Abstract
We found that the immunohistochemical distribution of TGF-α varied in rodent tissues depending on the antibody used, suggesting that the specificity of anti-TGF-α antibodies differs significantly. To address this issue, we compared the specificity of two representative antibodies that have been widely used to detect rodent TGF-α. In a competition study, the antibodies were preincubated with an excess of synthetic rat TGF-α34-50 and were used for staining of rat and mouse kidneys and/or uterus. The results revealed that one of the antibodies, anti-rat TGF-α polyclonal antibody, was neutralized by the peptide, whereas the other, anti-human TGF-α monoclonal antibody, was not absorbed by the peptide up to an excess of 100-fold. Western blotting analysis showed that the anti-rat TGF-α polyclonal antibody recognized both human and rat purified TGF-α. However, the anti-human TGF-α monoclonal antibody did not detect purified rat TGF-α, although the antibody reacted with mouse proteins other than TGF-α from kidneys and uterus, purified human TGF-α, and mouse carbonic anhydrase II. These data indicate that the anti-human TGF-α monoclonal antibody does not recognize rodent TGF-α under our experimental conditions and suggest that distribution of TGF-α in rodent tissues may need to be reexamined.
I
In the present study, we report the specificity of antibodies against rodent transforming growth factor-α (TGF-α). TGF-α is a 50-amino-acid polypeptide mitogen synthesized as an internal part of a 160 (human)-or 159 (rat and mouse)-amino-acid precursor (Vaughan et al. 1992; Lee et al. 1985; Derynck et al. 1984). The structure of TGF-α is highly conserved among mammalian species. The amino acid sequence of mouse TGF-α is exactly the same as that of rats, and only four amino acids are different between rodent and human TGF-α. Because TGF-α has been detected in a variety of tumors, this protein is postulated to play an important role in cell transformation, mainly by interacting with the producer cells in an autocrine fashion (Derynck 1986,1988).
During studies to localize TGF-α in some rodent cancer tissues, we found that the immunohistochemical distribution of this protein varied depending on the antibody used, suggesting that the specificity of anti-TGF-α antibodies differed significantly. To address this issue, we compared the specificity of two representative antibodies that have been widely used to detect rodent TGF-α. Our results revealed that one of these antibodies was specific for rodent TGF-α, whereas another failed to show reactivity with purified rodent TGF-α, at least under our experimental conditions, suggesting that the distribution of TGF-α in rodent tissues may need to be reexamined using an antibody that has been proved to have specificity for rodent TGF-α.
Materials and Methods
Antibodies
A monoclonal antibody (MAb), a mouse IgG2a raised against amino acid recidues 34-50 of human TGF-α (Sorvillo et al., 1990), was obtained from Oncogene Science (Ab-2, Lot no. 35900114; Uniondale, NY). The concentration of IgG2a in an original vial was 100 μg/ml. The other was a rabbit polyclonal antiserum against rat TGF-α obtained from Peninsula Laboratories (Lot no. 031873; Belmont, CA). The epitope is not known, but at least the antibody does not recognize amino acid residues 34-43. The estimated concentration of IgG in an original vial was 32 μg/ml.
A polyclonal antiserum against mouse carbonic anhydrase II (CAII) was a kind gift from Dr. Richard E. Tashian (University of Michigan, Ann Arbor, MI). The antiserum was derived from goat (Lewis et al., 1988) and was used for certifying proteins recognized by the anti-human TGF-α MAb.
Immunohistochemistry and Competition Study
Synthetic rat TGF-α (amino acid residues 34-50) was kindly given to us by Dr. Richard P. DiAugustine at our Institute. The peptide was dissolved in 0.01 M PBS at a concentration of 200 μg/ml and was used to neutralize the antibodies. As shown in Table 1, a constant amount of antibodies was mixed with 0 (positive control), 10-, 50-, or 100-fold excess rat TGF-α34-50, with the volume adjusted to 100 μl by adding 0.01 M PBS, and was incubated overnight at 4 C. The solution was diluted with 0.01 M PBS containing 1% non-fat dry milk (Carnation; Los Angeles, CA) and 1% bovine serum albumin (BSA; Sigma Chemical, St Louis, MO) next morning and was used for subsequent immunohistochemistry.
