Abstract
In the olfactory bulb (OB) of a transgenic mouse line that carries the bacterial LacZ gene under the control of the 5'-regulatory region of the GAD67 gene, expression of the β-galactosidase was confined almost exclusively to the non-GABAergic mitral and tufted cells. By light microscopy, enzyme histochemistry showed strong staining in the cell bodies and faint diffuse staining in the axons and dendrites. With immunohistochemistry for β-galactosidase the entire cytoplasm, including the axons and dendrites, was strongly stained. By electron microscopy, β-galactosidase enzyme histochemistry resulted in a submicroscopic reaction product that was diffusely distributed in the cytoplasm of neurons. In addition, large deposits of the reaction product were also seen attached to the cytoplasmic side of the membranes. In contrast, when the intracellular localization of β-galactosidase was determined by immunohistochemistry, homogeneous cytoplasmic staining was obtained that filled the entire cytoplasm including the terminal dendrites and fine axons. Therefore, synaptic contacts of the β-galactosidase-positive output neurons with other β-galactosidase-negative neuronal cells were readily recognized in the OB. As we demonstrated, transgenic mouse lines expressing the LacZ reporter gene in a well-defined neuronal subpopulation can be used to follow β-galactosidase-positive neurons and to directly identify their synaptic connections.
Transgenic mouse lines expressing the LacZ reporter gene in a well-defined neuronal population have already been used to explore the mechanisms involved in cortical development (Tan and Breen 1993; Cohen–Tannoudji et al. 1994; Soriano et al. 1995) and to follow the migration of β-galactosidase-expressing cells originating from the subventricular zone that were grafted into adult brain (Lois and Alvarez-Buylla 1994; Lois et al. 1996).
Targeting of bacterial β-galactosidase as a marker for CNS neurons can be achieved by using promoters of neuron-specific genes such as neuron-specific enolase (Forss–Petter et al. 1990), β-amyloid precursor protein (Wirak et al. 1991), aldolase C (Arai et al. 1994), GAP-43 (Vanselow et al. 1994), and neurofilament light chain (Charron et al. 1995). β-Galactosidase expression can also be restricted to a defined subpopulation of neuronal cells dependent on the specificity of the promoter (Hoyle et al. 1994; Huh et al. 1994; Min et al. 1994; Rincon–Limas et al. 1994; Bessis et al. 1995; Morgan et al. 1996).
Some of the transgenic mouse lines express β-galactosidase in the OB (Banerjee et al. 1992; Smeyne et al. 1992; Morita et al. 1993; Arai et al. 1994; Hoyle et al. 1994; Masters et al. 1994; Ponte et al. 1994; Rincon–Limas et al. 1994; Vanselow et al. 1994; Erickson et al. 1995; Morgan et al. 1996), but in none of these cases was the intracellular distribution of β-galactosidase determined.
The aim of our study was to find a reliable method to identify synaptic connections of a transgenically labeled neuronal population. For this purpose, here we characterize the intracellular distribution of the transgene product in the output neurons within the OB of a transgenic mouse line Tg(GAD67lacZ7.5)1 (Katarova et al., submitted for publication) by using enzyme and immunohistochemical detection of β-galactosidase at the light and electron microscopic levels.
Materials and Methods
Transgenic Mice
A number of transgenic mouse lines were generated that carry the bacterial β-galactosidase marker gene attached to a segment of the mouse gene encoding the 67-kD isoform of the GABA biosynthetic enzyme glutamic acid decarboxylase (GAD67) (Katarova et al., submitted for publication). The constructs included the first intron and exon, as well as progressively longer portions of the 5'-flanking region of the regulatory DNA region of the GAD67 gene (Katarova et al., submitted for publication). In this study, one of the transgenic mouse lines with 5
Histological Processing
Two-month-old heterozygous transgenic males (n = 4) were deeply anaesthetized and transcardially perfused with PBS (137 mM sodium chloride, 2.7 mM potassium chloride, 8 mM sodium phosphate dibasic, and 1.47 mM potassium phosphate monobasic), followed by a mixture of 2% paraformaldehyde and 0.25% glutaraldehyde in 0.1 M PIPES buffer (pH 6.9). Brains were removed and postfixed for 1 hr in the same fixative. For light microscopic observation, 15–30-μm-thick sections were cut on a cryostat after cryoprotection in 20% sucrose with 0.02% sodium azide in PBS. For electron microscopy, 100-μm-thick vibratome sections were prepared. Until further use, all the sections were kept in PBS at 4C. Half of both kinds of sections were processed for enzyme histochemistry, and the rest of the sections were prepared for β-galactosidase immunohistochemistry. Alternate sections processed by enzyme histochemistry were counter-stained with 1% neutral red. For routine histology, all stained cryostat sections were dehydrated in an ascending series of ethanol, cleared in xylol, and mounted in Entellan (Merck; Darmstadt, Germany).
β-Galactosidase Enzyme Histochemistry
Enzyme histochemical detection of β-galactosidase was performed by incubating the sections in PBS containing 5 mM potassium ferricyanide, 5 mM potassium ferrocyanide, 2 mM magnesium chloride, 0.02% Nonidet P-40 (w/v), 0.01% sodium deoxycholate (w/v) (detergents were omitted during processing for electron microscopy), and either 1 mg/ml X-gal (5-bromo-4-chloro-3-indolyl-β-
β-Galactosidase Immunohistochemistry
For β-galactosidase immunohistochemistry, sections were kept for 30 min in 0.6% H2O2 in 10% methanol to block endogenous peroxidase activity. After brief rinsing, nonspecific antibody binding was suppressed by normal goat serum diluted 1:20 in Tris-buffered saline (TBS; 10 mM Tris-HCl, pH 7.6, 150 mM NaCl) with 1% bovine serum albumine (BSA) for 1 hr. Then sections were incubated with monoclonal anti-β-galactosidase antibody (Promega; Madison, WI) diluted 1:1500 in TBS with 1% BSA for 1 hr at room temperature (RT). After rinsing in TBS the sections were incubated with biotinylated sheep anti-mouse IgG F(ab') (Amersham; Poole, UK) diluted 1:2000 in TBS with 1% BSA for 1 hr at RT. After several rinsings, the bound antibody was visualized by the avidin–biotin–peroxidase procedure using the Vectastain ABC Elite kit (Vector Laboratories; Burlin-game, CA) and 3,3'-diaminobenzidine tetrahydrochloride (DAB) as chromogen.
Control sections incubated without the primary antibody, as well as sections from non-transgenic animals, showed very low background and no specific cell staining.
Electron Microscopy
For electron microscopy, the vibratome sections from transgenic and control animals stained with anti-β-galactosidase were postfixed in a mixture of 2% paraformaldehyde and 2% glutaraldehyde in PBS. Free aldehydes were carefully removed by several changes of PBS and in freshly prepared 1% sodium borohydride in PBS for 30 min. The tissue was postfixed for 1 hr in 1% OsO4 (Sigma; St Louis, MO) in PBS, washed in PBS, dehydrated in ethanol (30%, 50%, 70%), stained with 1% uranyl acetate in 70% ethanol for 1 hr, dehydrated in ethanol (80%, 90%, 100%), infiltrated, and embedded in Durcupan (Fluka; Buchs, Switzerland). Ultrathin sections were cut and stained with uranyl acetate and lead citrate. The sections were observed in Tesla Bs500 and Zeiss EM−10C electron microscopes. In the control sections, we did not observe any electron-dense precipitate.
Results
To study the transcriptional regulation of the mouse GAD67 gene, we have constructed several transgenic mouse lines carrying the bacterial LacZ gene under the control of progressively longer portions of the 5'-upstream regulatory region of the GAD67 gene (Katarova et al., submitted for publication). Preliminary results indicated a striking but incorrect expression pattern of the transgene in the non-GABAergic mitral and tufted cells of the OB of the transgenic line Tg(GAD67lacZ7.5)1. Here we use this transgenic line as a model to characterize the intracellular distribution of β-galactosidase staining within the large output neurons at light microscopic and electron microscopic level by using β-galactosidase enzyme and immunohis-tochemistry.
Light Microscopy
The laminar organization of the OB from the Tg(GAD67lacZ7.5)1 mouse line was indistinguishable from that of the OB from non-transgenic animals (Figure 1). The transgene expression was predominantly detected in the majority of mitral and tufted cells by the prominent bright blue X-gal reaction product, and occasionally in some granule and periglomerular cells (Figures 1 and 2). The superficial tufted cells showed the strongest staining among the neuronal populations of the OB (Figure 2). Nuclei of mitral and tufted cells were strongly stained, whereas perikarya and dendrites of these cells were less intensely labeled. Diffuse staining without well-defined cell localization was seen in the glomeruli, the external plexiform layer (especially in its superficial segment), and the internal plexiform layer. In the granule cell layer, patches were labeled with diffuse blue staining. In addition, small punctate-like accumulation of the reaction product was observed in the perikarya and along the dendrites of the output neurons (Figure 2 and inset).
