Abstract
Casein kinase 1 epsilon (CK1ε) is involved in various cellular processes, including cell growth, differentiation, and apoptosis, vesicle transport, and control of the circadian rhythm. Deregulation of CK1ε has been linked to neurodegenerative diseases and cancer. To better understand the cell type-specific functions of CK1ε, we determined its localization by immunhistochemistry in tissues of healthy, young adult BALB/c mice and in mammary tumors of SV40 T-antigen-transgenic mice. CK1ε expression was found to be highly regulated in normal tissues of endodermal, mesodermal, and ectodermal origin and in neoplastic tissue of mammary cancer. The data presented here give an overview of CK1ε reactivity in different organs under normal conditions and outline changes in its expression in mammary carcinomas. Our data suggest a cell/organ type-specific function of CK1ε and indicate that tumorigenic conversion of mammary glands in SV40 T-antigen-transgenic mice leads to downregulation of CK1ε. This manuscript contains online supplemental material at http://www.jhc.org. Please visit this article online to view these materials.
Keywords
C
In the present study, we examined the distribution of CK1ε in various tissues and organs of young adult, healthy BALB/c mice by immunohistochemistry (IHC) and Western blot analyses. In addition, we used real-time PCR and IHC of mammary tumors in SV40 T-Ag-transgenic mice to determine how neoplastic transformation affects CK1ε expression. Our immunohistochemical data describing the cell type- and organ-specific expression of CK1ε in BALB/c mice can be used as an anatomical basis for understanding its physiological functions. Furthermore, our data point to the possibility that down-regulation of CK1ε may potentially be involved in tumorigenesis of the mammary gland.
Materials and Methods
Animals and Tissue Processing
Animal studies were performed in accordance with the guidelines of the authority of animal use and were approved by the local authorities (Regierungspräsidium Tübingen). BALB/c mice and SV40 T-Ag-transgenic mice, strain WAP-T-NP8 (Schulze-Garg et al. 2000) (henceforward called WAP-T mice), were bred in the Animal Research Center of the University of Ulm. WAP-T mice are a well-established animal model for mammary cancer, allowing us to study morphological and biological qualities of different intraductal and invasive carcinomas. In these mice, an epitope-tagged T-Ag controlled by the WAP promoter, is induced in lactating mammary epithelium by mating. On average, 5.5 months after induction, female WAP-T mice have developed mammary tumors that typically display a poorly differentiated or even anaplastic morphology.
Tissue samples from 4- to 6-week-old BALB/c male and female mice (5/5) and normal mammary glands and mammary tumors of 18 induced female WAP-T mice were immediately excised after sacrifice, and shock frozen or fixed by immersion in either 1% acetic acid in formalin, or 10% buffered neutral formalin. Bone tissue was decalcified with EDTA for several days at 4C. Fixed tissues were then dehydrated in a graded ethanol series, cleared in methyl benzoate, and embedded in paraffin. Paraffin-embedded sections were cut at 3 μm and mounted on glass slides. Frozen tissue was embedded in Tissue-Tek (Sakura; Heppenheim, Germany). Sections (5–8 μm) were cut on a cryostat microtome (Leica; Bensheim, Germany), mounted on dry glass slides, and fixed in 100% acetone for 10 min at 4C.
Primary Antibodies
The following primary antibodies were used: CK1ε-specific polyclonal rabbit antisera 712 (Brockschmidt et al. 2008) and sc25423 (H60) (Santa Cruz Biotechnology; Santa Cruz, CA), mouse monoclonal CK1ε-specific antibody 610445 (BD Biosciences; San Jose, CA), T-Ag-specific polyclonal rabbit antiserum R15 (Deppert and Pates 1979), and mouse monoclonal anti-β-actin specific antibody A5316 (Sigma-Aldrich; Hamburg, Germany).
Immunohistochemistry
Staining of Paraffin Sections. Staining procedures included deparaffinization in xylene, followed by rehydration via transfer through graded alcohols. To inhibit endogenous enzyme activity, peroxidase blocking reagent (DAKO; Glostrup, Denmark) was used. The sections were treated with the antigen retrieval solution Citra Plus, pH 6.03 (BioGenex; San Ramon, CA) in a microwave oven according to the manufacturer's instructions. Sections were then incubated with one of the CK1ε-specific antibodies (712, 1:1200; or SC25423 (H60) 1:150) at 4C overnight. After washing in Tris-HCl buffer, horseradish peroxidase-containing polymer-conjugated anti-rabbit IgG antibody (N-Histofine; Nichirei Corporation, Tokyo, Japan) was applied at room temperature for 30 min. The enzymatic reaction was developed in a freshly prepared solution of 3,3'-diaminobenzidine using DAKO Liquid DAB Substrate-Chromogen solution as a chromogen for horseradish peroxidase. The sections were then counterstained with hematoxylin and permanently mounted in Entellan (Merck; Darmstadt, Germany). Positive and negative controls were included for each case. As a negative control, the primary antiserum was omitted, with Tris-HCl buffer as a substitute. Immunohistochemical detection of T-Ag was performed as described earlier using T-Ag-specific polyclonal rabbit antiserum R15 (Schulze-Garg et al. 2000).
Staining of Frozen Sections. Frozen sections were quickly rehydrated in Tris-HCl buffer. Endogenous enzyme activity was blocked as described above. Sections were then incubated with CK1ε-specific polyclonal serum 712 (1:1000) for 40 min at room temperature. Slides were washed in Tris-HCl buffer, and the DAB reaction was performed as described above. Next, sections were counterstained with hematoxylin.
Grading System. Sections were graded according to the intensity of the CK1ε-specific staining. Intensity levels of the CK1ε-specific staining were graded as: -, negative; +, weak; ++, moderate; or +++, strong. A slash indicates simultaneous expression of different intensities, e.g., -/++ indicates negative and moderately positive staining in one cell type or micro-anatomical region.
Western Blot Analysis. For the detection of CK1ε and β-actin protein, murine tissues were lysed in NP40 lysis buffer. Extracts were clarified by centrifugation, and the protein concentration of the lysates was determined. Seventy-five μg of the protein extract was separated on 12.5% SDS-polyacrylamide gels and transferred onto a nitrocellulose blotting membrane (Hybond C super; GE Healthcare, Chalfont St. Giles, UK). The membranes were probed with either CK1ε-specific monoclonal antibody 610445 (BD Biosciences) or with the β-actin-specific antibody (Sigma, Germany). Immunocomplexes were detected using anti-mouse or anti-rabbit peroxidase-conjugated IgG, followed by chemiluminescence detection (ECL; GE Healthcare).
