Abstract
We investigated the cells that express platelet-derived growth factor receptor α (PDGFRα) during mouse embryogenesis. PDGFRα expression has been identified by in situ hybridization or immunohistochemistry using polyclonal antibodies on tissue sections. Because no immunostaining study using whole-mount specimens has been published to date, we established a new monoclonal antibody (MAb), APA5, for this purpose. Our results differed in that APA5 stained only the paraxial mesoderm, whereas other investigators concluded that most if not all mesodermal cells expressed PDGFRα. Moreover, we did not find PDGFRα expression in embryonic erythrocytes, which have been previously suggested to express PDGFRα. On the basis of our present results, we wish to revise the proposed PDGFRα expression as follows. At the pregastrulation stage, PDGFRα is expressed only in primitive endoderm, particularly that in the ectoplacental cone. On gastrulation, it is expressed at high levels in the paraxial mesoderm. This expression continues after its differentiation into the somite. Along with the differentiation and migration of the sclerotome, PDGFRα+ cells begin to become distributed throughout the embryonal mesenchyme. During organogenesis, particularly intense staining is detected in regions of epithelial and mesenchymal interaction, such as the tooth bud and bronchi. In addition to mesodermal derivatives, the developing lens, apical ectodermal ridge, glial precursor, cardiac valves, and choroid plexus express PDGFRα. Our results with whole-mount immunostaining show that PDGFRα is abundantly expressed and may play important roles during embryogenesis.
P
We therefore established an MAb that specifically recognizes PDGFRα. Here we describe MAb APA5, which can be used for Western blotting, immunoprecipitation, immunostaining of tissue sections, whole mount immunostaining and the surface staining of living cells. Using this MAb, we investigated the immunolocalization of PDGFRα during murine embryogenesis. Most of the staining profiles obtained here were comparable to those of in situ hybridization analyses (Pringle and Richardson 1993; Morrison-Graham et al. 1992; Orr-Urtreger and Lonai 1992; Orr-Urtreger et al. 1992; Schatteman et al. 1992; Holmgren et al. 1991). However, our results differed in that APA5 stained only the paraxial mesoderm, whereas previous reports concluded that most if not all mesodermal cells expressed PDGFRα.
Materials and Methods
Animals
C57BL/6 mice and Wistar rats were purchased from Japan SLC (Shizuoka, Japan). Ph/+ mutant mice were a gift from Dr. L.D. Shultz of the Jackson Laboratory and were maintained in our animal colony for a few generations. Females were paired with males overnight and vaginal plugs were checked in the following morning. The noon when a vaginal plug was detected was designated as day 0.5 of gestation (0.5 day postcoitum, 0.5 dpc).
Production of Anti-murine PDGFRα Antibody
The production of anti-murine PDGFRα MAb and its characterization by Western immunoblotting were previously described (Takakura et al. 1996). Briefly, a cDNA fragment corresponding to the extracellular domain of murine (m)PDGFRα (Do et al. 1992) was generated by reverse transcription-PCR (RT-PCR) amplification of mRNA prepared from mouse brain. The fragment was cloned into pCRII vector (Invitrogen; San Diego, CA). A cDNA fragment of the cloned PDGFRα gene was inserted into CD4Rg (Sudo et al. 1993) from which CD4 gene was removed. Subsequently, a fusion gene containing the extracellular domain of PDGFRα and the Fc portion of human IgG1 was inserted into pEFBOS (a gift from S. Nagata, Osaka Bioscience Institute) (Mizushima and Nagata 1990) and transfected into COS1 cells in Dulbecco's modified Eagle's medium/F-12 medium (GIBCO; Grand Island, NY) supplemented with 1% Nutridoma-NS (Boehringer; Mannheim, Germany). The mPDGFRα-human(h) IgG1 recombinant fusion protein was purified from the culture supernatant of the transfected COS1 cells using Procep-A (Bioprocessing; Princeton, NJ). The spleen cells from a Wistar rat that was immunized and boosted with this fusion protein were fused with X63.Ag8 cells as described (Sudo et al. 1993). The hybridoma supernatants were screened by their binding to the fusion protein and by staining a PDGFRα-positive BALB/c-3T3 cell line (Olashaw et al. 1991).
Immunohistochemistry
Tissue fixation procedures were basically the same as described (Yoshida et al. 1993), but microwave irradiation for prefixation proceeded for 20 sec at 600 W in a microwave oven (Toshiba ER-365S; Tokyo, Japan). Fixed specimens were embedded in polyester wax and sectioned at 5 μm as described (Kusakabe et al. 1984). Immunostaining procedures were the same as described (Takakura et al. 1996).
Whole-mount immunohistostaining was performed according to the protocol described by Davis (1993), with some modifications. In brief, embryos were prefixed in 0.1 M phosphate buffer, then incubated in fixing solution (methanol:dimethyl sulfoxide 4:1) for 2 hr to overnight at 4C according to the size of specimen. To block endogenous peroxidase, the fixed specimens were bleached (methaol: dimethylsulfoxide:30% H2O2 4:1:1) for 2 hr at room temperature (RT). For staining, the rehydrated specimens were first blocked by incubating twice in PBSMT (2% skim milk and 0.1% Triton X-100 in PBS) containing 1% normal goat serum and 0.2% BSA for 1 hour at RT, incubated with PBSMT containing anti-PDGFRα MAb APA5 (0.5 μg/ml) overnight at 4C, then washed five times in PBSMT each for 1 hr at 4C for the initial three washes and at RT for the final two. The primary antibody was developed by incubating 1 μg/ml HRP-conjugated anti-rat Ig antibody (Biosource; Camarillo, CA) overnight at 4C. After extensive washing with more than five exchanges of PBSMT, including the final 20-min wash in PBST (0.1% Triton X-100 in PBS) at RT, the specimens were soaked in PBST containing 0.05% NiCl2 and 250 μg/ml diaminobenzidine (Dojin Chem; Kumamoto, Japan) for 10–30 min, and hydrogen peroxide was added to 0.01%. The enzymatic reaction was allowed to proceed until the desired color intensity was reached, and the specimens were rinsed three or four times in PBST. Finally, the specimens were dehydrated in 100% methanol and stored at −20C until photographed.