Formalin-fixed paraffin sections (5 μm) of uterus and/or kidneys of mice and rats were stained immunohistochemically using the neutralized antibodies. Tissues were first incubated with 3% H2O2 (Cumberland-Swan; Smyrna, TN) in methanol to inactivate endogenous peroxidase activity, which was followed by incubation with trypsin-EDTA (GIBCO Laboratories; Grand Island, NY) in 0.01 M PBS at a dilution of 1:2. After blocking for nonspecific staining with normal horse serum (Oncogene Science; for MAb) in 1% milk, 1% BSA, and 0.01 M PBS or with normal goat serum (Vector Laboratories, Burlingame, CA; for polyclonal antibody) in 5% milk, 1% BSA, and 0.01 M PBS, tissues were incubated with the neutralized primary antibodies for 24 hr at 4 C at a dilution of 1:1000 (monoclonal antibody) or 1:500 (polyclonal antibody). Adjacent sections incubated with the same diluent without primary antibodies served as a negative control. Tissues were then incubated with a biotinylated secondary antibody (anti-mouse IgG horse serum for the MAb, Oncogene Science; or anti-rabbit IgG goat antiserum for the polyclonal antibody, Vector Laboratories), and reactivity was visualized by the avidin-biotin-immunoper-oxidase system (ABC kit; Vector Laboratories) according to the manufacturer's directions using 3,3'-diaminobenzidine (Sigma) as the chromogen. Mayer's hematoxylin was used for counterstaining.
Western Blotting
Samples used to examine the specificity of antibodies against human and rodent TGF-α were recombinant human TGF-α (Biomedical Technology; Stoughton, MA), synthetic rat TGF-α (Peninsula Laboratories; Belmont, CA), and protein extracts from the uterus and kidneys of a female B6C3F1 mouse. In the second blotting experiment, purified mouse CAII protein (a gift from Dr. Tashian at the University of Michigan) and protein extracts from the uterus and kidneys of a female F344 rat, as well as the above-mentioned mouse proteins) were used to certify the crossreactivity of anti-human TGF-α MAb with rodent CAII, since the antibody had detected a clear signal for bovine carbonic anhydrase (CA), which had been used as a molecular marker of 30 kD. The samples were dissolved in a lysis buffer (10 mM Tris-HCl, pH 8.0, 150 mM NaCl, 3 mM MgCl2, 0.5% Nonidet P-40, 100 mM dithiothreitol, 2 mM phenylmethylsulfonyl fluoride, and 10 μg/ml of aprotinin) and then mixed with a sample buffer (10% glycerol, 60 mM Tris-HCl, pH 6.8, 2% SDS, and 0.02% bromophenyl blue).
Protocol for competition studies to examine the specificity of antibodies against rodent TGF-α
In the first experiment, sample proteins were subjected to SDS-PAGE on a 17.5% acrylamide gel together with a molecular marker (Rainbow Marker; Amersham Life Science, Poole, UK) and transferred to a polyvinyl membrane (Immobilon-P; Millipore, Bedford, MA). After blocking with 10% milk in 1 X PBS (Boehringer Mannheim; Indianapolis, IN) with 0.1% Tween 20 (PBS-T) overnight at 4C, the membrane was incubated with the primary antibody, an anti-human TGF-α MAb, for 2 hr at room temperature at a dilution of 1:1000 in 1% milk and PBS-T. The following immunoperoxidase staining was done by using an ECL chemiluminescence kit (Amersham) according to the manufacturer's direction.
The same membrane was blotted by a rabbit polyclonal antiserum after removal of bound antibodies by a stripping buffer (100 mM 2-mercaptoethanol, 2% SDS, and 62.5 mM Tris-HCl, pH 6.8). The blotting procedure was the same as those described above, except that the blocking solution contained 0.1% normal donkey serum (Accurate Chemical & Scientific; Westbury, NY). The concentration of the primary antibody was 1:2000.
In the second blotting experiment, signals were first detected by using the anti-human TGF-α MAb. Then the membrane was stripped and stained with an anti-mouse CAII antiserum by the same methods, with the exception that an ABC kit was applied before visualizing the signals by an ECL kit. The concentration of the primary antibody was 1:5000.