Gene Expression Analysis. Total RNA was isolated from normal mammary glands and breast tumors of induced WAP-T mice using the RNeasy Kit (Qiagen; Hilden, Germany). Total RNA (1.5 μg) was reverse transcribed into cDNA using the RT2 First Strand Kit (SuperArray Bioscience Corp.; Frederick, MD). Profiling of CK1ε expression levels was done as described by the manufacturer (SuperArray Bioscience Corp.). The reactions were carried out in an Applied Biosystems 7500 Fast Real-Time PCR System (Foster City, CA). The results were read out with the 7500 Fast System SDS Software.
Results
CK1ε Expression Levels in Young Adult Mice
To characterize the CK1ε expression levels in various tissues and organs of BALB/c mice, equal amounts of the different protein extracts were analyzed by Western blotting. Although CK1ε is widely expressed, expression levels varied significantly among tissues (Figure 1). CK1ε was markedly expressed in the lung and brain; intermediate expression levels of CK1ε were found in the heart, ovary, muscle, liver, uterus, and testis; and low expression levels could be observed in the thymus, mammary gland, eye, colon, pancreas, bladder, and kidney. An increased amount of a faster migrating form of CK1ε was seen in the spleen (Figure 1).
Comparison of the Immunoreactivity of Two CK1ε-specific Antibodies in Paraffin-embedded Tissues
To analyze the cell type-specific expression of CK1ε in histological sections of different tissues and organs, immunohistology was carried out with either CK1ε-specific polyclonal rabbit antiserum 712 (Brockschmidt et al. 2008) or rabbit polyclonal CK1ε-specific antibody sc25423 (Santa Cruz Biotechnology). Comparative analysis revealed a similar reactivity of both antibodies in all paraffin-embedded tissues, as illustrated by immunohistochemical staining of the bladder and prostate (see supplementary Figure SF1).
Detection of CK1ε in Frozen and Paraffin-embedded Tissues
CK1ε-specific antiserum 712 detected CK1ε in both frozen and paraffin-embedded tissues, with similar staining strength and specificity for the heart, lung, liver, and colon (see supplementary Figure SF2). However, the quality in regard to the morphology of the frozen sections was quite variable, whereas the paraffin sections exhibited reliable and consistent results of good quality. In areas of dense staining, individual cells could still be identified in paraffin sections, but not in frozen sections, as shown in supplementary Figure SF2.
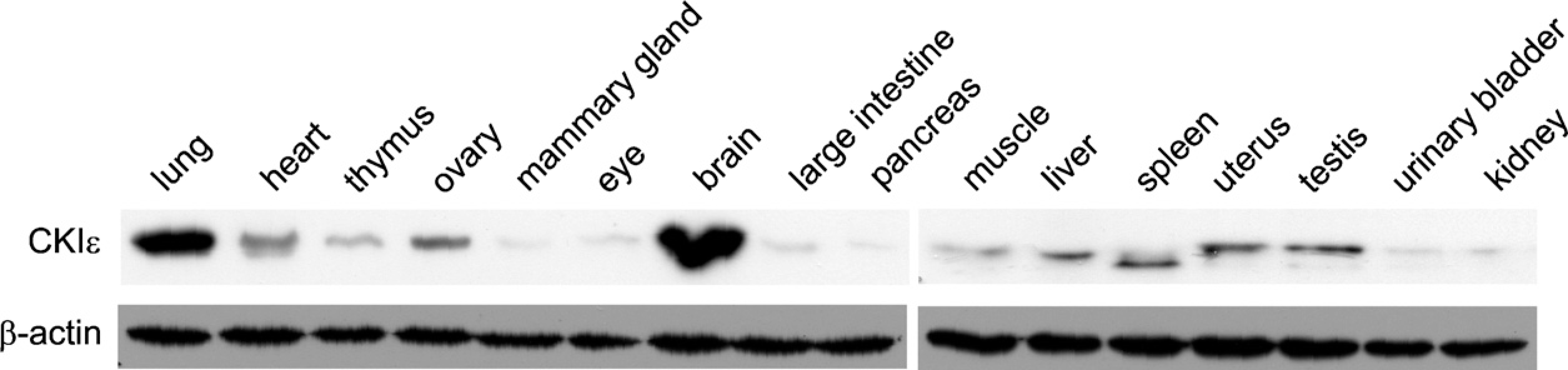
Expression level of CK1ε in different tissues and organs of 4–6-week-old BALB/c mice. Specimens were homogenized, and equal protein amounts (75 μg) were separated by SDS-PAGE. CK1ε protein levels were detected by Western blotting using a CK1ε-specific monoclonal antibody (BD Biosciences). Equal loading of extracted proteins was determined by probing a second membrane with a β-actin-specific monoclonal antibody.
PGD results



Intensity levels of the CK1ε-specific staining were graded as: -, negative; +, weak; +, moderate; or +++, strong. Slash indicates simultaneous expression of different intensities, e.g., -/++ indicates negative and moderately positive staining in one cell type. Staining results were nearly identical independent of the CK1ε-specific antibody [712 and (H60) sc-25423] having been used. MALT, mucosa-associated lymphoid tissue.
Detection of CK1ε Immunoreactivity in Paraffin-embedded Tissues of Young Adult BALB/c Mice
CK1ε reactivity in various paraffin-embedded tissues of 10 young adult BALB/c mice (5 males/5 females) determined by IHC is summarized in Table 1. These results correspond well with our results obtained by Western blot analysis.
Subcellular Antigen Distribution
In most immunoreactive cells, CK1ε was localized to the cytoplasm. Only some cell populations, e.g., male germ cells, displayed a nuclear immunoreactivity (Figure 2S).
Alimentary Tract
In the keratinized squamous epithelium lining, the esophagus, and parts of the murine stomach, CK1ε was moderately positive in the basal, parabasal, and intermediate cell layers, increasing to strong staining levels in the superficial stratum (Figure 2A). The various cell types that constitute the epithelium of the glandular part of the murine stomach showed a heterogeneous CK1ε expression pattern. At the base of the crypts, chief cells were strongly CK1ε positive, in contrast to parietal cells, which expressed only low to intermediate amounts of CK1ε. Toward the more superficial parts of the gastric mucosa, mucous neck cells and surface epithelial cells were weakly CK1ε positive (Figures 2B1, 2B2). Intense CK1ε positivity was observed in the absorptive epithelium of the small and large intestine (Figure 2C). Some goblet cells expressed moderate levels of CK1ε, whereas a subset of goblet cells lacked CK1ε (Figures 2C and 2D). In the small intestine, Paneth cells were CK1ε negative or weakly positive (Figure 2C).