Cell Preparation and Flow Cytometry
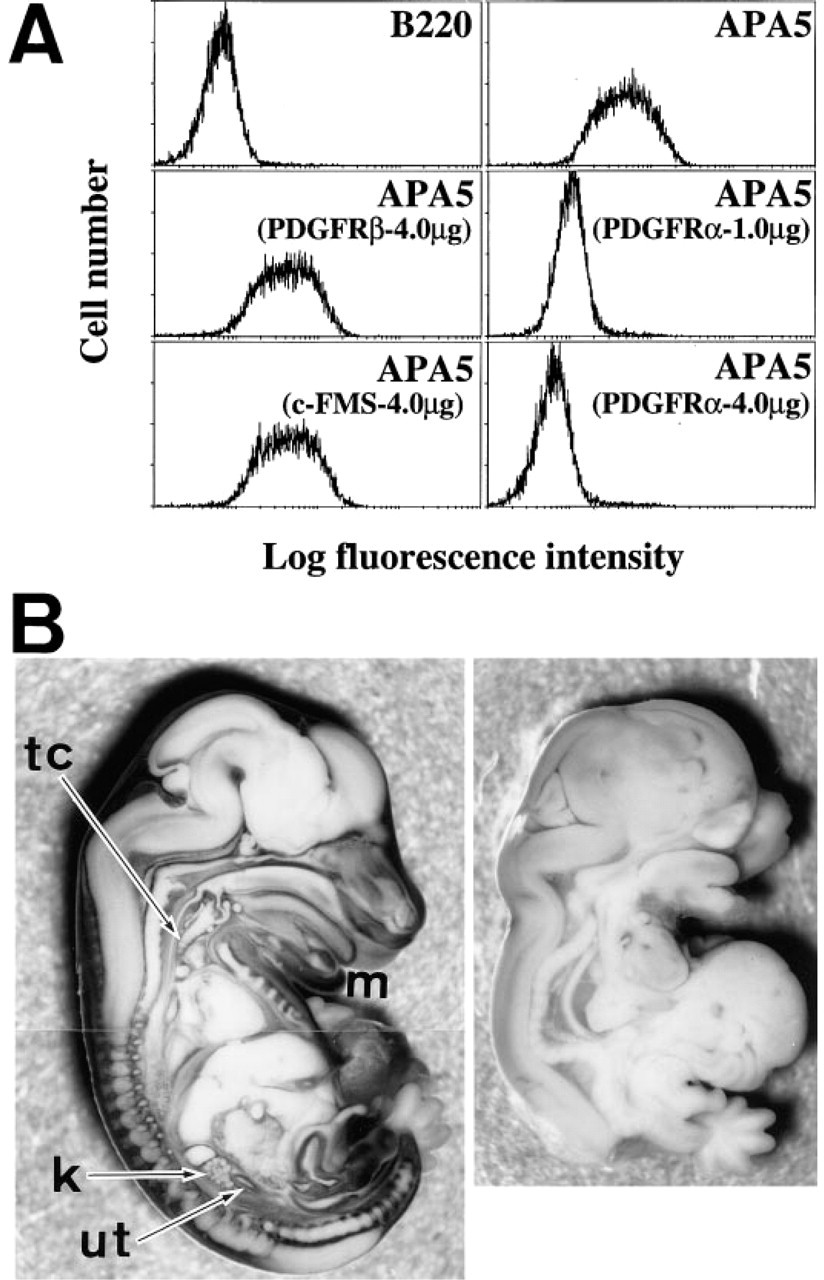
The fibroblast cell line BALB/c-3T3 was maintained in DMEM (GIBCO) containing 10% FCS (Bio Whittaker, lot no. 1M1250; Walkersville, MD). Exponentially growing BALB/c-3T3 cells on 10-cm tissue culture plates were harvested by an incubation in 10 mM ethylenediamine tetraacetic acid containing PBS for 10 min at 37C. Cells (5 × 105) were suspended in 50 μl of 0.1% BSA containing Hanks' solution (Nissui; Tokyo, Japan) (HANKS/BSA) and nonspecific binding of the MAb was blocked with 10 μl of normal mouse serum. The cells were then stained with FITC-conjugated APA5 in the presence or absence of either mPDGFRα, mPDGFRβ (unpublished), or mC-FMS (Sudo et al. 1995) hIgG1 chimeric protein for 15 min at 4C. Finally, the stained cells were washed twice in HANKS/BSA and analyzed using an EPICS XL (Coulter; Hialeah, FL).
Results
Specificity of APA5
The specificity of APA5 was first tested by staining BALB/c-3T3 cells that expressed both PDGFRα and PDGFRβ. This staining was inhibited by the recombinant hIgG/PDGFRα but not by hIgG/PDGFRβ (Figure 1A). APA5 staining was then compared with whole-mount specimens of the Ph/Ph and normal embryos from the same litter at 13.5 days of gestation. The Ph/Ph mouse bears a deletion in the region encoding the PDGFRα gene (Stephenson et al. 1991). As shown in Figure 1B, APA5 did not react to any tissues of the Ph/Ph mouse, whereas it stained many tissues of normal embryos (see also following sections). Moreover, as described previously (Takakura et al. 1996), MAb APA5 recognized 180-kD surface proteins from PDGFRα+ BALB/c-3T3 cells in Western blotting, but not from the CYG/SCID7 pro-B cell line (Ogawa et al. 1989), which does not express PDGFRα.
Immunolocalization of PDGFRα in Presomitic Stages
Figure 2 shows a sectioned specimen of a 6.5 day old (E6.5) embryo before mesoderm induction. APA5 stained both the visceral and the parietal endoderm overlying the extraembryonic ectoderm, but not ectodermal cells. On generation of mesoderm cells in the posterior portion of the egg cylinder (E7.0), the embryonic mesoderm localizing the proximal posterior part of the embryo and ectoplacental cone was stained with APA5 (Figures 3A and 3B). At this stage it was difficult to determine if all nascent mesodermal cells express PDGFRα. In the E7.5 embryo (Figures 3C and 3D), however, at this early head fold stage, the proximal lateral and extraembryonic mesoderm, except the allantois, were not stained with APA5, whereas the paraxial mesoderm was strongly positive. The staining in the ectoplacental cone extends as it develops, whereas only faint staining is detectable in the allantois. This staining distribution strongly suggests that not all mesodermal cells express PDGFRα protein in the E7.5 embryo.
Specificity of APA5.
