Abstract
It has recently been shown that several members of the tetraspan superfamily, including CD9 and CD63, associate with each other and with β1 integrins. In this study, we examined the distribution of a recently identified tetraspan, PETA-3 (CD151), and of CD9, CD63, α5β1, and the integrin β1 chain in normal human tissues by the indirect immunoperoxidase and alkaline phosphatase-anti-alkaline phosphatase techniques. PETA-3 showed a broad distribution and was expressed by endothelium, epithelium, Schwann cells, and dendritic cells and by skeletal, smooth, and cardiac muscle. Expression in skin was mostly restricted to the basal cells of the epidermis and was downregulated on differentiation. In the small intestine, PETA-3 was expressed by crypt and villous enterocytes with a mostly basolateral distribution, but was not detectable on the brush border. CD9 was expressed on the plasma membrane of enterocytes in crypts and at the bases of the villi whereas CD63 demonstrated a unique granular appearance concentrated in the apical cytoplasm below the brush border. The findings of this study show co-localization of PETA-3 with CD9, CD63, α5β1, and β1 in particular tissues, demonstrating that tetraspan/integrin complexes may occur. However, the lack of co-localization of these antigens in other tissues also implies distinct roles for these molecules.
Members of the TM4SF are characterized by four highly conserved hydrophobic regions that are believed to be membrane-spanning which show little homology with other proteins containing four transmembrane domains (Wright and Tomlinson 1994; Horejsi and Vlcek 1991). Also characteristic of tetraspan proteins is the presence of a large divergent extracellular loop between the third and fourth membrane-spanning domains, which has been suggested to define the functional specificity of the particular tetraspan. Members of the TM4SF have been identified in a diverse range of organisms, including Schistosoma (Wright et al. 1990), Drosophila (Kopczynski et al. 1996), and humans (Wright and Tomlinson 1994). This evolutionary conservation of tetraspans suggests an integral role for these molecules in cell biology.
Although the biochemical functions of the TM4SF are unknown, several studies using monoclonal antibodies (MAbs) have shown tetraspans to mediate a broad range of biological responses. Anti-CD9 and anti-PETA-3 MAbs are able to induce platelet activation and mediator release by crosslinking of FcgammaRII, the low-affinity IgG receptor (Roberts et al. 1995; Ashman et al. 1991; Worthington et al. 1990). More recent studies in platelets have demonstrated intrinsic signaling capacity for CD9, with anti-CD9 F(ab')2 fragments inducing phosphorylation of the tyrosine kinase syk (Ozaki et al. 1995). Signal transduction events have also been demonstrated in B-cells, granulocytes, and monocytes by anti-CD53 MAbs (Olweus et al. 1993). MAbs against CD9 and CD81 can cause homotypic adhesion of pre-B-cells (Masellis-Smith et al. 1990; Takahashi et al. 1990). Increased adhesion of neutrophils to endothelium (Forsyth 1991) and adhesion of pre-B-cells to bone marrow fibroblasts through the modulation of fibronectin receptors has also been demonstrated for anti-CD9 MAbs (Masellis-Smith and Shaw 1994). A potential role for tetraspans in modulating adhesion events has been further supported by the finding that several members are associated with integrin complexes. Initially, CD9 was demonstrated to associate with the αIIb/β3 complex in platelets (Slupsky et al. 1989), and subsequent studies have highlighted associations with integrins of the β1 subfamily. For example, immunoprecipitation studies have shown CD9 to be associated with both α4β1 and α5β1 in the pre-B-cell line NALM-6, whereas in the erythroleukemia-derived cell line HEL, CD9 was associated only with α4β1 (Rubinstein et al. 1994). CD63 has been demonstrated to associate with α3β1 and α6β1 integrins in HT1080 fibrosarcoma cells and in transfected K562 cells expressing either the α3 or α6 chain (Berditchevski et al. 1995). Furthermore, CD9, CD63, and CD81 were demonstrated to associate with each other and α3β1 to form tetraspan/integrin and tetraspan/tetraspan/integrin complexes (Berditchevski et al. 1996). Similarly, PETA-3 has recently been shown to associate with α5β1, and CD63 in MO7e and HEL cells (S. Fitter, unpublished data) and CD9 and the integrin β1 chain in platelets (P. Sincock, unpublished data). The physical association of several tetraspan members with integrins suggests a possible role for tetraspans in modulation of integrin function.
Previous studies using MAb 14A2.H1 have shown expression of PETA-3 on platelets, endothelium, some myeloid leukemia cells, and epithelium of tonsil sections (Ashman et al. 1991). Furthermore, studies of PETA-3 mRNA distribution by Northern blotting analysis showed mRNA expression in all tissue homogenates examined, with the exception of brain (Fitter et al. 1995). Because PETA-3 is expressed by endothelium, it was unclear whether or not the vasculature component of the tissues examined accounted for their positivity by Northern analysis. Furthermore, the association of PETA-3 with α5β1, CD9, and CD63 by biochemical studies suggests that these antigens may be co-expressed with PETA-3 in particular tissues. This study investigated the tissue localization of the newly described PETA-3 and the possibility that it is co-localized with CD9, CD63, α5β1, and β1 chain in normal human tissues.