Large Exocrine Glands of the Alimentary Tract
The large salivary glands draining in the oral cavity were composed of CK1ε-negative serous and myo-epithelial cells and mucous cells that were weakly positive for CK1ε. The duct epithelia displayed a moderate CK1ε staining intensity (Figure 2E). In the serous glandular epithelium of the exocrine part of the pancreas, CK1ε expression was low and in some cells negative. The cuboidal/columnar epithelium lining the pancreatic ducts showed weak CK1ε expression (Figure 2F and inset). In the liver, moderate CK1ε staining was seen in the hepatocytes (Figure 2G). Staining of the parenchyma was diffuse throughout the different zones of the liver acinus. The intrahepatic bile ducts demonstrated intermediate CK1ε immunoreactivity (Figure 2G inset).
Endocrine Organs
We confined our immunohistochemical analysis of the endocrine organs to the pancreas (Figure 2F), the adrenal (Figure 2H), the thyroid (Figure 2I), and the anterior lobe of the hypophysis (Figures 3A and 3B). Other components of the endocrine system, i.e., the parathyroid gland and the diffuse endocrine system of the mucosal surfaces, were excluded from our studies, owing to difficulties in anatomical preparation or in histological detection, respectively.
Cells of the endocrine pancreas, i.e., the Islets of Langerhans, were moderately to strongly CK1ε positive (Figure 2F). Without resorting to double immunostaining techniques, it was not possible to further specify CK1ε expression in different Langerhans cell subsets, as defined by the peptide hormone produced. In young adult mice, the adrenal cortex consisted of the moderately CK1ε-positive zona glomerulosa, the weakly stained zona fasciculata, and the strongly CK1ε-expressing X zone (Figure 2H). CK1ε expression was markedly heterogenous in the adrenal medulla, which contained strongly CK1ε-positive cells as well as a CK1ε-negative subset (Figure 2H). The thyroid gland showed low to intermediate levels of CK1ε expression in the follicular epithelium (Figure 2I).
All three parts of the pituitary gland exhibited cytoplasmic CK1ε expression. There was no obvious variation in expression among the different cell types of the adenohypophysis (Figures 3A and 3B). In the neuro-hypophysis, pituicytes showed strong cytoplasmic CK1ε immunoreactivity (Figure 3C). The neuropil was moderately labeled.
Respiratory Tract
The ciliated respiratory epithelium lining the trachea (Figure 2J) and the proximal branches of the bronchial tree (Figure 2K1) was intensely CK1ε positive. CK1ε expression was reduced to weak levels upon transition to the type I pneumocytes of the respiratory bronchioles and the lung alveoli, whereas surfactant-producing type II pneumocytes retained moderate CK1ε levels (Figures 2K1 and 2K2). The small glands of the respiratory tract contained CK1ε-negative serous epithelial cells and mucous cells that displayed low amounts of CK1ε.
Skin and Skin Appendages
The squamous epithelium of the skin was moderately CK1ε positive in the basal cell layers, increasing to strong antigen expression toward the superficial parts of the epidermis. Hair follicles displayed intermediate levels of CK1ε expression. Sebaceous glands and Haderian glands were weakly CK1ε positive (Figure 2L). In the non-lactating mammary gland, moderate CK1ε expression was seen in the secretory epithelium, whereas the myoepithelium was CK1ε negative (Figures 2M1 and 2M2).
Urinary Tract
Cells of the renal glomerulum were CK1ε negative or weakly positive (Figure 2N2). This low staining intensity, in conjunction with the morphology obtainable in CK1ε immunohistochemical preparations, did not permit a more detailed assignment of CK1ε expression to specific glomerular cell populations. The tubular epithelium was strongly CK1ε positive in the intermediate part, and moderately positive in the proximal and distal sections, whereas low CK1ε expression was found in the epithelium of the collecting ducts (Figures 2N1, 2N2, and 2N3). The transitional epithelium of the ureter and the urinary bladder was weakly CK1ε positive, increasing to moderate levels in the superficial (umbrella) cells (Figure 2O).
Female Genital System
Immunohistochemical staining of the ovary revealed weak to intermediate CK1ε expression in the oocytes (Figures 2P1 and 2P2). The follicular epithelium was moderately CK1ε positive at immature stages, dropping to no or only weak staining in more-mature follicles (Figures 2P1 and 2P2). Theka cells surrounding the ovarial follicles were weakly CK1ε positive (Figures 2P1 and 2P2). The epithelia of the oviducts and the uterine mucosa displayed intermediate to strong levels of CK1ε expression, whereas the endometrial stroma cells were only weakly CK1ε positive (Figure 2Q). The squamous epithelium of the vagina was strongly CK1ε positive (Figure 2R).
Male Genital System
In the germinal epithelium of the testis, CK1ε expression was intense in the spermatogonia and slightly decreased in the course of maturation toward spermatocytes (Figure 2S). Sertoli cells were weakly CK1ε positive, and Leydig cells of the interstitium were moderately CK1ε positive (Figure 2S). Strong CK1ε expression was observed in the epithelial lining of the epididymis and the spermatic duct (Figure 2T). The epithelial cells of the seminal vesicle and the prostate gland were strongly to moderately CK1ε positive (Figure 2U).