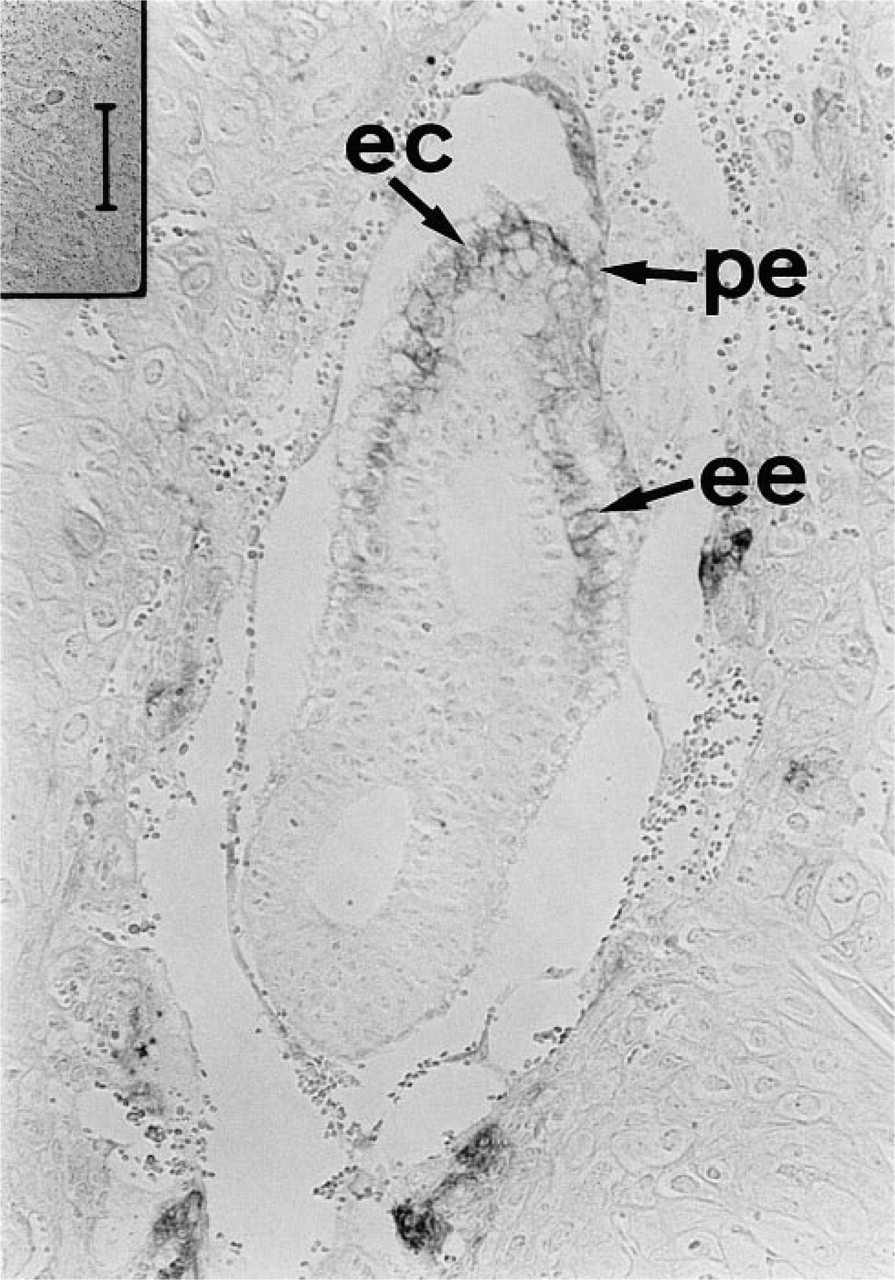
Immunolocalization of PDGFRα in the 6.5 dpc embryo. A section of a 6.5 dpc embryo with the uterus was stained with APA5. APA5 immunoreactive cells were found in the parietal (pe), extraembryonic (ee) endodermal cells, and in the ectoplacental cone (ec). Bar = 50 μm.
Immunolocalization of PDGFRα During Somitogenesis
APA5+ cells were diffused throughout the paraxial region as it started to change into developing somites. Whole-mount immunohistostaining showed that diffuse staining in the anterior region became divided into the outer and inner regions (Figure 3C). The inner APA5+ region invaginated and extended forward under the outer region, and probably develops into head fold mesenchyme. At the same time, the APA5+ region in the trunk started to segregate and develop into the somites (Figures 4A and 4B). Diffuse staining in the head mesenchyme continued. No APA5 staining was found in the neural fold except for the neural crest cells at prorhombomeres A and B (Figure 4A, arrowheads). After formation of the neural tube, APA5 stained the dorsal midline (Figure 4C, arrows), particularly at the head region.