Immunohistochemical detection of β-galactosidase labeled the same subpopulation of neurons as revealed by enzyme histochemistry. The mitral and tufted cells were strongly stained and the OB was devoid of diffuse staining (Figures 3–5). The reaction product was mainly confined to the cytoplasm of neurons, although some nuclei were also stained (Figures 3–5). In addition to the perikarya, the dendrites and axons were also filled with reaction product (Figure 4). The strong dendritic staining was most evident in the superficial segment of the external plexiform layer, whereas the internal plexiform layer was slightly labeled (Figures 3 and 4). The punctate-like accumulation of reaction product was also present in the cytoplasm of β-galactosidase-positive neurons (Figure 4), as in Figure 2.
Electron Microscopy
Using electron microscopy, neurons expressing the LacZ fusion gene can be directly identified, because the 5-bromo-3-indolyl precipitate formed on cleavage of Bluo-gal by the β-galactosidase reaction is highly electron-dense.
The product of the enzyme histochemical reaction was dispersed throughout the cytoplasm and nuclei of β-galactosidase-positive neurons as submicroscopic grains with an average diameter of 30 nm. Within the same cells, large 0.2–0.5-μm-long electron-dense deposits of the reaction product were also present. Clusters of these electron-dense deposits were generally attached to intracellular membranes, including the nuclear envelope, mitochondria, and endoplasmic reticulum (Figure 6). On the somata of the labeled output neurons, several synapses were found with granule cells, although their presence was often masked by massive precipitate (Figure 6 and inset). Both types of precipitate were visible in the dendrites of the output neurons in the external plexiform layer (Figure 7), where dendro–dendritic synaptic contacts could be detected between labeled β-galactosidase-positive output neurons and unlabeled β-galactosidase-negative granule cells (Figure 7). The terminal segments of mitral/tufted cell dendrites contained very little reaction product (Figure 8). Because of the diffusion of the reaction product, we also found some precipitate in the extracellular space and in neighboring structures (Figures 7 and 8).
Immunohistochemical detection of β-galactosidase resulted in a fine reaction product that almost completely filled the cytoplasm of neurons expressing the LacZ marker gene, including perikarya (Figure 9), dendrites (Figure 10), and axons (data not shown). The reaction product was also detected in some nuclei (Figure 9). In contrast to the enzyme histochemistry, cytoplasm of terminal dendrites of output neurons entering the olfactory glomeruli contained heavily electron-dense reaction product (Figure 11; compare to Figure 8). Several synaptic contacts were found between the labeled terminal segments of output neurons and axons of the unlabeled olfactory receptor neurons (Figure 11). Synaptic specializations were also identified on the somata as well as on the dendrites of labeled output neurons (Figures 9, 12, and 13). At high resolution, individual and reciprocal synaptic contacts between the labeled dendrites of mitral/tufted cells and the unlabeled β-galactosidase-negative granule cells were readily detected (Figures 12 and 13).
Often, β-galactosidase was found sequestered in large lysosome-like structures (Figure 6), which were identical to the punctate-like staining seen by light microscopy (Figures 2 and 4).
Discussion
In the OB of the transgenic mouse line Tg(GAD67lacZ7.5)1, the majority of the mitral and tufted cells express the bacterial enzyme β-galactosidase. This type of expression is regarded as incorrect (ectopic) because the large output neurons of the OB operate with glutamate rather than GABA as a neurotransmitter (Halász 1990). A small subpopulation of the interneurons, granular and periglomerular cells, is also stained in the OB, which might be of the correct type, as these cells are considered to be GABAergic (Mugnaini et al. 1984). The incorrect expression of the transgene, frequently found in transgenic mice, most probably results from site of integration effects (Al-Shawi et al. 1990; Weis et al. 1991; Bessis et al., 1995). The purpose of this study was to find a reliable method for direct identification of the synaptic interactions of well-defined neuronal cell types, ectopically labeled, β-galactosidase-positive output neurons in particular. Here, the use of the transgenic mouse line Tg(GAD67lacZ7.5)1 provides a new means to intrinsically label a specific neuronal population within the OB.
We have used β-galactosidase enzyme and immunohistochemistry to identify β-galactosidase-expressing output neurons and their synaptic connections in the OB at both light and electron microscopic levels. Using enzyme histochemistry, the blue reaction product accumulated mostly in neuronal nuclei, perikarya, and to some extent in the processes. Diffuse staining in the glomerular layer and external plexiform layer probably represents labeled processes of the output neurons. Especially strong staining of the superficial part of the external plexiform layer can be attributed to stained dendrites of the superficial tufted cells. On the other hand, the presence of diffuse patches in the internal plexiform and granule cell layers suggests axonal staining of the mitral and tufted cells. However, one cannot exclude the possibility that the diffuse staining is at least in part due to limitations of this method, i.e., diffusion of the reaction product out of the positive cells, as reported by Sanes et al. (1986) and Wirak et al. (1991). Diffusion is more pronounced with higher level of β-galactosidase expression and with extended incubation time.
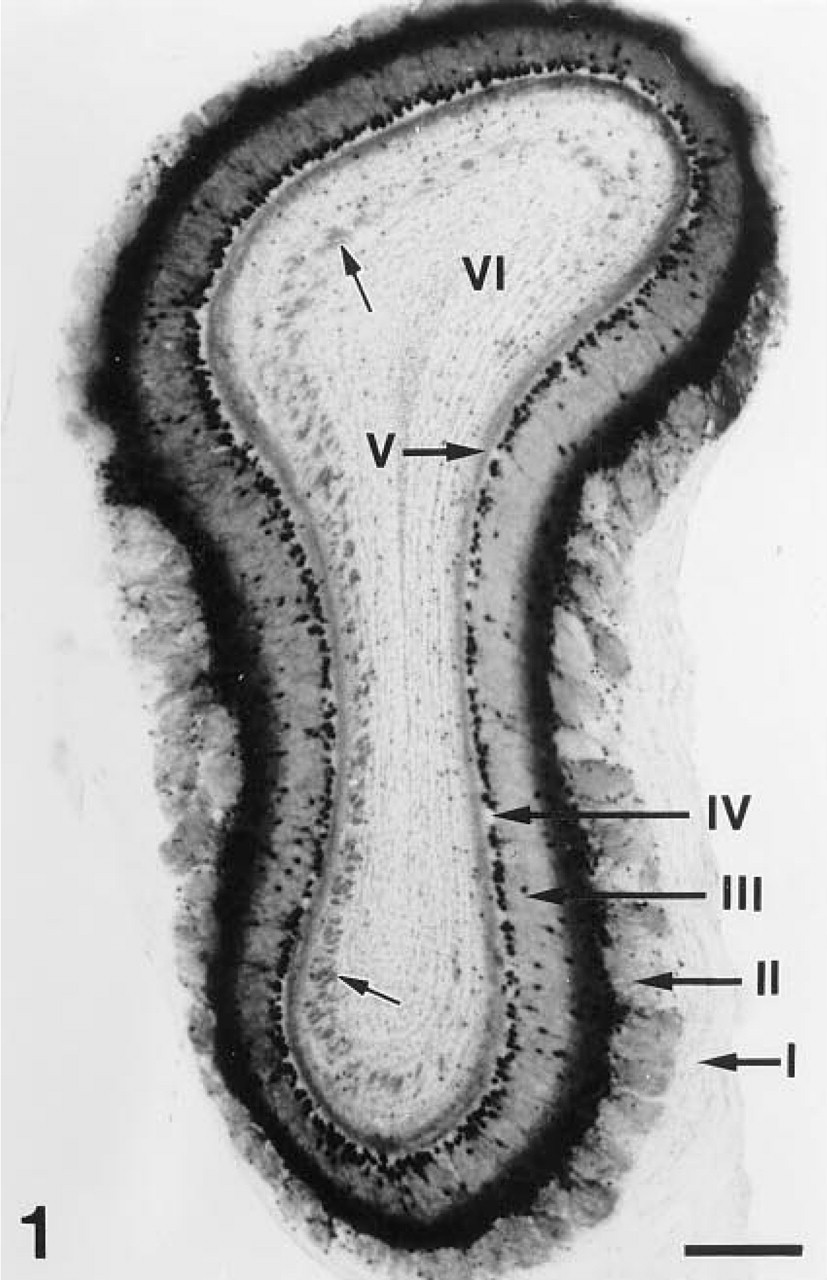
Enzyme histochemical detection of β-galactosidase activity in the olfactory bulb of a Tg(GAD67lacz7.5)1 transgenic mouse. Staining is present in all layers of OB except for the olfactory nerve layer (I). The glomerular (II), external plexiform layer (III), and internal plexiform layer show (V) diffuse staining, whereas in the granule cell layer (VI) stained patches (small arrows) are present. Cellular staining is obvious in the mitral (IV, mitral cell layer) and tufted (layer III) cells. Only few labeled periglomerular (layer II) and granular (layer VI) cells could be seen. Bar = 500 μm.
