Abstract
We present a method that permits extremely simple and rapid screening of proteolytic enzyme activity in sectioned tissues. Enzyme overlay membranes (EOMs) are custom-made membranes designed to fluoresce at sites of specific proteolytic enzyme activity after separation of proteins by gel electrophoresis. EOMs, selected to detect either plasmin-like or cathepsin B-like activity, have been used in a novel way to document the distribution of enzyme activity in frozen sectioned tissues. When moistened membranes were placed in contact with sectioned regenerating newt limbs, a fluorescent pattern of enzyme activity was generated. In limbs at 3 hr post amputation, cathepsin B-like activity was prominent across the amputation site but plasmin-like activity was distributed in dermal and deeper proximal tissues, suggesting different roles for these two classes of enzymes. EOM enzymology in situ (EEI) on frozen sectioned tissues may be a widely useful technique to display distribution and level of activity of proteolytic enzymes in various systems.
P
The process of newt limb regeneration also depends on proteolytic enzyme activity. After amputation of a limb, the extracellular matrix (ECM) of damaged adult tissues near the amputation site is eroded, liberating cells to form the growth bud (blastema) which gives rise to the new limb (reviewed by Stocum 1995; Rose 1970). Several earlier studies have documented a general role for proteolytic enzymes (Dresden and Gross 1970; Grillo et al. 1968; Gross and Lapiere 1962) in this process. Recent studies have renewed investigation of proteolytic activities (Lee and Kim 1996; Ju and Kim 1994) and the involvement of ECM-specific matrix metalloproteases (MMPs) in appendage regeneration (Miyazaki et al. 1996; Yang and Bryant 1994). Knowledge of the distribution and relative contributions of both lysosomal and nonlysosomal enzymes in histolysis of ECM at the amputation site may clarify the way in which blastema formation and regeneration differ from scar formation during mammalian postamputational healing.
EOMs were used in this study to investigate tissue enzyme activities in regenerating newt limbs. EOMs are cellulose membranes impregnated with specific amino acid sequences coupled to a fluorescent label. They are designed to detect proteases that have been separated by gel electrophoresis. This study demonstrates, however, that they may also be used in a simple procedure to screen enzyme activity in frozen tissue sections. Regenerating newt limbs were cryosectioned and EOMs used to assess activity of a lysosomal protease (cathepsin B-like activity) and a proposed MMP activator (plasmin-like activity). Results have demonstrated differences in the activities of these enzymes and suggest that this method may be widely useful elsewhere.
Materials and Methods
Adult newts, Notophthalmus viridescens, were obtained from Connecticut Valley Supply (Southhampton, MA), housed in glass aquaria at room temperature, and fed diced tubifex twice weekly. Newts were anesthetized in 1% ethyl m-aminobenzoate methanesulfonate and forelimbs amputated by scalpel blade through the mid-radioulnar level. No hemostatic agents or antibiotic agents were applied to the wound surface. On selected post-amputation days, animals were anesthetized as above, sacrificed by decapitation, and tissues removed for processing. More than 15 amputated limbs were serially sectioned and analyzed. Protocols for animal handling, as detailed above, were approved by the University of South Dakota Institutional Animal Care and Use Committee.
Tissues were embedded in OCT compound (Miles; Elkhart, IN) and immediately frozen and sectioned at 10 μm. Sections were serially collected on 10 slides, beginning with Slide 1 and continuing through Slide 10, the process being repeated until sections were exhausted (typically approximately 60 sections per limb). Slides were air-dried for 1 hr, fixed in 100% methanol at −20C for 10 min, and rinsed in PBS to remove OCT. Enzyme overlay membranes (Enzyme Systems Products; Dublin, CA) were dipped in distilled water with forceps and carefully applied over sections. A sandwich was prepared by covering with a second microscope slide and inverting on the microscope stage. Enzyme activity is visualized as blue-green fluorescence under UV excitation. Localization and intensity were monitored and photographed at 5–10-min intervals for 30 min to 1 hr.
To demonstrate enzyme specificity, several types of protease inhibitors were used. General inhibitors of proteolysis, PIC I [a cocktail of leupeptin (1 mg/ml), antipain (2 mg/ml), and benzamidine (10 mg/ml)] and PIC II [chymostatin (1 mg/ml) and pepstatin (1 mg/ml)] (Deutscher 1990) were used at a concentration of 1 μl/ml. Plasmin inhibitors (serine protease inhibitors), 4-(2aminoethyl)-benzenesulfonylfluoride (AEBSF) and phenylmethylsulfonylfluoride (PMSF) and a cathepsin inhibitor (cysteine protease inhibitor), p-aminophenylmercuric acetate (APMA) were used at 1-mM concentrations. Inhibitors were added to tissue sections before EOM placement or EOMs were soaked in inhibitors. All reagents were obtained from Sigma (St Louis, MO).