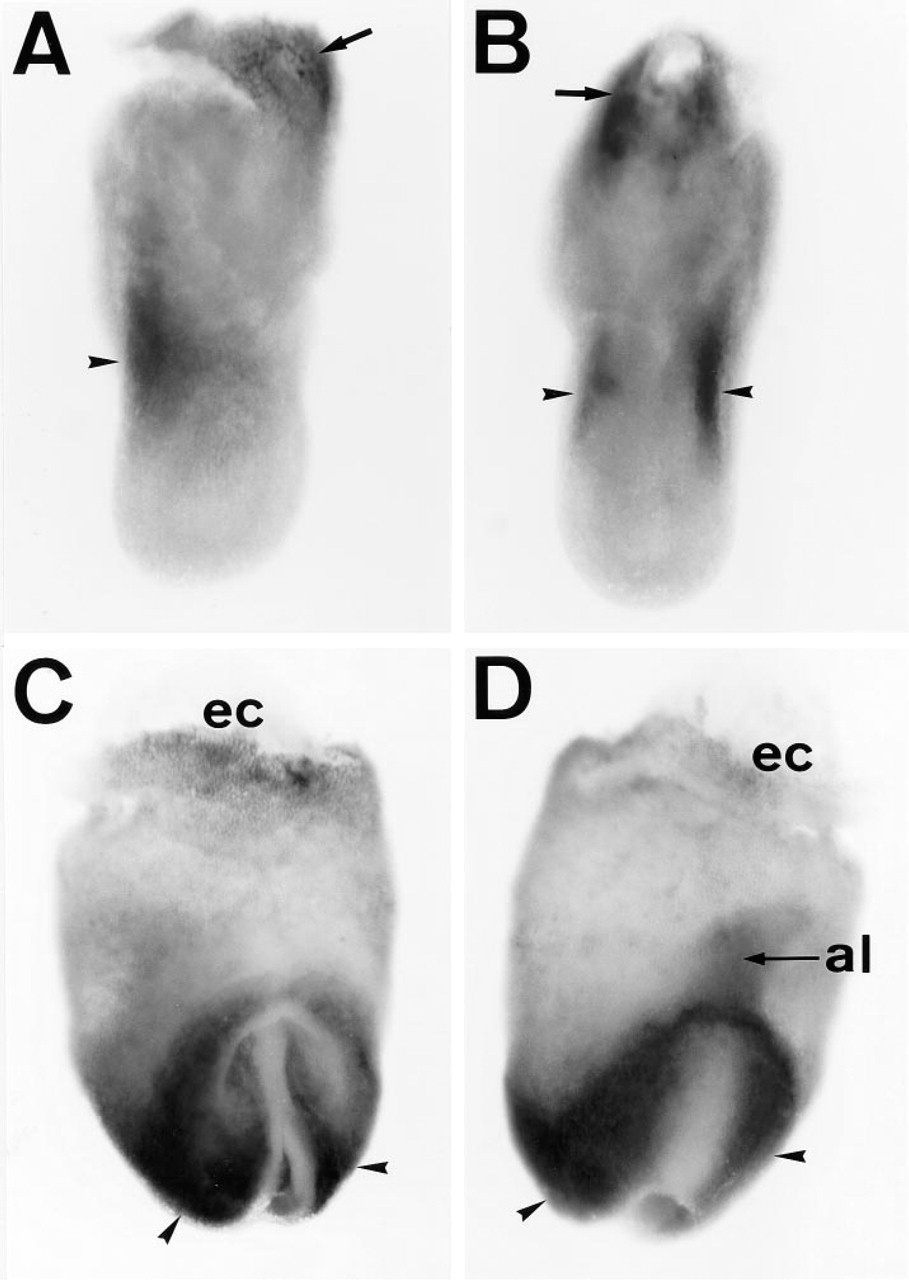
Immunolocalization of PDGFRα in the gastrulating embryo. Embryos at 7.0
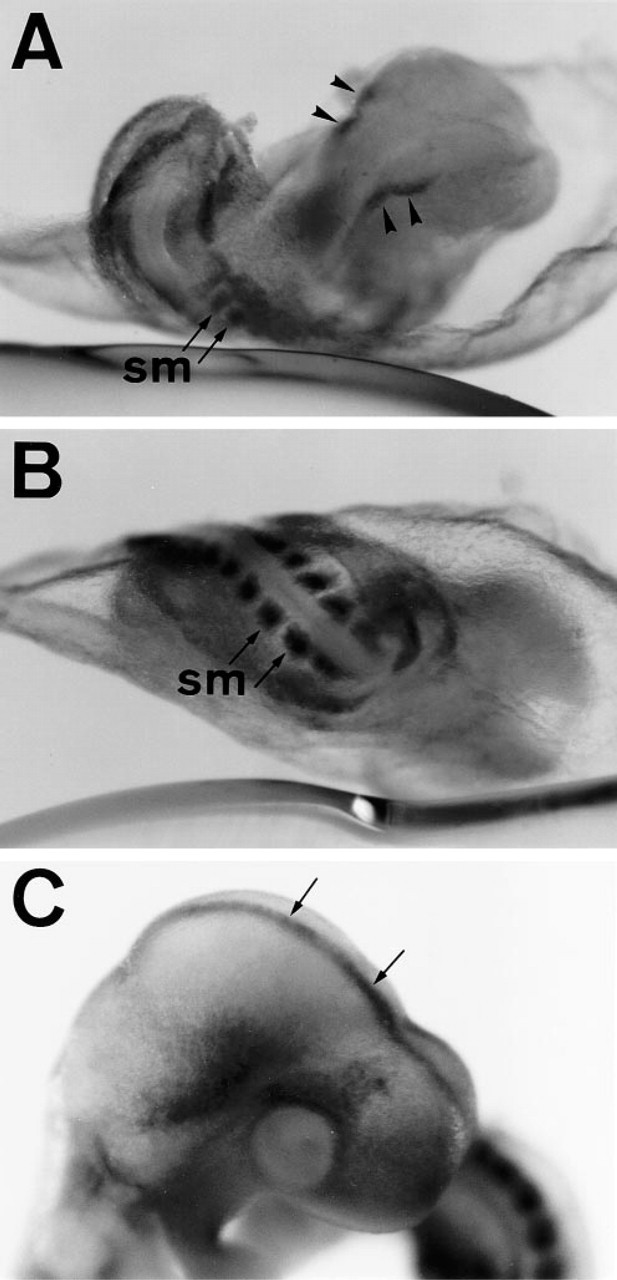
Immunolocalization of PDGFRα during somitogenesis. Eight-somite
Immunolocalization of PDGFRα During Early Organogenesis
In the E9-10 embryo (Figures 5A and 5B), when only posterior somites were stained by APA5, only the basal part of anterior somites were APA5+. Owing to the formation of vertebrae and intervertebral discs, APA5 staining was concentrated in the latter. At the same stage, PDGFRα immunolocalization in the anterior region was seen in the mesenchyme of the most anterior and basal parts of the head, branchial arch, and lens. In the trunk region, staining in the heart and basal part of the fore- and hindlimbs was most conspicuous. Transient expression of PDGFRα was detected also in the apical ectodermal ridge (AER) of the fore- and hindlimbs. In the posterior region, the splanchnic mesenchyme was strongly APA5+.
Immunolocalization of PDGFRα During the Later Stages of Organogenesis
In sagittal sections of E12.5-E16.5 embryos, PDGFRα was diffusely immunolocalized in the connective tissue. At this stage, several areas were conspicuously stained. These included the mesenchyme around vertebrae (Figures 6B and 7A), kidney (Figures 6C and 7B), and thymus (Figures 6D and 7C). During renal development, mesenchymal cells within the kidney expressed PDGFRα (Figures 1B and 7B). Also notable was the mesenchyme staining surrounding the urethric tubes (Figures 1B and 6C). The heart was markedly stained in cells covering valves and the pericardium (Figures 6D and 7D). The trachea was also lined by APA5+ mesenchyme (Figure 6D). Within the trachea, stripes of APA5 staining were present in the regions where tracheal cartilage develops (Figure 1B). The developing lung was intensely stained around the developing bronchial tree (Figures 8C and D).