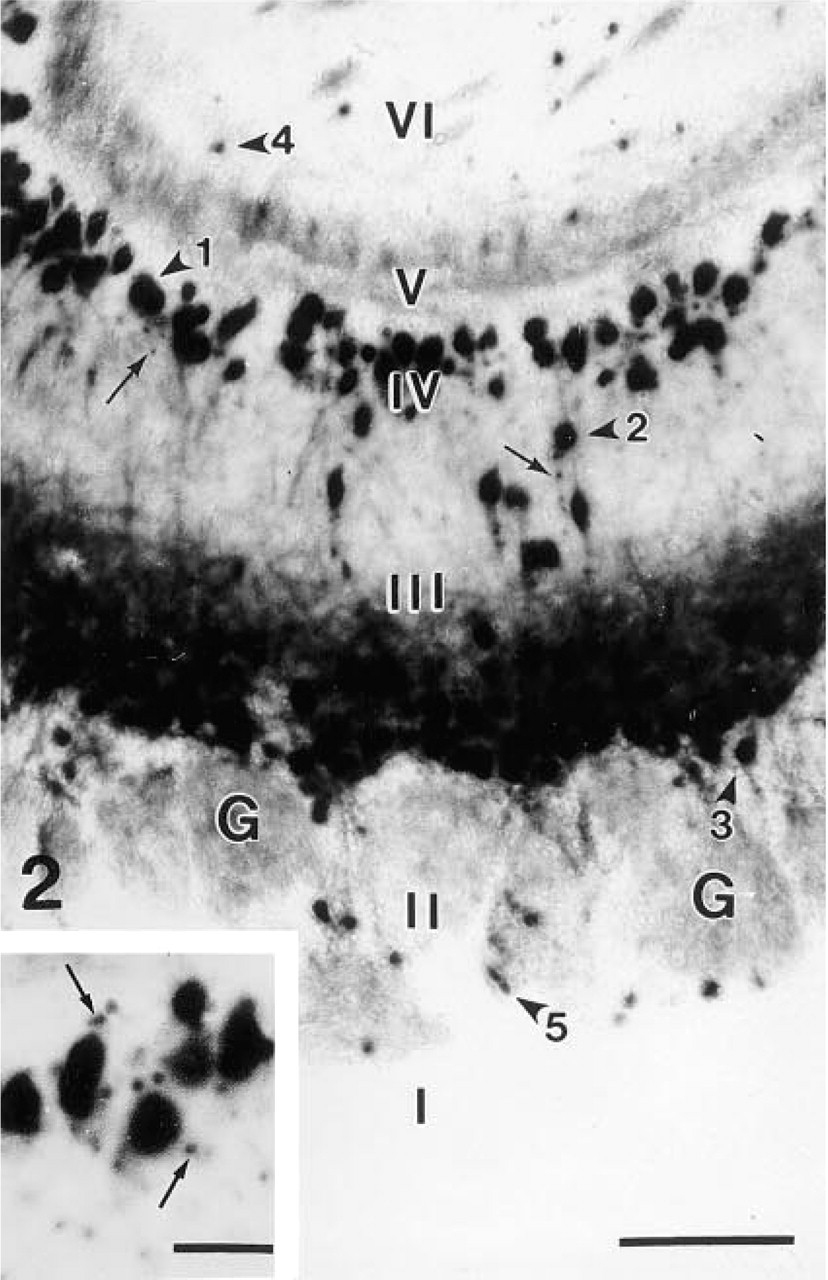
Higher magnification shows that the nuclei of the mitral (arrowhead 1), deep, intermediate (arrowhead 2), and superficial (arrowhead 3) tufted cells are prominently stained, whereas the perikarya and the dendrites are less intensely labeled. There is intense diffuse staining in the superficial part of external plexiform layer (III). A few granule (arrowhead 4) and periglomerular (arrowhead 5) cells are also stained. Arrows point to small punctate-like accumulation of the reaction product.
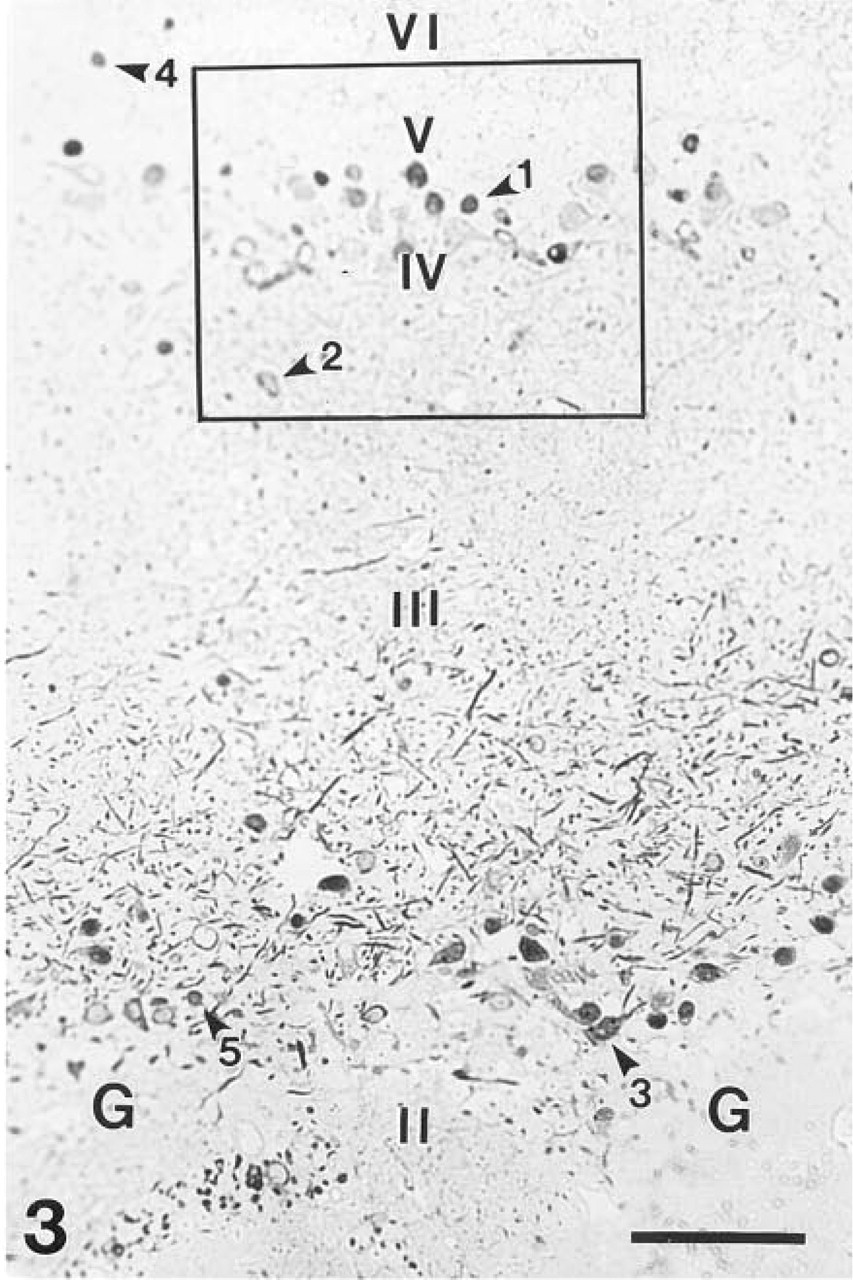
Microphotograph of a semithin section stained with anti-β-galactosidase. The reaction product is focally localized in neurons spread throughout the entire cytoplasm. Staining is present in the mitral (arrowhead 1) and tufted (arrowheads 2,3) cells as well as in a small subpopulation of the granule (arrowhead 4) and periglomerular (arrowhead 5) cells. Many strongly stained dendrites of the mitral and tufted cells, especially of the superficial tufted cells, are present in the superficial part of the external plexiform layer (III). G, glomerulus. Bar = 200 μm.