Results
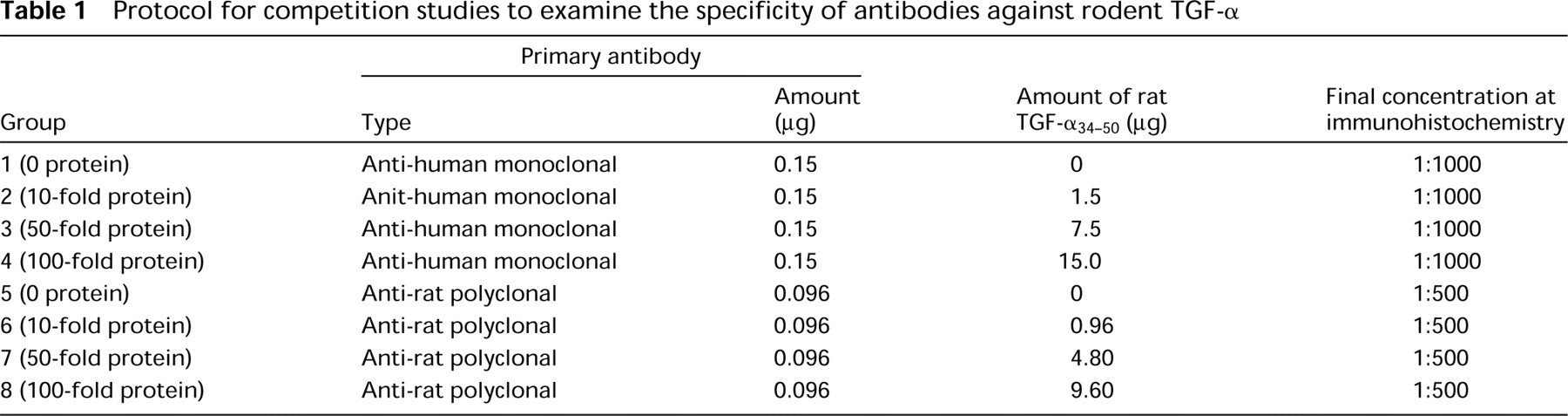
Localization of signals in the positive control group of the competition study (tissues were stained by antibodies preincubated with PBS) varied significantly depending on whether an anti-human TGF-α MAb or an anti-rat TGF-α polyclonal antibody was used. In the mouse uterus, faint staining was found in some endometrial luminal and glandular epithelia when the anti-human TGF-α MAb was applied (Figure 1A), whereas the anti-rat TGF-α polyclonal antibody did not detect any positive signals (Figure 1D). In the kidneys stained with the anti-human TGF-α MAb, clear cytoplasmic and/or nuclear staining was observed, mainly in some of the renal tubule epithelia in both rats and mice (Figures 1B and 1C). However, only the walls of blood vessels showed faint positive staining when the same tissues were stained with the anti-rat TGF-α polyclonal antibody (Figures 1E and 1F). No positive staining was seen in negative control tissues for both of the antibodies (data not shown), suggesting that the signals were specific with each antibody. However, there was no overlapping positive site between the staining with the two antibodies.
Staining results were also different between these two antibodies when they were preincubated with an excess amount of rat TGF-α34-50 before application to sections. In the case of the anti-human TGF-α MAb, no apparent change was found in the intensity of positive signals between sections stained with an intact antibody and those preincubated with rat TGF-α34-50 up to a 100-fold excess (Figures 1G and 1H), indicating that this antibody was not absorbed by rat TGF-α34-50. On the contrary, the anti-rat TGF-α polyclonal antibody was neutralized by a 10-fold or more excess of rat TGF-α34-50. When the antibody was applied to mouse and rat kidney sections after preincubation with the peptide, signals disappeared or at least became very faint (Figures 1I and 1J).