In the head region, the most intense staining was noted in the mandibular and palate regions (Figures 1B, 6A, and 8A). Although most APA5+ cells were present in the mesenchyme beneath the ectodermal surface, those surrounding the tooth bud were highly positive (Figure 8A). Although weakly stained, repetitive strings ran vertically in the muscle region of the tongue (Figures 8A and 8B).
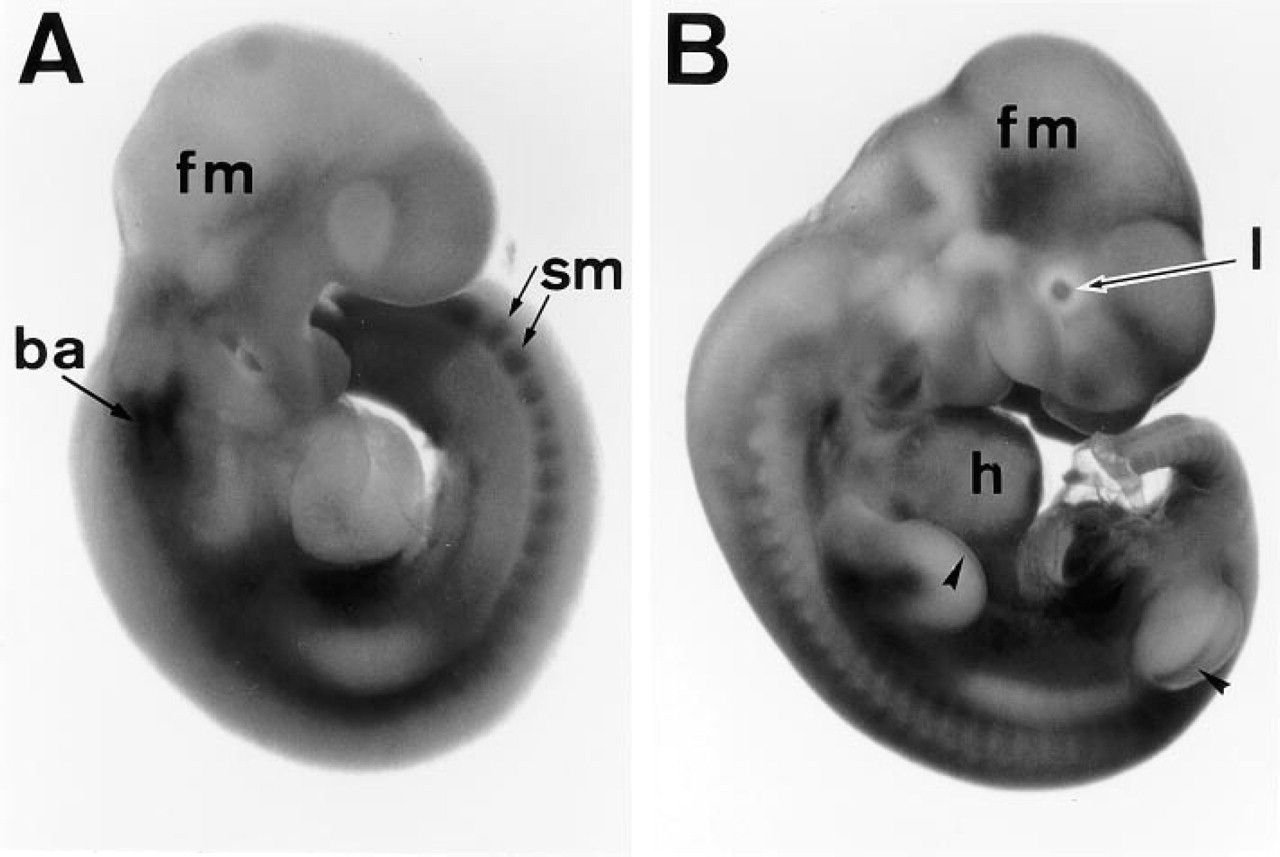
Immunolocalization of PDGFRα during early organogenesis. Embryos at 9.5 and 10.5 dpc were whole-mount immunostained with APA5. In the 9.5 dpc embryo
The nervous system was basically APA5-negative. However, the midportion of the spinal cord was faintly stained. A magnified view of this region in E12.5 embryo shows cells with dendrites (Figures 6E and 7E). Similarly, an array of APA5+ cells was concentrated in the dorsal part of the eye, emanating downwards along with the presumptive optic nerve (Figures 6F and 7F). These APA5+ cells represent glial precursors. Similar APA5+ regions were also seen in the central nervous system. In the cranial region, highly immunoreactive cells were found in the epithelial surface of the choroid plexus (Figures 8E and 8F).