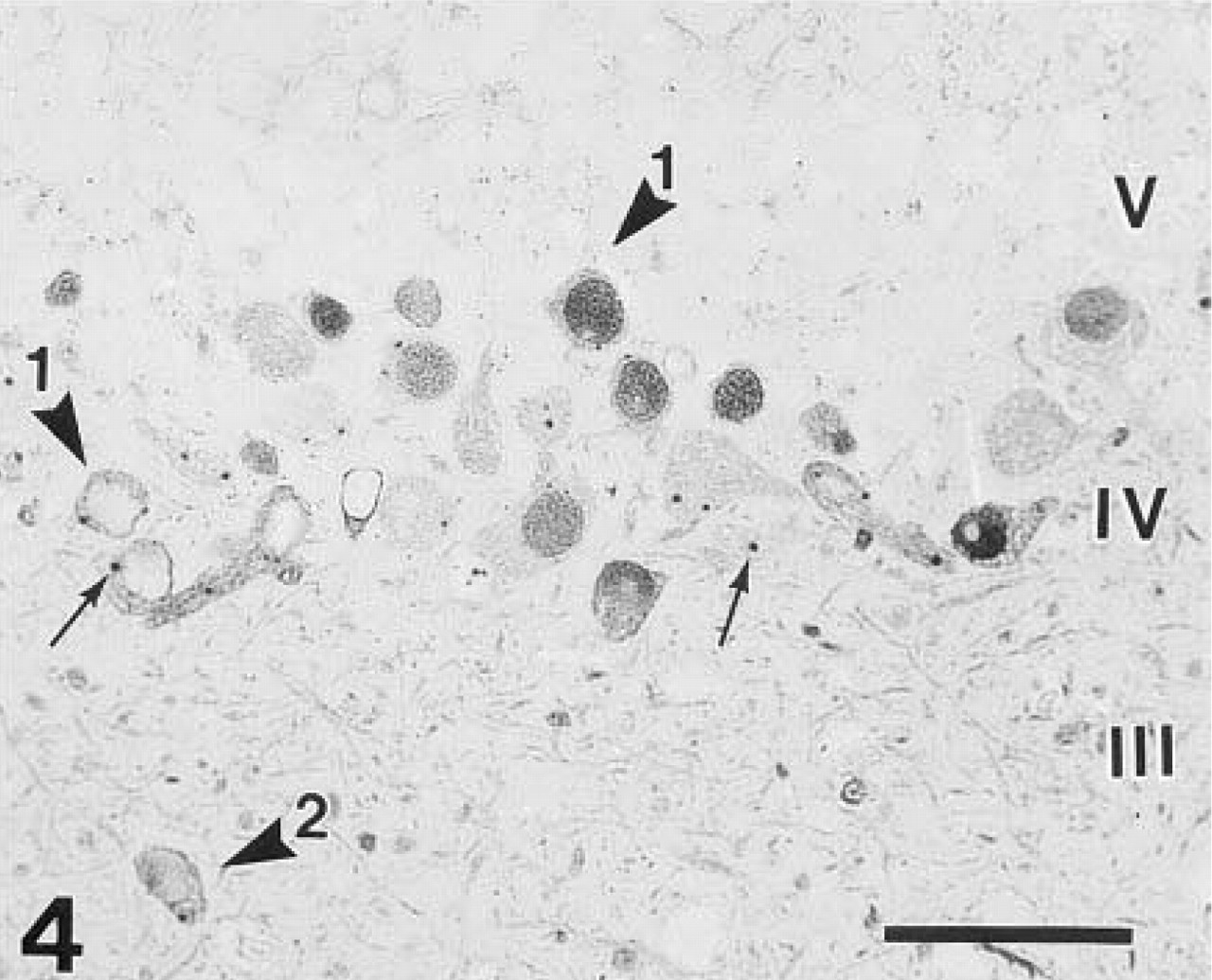
Higher magnification of the boxed area in Figure 3 illustrates that the reaction product filled the entire cytoplasm of the mitral cells, whether in the perikaryon, dendrite, or in the axon. Differences in nuclear staining of the mitral cells (arrowhead 1) are obvious. Arrowhead 2, intermediate tufted cells; arrows, punctate-like staining. β-Galactosidase immunohistochemistry. Bar = 100 μm.
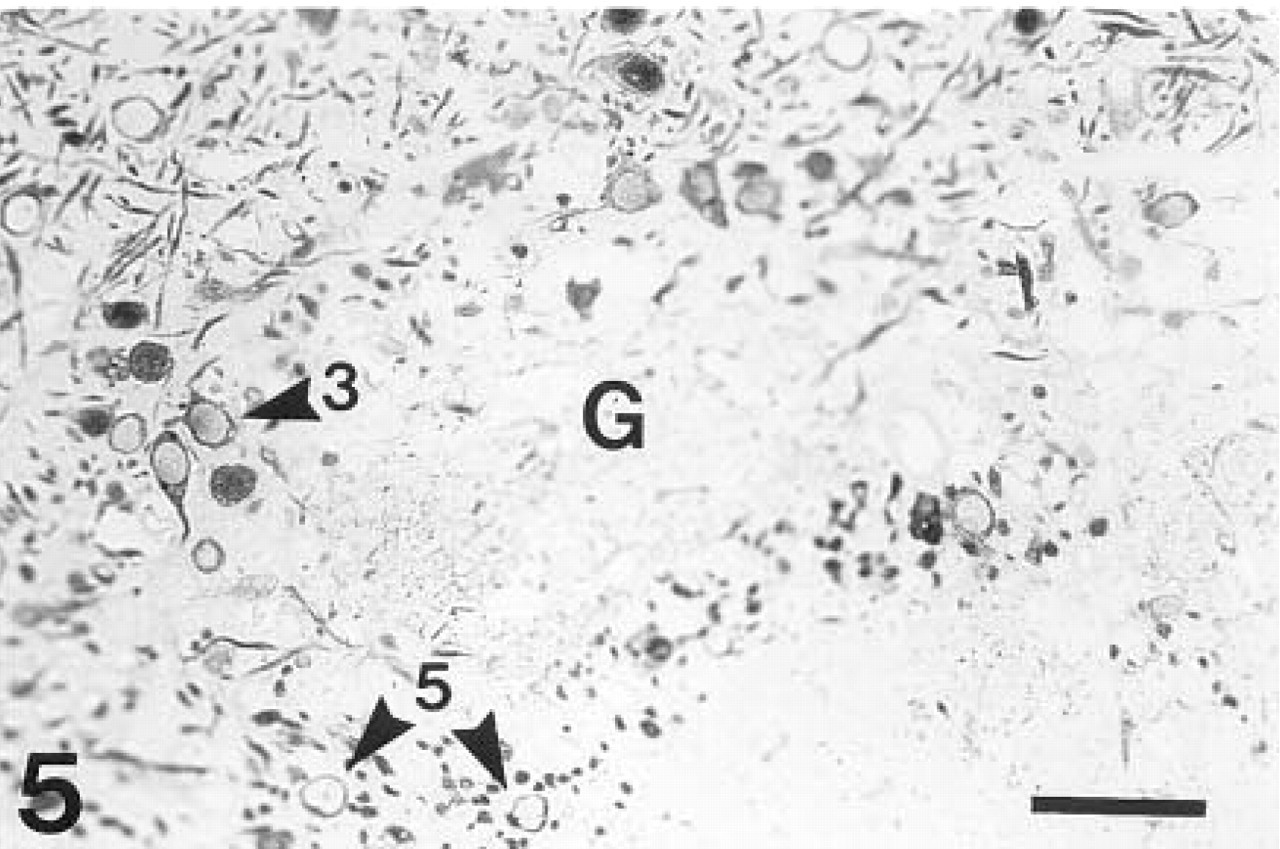
A glomerulus (G) surrounded by immunostained tufted (arrowhead 3) and periglomerular (arrowhead 5) cells. β-Galactosidase immunohistochemistry. Bar = 100 μm.