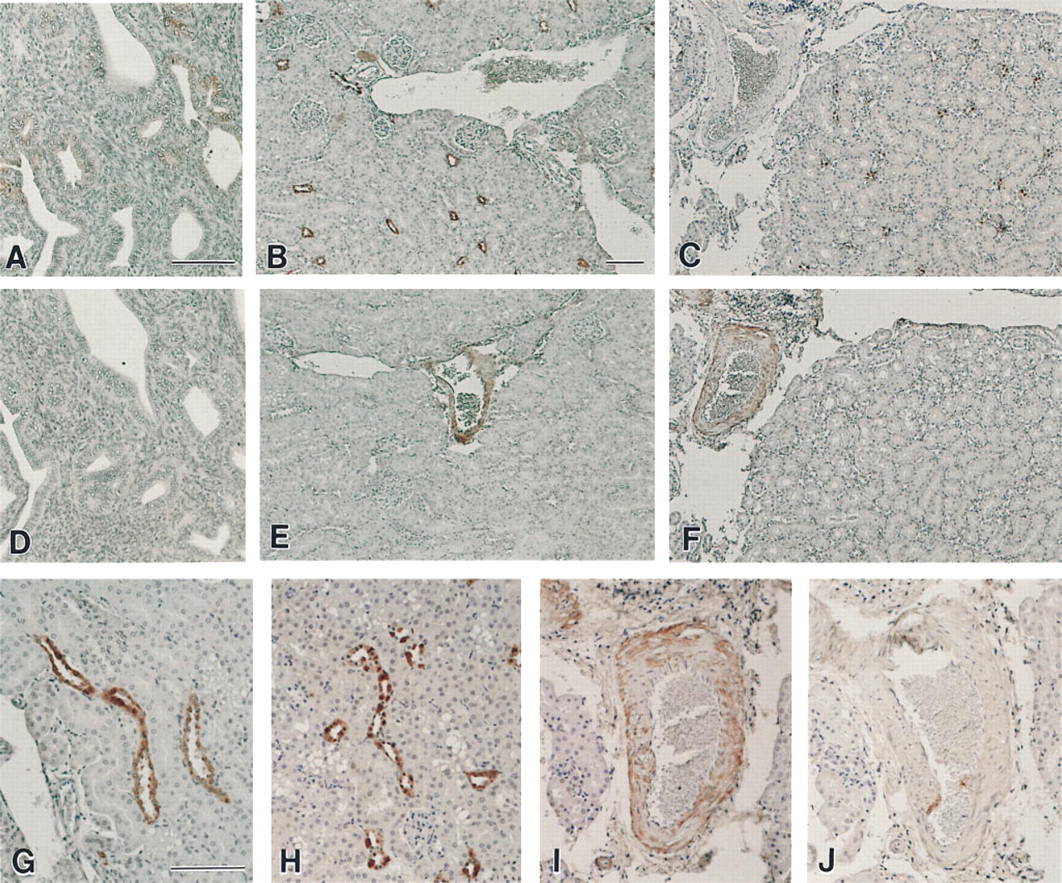
Differences in reactivity between these two antibodies were again seen in Western blotting analysis. The anti-human TGF-α MAb detected 100 ng or more of human TGF-α of which the molecular weight was approximately 5.5 kD, whereas it did not recognize synthetic rat TGF-α up to an amount of 400 ng (Figure 2A). The antibody showed strong crossreactivity with bovine CA used as a molecular marker of 30 kD. Several bands were also found on lanes of proteins extracted from normal mouse uterus and kidneys, one of whose molecular weight was approximately the same as that of bovine CA. However, there was no signal on these lanes at approximately 5.5 kD. When the same membrane was stripped and again blotted by the anti-rat TGF-α polyclonal antibody, signals appeared on lanes of both human and rat purified TGF-α with detectable limits of 100 ng and 25 ng, respectively (Figure 2B). This indicated that the antibody was reactive with TGF-α of both species. However, the antibody did not detect any signals on mouse protein sample lanes. This could be interpreted as indicating that TGF-α was not expressed, or at least that the level was lower than detectable limits under our experimental conditions.