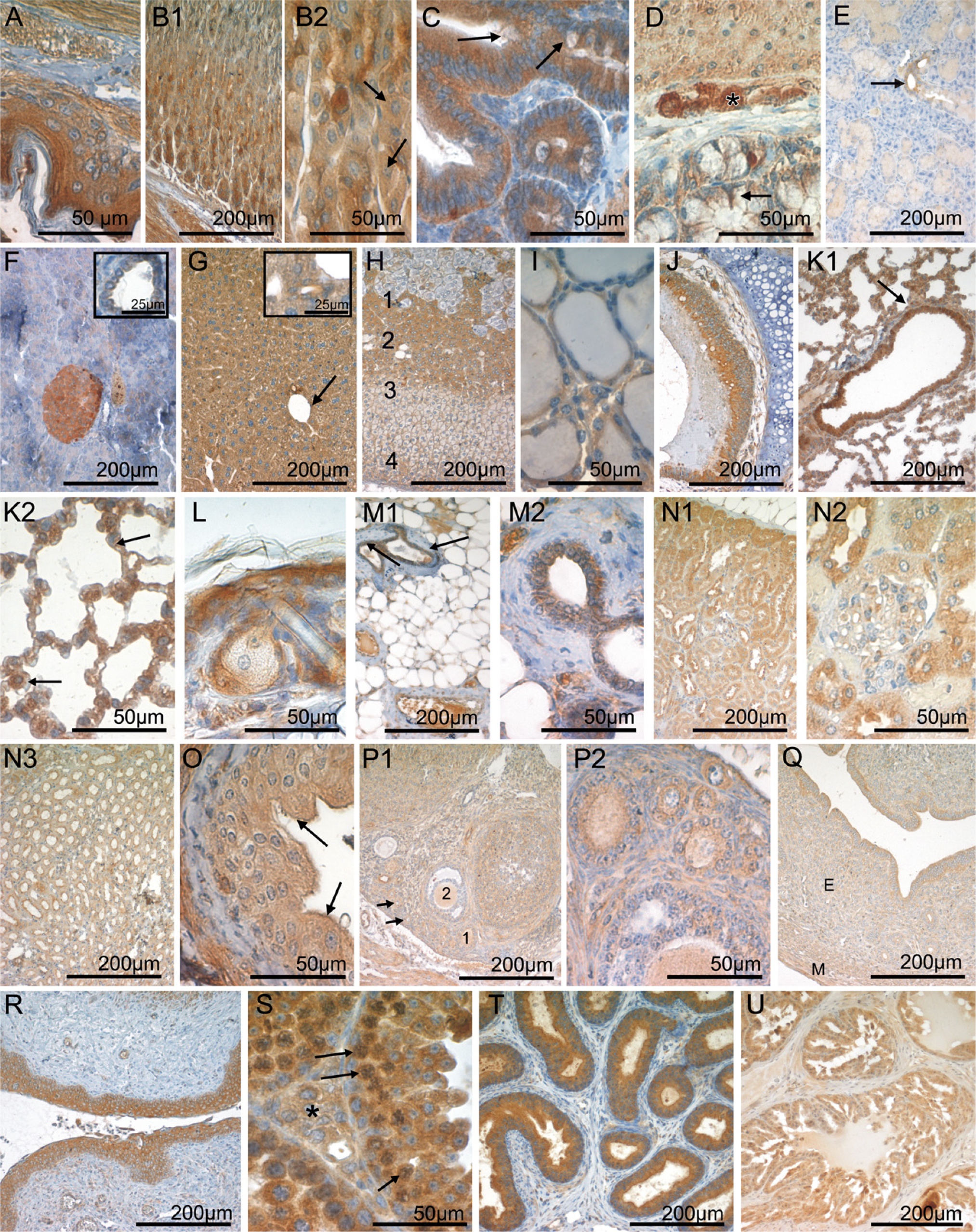
CK1ε expression in the gastrointestinal tract, endocrine glands, lung, mammary gland, skin, and urogenital tract. Fixative: acid formalin, fixation by immersion. Peroxidase reaction, dye: DAB. CK1ε-specific serum 712 was used for immunohistochemical staining of the following organs: esophagus (
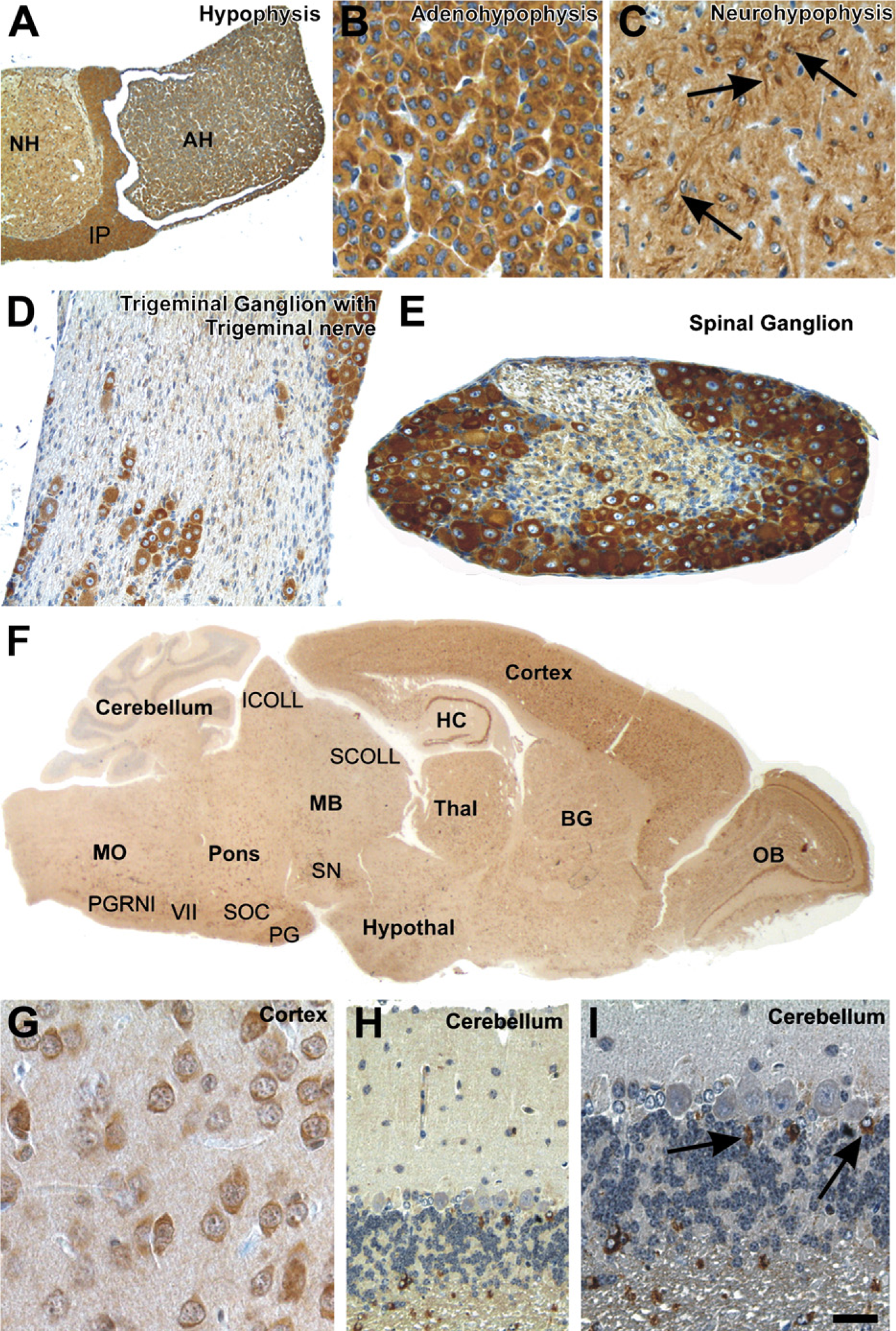
CK1ε expression in the nervous system. Fixative: acid formalin, fixation by immersion. Peroxidase reaction, dye: DAB. CK1ε expression in the hypophysis, the peripheral nervous system, and the central nervous system. (
Immobile Cells of Mesenchymal Origin
Skeletal and cardiac muscle cells showed a heterogeneous staining pattern consisting of CK1ε-negative and CK1ε-moderately positive myocytes (Figures 4A1, 4A2, and 4B). The staining in these cell populations was mostly diffuse; however, in the myocardium, an increased staining intensity was observed in the intercalated discs. Smooth-muscle cells at the various locations showed an intermediate level of CK1ε expression (Figure 4C). The vascular endothelium of arterial vessels and high endothelial venoles (as seen in secondary lymphatic organs) (Figures 4D1 and 4D2) contained intermediate amounts of CK1ε, whereas veins and lymphatics harbored a weakly CK1ε-stained endothelium (Figure 4D1). Mesothelial cells lining the peritoneal and the pleural cavity as well as the pericardium were invariably CK1ε weakly positive (Figure 4E).