Diffusion was limited when Bluo-gal was used as a substrate, although X-gal was preferred for light microscopic visualization because it usually gave a brighter color reaction. Bluo-gal is a more suitable substrate for identifying β-galactosidase-positive cells at the ultrastructural level than X-gal, as the 4-chloro-3-indolyl precipitate is more electron-dense and less diffusable. At the electron microscopic level, the Bluo-gal reaction product formed fine submicroscopic grains as well as larger electron-dense deposits bound to internal and occasionally to external membranes, which were in part also detected in some dendrites. The precipitate accumulating on the membranes makes it difficult to identify synaptic clefts on labeled neurons. In addition, diffusion of the reaction product into neighboring structures interferes with proper distinguishing between the terminal segments of the β-galactosidase-positive output neurons with little reaction product and the β-galactosidase-negative neuronal cells. Another obvious disadvantage of this detection system, reported also by Friedrich et al. (1993), is that under slightly different reaction conditions the appearance of the precipitate and its distribution within neuronal cells are variable. Nevertheless, this technique can be improved by optimizing the reaction conditions. β-Galactosidase enzyme histochemistry can still be the method of choice to label specific neuronal cell types in combination with different immunohistochemical reactions.
Comparison of staining of the mitral and tufted cells after enzyme histochemistry with that after immunohistochemistry revealed several advantages of using β-galactosidase immunohistochemistry, at least for electron microscopic studies, most notably strict intracellular localization of the reaction product and homogeneous labeling of the entire cytoplasm, including processes and their terminal segments. Using immunohistochemistry for β-galactosidase to label output neurons expressing LacZ enabled us to readily identify synaptic connections between the β-galactosidase-negative olfactory receptor neurons and the labeled β-galactosidase-positive mitral/tufted cells within the glomerular layer, as well as synaptic junctions between labeled soma/dendrite of mitral/tufted cells and β-galactosidase-negative granule cells in the external plexiform layer.
The expression of β-galactosidase in well-defined neuronal subpopulations in transgenic mouse lines carrying the LacZ reporter gene may prove useful in providing a neutral intrinsic marker for these neuronal populations that may help their identification after experimental manipulations such as transplantation, on ablation, and in cell culture. Here we show that this marker could successfully be used for identification of labeled neurons and their synaptic contacts with unlabeled neurons at the ultrastructural level, which may prove indispensible in cases where a specific marker is not available, as in the case of mitral and tufted neurons of the OB.
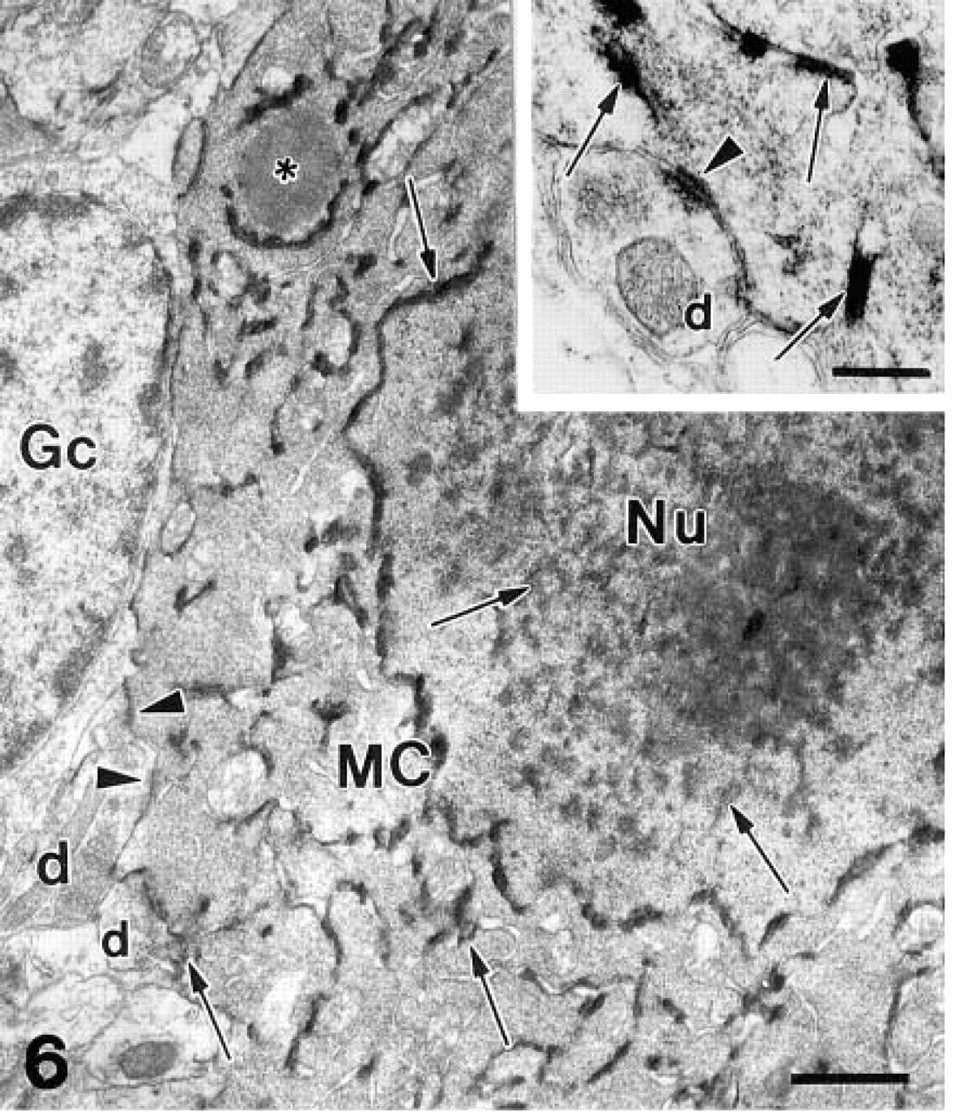
Enzyme histochemistry of β-galactosidase activity at the electron microscopic level. The cytoplasm and the nucleus (Nu) of the mitral cell (MC) are filled with a submicroscopic precipitate of Bluo-gal. Large electron-dense deposits of reaction product (arrows) are obvious within the nucleus and also in the cytoplasm, where they are attached to the nuclear envelope and the cellular membranes, mitochondria, and endoplasmic reticulum. Asterisk, lysosome; d, dendritic terminals of the granule cells; Gc, a granule cell in satellite position to a mitral cell; arrowheads, postsynaptic membranes.
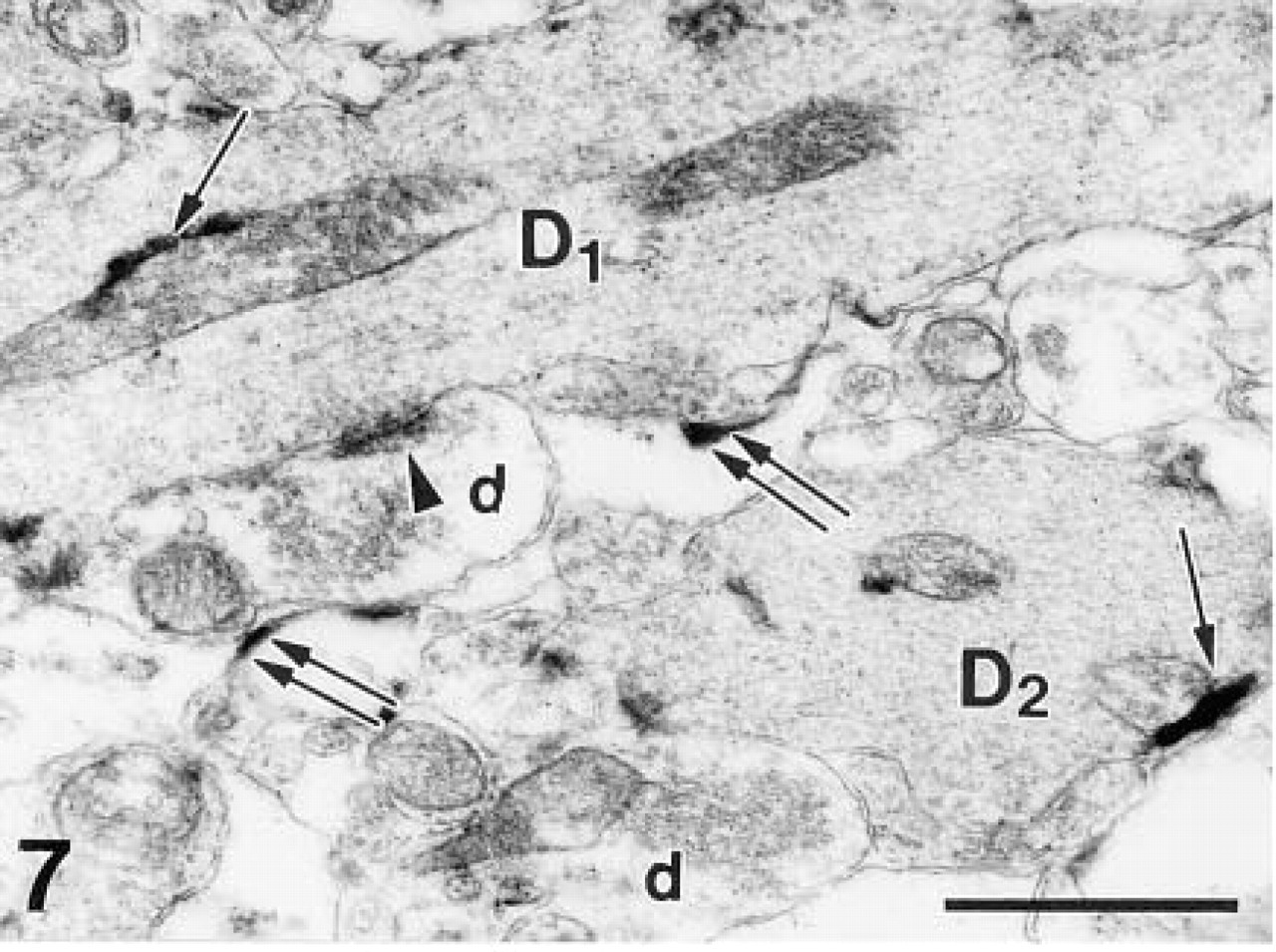
Fine submicroscopic precipitates and large deposits (arrows) are also present in the dendrites (D1,2) of the output neurons in the external plexiform layer. A dendro–dendritic synapse (arrowhead points to postsynaptic densities) between a labeled dendritic profile of mitral/tufted cell and a non-labeled terminal of a granule cell (d) is seen. Note diffusion of the precipitate to the extracellular space (double arrows). β-Galactosidase enzyme histochemistry. Bar = 1 μm.