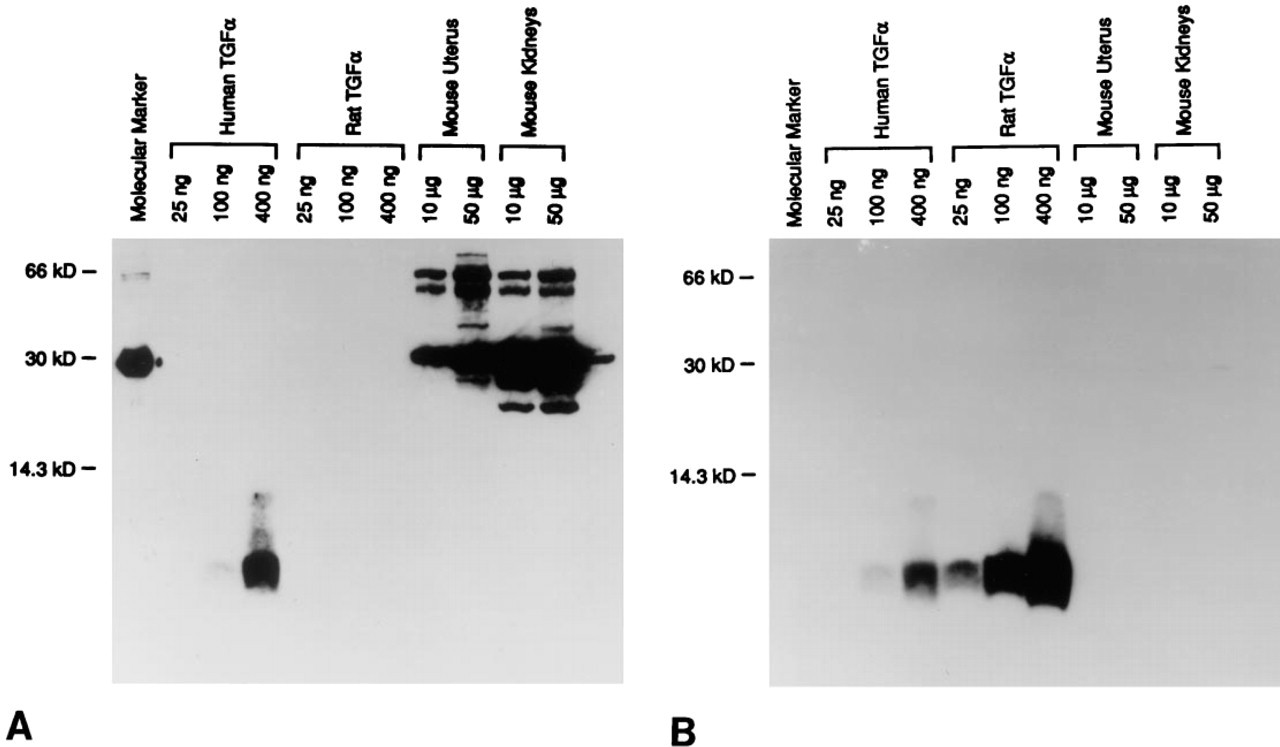
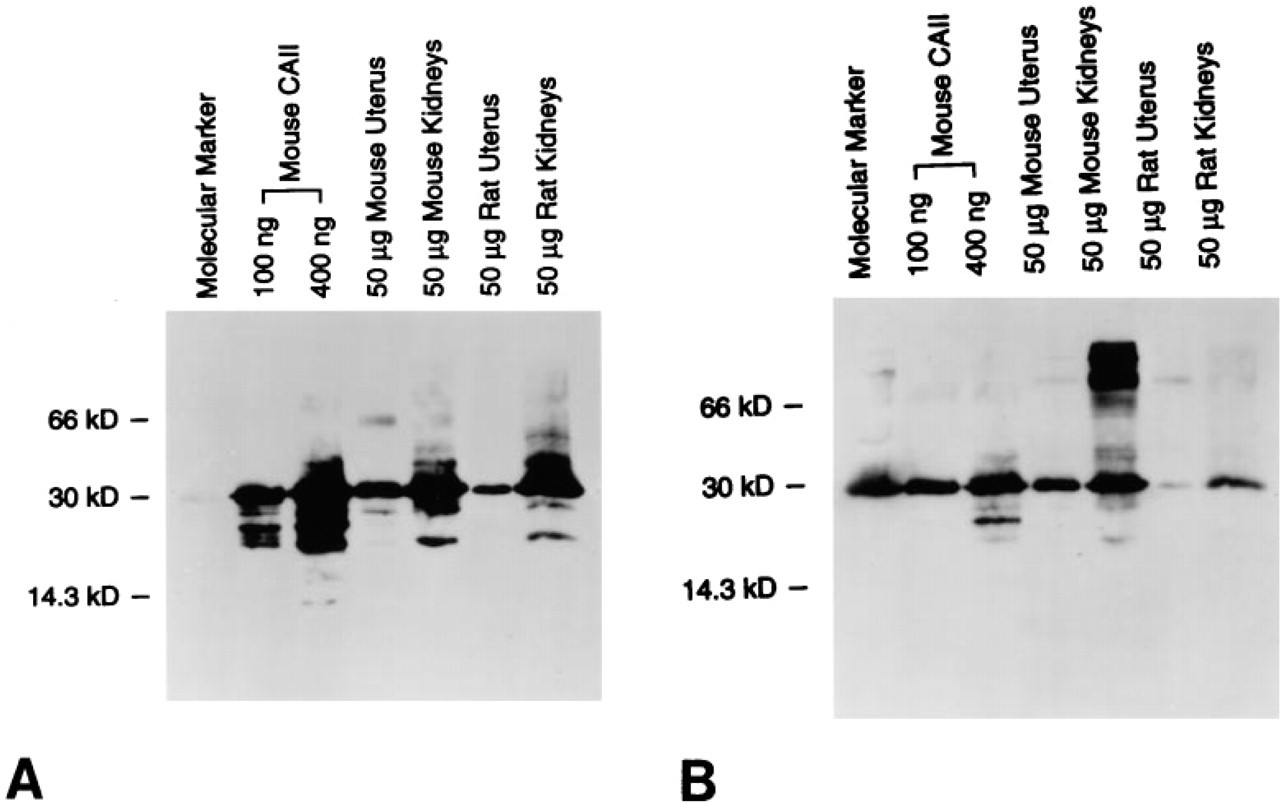
The crossreactivity of anti-human TGF-α MAb with CA was further characterized by the second blotting experiment, in which purified mouse CAII was examined together with mouse and rat proteins extracted from the uterus and kidneys. When a protein-transferred membrane was blotted with the anti-human TGF-α MAb, several bands appeared on all lanes, including the molecular marker lane as observed previously (Figure 3A). Among them, the most intense signal had a common molecular weight of approximately 30 kD, although the total number and/or position of detected bands on each lane differed from one another. This intense signal was confirmed to be CAII by observations in the subsequent blotting using an anti-mouse CAII polyclonal antibody (Figure 3B). Although this antibody again detected several bands on all lanes, including the marker lane, the main signal universally found in all lanes had the same molecular weight of approximately 30 kD, which entirely coincided with those detected by the anti-human TGF-α MAb.
(
Discussion
Many antibodies against a mature or precursor form of TGF-α have been made for use in immunocytochemical experiments, including immunohistochemistry (Paria et al. 1994; Barton et al. 1991; Sorvillo et al. 1990; Gentry et al. 1987). Among them, an MAb against human TGF-α primarily prepared by Sorvillo et al. (1990) is now commercially available (Ab-2; Oncogene Science) and has become one of the most commonly used antibodies in immunohistochemistry. This antibody is believed to detect rodent TGF-α on the basis of the results of competition studies in which preincubation of this antibody with an excess amount of human or rat TGF-α abolished positive signals in paraffin sections of rat tissues (Hormi et al. 1995; Deguchi et al. 1993; Diaz-Ruiz et al. 1993; Walker et al. 1991). However, it is undeniable that the specificity of the antibody has not yet been fully determined, since no competition study using only a target protein to neutralize a certain antibody can ensure that the antibody has no crossreactivity with proteins other than the target.