Materials and Methods
Tissues
Normal adult human tissue specimens were obtained with approval from the Royal Adelaide Hospital Ethics Committee. Tonsil and gut were obtained through surgical procedures and embedded within 1 h. Postmortem tissues, including brain, skeletal muscle, cardiac muscle, skin, kidney, liver, lung, pancreas, spleen, thyroid, and adrenal gland, were obtained within 6 hr of death. Solid tissues were placed in Tissue-Tek OCT compound (Miles; Elkhart, IN) and frozen by immersion into isopentane precooled in liquid nitrogen. Skeletal and cardiac muscle were not embedded in OCT but were plunged directly into liquid nitrogen and stored at — 70C. Fresh frozen air-dried sections (5 μm) were fixed in 95% ethanol for 10 min at 4C and washed with three changes of cold (4C) PBS before staining by the indirect immunoperoxidase technique. Peripheral blood was collected in heparin and bone marrow mononuclear cells (BMMNCs) were obtained and Ficoll-purified as previously described (Simmons et al. 1994). Blood and cytocentrifuge slides were allowed to air-dry and were stored at — 70C before staining with the alkaline phosphatase–anti-alkaline phosphatase (APAAP) technique.
Monoclonal Antibodies
The anti-PETA-3 MAb 11B1.G4 (IgG2a) was raised by fusion of splenocytes from Balb/c mice immunized with MO7e megakaryoblastic leukemia cells with the X-63.Ag8.653 myeloma cell line. The resultant hybridoma supernatants were screened on FDCP1 cells transfected with PETA-3 cDNA (Fitter et al. 1995). Anti-CD9 MAb 1AA2.H9 (IgG1) (Cole et al. 1989) and anti-CD63 MAb 12F12.1G2 (IgG1) (Zannettino et al. 1996) were raised as described. The MAb 61.2C4 (IgG1) against the β1 integrin chain (unpublished) was a kind gift from Dr. J. Gamble (Hanson Centre for Cancer Research) and the anti-α5β1 MAb PHM2 (IgG1) (Hancock et al. 1983; Becker et al. 1981) was kindly supplied by Dr. Randall Faul (Renal Unit, Royal Adelaide Hospital). Isotype-matched negative control MAbs 1B5 (IgG1), anti-Giardia (G. Mayrhofer, unpublished) and Sal-1 (IgG2a), anti-Salmonella (O'Connor and Ashman 1982) were also included in all experiments.
Immunohistochemistry
All solid tissues were examined by the indirect immunoperoxidase technique, and peripheral blood smears and bone marrow aspirates were examined by the APAAP technique. To block Fc-mediated binding, MAb culture supernatant was supplemented with 10% heat-inactivated normal rabbit serum before application to sections. For indirect immunoperoxidase staining, sections were allowed to incubate for 1 hr at 4C with MAb. After washing in three changes of cold PBS, sheep anti-mouse immunoglobulin (Ig)-peroxidase (Amersham; Poole, UK) diluted 1:20 in 10% NRS was applied and incubated for 1 hr at 4C. After further washes, sections were brought to room temperature and peroxidase activity was detected by incubation with 1.3 mM 3,3′-diaminobenzidine tetrahydrochloride (Sigma; St Louis, MO) containing 0.07% hydrogen peroxide (Sigma). Sections were then washed in PBS, counterstained with Mayer's hematoxylin, dehydrated through alcohol and safsolvent (Ajax Chemicals, Australia) gradients, and mounted with Depex (BDH Gurr; Sydney, Australia). APAAP staining was performed as previously described (Ashman et al. 1991), with the exception that cells were fixed with methanol.