Adipocytes of the plurivacuolated (“brown”) and univacuolated (“white”) type were weakly to moderately CK1ε positive (Figures 4F1 and 4F2). Fibrocytes, chondrocytes, and osteocytes showed no or only weak CK1ε expression (Figures 4G, 4H, and 4I). In contrast, osteoblasts were intensely CK1ε positive (Figures 4H and 4I).
Hematopoietic Cells and Immune System
Immature hematopoietic cells in the bone marrow (Figure 4J) and at sites of extramedullary hematopoiesis [i.e., the spleen (Figure 4K)] displayed varying levels of CK1ε expression, ranging from negative to strongly positive. Although strongly CK1ε-expressing megakaryocytes were readily identified by their size and distinctive morphology, it was not possible to distinguish precisely the different cell types in the bulk of hematopoietic cells on the basis of single immunostainings carried out in the present study.
In the thymus, a large population of small lymphoid cells probably representing thymocytes was CK1ε negative to weakly positive (Figure 4L). In the thymic medulla, a moderately to strongly CK1ε-positive stroma cell population was observed (Figure 4L). Lymphocytes of the secondary lymphatic organs displayed varying amounts of CK1ε expression, depending on the organ type and the microanatomical compartment. CK1ε expression tended to be strongest in the germinal centers of secondary lymphoid follicles seen in lymph nodes, whereas mantle zones and primary follicles were CK1ε negative (Figures 4M and 4N). In T zones, weak CK1ε expression was observed (Figures 4M, 4N, and 4O). In the white pulp of the spleen, no or only weak CK1ε expression was observed, corresponding to a predominating population of resting B and T lymphocytes (Figure 4K). At the border between the T/B zone and the marginal zone, macrophages, here called sinusoidal lining cells, were conspicuous by their strong CK1ε positivity. In the red pulp, the majority of cells showed intermediate CK1ε staining.
Eye
The microanatomical complexity of the retina was mirrored by a differential CK1ε expression in its various layers. The ganglion cell layer was weakly CK1ε positive (Figures 4P and 4Q). Fluorescent double staining revealed a colocalization of CK1ε staining with βIII-positive retinal ganglion cells (Figures 4R-4T). No CK1ε staining was observed in the inner nuclear layer, whereas the outer plexiform layer displayed a significant CK1ε staining intensity. The outer nuclear layer and the outer segment of the photoreceptors lacked CK1ε expression. In contrast, the inner segment of photoreceptors was CK1ε positive. CK1ε was weakly expressed by the retinal pigment epithelium (Figure 4P). Adjacent to the retina, the vascularized connective tissue of the choroidea was moderately CK1ε positive (data not shown). No CK1ε staining was seen in the sclera or the cornea (data not shown). The capsule of the lens was CK1ε negative, and the underlying cuboidal epithelium stained weakly. The fibers in the center of the lens lacked CK1ε, whereas intermediate expression levels were detected in the cortical fibers. The stroma of the iris was moderately CK1ε positive. The epithelial cells lining the posterior surface of the iris displayed weak CK1ε staining. In contrast, the epithelium covering the ciliary body was moderately CK1ε positive, whereas weak antigen expression was found in the smooth ciliary muscle (Figure 4P).
Peripheral and Central Nervous System
Peripheral Nervous System. The peripheral nerve fibers were only weakly labeled for CK1ε. Primarily the Schwann cells exhibited a slight intracytoplasmatic immunoreactivity, whereas the axons did not appear to be marked (Figure 3D). In the peripheral ganglia, the nerve cells showed a strong immunoreaction with anti-CK1ε in the soma (Figures 2D and 3D).
Central Nervous System. The cross-section of the hypophsis shows strong CK1ε expression in the adenohypophysis, the neurohypophysis, and in the intermediate part (Figures 3A-3C). CK1ε was expressed in all neurons of the olfactory bulb, cerebral cortex, and hippocampus (Figures 3F and 3G). Most neurons in the basal ganglia, hypothalamus, thalamus, midbrain, pons, and medulla oblongata also exhibited cytoplasmic staining for CK1ε (Figure 3F). Only the Purkinje and granule cells in the cerebellum did not show significant amounts of CK1ε labeling (Figures 3H and 3I). However, a subset of astrocytes was strongly marked with anti-CK1ε (Figure 3I). The neurons of the spinal cord exhibited a moderate cytoplasmatic expression of CK1ε.
CK1ε Expression Is Altered in Ductal Carcinoma of the Mammary Gland in SV40-transgenic Mice
To get hints for a role of CK1ε in tumorigenesis of mammary cancer, we used an SV40 T-Ag-inducible transgene animal model for ductal carcinoma. In this model, the tumors mostly display a poorly differentiated or even anaplastic morphology (see Figure 5; Table 2). Immunostaining revealed that expression of nuclear T-Ag in mammary epithelium preceded the development of morphologically detectable tumors. Of note, T-Ag expression was associated with a high mitotic rate. In overt neoplasia, i.e., intraductal and invasive carcinoma, T-Ag expression lingered in most tumors (Figure 5A), although in a small number of tumors, T-Ag was downregulated. CK1ε was detected in the cytoplasm of normal and neoplastic epithelial cells. In normal breast tissue of lactating mice, a strong CK1ε immunoreactivity was observed within the cytoplasm of ductal and acinar epithelial cells (Figure 5A). Upon neoplastic transformation to ductal carcinoma in situ (DCIS), CK1ε staining was downregulated. The lowest CK1ε immunoreactivity was observed in high-grade invasive carcinomas, i.e., those that are poorly differentiated (Figure 5A; Table 2). Correspondingly, on the RNA level, the expression of CK1ε was reduced in tumors of induced transgenic mice, compared with normal tissue of induced transgenic mice and non-transgenic mice (Figure 5B).
Discussion
CK1ε exhibits a variety of functions in eukaryotic cells. It is involved in the control of developmental processes, cell division, and circadian rhythm. Deregulation of CK1ε has been linked to pathophysiological processes such as carcinogenesis and neurodegeneration (Knippschild et al. 2005; Price 2006). However, amazingly little is known about the organ and cell type-specific distribution of CK1ε. To fill this gap and to obtain more information on its physiological role, we determined the expression of CK1ε in tissues and organs of young adult BALB/c mice. Furthermore, we assessed changes in the expression of CK1ε during tumorigenesis in a murine model of mammary carcinoma.