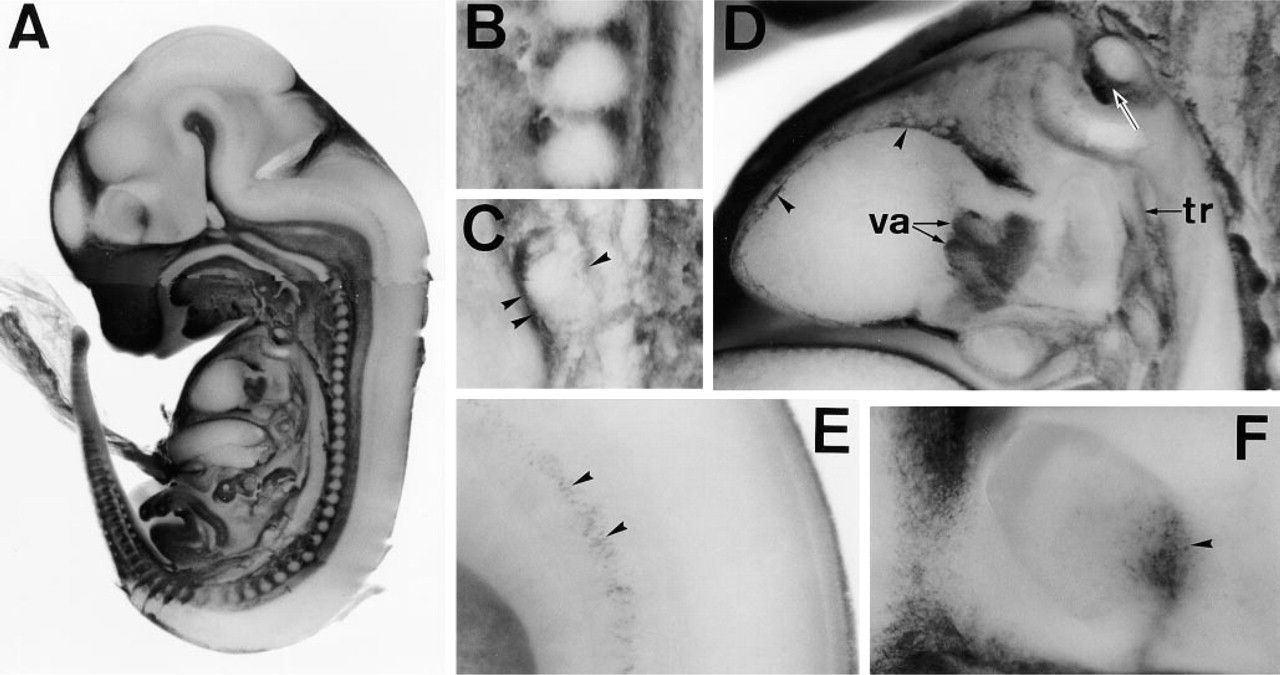
Immunolocalization of PDGFRα during the later stage of organogenesis. A 12.5 dpc embryo was cut sagittally in half and stained with APA5
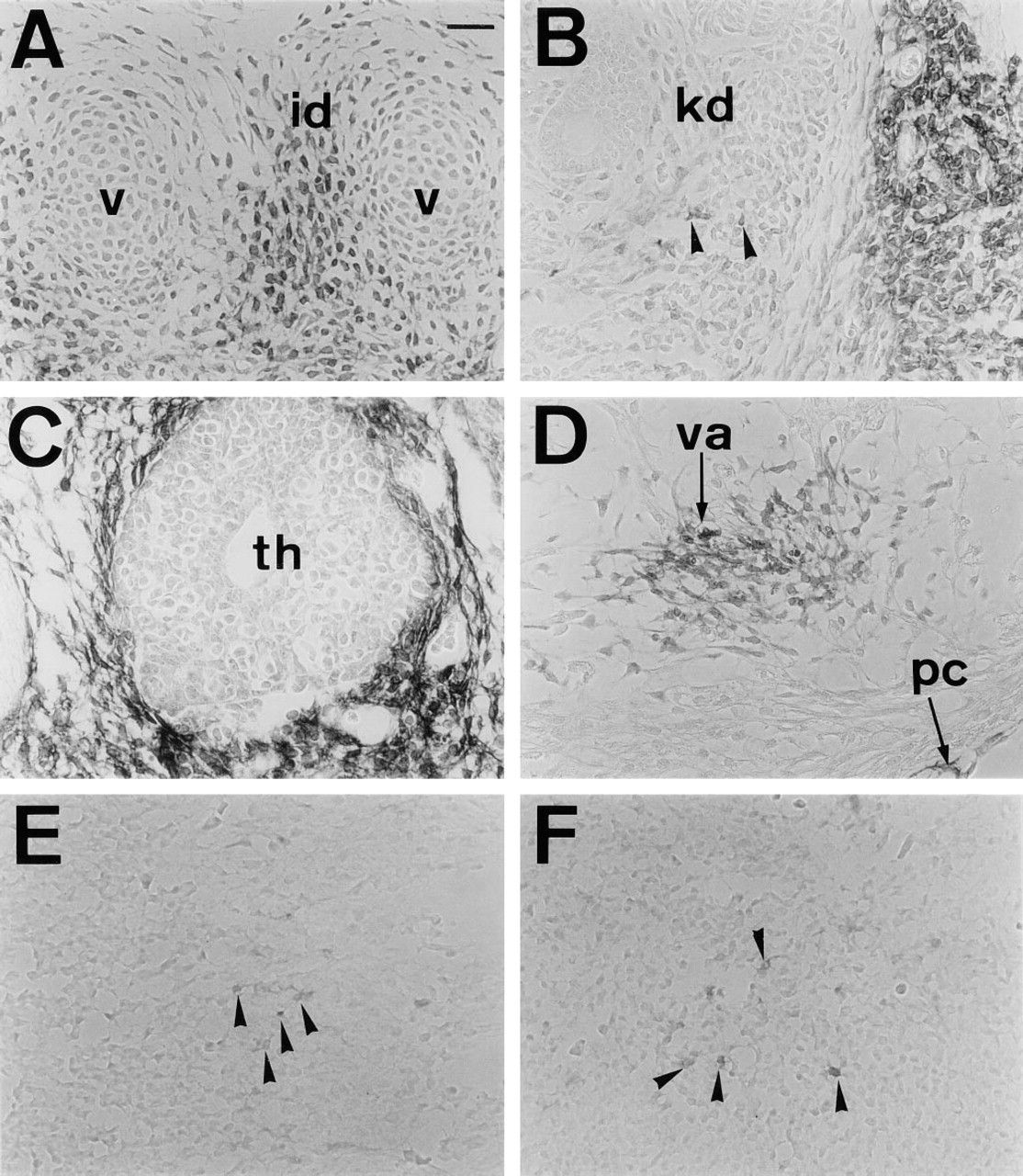
Histological analysis of PDGFRα+ cells in the 12.5 dpc embryo. Immunoreactive cells with APA5 were found in mesenchymal cells in the intervertebral disc
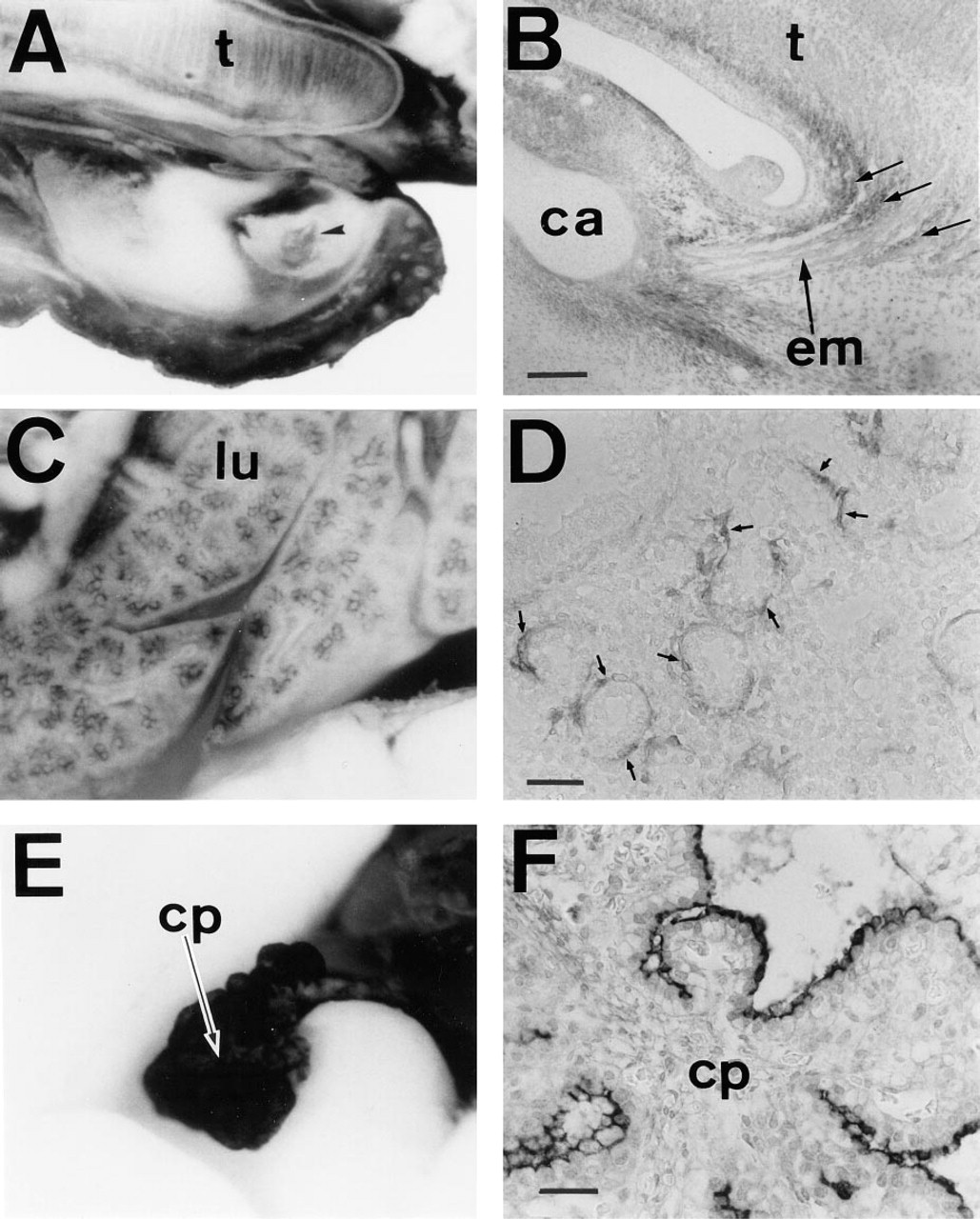
Immunolocalization of PDGFRα in the 16.5 dpc embryo.
Discussion
The aim of this study was to show the immunolocalization of PDGFRα in murine embryos and to compare it with the result of in situ hybridization studies (Pringle and Richardson 1993; Morrison-Graham et al. 1992; Orr-Urtreger and Lonai 1992; Orr-Urtreger et al. 1992; Schatteman et al. 1992; Holmgren et al. 1991). Although a few reports describing the immunolocalization of PDGFRa have been published (Bidwell et al. 1995; Han et al. 1992), none used monospecific MAbs.
Table 1 compares our results with those of in situ hybridization. Basically, most regions that were PDGFRα-positive by in situ hybridization were also positive by APA5 staining. However, the two methods gave entirely different results with respect to PDGFRα expression in E7.5 embryos. Studies by Orr-Urtreger et al. (1992) and Schatteman et al. (1992) have concluded that most if not all mesodermal cells exfoliating from the primitive streak are PDGFRα+ from the onset of mesoderm formation during gastrulation. In contrast, whole-mount immunostaining of the E7.5 embryo with APA5 demonstrated that only the mesoderm in the paraxial region was positive, although a few APA5+ cells might present in the proximal lateral and extraembryonic mesoderm. Our results are in contrast to the conclusion by Orr-Urtreger et al. (1992) that blood islands express PDGFRα transcripts and also to that of Bidwell et al. (1995), who showed that embryonic erythrocytes were stained by a polyclonal anti-PDGFRα antibody. APA5 hardly stained mesodermal layers of the extraembryonic tissues, including blood islands, whereas they were stained by anti-c-Kit MAb (data not shown). The allantois is the only notable APA5+ extraembryonic mesoderm in the gastrulating embryo, although the expression in E7.5 embryo was faint. In contrast, the ectoplacental cone was APA5+ from the pregastrulation stage and the stained area was extended. We concluded that most if not all cells of proximal lateral and extraembryonic mesoderm do not express PDGFRa protein, although the allantois expresses PDGFRα at later stages.
In addition to PDGFRα expression, several studies have suggested that Flk1 expression can be used to mark the nascent mesoderm (Shalaby et al. 1995; Millauer et al. 1993; Yamaguchi et al. 1993). By whole-mount in situ hybridization with the flk1 gene, Yamaguchi et al. (1993) have shown that it is specifically expressed in the proximal lateral and extraembryonic mesoderm of the E7 to 7.5 embryo. Because the flk1-expressing expressing area shown in their study appeared complementary to the area stained by APA5, Flk1 expression may mark a distinct subset of mesoderm cells that are APA5 negative. Establishment of an MAb that specifically recognizes murine Flk1 is in progress to examine this notion.