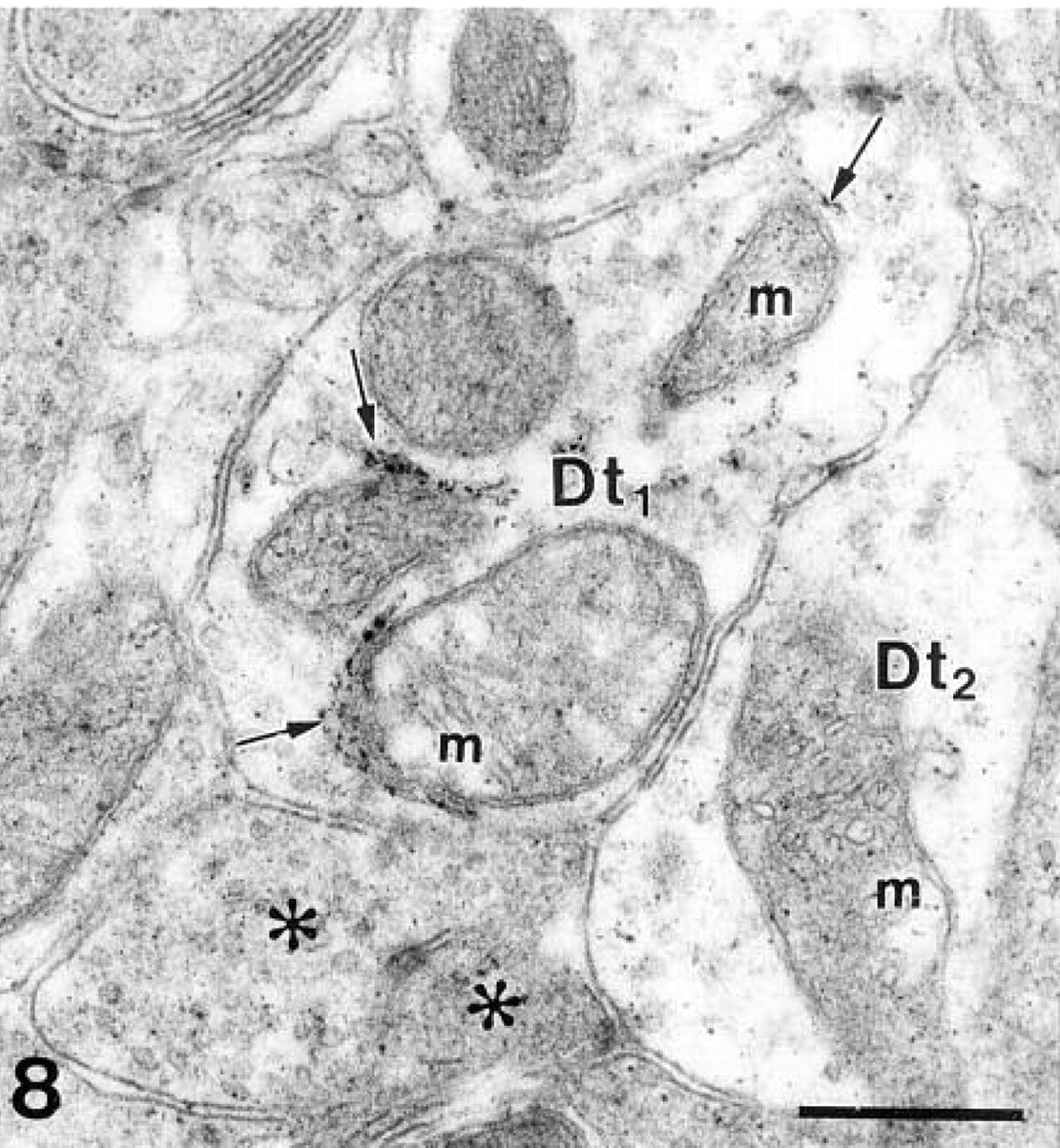
A labeled dendritic terminal (Dt1) in a glomerulus. Deposits of electron-dense precipitate (arrows) are present in the endoplasmic reticulum near the mitochondria (m). The presence of fine precipitate in the neighboring structures, even in β-galactosidase-negative structures such as olfactory axons (asterisks), is due to the diffusion of the reaction product. Dt2, dendritic terminal without large deposits. β-Galactosidase enzyme histochemistry. Bar = 0.5 μm.
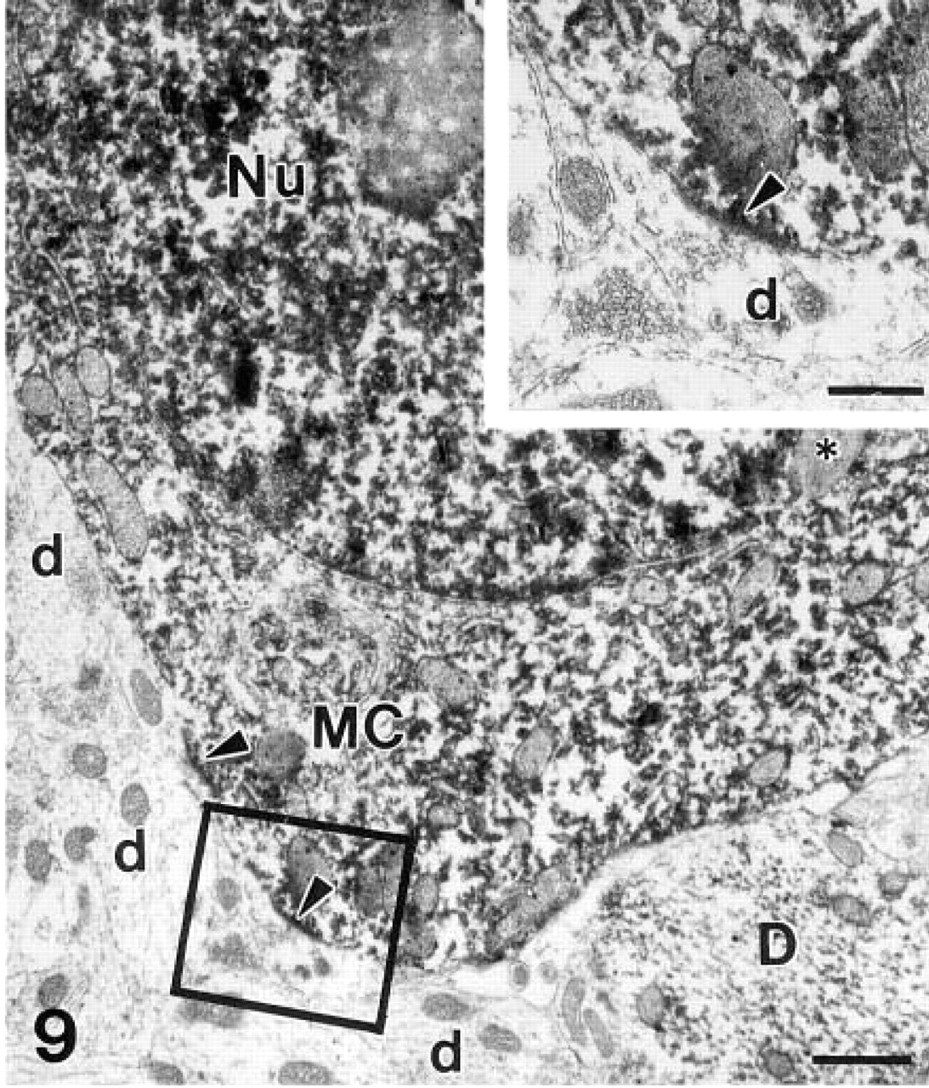
β-Galactosidase immunohistochemistry for electron microscopy. The mitral cell (MC) cytoplasm and nucleus (Nu) are filled with the electron-dense precipitate of DAB–peroxidase complex. Arrowheads point to postsynaptic membranes of synapses between the soma of the mitral cell and unlabeled dendritic terminals of the granule cells (d). Asterisk, lysosome; D, a labeled dendrite of another output neuron.