Results of Western blotting analysis using an anti-human TGF-α MAb (
In the positive control group of the present competition study, an anti-human TGF-α MAb showed a staining pattern in kidney sections of rats and mice similar to those reported by Deguchi et al. (1993) and Walker et al. (1991), in which the same antibody was applied to the same tissue in rats. When the same sections were stained by an anti-rat TGF-α polyclonal antibody, however, the localization of signals differed significantly and never overlapped, suggesting that these two antibodies detected different proteins.
Our experiments also demonstrated that the anti-human TGF-α MAb was not absorbed by rat TGF-α34-50 up to an excess of 100-fold and failed to detect synthetic rat TGF-α, although it reacted with recombinant human TGF-α and several rodent proteins other than TGF-α as well as with bovine carbonic anhydrase (CA). Further blotting analysis revealed that one of the most intense signals on rodent protein sample lanes was CAII, suggesting that the signals detected in rodent tissues by immunohistochemistry might be CAII rather than TGF-α, at least under our experimental conditions. This is supported by the results of immunohistochemical studies using antibodies against CA or CAII, in which rat and mouse renal tubule epithelium showed clear positive staining (Ridderstrale et al. 1992; Nitta et al. 1989; Holthofer et al. 1987). Careful comparison revealed that distribution of CA or CAII in the rodent kidneys (Ridderstrale et al. 1992; Nitta et al. 1989; Holthofer et al. 1987) coincided with that of positive signals detected by the anti-human TGF-α MAb in both our present study and in rat studies reported by Deguchi et al. (1993) and Walker et al. (1991). The cause of nonspecific staining shown by the anti-human TGF-α MAb is unclear at present, and there appears to be no sequence homology between CAII and TGF-α.