PDGFRα expression patterns in early stage of embryogenesisa
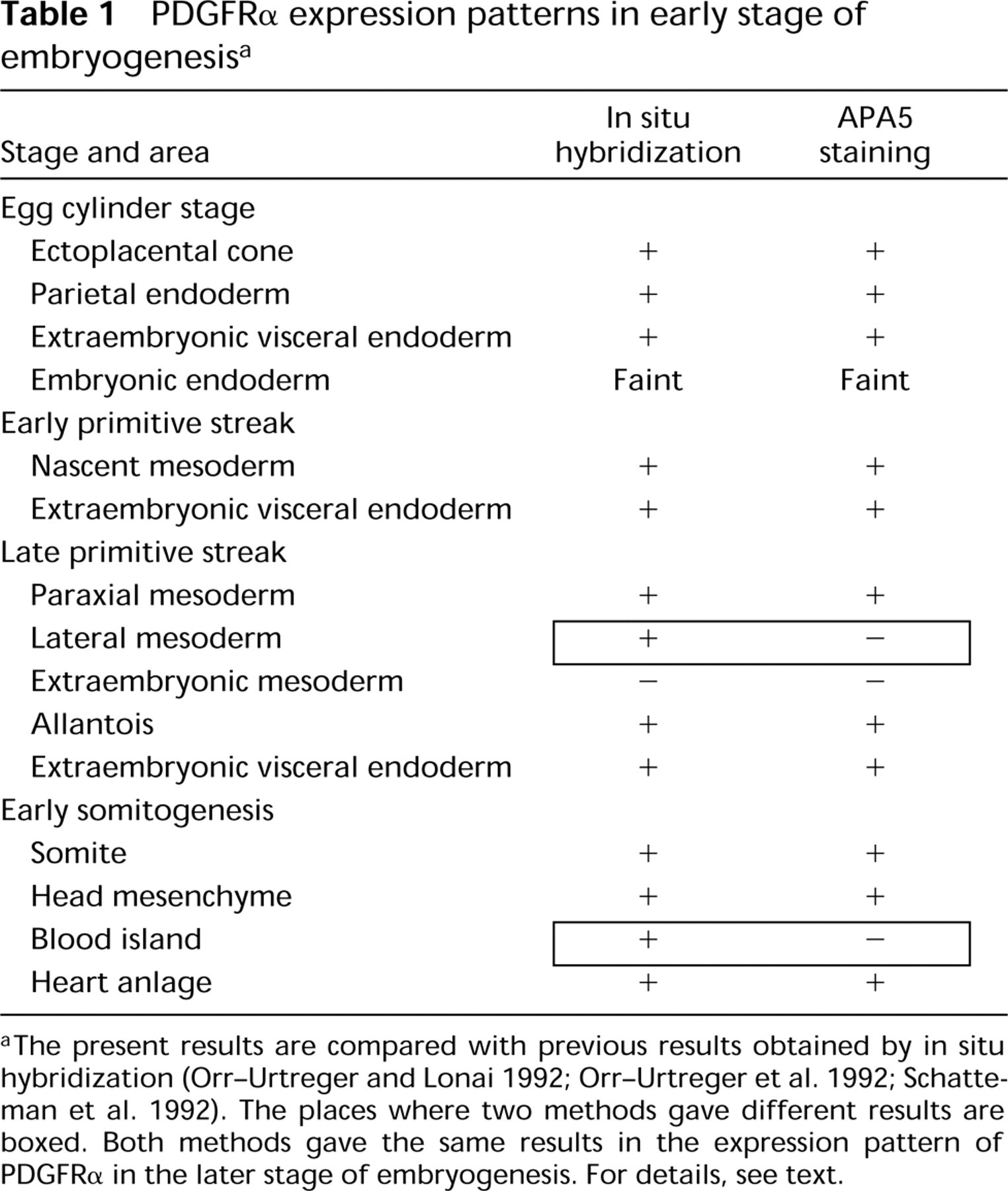
aThe present results are compared with previous results obtained by in situ hybridization (Orr-Urtreger and Lonai 1992; Orr-Urtreger et al. 1992; Schatteman et al. 1992). The places where two methods gave different results are boxed. Both methods gave the same results in the expression pattern of PDGFRα in the later stage of embryogenesis. For details, see text.
It is difficult to explain the discrepancy in the results obtained by the two methods. Certainly, this discrepancy is due to inherent differences in the level of sensitivity between nucleic acid in situ hybridization and immunohistochemical techniques. Alternatively, in situ hybridization may detect untranslated PDGFRα transcripts present in cells that do not express the PDGFRα protein. APA5 can block the binding of PDGFs to PDGFRα, thus recognizing its ligand binding site. Hence, all functional PDGFRα proteins on the cell surface should be detected by APA5, whereas truncated molecules without the ligand binding will not be recognized.
The other plausible possibility is that our whole-mount immunostaining result simply gives a clearer view of PDGFRα expression in the whole embryo. In other words, which part of the embryo expresses more or less PDGFRα can be determined easily by whole-mount immunostaining. Thus, although it could be that all mesodermal cells express PDGFRα at a level that is above the detection sensitivity of in situ hybridization, regional differences in its expression level cannot be noted by the in situ hybridization of tissue sections.
It is important to emphasize that there was no significant discrepancy in the PDGFRα expression profiles of later embryos determined by these two methods. Much has been discussed regarding the correlation between PDGFRα expression and phenotypes of the Ph/Ph mutant mice, which have a large deletion in the region containing the PDGFRα gene (Stephenson et al. 1991). Basically, our results agree with those of published studies: that PDGFRα is involved in many events of organogenesis.
One important aspect of PDGFRα that has not been addressed is its expression in the spinal cord of the E12.5 embryo. PDGFRα has been implicated in the development of glial precursors (Barres et al. 1994; Pringle and Richardson 1993; Yeh et al. 1991). Miller (1996) has shown that the glial precursor appears first at the ventral ventricular zone of the developing vertebrate spinal cord, then subsequently migrates dorsally and radially to populate presumptive white matter. The staining profile in the spinal cord implies that PDGFRα plays a role in this early process of glial cell differentiation. Likewise, the PDGFRα staining in the eye region of the same embryo supports the notion that timely expression of PDGFRα is important for glial cell differentiation and neuronal development. Sretavan et al. (1994) have shown that E12 is the stage at which the first retinal axons entering the ventral hypothalamus encounter a population of early generated neurons arranged as an inverted V-shaped array pointing anteriorly at the future site of the chiasm.
Accepting the notion that PDGFRα plays a diverse role in murine embryogenesis, the most important issue is to determine how it regulates the processes of organogenesis. APA5 is an antagonistic MAb that can be used to inhibit the function of PDGFRα both in vitro and in vivo. Therefore, studies to elucidate the function of PDGFRα in the formation of various organs are now in progress.
Footnotes
Acknowledgements
Supported by grants from the Japanese Ministry of Education, Science and Culture (nos. 07CE2005, 07457085, and 06277103), by a grant from the Ministry of Science and Technology (no. 13073–2125–14), and by a grant from RIKEN.
We would like to thank Dr L. Shultz (The Jackson Laboratory) for providing the Ph/Ph mice.