Cathepsin-like activity was detected using a membrane impregnated with the substrate Z-Val-Lys-Lys-Arg-AFC (Smith et al. 1992) and plasmin-like activity was detected with the substrate D-Val-Leu-Lys-AFC (Kato et al. 1980). Activities were observed in intact limbs and at various times after amputation.
Results
Validation of the use of overlay membranes to observe enzyme profiles is shown in Figure 1. To demonstrate that emitted fluorescence was not an artifact and that it represented the pattern and intensity of protease activity in sectioned tissues, several inhibitors were used. Initially, to block all protease activity, sectioned tissues (Figure 1A) were exposed to a cocktail of inhibitors (PIC I and PIC II) at the time of contact with the EOM (Figure 1B). In such experiments, no fluorescence was seen as long as 1 hr after EOM application. These results can be contrasted with fluorescence emission seen routinely after a 20-min incubation of sectioned tissue without inhibitors (Figures 1C and 1D).
Specificity of reaction product was confirmed in other experiments in which, as above, reaction product was suppressed by appropriate inhibitors but unaffected by inappropriate inhibitors. AEBSF and PMSF each blocked plasmin-like activity but did not affect cathepsin-like activity. Similarly, APMA blocked cathepsin activity but did not affect plasmin-like activity.
Different patterns of activity for the two categories of proteases can be seen in paired sections from a limb 3 hr post amputation (Figure 2). In unamputated limbs (not shown), minimal staining of deeper soft tissues was seen with either type of protease, although visible background levels were seen in epidermis (cathepsin-like) and dermis (plasmin-like). After amputation, both types of enzymes appeared to be upregulated, although in different patterns. Cathepsin B-like activity appeared somewhat more diffuse but was distinctly increased across the amputation site. It was also generally increased over background in the soft tissue subjacent to the amputation site. Plasmin-like activity was increased in the dermis and also was focally increased within deeper tissues. Closer examination suggested activity associated with vessels and with selected tissues, the precise significance of which is being pursued in other studies. This pattern shifts, however, such that by 4 days post amputation there is very little plasmin-like activity in the vicinity of tissues near the amputation site that will participate in regeneration (compare Figures 1D and 2D).
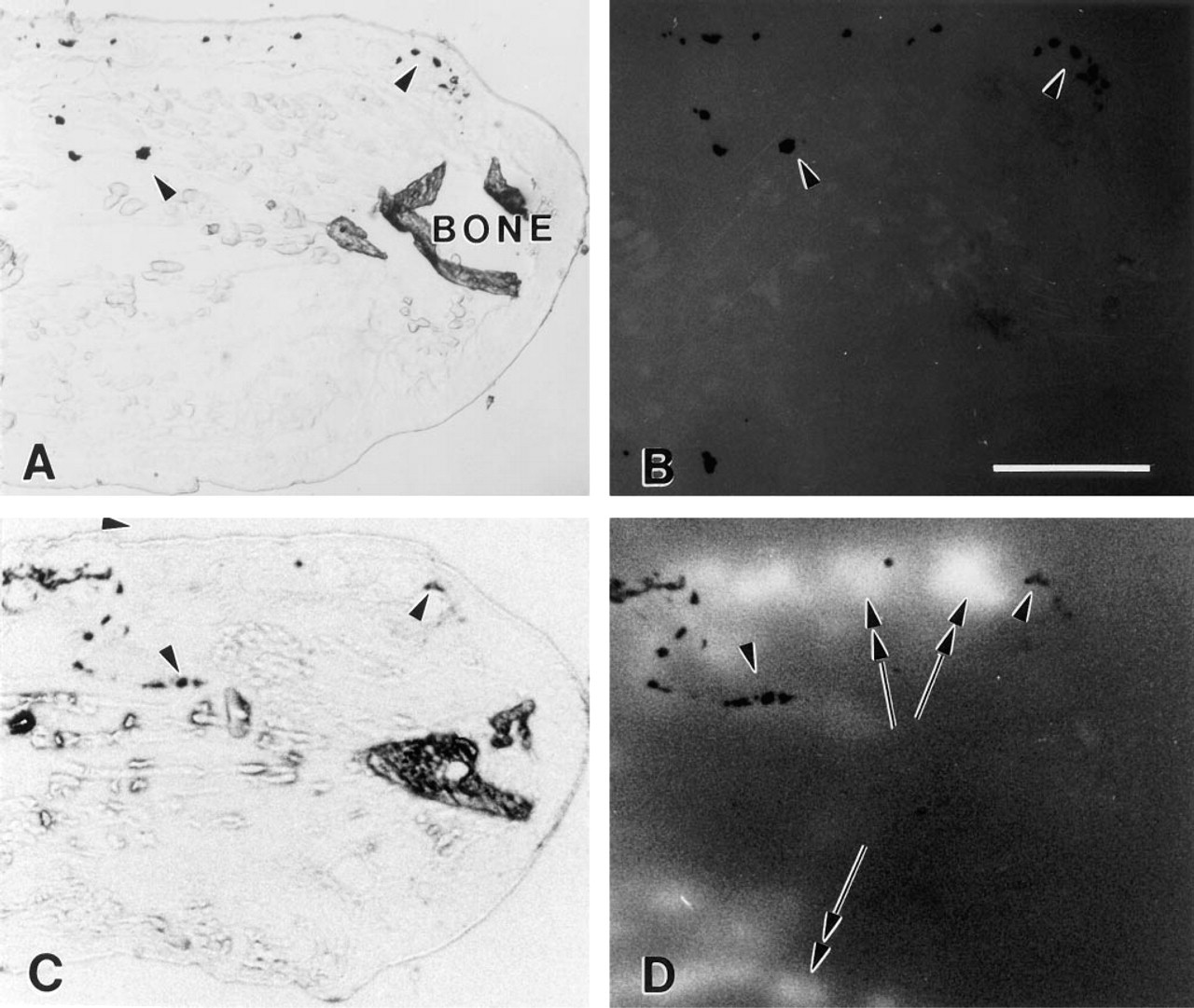
Demonstration of in situ enzymology using EOMs on control
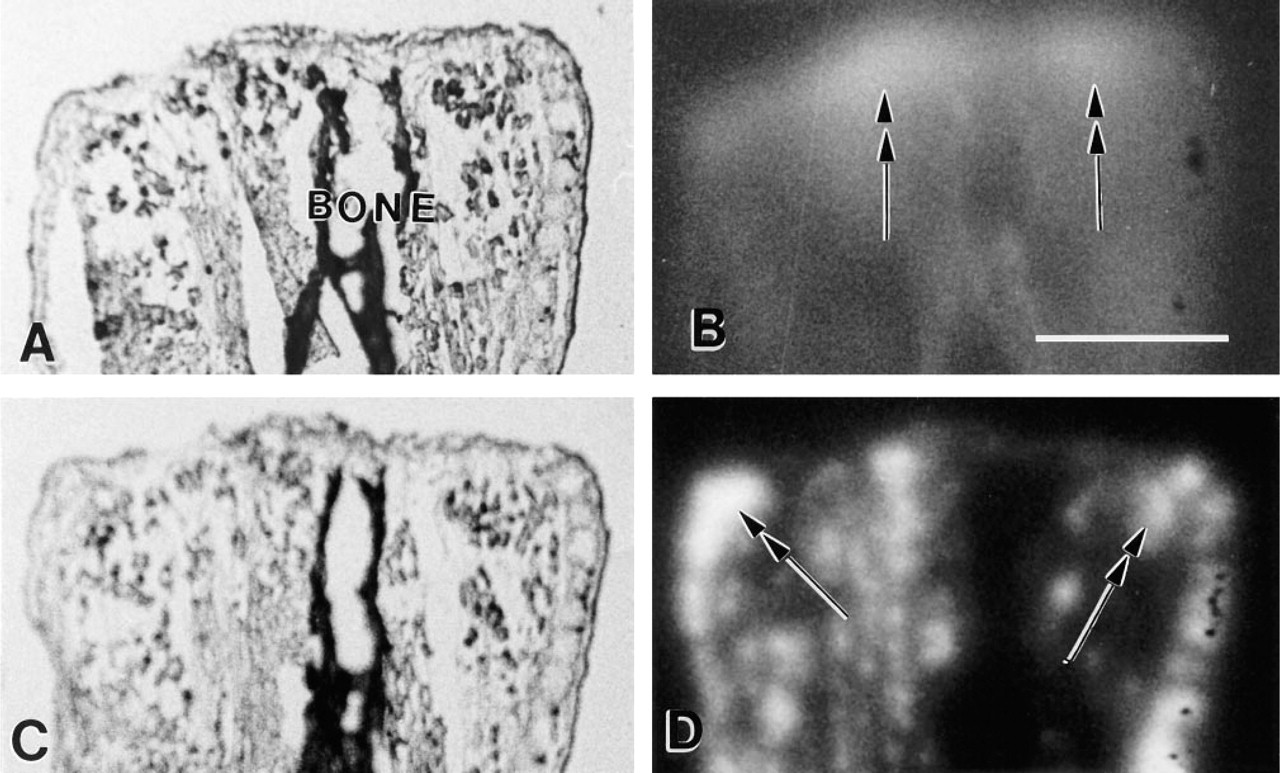
EOM localization of cathepsin-like and plasmin-like activity in paired sections from an amputated stump of a newt limb 3 hr after amputation. The distal end of the limb (the amputated surface) is at the top of both brightfield
Discussion
Enzyme overlay membranes have been used, to our knowledge for the first time, to assess patterns and intensity of protease activity in sectioned tissues.