Diaz-Ruiz et al. (1993) and Hormi et al. (1995) reported that the anti-human TGF-α MAb was neutralized by rat TGF-α, giving rise to a discrepancy from our results. There were several differences between their experiments and ours: (a) their peptide for neutralizing the antibody was an entire sequence of mature TGF-α, whereas we used TGF-α34-50; (b) the concentration of their primary antibody was much higher than ours; and (c) buffers used in their experiments differed from ours (Hormi et al. 1995; Diaz-Ruiz et al. 1993). These points suggest that the antibody may detect rat TGF-α under certain experimental conditions. Even if this is the case, it is still unclear whether the signal detected in their immunohistochemical studies was TGF-α or CAII (or other proteins), owing to a lack of direct evidence that the antibody had no cross-reactivity with proteins other than TGF-α under their study conditions.
Results of Western blotting analysis using an anti-human TGF-α MAb (
The present competition study revealed that the signal detected by the anti-rat TGF-α polyclonal antibody was abolished by preincubating the antibody with a 10-fold excess or more of TGF-α34-50. The result coincided with that obtained by Snedeker et al. (1991), in which positive staining found in the mouse mammary gland was blocked by preincubation of the antibody with the same peptide. In the Western blotting analysis, the antibody detected both human and rat purified TGF-α without showing any signals on mouse and rat protein sample lanes. These observations lead to the conclusion that the anti-rat TGF-α polyclonal antibody is specific to both human and rat TGF-α without crossreactivity with other rodent proteins.
Another way to ensure the specificity of antibodies may be to compare data from immunocytochemical studies with those obtained by Northern blotting analysis. According to Nelson et al. (1992), the uterus of ovariectomized mice expressed TGF-α mRNA in response to estrogen treatment. However, the level of TGF-α protein was extremely low even in these estrogen-treated uteri, and no TGF-α mRNA was detected in the untreated uterus. TGF-α mRNA has also been reported to be expressed in the uterus of rats and mice during the early stage of pregnancy (Tamada et al. 1991; Han et al. 1987). Detailed observations have suggested that the mRNA was expressed predominantly in the maternal decidua but not in the uterus itself in rats (Han et al. 1987). In the kidneys, expression of TGF-α mRNA has not been found in normal mice and rats (Deguchi et al. 1993; Vaughan et al. 1992) or at least was barely detectable (Walker et al. 1991), although in some cases simultaneous immunohistochemistry using the anti-human TGF-α MAb showed intense signals as described previously.
In the present immunohistochemical study, the polyclonal antibody against rat TGF-α showed only weak staining in mouse and rat kidneys but not in normal mouse uterus. The antibody also detected no obvious signals in protein samples extracted from these tissues in Western blotting analysis. These results, absent or very limited expression of TGF-α protein in the normal uteri and kidneys, appear to be consistent with above-mentioned mRNA data (Deguchi et al. 1993; Nelson et al. 1992; Vaughan et al. 1992; Tamada et al. 1991; Walker et al. 1991; Han et al. 1987), suggesting again that the anti-rat TGF-α polyclonal antibody used in our experiments is specific for rodent TGF-α and that the other antibody recognizes some other protein, such as CAII. It is very likely, therefore, that the level of TGF-α protein is extremely low and is almost equal to or less than the detectable limit in the normal uteri and kidneys of mice and rats. Further studies are needed to clarify the distribution of TGF-α in rodent tissues using an antibody that has been proved to be specific for rodent TGF-α.
Acknowledgments
We thank Dr Richard P. DiAugustine (NIEHS) for his kind gift of synthetic rat TGF-α34-50 and Dr Richard E. Tashian (University of Michigan) for purified mouse CAII and goat anti-mouse CAII serum. We also thank Drs Izumi Horikawa (NIEHS) and David Malarkey (NIEHS) for critical review of this manuscript.