Immunohistochemical localization of PETA-3, CD9, CD63, α5β1, and the β1 integrin chain in normal human tissues a
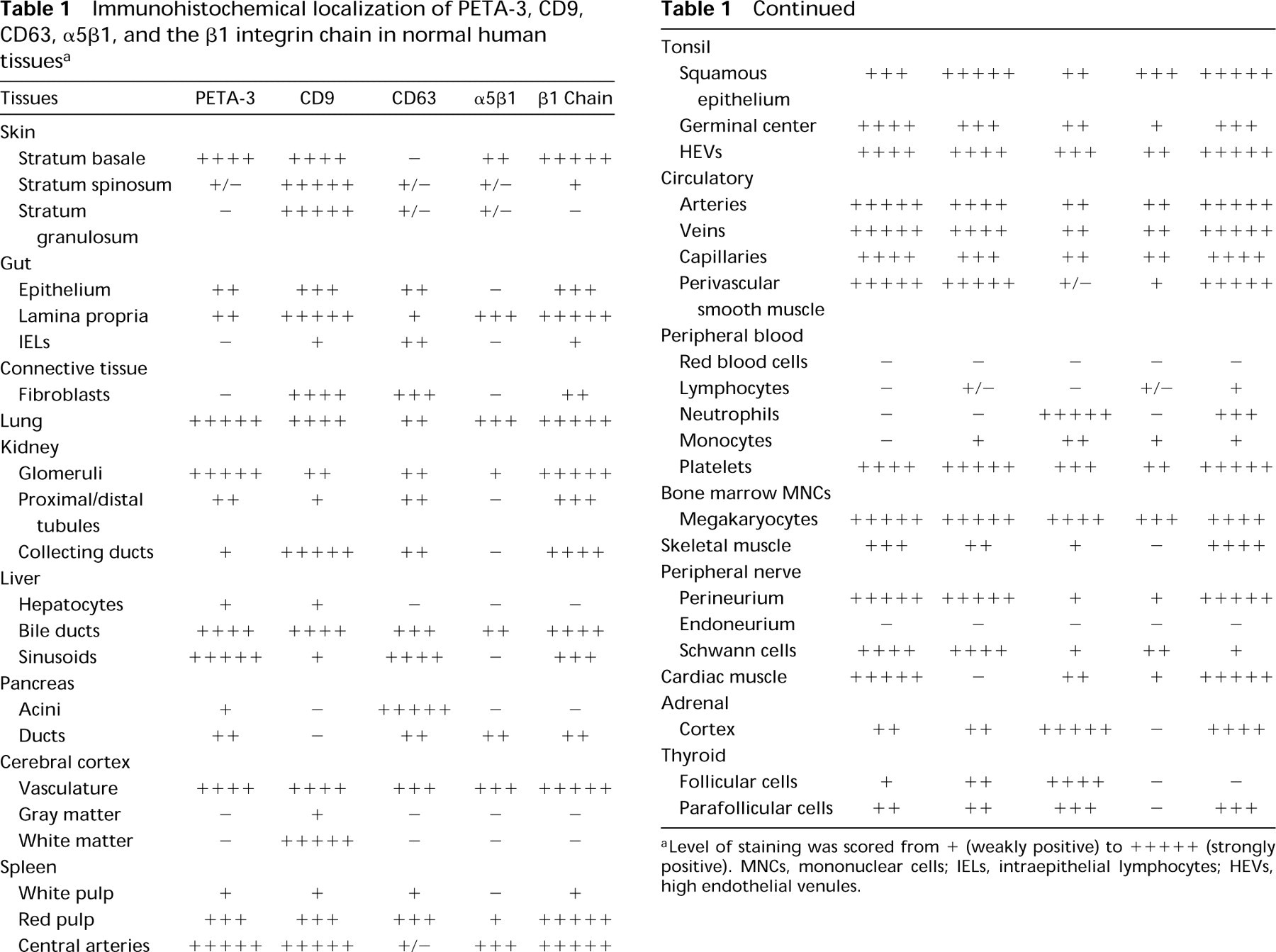
Level of staining was scored from + (weakly positive) to +++++ (strongly positive). MNCs, mononuclear cells; IELs, intraepithelial lymphocytes; HEVs, high endothelial venules.
Results
All tissues were examined for expression of PETA-3 using MAb 11B1.G4 in the indirect immunoperoxidase assay. Adjacent sections were stained with MAbs against CD9 (1AA2.H9), CD63 (12F12.1G2), the β1 integrin family (anti-β1 MAb 61.2C4), and the α5β1 integrin (PHM2). The findings are summarized in Table 1. Isotype-matched negative control MAbs were included in all experiments as specificity controls. Apart from the endogenous peroxidases of eosinophils, alveolar macrophages, and Kupffer cells, no other cells in any of the tissues stained with these MAbs by the indirect immunoperoxidase technique. One exception was tonsillar follicular dendritic cells, which showed low levels of binding of the IgG2a control Sal-1. No staining of peripheral blood/bone marrow cells was seen with negative control MAbs by the APAAP technique.
Tissue Distribution of PETA-3, CD9, CD63, α5β1, and the Integrin β1 Subfamily
Vascular endothelium was stained strongly with MAbs against PETA-3, CD9, and β1. Perivascular smooth muscle and several connective tissue components were also positive. A similar distribution was seen for α5β1. However, the degree of staining was much weaker and connective tissue components failed to react. CD63 was identified on endothelium and connective tissue components, but was not detected on smooth muscle. The following descriptions are of PETA-3, CD9, CD63, α5β1, and β1 integrin localization within the tissues examined. The results obtained are summarized in Table 1.