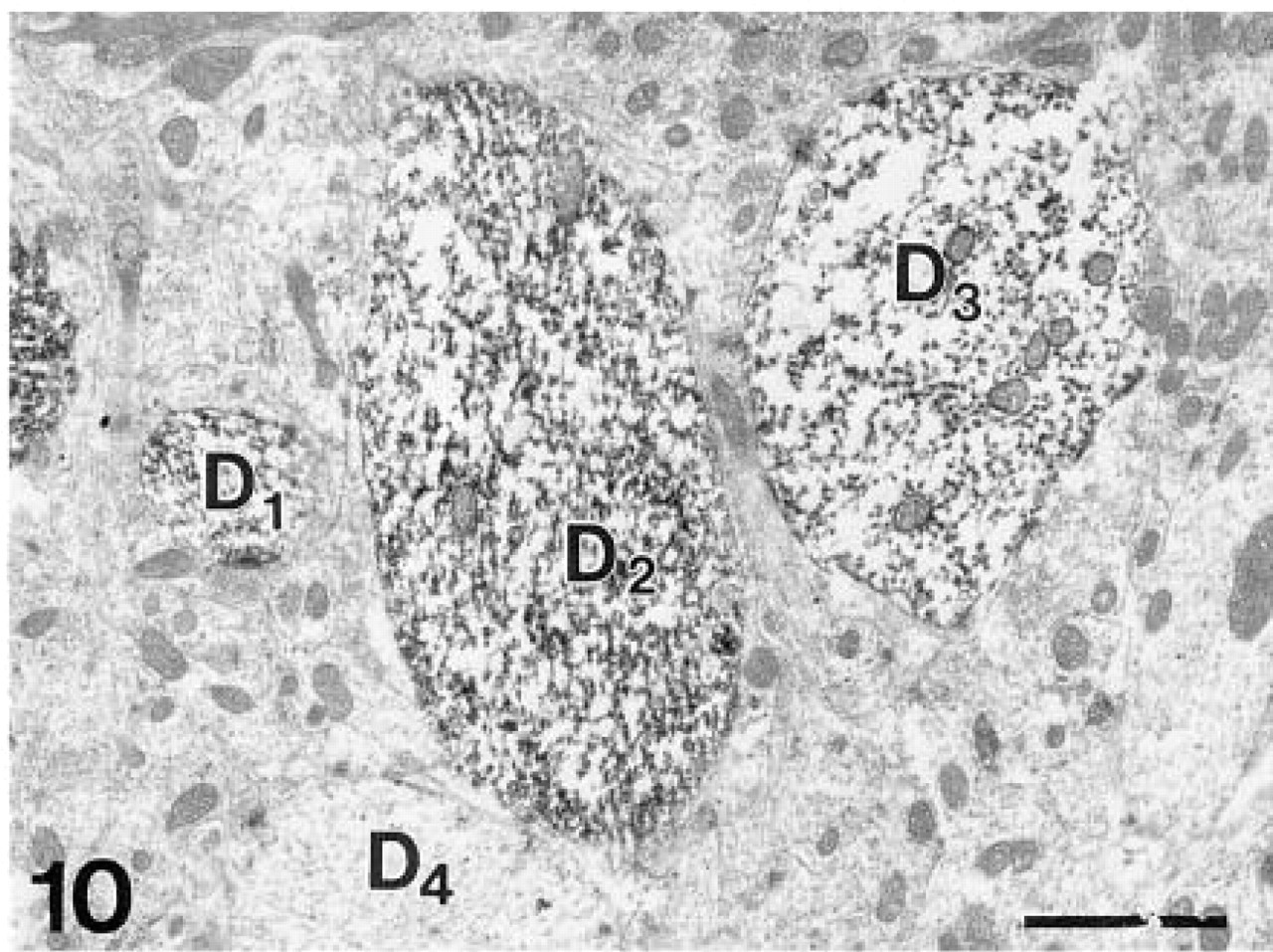
Stained dendritic profiles (D1,2,3) of output neurons in the external plexiform layer. D4, unstained dendrite. β-Galactosidase immunohistochemistry. Bar = 1 μm.
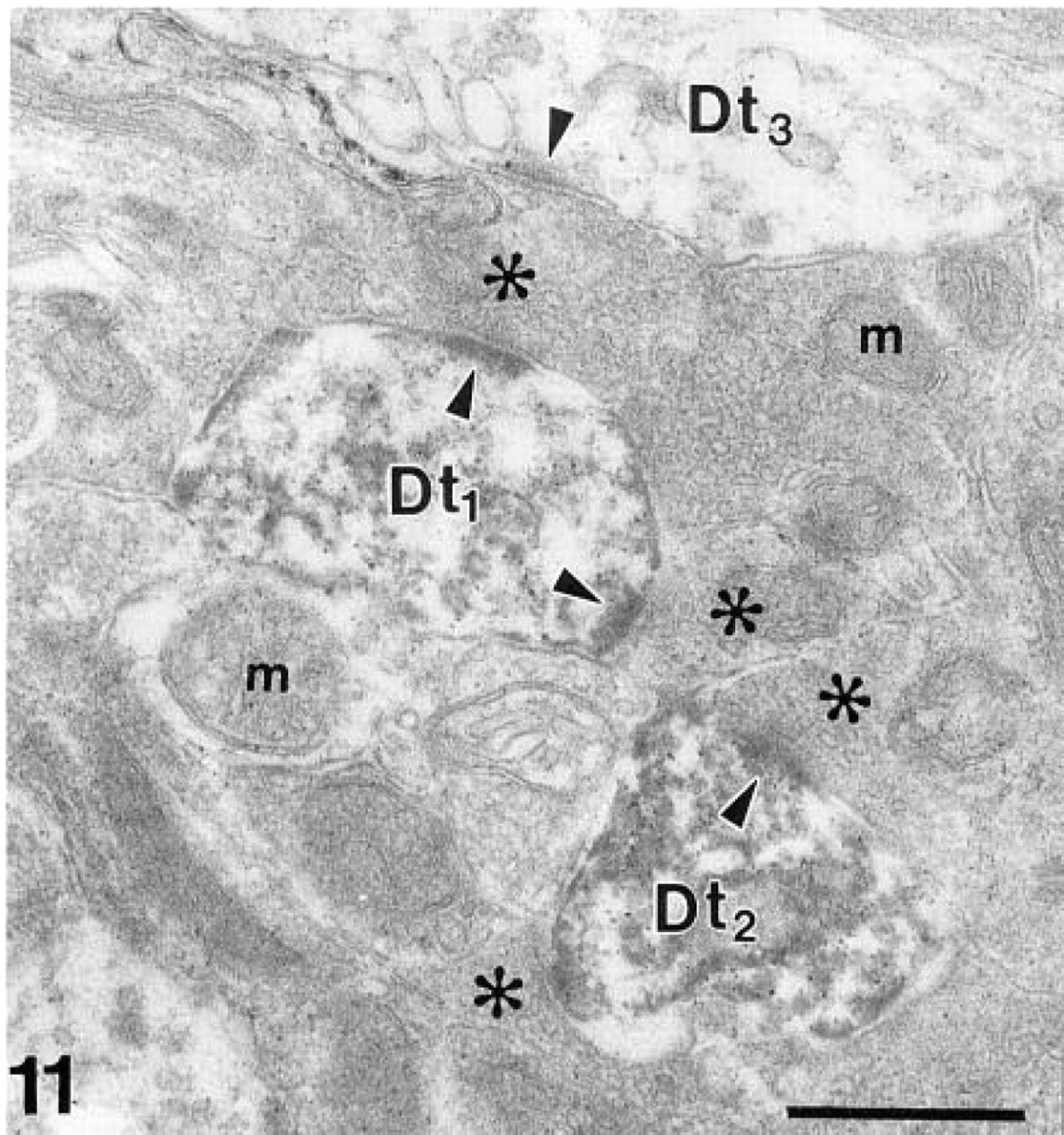
Synaptic contacts (arrowheads point to postsynaptic densities) between olfactory axons (asterisks) and labeled (Dt1,2) as well as unlabeled dendritic terminals (D3) of the mitral/tufted cells within a glomerulus. m, mitochondria. β-Galactosidase immunohistochemistry. Bar = 0.5 μm.