Western blot analysis revealed expression of CK1ε in all analyzed organs and tissues, with notably high protein levels in the brain and lung. Immunohistochemical staining using two different CK1ε-specific antibodies confirmed the ubiquitous expression patterns of CK1ε. We observed a distribution pattern for CK1ε similar to that recently described for CK1δ (Lohler et al. 2009). This may not be surprising in view of the high homology between CK1δ and CK1ε. A differential expression of CK1δ and CK1ε was observed in only a few cell types (summarized in Table 3). In the following, we will discuss our data on the background of previous findings describing cell type-specific functions of CK1ε.
Endocrine Tissue
CK1ε is highly expressed in endocrine organs, pointing to an involvement in the production, storage, and/or secretion of hormones, as previously described for other CK1 family members (Gross et al. 1997; Gross and Anderson 1998; Murakami et al. 1999; Behrend et al. 2000b; Knippschild et al. 2005; Lohler et al. 2009). We observed strong cytoplasmic CK1ε positivity in all three parts of the pituitary gland, indicating that CK1ε might be involved in specific functions of the SNARE complex and in neurotransmitter release, like CK1α and -δ (Gross et al. 1995; Shimazaki et al. 1996; Reisinger and Allerberger 1999; Kataoka et al. 2000; Pyle et al. 2000; Chheda et al. 2001; Dubois et al. 2002; Snyder et al. 2006; Wolff et al. 2006; Pozzi et al. 2008). In the testis, CK1ε positivity of Leydig cells suggests regulatory functions of CK1ε in vesicle transport and hormone release (Knippschild et al. 2005, and references therein). In the pancreas, CK1ε was weakly expressed in the exocrine part. In contrast, a high CK1ε immunoreactivity was observed in the Islets of Langerhans, with gradual differences in the expression level in individual islet cells. Similarly, gradual differences in CK1ε immunoreactivity were seen in cells of the adrenal and pituitary glands. Additional experiments are necessary to clarify whether CK1ε expression depends on the level of stimulation or the type of hormone-producing cell.
Immune System
Like CK1δ, CK1ε was found to be expressed in all lymphatic organs. Resting B lymphocytes in primary follicles and in the mantle zone of secondary follicles lacked CK1ε, whereas the germinal centers of secondary follicles, where activated B-cells predominate, expressed substantial levels of CK1ε. Thus, CK1ε positivity and activation status seemed to correlate in the B-cell compartment. Because several components of the B-cell activation process critically depend on nuclear factor ‘kappa-light-chain-enhancer’ of activated B-cells (NFκB), it is tempting to speculate that CK1ε might interfere with the regulation of the NFκB pathway in B lymphocytes. The CK1ε immunoreactivity seen in germinal centers might also indicate regulatory functions of CK1ε in proliferation, mobility, and antigen recognition. Our data are in line with previous reports showing an involvement of CK1δ/ε in lymphocyte physiology and a role of CK1ε in cytokine-induced differentiation of granulocytes (Maritzen et al. 2003; Okamura et al. 2004a). The adaptive immune system is remarkable for the high level