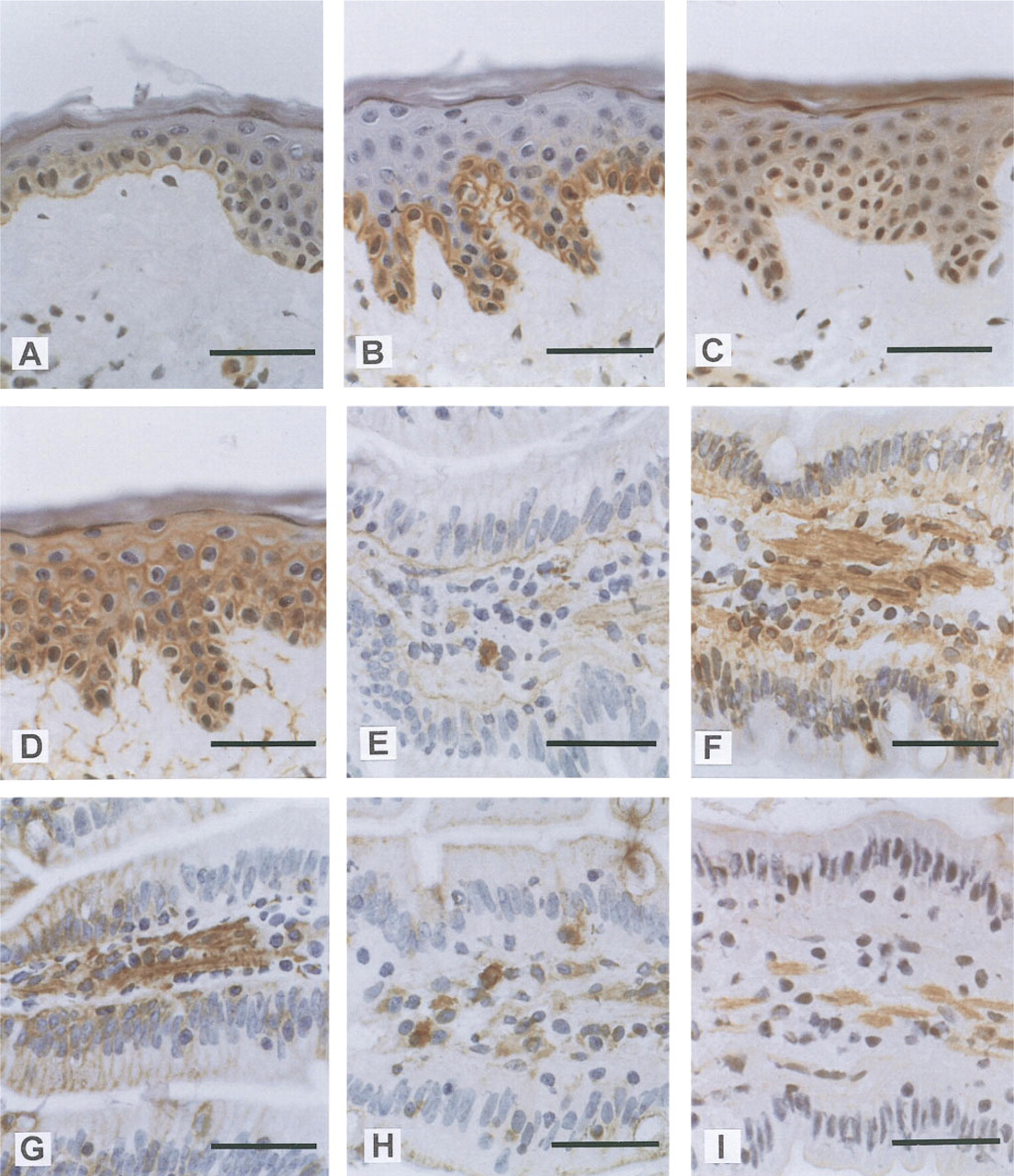
Immunohistochemical localization of tetraspans and β1 integrins in human skin and small intestine.
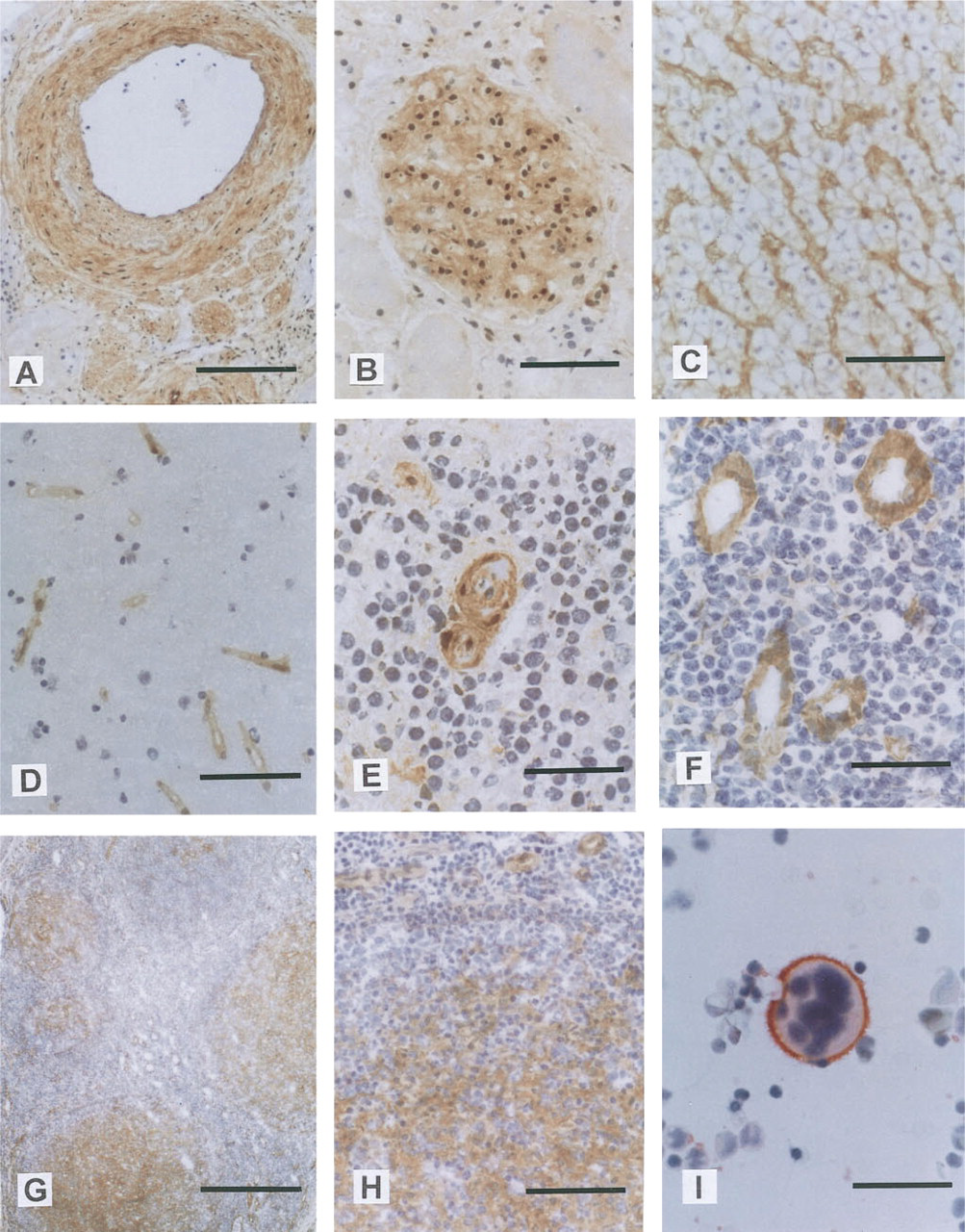
Immunohistochemical localization of PETA-3 in blood vessels, including specialized endothelium, in tonsil, and in BMMNCs.
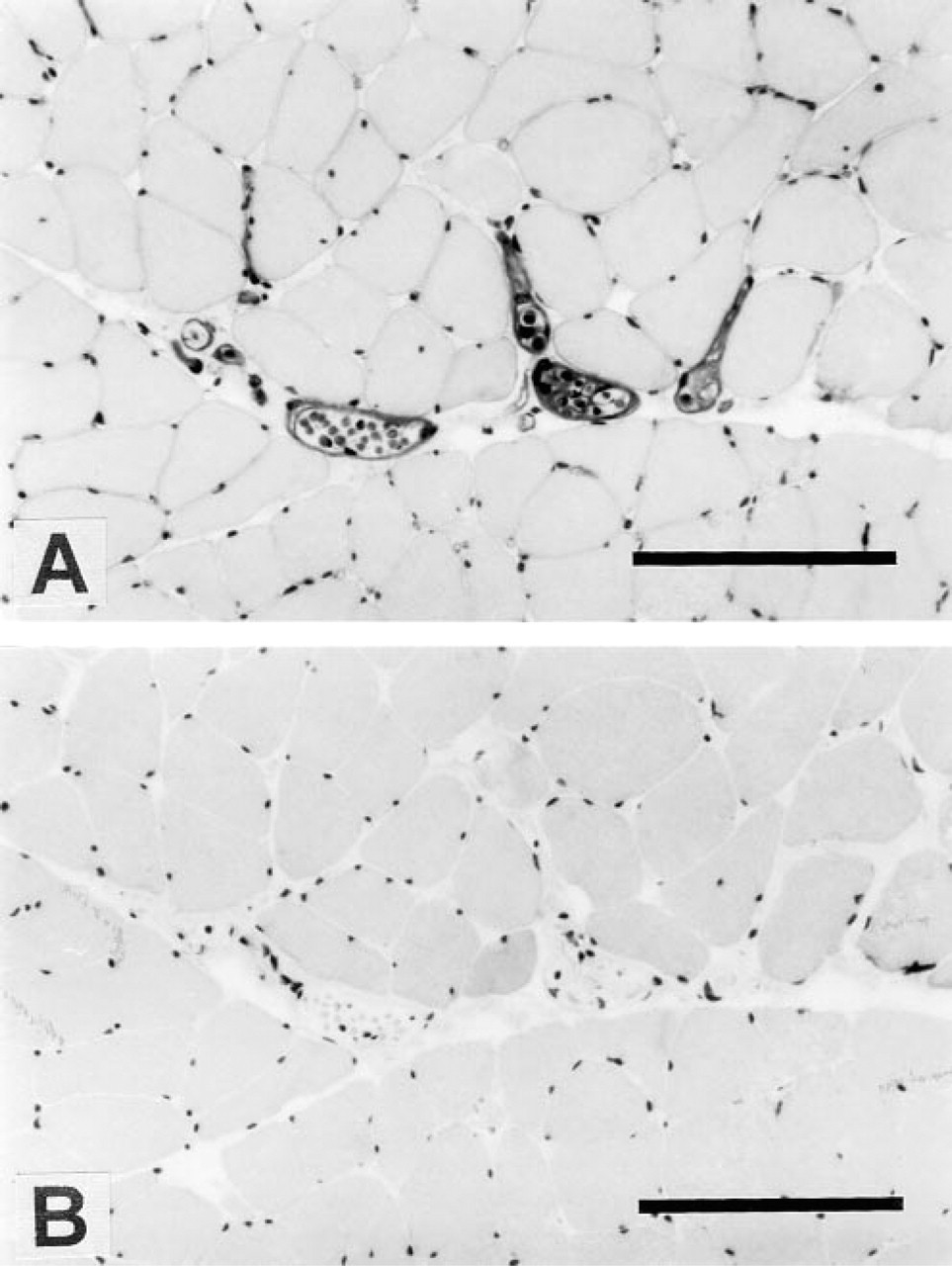
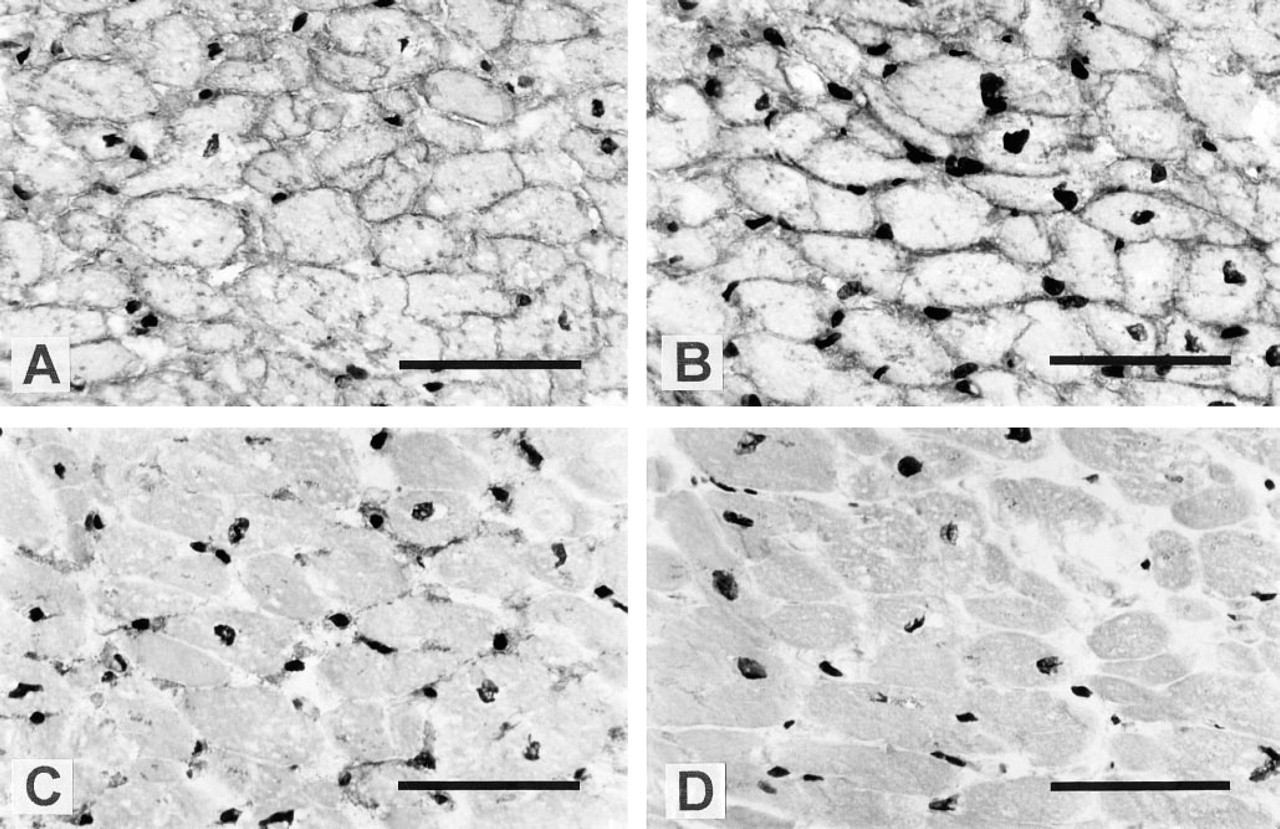
Immunohistochemical localization of
Discussion
The association of tetraspan molecules with each other and with β1 integrins has suggested a role for these proteins in the modulation of integrin function (Berditchevski et al. 1995,1996; Rubinstein et al. 1994). PETA-3, like other members of the TM4SF, appears to share these associations. Complexes containing PETA-3 in association with CD63 