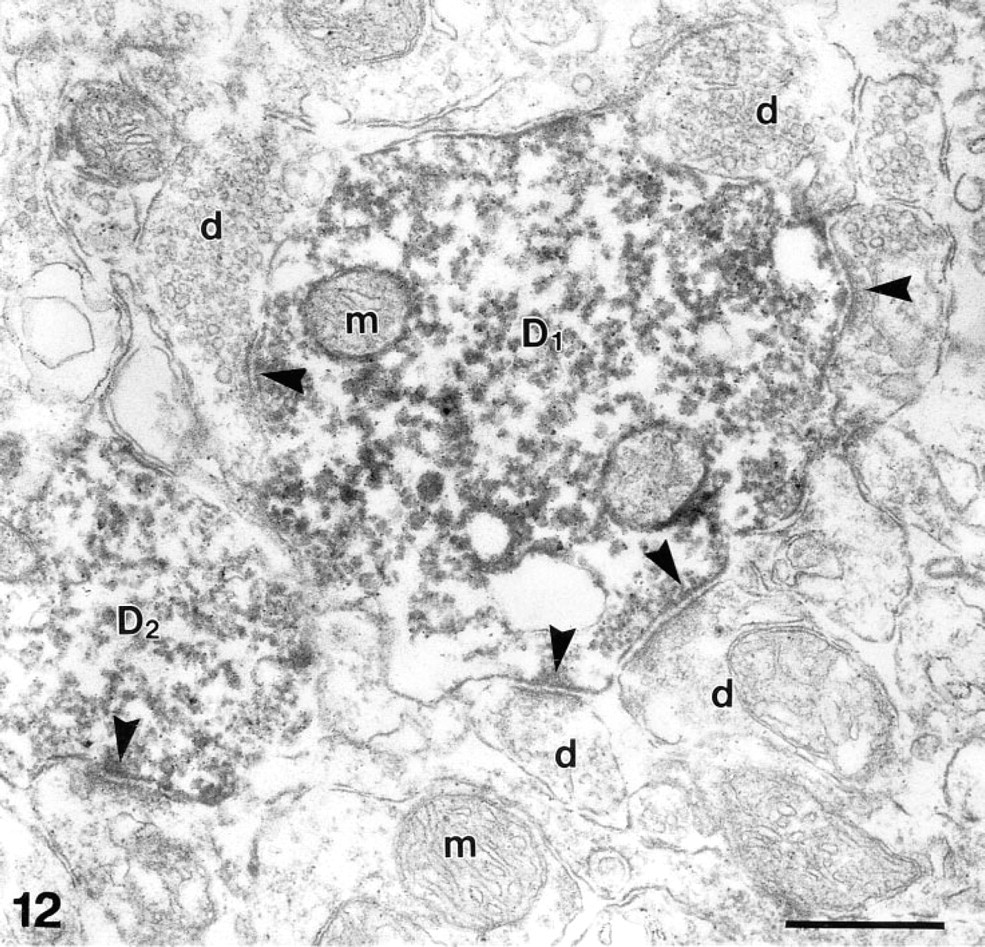
Synaptic junctions (arrowheads point to postsynaptic densities) between β-galactosidase-positive dendrites (D1,2) of output neurons with unlabeled dendritic terminals of granule cells (d). m, mitochondria. β-Galactosidase immunohistochemistry. Bar = 0.5 μm.
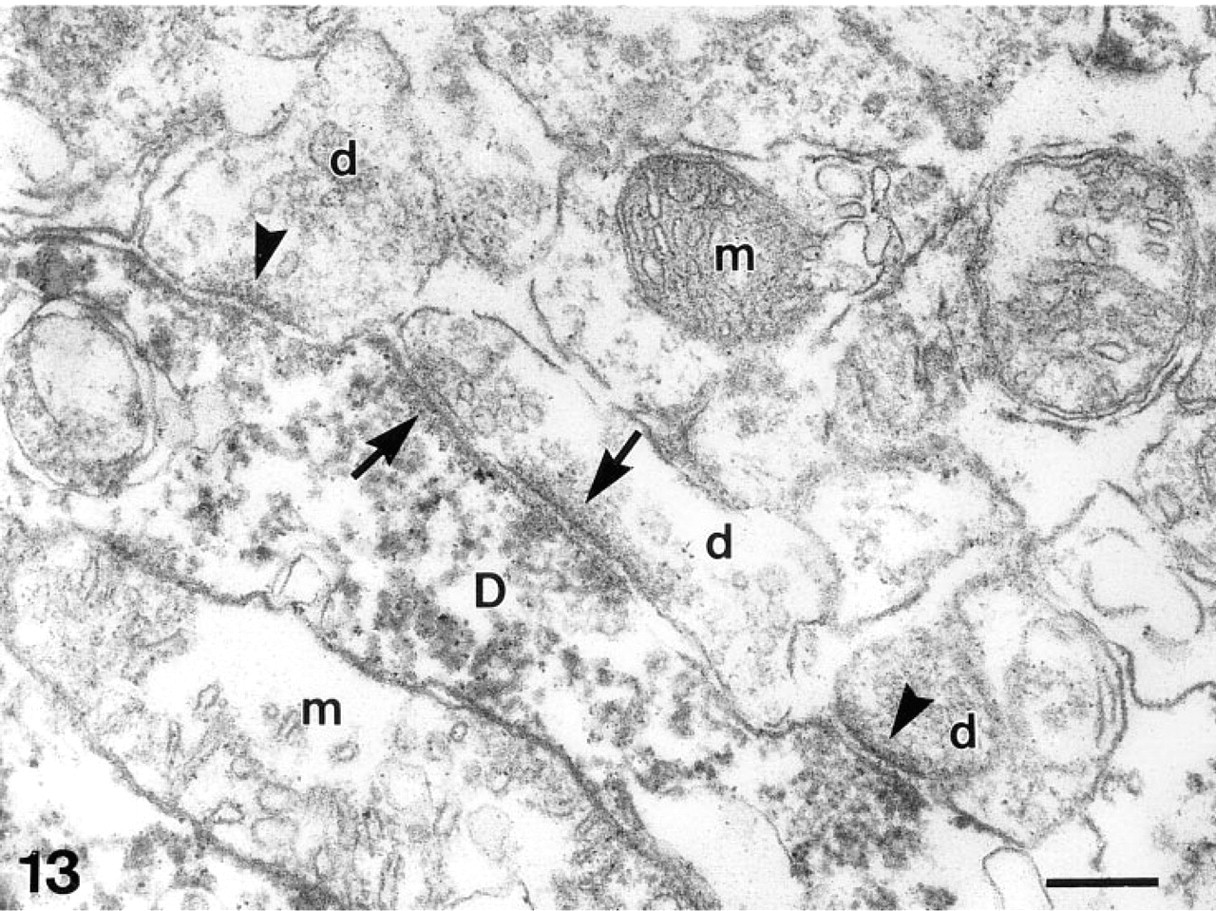
Dendro–dendritic synaptic connections between a labeled dendrite (D) of a β-galactosidase-positive mitral/tufted cell and dendritic terminals of β-galactosidase-negative granule cells (d). Arrowheads indicate postsynaptic densities of individual synapses; arrows point to postsynaptic densities of reciprocally arranged synapses. m, mitochondria. β-Galactosidase immunohistochemistry. Bar = 0.2 μm.
Footnotes
Acknowledgements
Supported in part by the Hungarian Research Fund (OTKA T14645, T006373, and T016971) and the Ministry of Public Welfare (ETT T-04 029/93), by the Slovak Academy of Sciences grant No.1319, and by grants from the Deutsche Forschungsgemeinschaft SFB 406 and from the Volkswagen Stiftung (No. I-7-777).
We thank Ms Ildikó Harmos, Ms Annelies Wolff, and Martha Syneková for excellent technical assistance.