of functional differentiation of its cellular components. No reliable morphological criteria are available to identify the multitude of immune cell types known today. Inasmuch as the data presented here provide a low-resolution image of the lymphoid system, it will be important to study CK1ε in immune cell subsets defined by a combination of their marker profile and microanatomical location. This could be achieved by using double immunofluorescence staining of lymphoid tissues or by examining CK1ε expression in distinct lymphoid tumor entities that represent the neoplastic counterpart of defined B-cell populations.
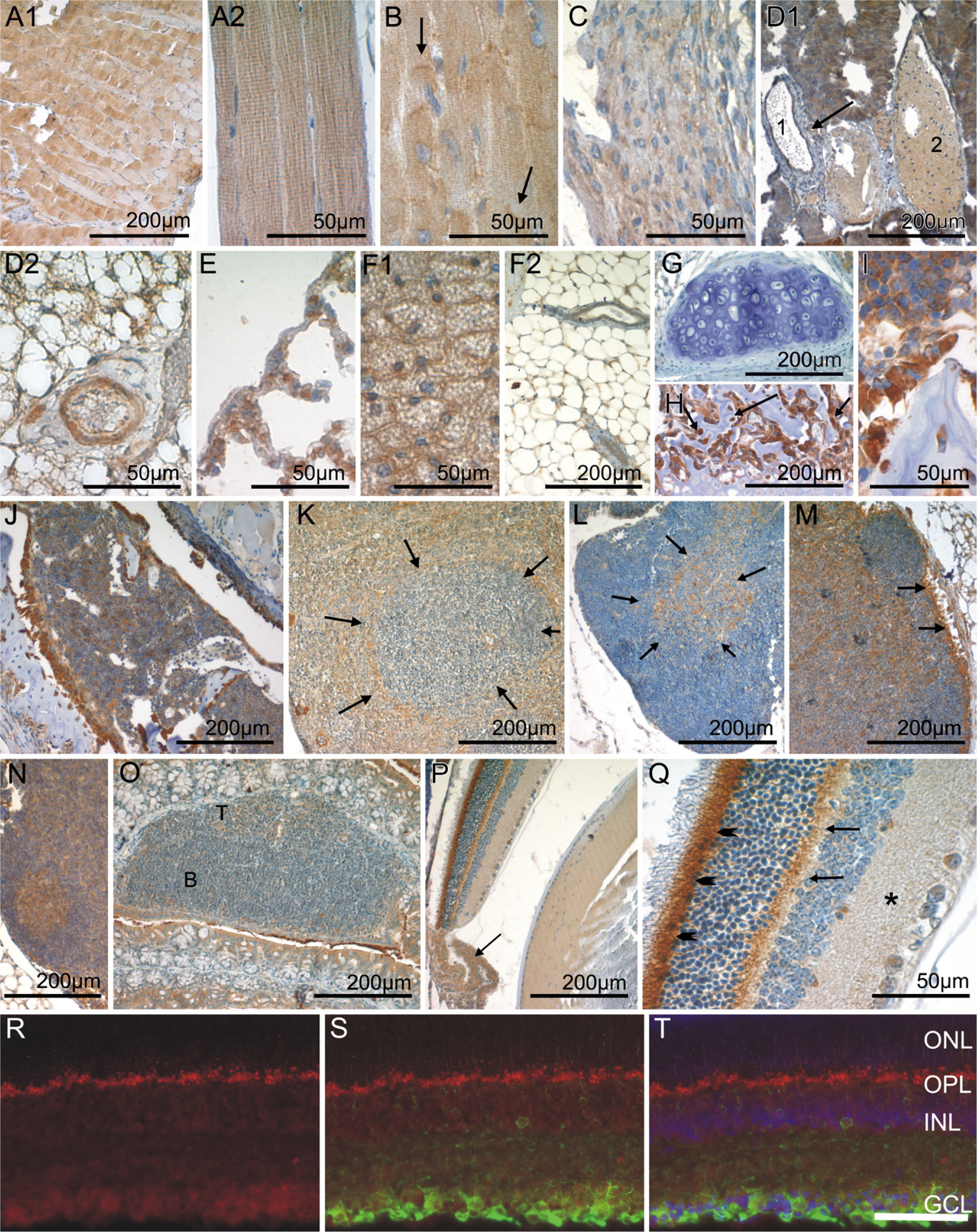
CK1ε expression in immobile cells of mesenchymal origin, in hematopoetic and lymphoid organs, and in the eye. Fixative: acid formalin, fixation by immersion. Peroxidase reaction, dye: DAB. CK1ε-specific antiserum 712 was used for immohistochemical staining of the following organs/tissues: skeletal muscle (
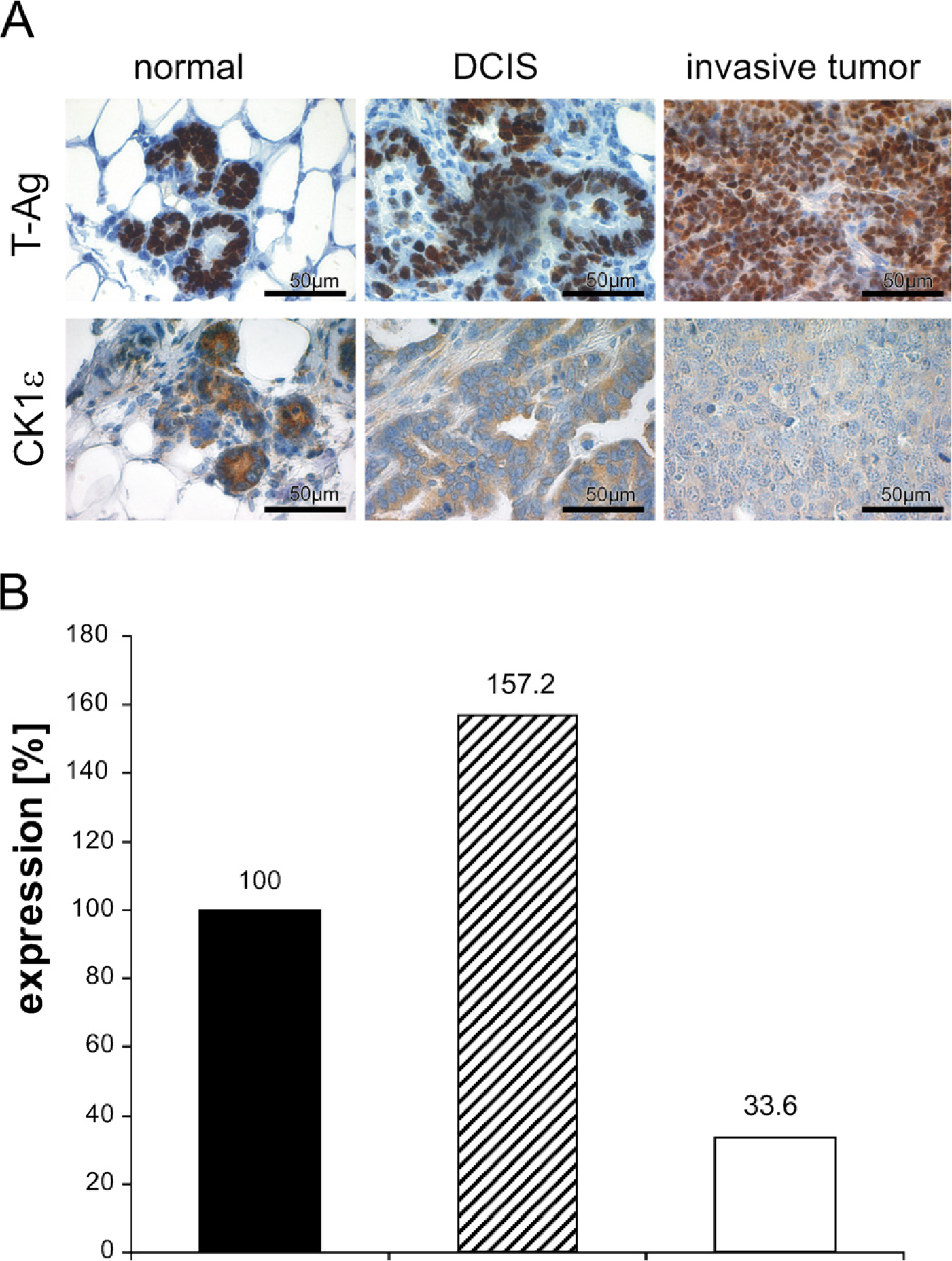
CK1ε expression in mammary epithelium is downregulated upon neoplastic transformation (
Central Nervous System
CK1ε expression in the brain revealed a widespread distribution similar to that described for CK1δ (Lohler et al. 2009), suggesting that the closely related CK1 isoforms exhibit similar biological functions. However, a few differences were detected (see also Table 3). In the cerebellum, Purkinje cells were strongly CK1δ positive, but negative for CK1ε. In contrast, astrocytes showed high CK1ε but no CK1δ immunoreactivity. The strong CK1ε positivity in neurons of various neuroanatomical compartments and the thalamus, and the heterogenous neuronal expression in the nuclei of the midbrain, pons, and medulla oblongata point to regulatory, cell type-specific functions of CK1ε in neuronal signal transduction and/or metabolism. These findings are in line with previous observations showing that CK1ε is involved in regulating the circadian rhythm and that deregulation of CK1 isoforms, especially those of CK1ε and -δ, lead to neurodegenerative disorders such as Alzheimer disease and circadian disorders (Greengard 2001; Takano et al. 2004; Chergui et al. 2005; Gould and Manji 2005; Meng et al. 2008; Bryant et al. 2009).
CK1ε expression in normal tissue, in situ carcinoma, and invasive tumors of SV40 T-Ag-induced transgenic mice

Intensity levels of CK1ε-specific staining were graded as: -, negative; +, weak; ++, moderate; or +++, strong. +/++ Indicates weakly and moderately positive staining in tumor cells. Ø, no in situ carcinoma detected in the specimen.
Reproductive Organs
In the testis, CK1ε expression was high in spermatogonia, i.e., the self-renewing pool of spermatogenesis characterized by a high mitotic rate. This indicates that CK1ε might be involved in the regulation of chromosome segregation and cell division, an assumption supported by several reports linking CK1 isoforms to mitosis and meiosis (Brockman et al. 1992; Horiguchi et al. 2005; Petronczki et al. 2006; Grozav et al. 2009). Upon maturation to spermatocytes, CK1ε was down-modulated to intermediate expression levels. Possibly, lower CK1ε amounts are engaged in meiotic cell division, compared with mitosis.