and α5β1 in both MO7e and HEL cells (S. Fitter, unpublished data) have been shown. In addition, PETA-3 has been demonstrated to co-precipitate with CD9 and the integrin β1 chain from platelet lysates (P. Sincock, unpublished data). Although much emphasis has been placed on the characterization of these associations using cell lines, the distribution of PETA-3 in vivo and its co-localization with associated antigens in various tissues have not been studied in detail. Recently, it was shown that CD9, CD63, CD81, and α3β1 co-localized in some cells in tissue sections of skin, breast, tonsil, and colon (Berditchevski et al. 1996). Furthermore, co-immunoprecipitation experiments provided direct evidence demonstrating the existence of tetraspan/tetraspan/integrin complexes involving these molecules (Berditchevski et al. 1996). Because PETA-3 has also been demonstrated to be involved in such complexes, the aim of our investigation was to determine the tissue distribution of PETA-3 and to compare systematically the localization of known associated molecules, including CD9, CD63, and the α5β1 integrin. PETA-3 was readily detected on vascular endothelium and smooth muscle in all tissues examined. Fibroblasts and some other undefined cells within connective tissues also expressed PETA-3, CD9, CD63, and β1 integrins, but α5β1 was undetectable. The widespread expression of these antigens throughout connective tissue probably reflects their interaction with components of the extracellular matrix such as collagen, laminin, and fibronectin.
