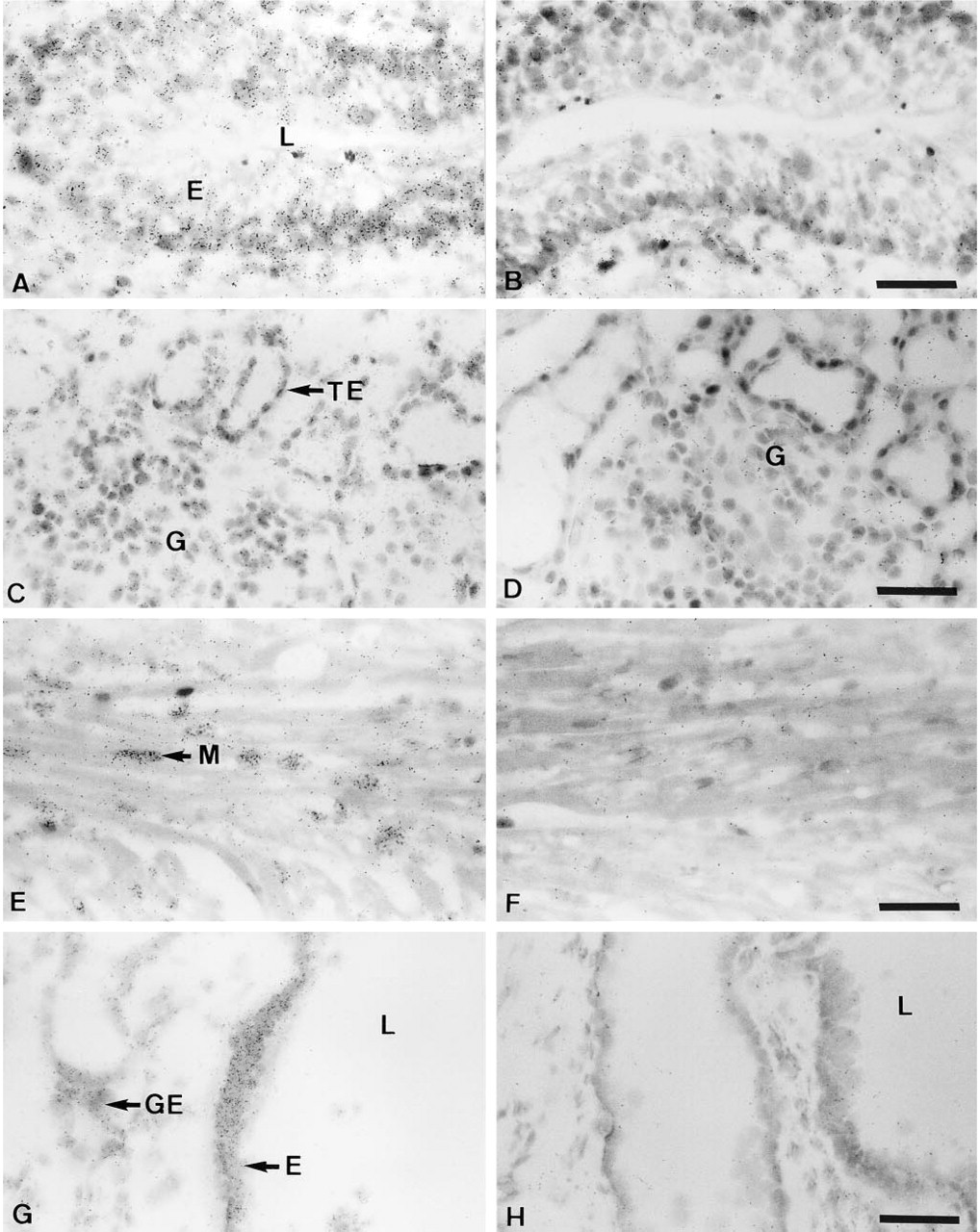
In situ hybridization analysis of BRCA1 mRNA expression in cynomolgus monkey bladder, kidney, heart, and trachea. (
BRCA1 expression in the monkey testis is predominantly observed in seminiferous tubules. This observation is in close agreement with our analysis in the rat testis showing a strong expression in primary spermatocytes and spermatids, whereas no specific labeling was detected in mature sperm cells (Durocher 1995). In the mouse testis, however, it was possible to detect a signal in all cell layers except the extreme periphery of the tubule (Marquis et al. 1995). The high levels of BRCA1 expression in the cynomolgus monkey, rat, and mouse testis is consistent with the initial Northern blot analysis showing a predominant expression of this transcript in the human testis (Miki et al. 1994). More recently, it has been demonstrated that in mouse testis BRCA1 expression is limited to the germ cells. The highest level was detected in meiotic cells, specifically pachytene spermatocytes and postmeiotic round spermatids (Zabuloff et al. 1996). This finding is in agreement with its suggested role in the control of recombination (Scully et al. 1997). Such high levels of expression in the testis were also observed for other tumor suppressor genes, I.e., p53 (Almon et al. 1993) and BRCA2, another breast cancer susceptibility gene recently isolated (Tavtigian et al. 1996).
In the present study, we have also found for the first time a very strong BRCA1 expression in the epithelium of the epididymis, whereas in the seminal vesicles its expression, which is relatively weaker than that observed in the epididymis, is almost solely restricted to the epithelial cells bordering the glandular lumen. This finding is also consistent with the predominant BRCA1 expression in epithelial cells.
Our analysis clearly demonstrates the relatively high levels of BRCA1 expression in epithelia of the stomach and of the small and large intestine. In general, in these tubular organs of the gastrointestinal tract, cells of the crypts, known to contain rapidly dividing, undifferentiated progenitor cells, exhibited a stronger positive reaction than that observed over surface epithelial lining cells. In agreement with our observation, it has been reported that in the mouse gastrointestinal tract BRCA1 expression is primarily confined to the basal epithelial cell layer located at the base of the mucosal epithelium and intestinal crypts (Marquis et al. 1995). Although it has been observed that carriers of a BRCA1 mutation possess a 4.11-fold increased risk for colon cancer (Ford et al. 1994), retroviral transfer of wild-type BRCA1 gene may inhibit growth in vitro of all breast and ovarian cancer cell lines tested, but not of colon or lung cancer cells (Holt et al. 1996). Further studies will therefore be needed to delineate the function of BRCA1 in the proliferation and/or differentiation of both normal and malignant colon epithelial cells.
Although BRCA1 mRNA was predominantly detectable in the epithelia of all the tissues analyzed, we have also demonstrated a weak but specific signal in several other cell types, including stromal cells of both male and female genital systems, in smooth muscle cells in various tissues, and in cardiac myocytes as well as in fibroblasts, thus illustrating a more generalized function of BRCA1 in primate tissues. The present data therefore provide a novel insight that may aid in the further understanding of the biological role of BRCA1 in normal tissues and how its dysfunction may be involved in malignant transformation in breast and ovarian tissues.
Footnotes
Acknowledgements
Supported by the Medical Research Council (MRC) of Canada and by Endorecherche. FD is recipient of a Studentship from the MRC and JS is an FRSQ scholar.
We thank Dr Jim Gourdon for his expert assistance.