Specific peptides coupled to fluorigenic substrates have been used to assay proteolytic activity since the early 1980s, when fluoromethyl coumarin was introduced as a sensitive leaving group (reviewed by Smith et al. 1992). The problem of endproduct diffusion was addressed by adsorbing the fluorogenic substrate onto a matrix that trapped the cleaved fluorescent product (Smith 1984). This concept was adapted into a commercially available product sold to detect electrophoretically separated enzymes by a fluorescent reaction product. Proteolytic enzymes, with specificity for the designated peptide sequence, hydrolyze the peptide and liberate the fluorescent label generated when the EOM is placed in contact with an enzyme-containing gel. However, there is no record to indicate that EOMs have been used to localize protease activity in sectioned tissues. The use of EOMs to distinguish between the distribution of proteases therefore represents an entirely new application for this product.
Cathepsin B-like and L-like activities have been associated with ECM degradation, their presence having been suggested to be a basis for cancer invasion (Chauhan et al. 1991). Cathepsin B has also been shown to degrade basement membrane (BM) (Guinec et al. 1993). In this study, several specific cysteine-type inhibitors (Woessner 1992) have blocked cathepsin-like activity but did not block plasmin-like activity, as seen by generation of fluorescence of appropriate membranes.
Plasmin-like activity has also been shown to erode components of BM (Alexander and Werb 1989; Liotta et al. 1981). In addition to demonstrated direct involvement in ECM breakdown, plasmin is also known to activate other enzymes and has been suggested to be instrumental in activation of a proteolytic enzyme cascade (reviewed by Roskelley et al. 1995) in which several MMPs are activated. Inhibitors of plasmin-type activity (serine-type proteases) have blocked the generation of fluorescent label on plasmin substrate, but did not affect cathepsin-like activity.
Amputated newt limbs undergo extensive local histolysis at the amputation site before formation of the growth bud that will produce the regenerated limb. Histolysis is known to be necessary to release cells near the amputation site, but proteolytic activity may also be important during blastemal outgrowth to remove ECM that might prematurely terminate outgrowth. It has recently been suggested (Neufeld and Day 1996; Neufeld et al. 1996) that the ability to prevent accumulation of BM and ECM bundles is central to the ability to regenerate amputated appendages. EOMs used in this study suggest that cathepsin B-like activity is found distally across the amputation site 3 hr after amputation and therefore is likely to be involved in debris removal during early healing stages. It may also be involved in BM removal at the same location in later stages. Distribution of plasmin-like activity is consistent with a role in tissue histolysis proximally, but not at the amputation site during the periods examined. These studies are continuing.
Because the EEI method (of EOM usage on serially sectioned tissues) has not been previously described, consideration of technical details is warranted. The sensitivity of EOMs depends on the substrate and details of the conditions used. For example, 0.01 to 0.02 microunits of cathepsin B activity can be detected in single bands in gels after a 15-min EOM incubation. In the current study, qualitative evaluation revealed that for each type of EOM the pattern of activity within a single section and on similar sections from different animals was consistently similar. Although plasmin-like activity appeared two to three times more rapidly than cathepsin-like activity, comparison of rates between enzymes is not valid under these conditions in which ionic, pH, and substrate conditions have not been optimized for each enzyme separately. Trapped air bubbles or disruptions in the tissue were further technical pitfalls that could be avoided by careful specimen handling. The potential benefits of this method dictate additional studies to refine its application.
Footnotes
Acknowledgements
Supported in part by the National Science Foundation under grant OSR-9108773 and by the South Dakota Future Fund.
We thank Diana Baker for technical assistance.