Expression of PETA-3 by vascular endothelium was constant throughout all tissues examined, including regions of specialized endothelium (e.g., glomeruli, HEVs, hepatic sinusoids, alveolar capillaries, and capillaries constituting parts of the blood-brain barrier). The widespread expression of PETA-3 in endothelium suggests that its primary role may be in the maintenance of vascular integrity. This could be achieved through cooperation with integrins in modulating adhesion events with extracellular matrix, between endothelial cells, or with formed elements of blood. In addition, the widespread expression of PETA-3 suggests that endothelial activation by locally produced proinflammatory cytokines does not regulate the level of expression. Such regulation of expression was seen with CD63, a known marker of endothelial activation (Vischer and Wagner 1993), which was not detected within central arteries of spleen yet was expressed within HEVs of the tonsil and most other vasculature. Interestingly, PETA-3 was expressed on the vasculature of brain tissue, although PETA-3 mRNA had not been previously detected by Northern analysis (Fitter et al. 1995). CD63, α5β1, and β1 were also restricted to the vasculature in brain tissue, whereas CD9 was expressed by other tissue components as well. Expression of CD9 in the brain parenchyma has been previously described (Rossler et al. 1992), and results of the present study are similar. Interestingly, the restriction of the β1 integrin chain to the vasculature in brain suggests that CD9, unlike the other tetraspans, may be predominantly associated with another integrin β-chain or that it has an integrin-independent function in these cells.
Throughout most tissues in this study, the tetraspans investigated showed similar patterns of distribution. The co-localization of the tetraspan antigens with each other and with β1 integrins, in some cases α5β1, is consistent with the concept that they can associate to form tetraspan/integrin or tetraspan/tetraspan/integrin complexes. PETA-3 did co-localize with α5β1 in several tissues examined, especially those that showed very high levels of PETA-3 expression. However, the stronger staining of β1 integrin throughout most tissues indicated the presence of other β1 integrins. Therefore, PETA-3 may associate with other β1 integrins in such cells.
PETA-3 was expressed at relatively high levels by cardiac muscle cells and was also expressed on striated skeletal muscle. The lack of co-localization of PETA-3 with α5β1 in muscle cells again suggests that other β1 integrins may associate with PETA-3 in vivo. Because it has been demonstrated that CD9 associates with more than one β1 integrin, i.e., α4β1 and α3β1, it is possible that PETA-3 also associates with several β1 integrins.
PETA-3 expression was also observed in a number of epithelia. In the skin and tonsil, staining was restricted to the basal layers of the stratified epithelia and exhibited polarity, with brightest staining adjacent to the basal membrane. In contrast, CD9 was expressed by all keratinocytes in skin and showed an opposite polarity in cells of the stratum basale. The presence of PETA-3 close to the basement membrane suggests that it is involved in anchoring of cells to the basal lamina. The loss of PETA-3 by keratinocytes as they leave the proliferative compartment and lose contact with the basal lamina further supports this hypothesis. In contrast, CD9 may be primarily involved in interactions between keratinocytes, as suggested by its expression by keratinocytes throughout the epidermis and decreased expression on the basal membrane in cells of the stratum basale. β1 integrin expression was also restricted to the basal layers, suggesting that association with PETA-3 is involved in anchoring to the basement membrane.