Cell type-specific differences in the expression level of CK1δ and -ε
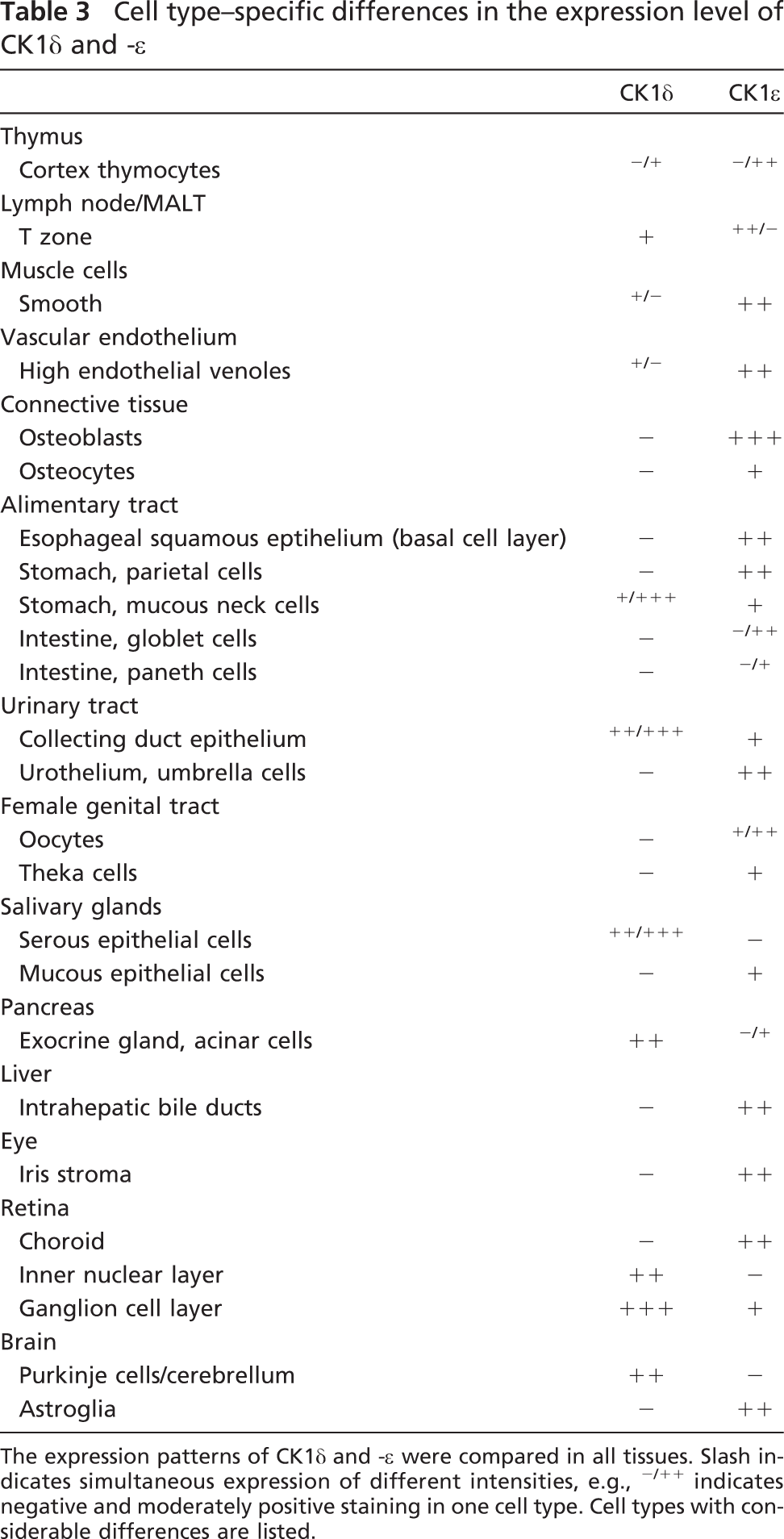
The expression patterns of CK1δ and -ε were compared in all tissues. Slash indicates simultaneous expression of different intensities, e.g., -/++ indicates negative and moderately positive staining in one cell type. Cell types with considerable differences are listed.
Mammary Tumors of Induced SV40 T-Ag-transgenic Mice
We observed changes in the immunoreactivity of CK1ε in preinvasive and invasive breast carcinoma of SV40 T-Ag-transgenic mice. Whereas CK1ε is highly expressed in the cytoplasm of normal ductal and acinar cells, lower levels were detected in ductal carcinoma in situ. The lowest expression was found in poorly differentiated invasive tumors. The observed downregulation of CK1ε might be due to lack of differentiation of the mammary tumors. Alternatively, downregulation of CK1ε could provide a potential mechanism leading to the development of the chemoresistance of tumor cells, especially to topoisomerase II drugs. The functions of topoisomerase IIα is regulated by site-specific phosphorylation, especially at Ser 1106 mediated by CK1δ and -ε. Hypophosphorylation of topoisomerase IIα at Ser 1106 correlates with resistance to topoisomerase II drugs and a reduced apoptosis rate (Grozav et al. 2009). Down-regulation of CK1ε has also been described for human mammary cancer. Fuja et al. (2004) observed a correlation between immunohistological CK1ε staining intensity and tumor differentiation. However, considering the similarities of DCIS and invasive mammary carcinoma development in transgenic mice to the respective human diseases, it seems worthwhile to analyze in detail the prognostic relevance of changes in the expression of CK1ε and its highly homologous CK1δ in tumor development and progression.
In summary, the widespread distribution of CK1ε indicates its involvement in the regulation of various cellular processes. Our detailed anatomical profiling of CK1ε expression can be used in further studies to characterize cell type-specific functions of CK1ε and to determine pathophysiological alterations in various diseases. It will be particularly interesting to determine whether high CK1ε expression levels in cell populations with a negligible proliferative activity, such as some endocrine cells, hint of a yet-unknown function of this enzyme. The observed downregulation of CK1ε in mammary tumors of SV40 T-Ag-induced mice is similar to that in human tumors, and thus could be of clinical interest in the study of the development and progression of breast cancer. Further biochemical, molecular, and functional analyses in the transgenic mice model and in humans are needed to more precisely elucidate the significance of changes in the expression of CK1ε and to correlate CK1ε expression with clinical outcome.
Footnotes
Acknowledgements
This work was supported by Grant 108489 from the Deutsche Krebshilfe, Dr. Mildred Scheel Stiftung (to UK). The Heinrich-Pette-Institute is financially supported by the Freie und Hansestadt Hamburg and the Bundesministerium für Gesundheit.
We thank Dr. Jürgen Löhler for his advice and helpful discussions. We are grateful to Arnhild Grothey for her technical support.