Expression of PETA-3 by simple columnar or cuboid epithelia also demonstrated a basolateral distribution, especially in villous and crypt enterocytes of the small intestine. Whereas α5β1 expression was not detected on these cells, the β1 integrin chain was present in a similar polarized distribution, again providing further evidence that PETA-3 may associate with other members of the β1 subfamily. The possibility of PETA-3 associations with other β1 integrins is presently being investigated. The basolateral distribution of PETA-3, CD9, and the β1 chain in epithelial cells implies that they may be involved in interactions of the cells with the basement membrane and possibly in intercellular adhesion. The expression of CD9 by gut epithelium was restricted to immature enterocytes of the crypts and the bases of the villi. Therefore, CD9 may be regulated during epithelial differentiation and its function may be limited to interactions with specific extracellular matrix components in the crypt microenvironment. In contrast to PETA-3 and CD9, CD63 expression was localized to granules or structures located in the apical cytoplasm, below the brush border. Ultrastructural studies would be necessary to determine whether CD63 is expressed on the brush border membrane. CD63 has been shown to possess a lysosomal targeting sequence (Metzelaar et al. 1991) and was demonstrated to localize in Weibel-Palade bodies of endothelial cells and platelet granules (Vischer and Wagner 1993; Nieuwenhuis et al. 1987). The apical region of the enterocyte cytoplasm is rich in multivesicular bodies that express major histocompatibility complex (MHC) Class II proteins (Mayrhofer and Spargo 1990). Therefore, CD63 may be expressed in the early endocytic pathway in enterocytes. Furthermore, other members of the TM4SF, CD37, CD53, CD81 (TAPA-1) and CD82 (R2/C3) have been shown to associate with MHC Class II molecules (Angelisova et al. 1994; Schick and Levy 1993), suggesting that CD63 is also involved in these complexes.
Simple epithelia in several other tissues also expressed very high levels of PETA-3. Such structures included bile ducts, terminal bronchioles, and pancreatic intralobular ducts. CD9, CD63, and β1 integrins showed similar localization in these tissues, although CD9 was not detected on pancreatic ducts. Interestingly, staining of bile duct and pancreatic duct epithelia for these antigens was diffuse, suggesting cytoplasmic localization. However, studies on biopsy material would be required to eliminate postmortem degeneration as a cause for this appearance.
Previous studies with MAb 14A2.H1 have characterized the expression of PETA-3 in hemopoietic cells within bone marrow and peripheral blood. These studies showed expression to be restricted to megakaryocytes and platelets (Ashman et al. 1991; von dem Borne 1989). In tonsil sections, PETA-3 expression was reported on the squamous epithelium and blood vessels (Ashman et al. 1991). The results of the present study are in general agreement with these previous findings on hemopoietic cells and tonsil. However, some immature cell types within the BMMNC population and follicular dendritic cells within tonsil follicles also showed PETA-3 expression. The most likely cause of these differences may be the use of MAb 14A2.H1 in the earlier study. 14A2.H1 displays weaker staining of the PETA-3 antigen than MAb 11B1.G4, which was used in the present investigation. Furthermore, the recognition of PETA-3 by 14A2.H1 has been demonstrated to be fixation- and glycosylation-sensitive. In the present study methanol rather than buffered formol acetone was used as fixative. Lymphocytes in blood and in organized secondary lymphoid tissues (spleen and tonsil) did not express PETA-3 or the other tetraspans investigated. An interesting exception was the population of IELs in the small intestine. These cells, which appear to be extrathymically-derived T-cells, express several unique antigens, including the αEβ7 integrin (Yuan et al. 1991). This appears to be the first report describing expression of tetraspan molecules CD9 and CD63 by IELs, and their restriction to this subset of lymphocytes may be related to the specialized microenvironment occupied by these cells.
The tissue distribution of CD9, as described in the present study, is in general agreement with previous work (Rossler et al. 1992; Bouchiex et al. 1985; Jones et al. 1982), with the exception of skeletal and cardiac muscle. Skeletal muscle had been reported as being negative and cardiac muscle positive by anti-CD9 MAb ALB6 or DU-ALL-1. The opposite was observed in this study. Such discrepancies may arise because different anti-CD9 MAbs may not necessarily be equivalent in their recognition of CD9. For example, the glycosylation state of CD9 in different cell types may vary, thus modifying antigen recognition by certain antibodies.
In summary, this study has shown that PETA-3 co-localizes with β1 integrins and the other tetraspans CD9 and CD63 in particular tissues. However, it is important to note that distinct localization of several antigens investigated occurred in several tissues. This may indicate that tetraspan molecules share common functions in a number of tissues but that they may also have specific functions in some cell types. This is exemplified by PETA-3 in cardiac muscle, CD9 in tissue components of the brain and medullary rays of the kidney, CD63 in pancreatic ancinar cells, and the apical localization of CD63 in enterocytes. Furthermore, co-expression of these antigens by a given cell type may not always imply that they form complexes. For example, compartmentalization of CD63 within intracellular granules in enterocytes would prevent its association with surface antigens. Whether the complexes formed by tetraspans and integrins are constitutive or inducible remains to be investigated. However, the co-localization of these antigens in normal tissues provides a basis for such interactions to occur.
Footnotes
Acknowledgements
We thank Kathy Cash for assistance with cutting the tissue sections, Antony Cambareri and Ly Nguyen for producing the 11B1.G4 antibody, and Stephen Fitter for valuable discussion.
PMS is a postgraduate scholar of the National Heart Foundation of Australia. LKA is a Senior Research Fellow of the National Health and Medical Research Council of Australia.
